Abstract
A novel Polyacrylamide superparamagnetic iron oxide nanoparticle platform is described which has been synthetically prepared such that multiple crystals of iron oxide are encapsulated within a single Polyacrylamide matrix (PolyAcrylamide Magnetic [PAM] nanoparticles). This formulation provides for an extremely large T2 and T2* relaxivity of between 620 and 1140 sec−1 mM−1. Administration of PAM nanoparticles into rats bearing orthotopic 9L gliomas allowed quantitative pharmacokinetic analysis of the uptake of nanoparticles in the vasculature, brain, and glioma. Addition of polyethylene glycol of varying sizes (0.6, 2, and 10 kDa) to the surface of the PAM nanoparticles resulted in an increase in plasma half-life and affected tumor uptake and retention of the nanoparticles as quantified by changes in tissue contrast using MRI. The flexible formulation of these nanoparticles suggests that future modifications could be accomplished allowing for their use as a targeted molecular imaging contrast agent and/or therapeutic platform for multiple indications.
Keywords
Introduction
Advances in nanoparticle technology over the last decade have shown that some of these materials have the potential to play an important role in the diagnosis and treatment of cancers. Nanoparticles are typically defined as particles in the nanometer size range (ranging typically from 10 to 200 nm) with colloidal properties so that particles do not settle out of the solution. Nanoparticles of many different compositions and purposes have been described [1]. Size, charge, hydrophilicity, and composition of nanoparticles can be manipulated to achieve optimal uptake within a tumor and specific targeting ligands (i.e., peptides, monoclonal antibodies, small molecules) can be attached to the surface to provide for tissue specificity. For molecular imaging, four classes of nanoparticles are of primary interest: (1) magnetic nanoparticles, (2) magneto fluorescent nanoparticles, (3) fluorescent nanoparticles (e.g., fluorochrome doped nanoparticles, quantum dots), and (4) isotope tagged nanoparticles.
Molecular imaging employs a variety of imaging modalities including positron emission tomography [2–4], optical imaging [5–8], and MRI [9–12]. Here, a platform technology to create biologically relevant nanoparticles having magnetic properties for the purpose of serving as an MRI contrast agent is described. Two generic types of magnetic nanoparticles have been used over the last two decades: primary iron oxide nanoparticles (the core of the nanoparticle consists of an iron oxide crystal, coated by a polymer) and polymeric nanoparticles that contain iron oxide crystals doped into their matrix. The former group of agents consists largely of dextran-coated iron oxide preparations, which are typically either monocrystalline (MION, CLIO) [13–19] or polycrystalline (PION) [13,19]. These agents have been used extensively for preclinical work and commercial preparations have been used in clinical trials. CLIO particles represent a stabilized dextran-coated iron oxide nanoparticle preparation specifically designed for targeting [13,17,18]. In these particles, the iron oxide core is caged by the cross-linked dextran coating so that there exists no equilibrium between free and iron oxide-associated dextran moieties.
An alternative method to developing targeted agents is to incorporate iron oxide crystals into polymer matrices, an approach investigated in this report (Figure 1). Using Polyacrylamide (PA) as the particle core and polyethylene glycol (PEG) to prolong blood half-lives, the in vitro and in vivo properties were investigated. Relaxivity, particle size, plasma and tissue half-lives, imaging characteristics, and potentially targeting principles were of primary interest in this study. Because the materials presented are somewhat larger (mean radii of 60 nm) than conventional monocrystalline iron oxide particles (mean diameter of 10 nm), they revealed a higher relaxivity per individual particle [20]. These particles exhibit superparamagnetic behavior at room temperature. The chemical flexibility offered by these nanoparticles provides an opportunity for incorporation of molecular targeting, therapeutic and contrast agents within a single preparation.

Schematic diagram of a PAM nanoparticle. This particle contains SPIO nanoparticle crystals encapsulated within the inner matrix of the core Also shown are surface attached PEG residues.
Materials and Methods
Chemistry—Synthesis and Characterization
Iron oxide encapsulated nanoparticles were synthesized by mixing an aqueous solution (4 mL) containing acrylamide (27% w/v), 3-aminopropylmethacrylamide (5% w/v), N,N-methylene(bis) acrylamide (9% w/v), and iron oxide nanocrystals (7.5% w/v, ˜10 nm average size, Ferrotec, Nashua, NH, USA) with hexane solution (90 mL) containing polyoxyethylene-4-laurylether (Brij 30, 7.1% v/v) and bis(2-ethylhexyl)sulfosuccinate (AOT 3–5% w/v). Nanoparticle synthesis was initiated by ammonium persulfate (10%, 0.06 mL) and N,N,N′,N′-tetra-methylethylenediamine (TEMED, 0.08 mL). The nanoparticle solution was evaporated and the resulting thick residue was thoroughly washed in ethanol, and then dried to produce a fine black powder of nanoparticles.
“PEGylation” was performed by suspending the nanoparticles in sodium phosphate buffer (pH 7.4) and an equimolar ratio of monomethoxy polyethylene succinimidyl ester. The reaction mixture was concentrated and the particles were thoroughly washed with water followed by washing with phosphate-buffered saline (PBS). The required amount of the iron oxide encapsulated nanoparticles was then resuspended in PBS. Particles were synthesized either with no PEG in the synthetic mixture (PAM0) or with PEG of the following sizes: 0.6 kDa (PAM0.6), 2 kDa (PAM2), and 10 kDa (PAM10).
Mean particle size, size distribution, and polydispersity of Polyacrylamide magnetic (PAMs; dried, then redispersed in aqueous buffer solutions) were measured at room temperature by a Dawn EOS Light-Scattering Instrument (Wyatt Technology, Santa Barbara, CA). Particles were first suspended at a concentration of 2 mg/mL in PBS and sonicated for 30 min to 1 hr to reduce any aggregation. Samples were filtered through a 1.2-μm filter (25-mm diameter, Pall, Ann Arbor, MI) before injection into the 20-mL sample loop of an asymmetrical flow field–flow fractionation liquid separation system (Consenxus, Germany). The output of the separation channel was directed into the multi-angle static light-scattering instrument, where 15 detectors simultaneously measured the scattered intensity every 0.5 sec. The multi-angle data were used at each time point to calculate the particle number density and volume within that sample slice.
Cell lines. Rat 9L glioma cells (Brain Tumor Research Center, University of California, San Francisco) were grown as monolayers in minimal essential medium supplemented with 10% fetal calf serum, 100 IU/mL penicillin and 100 μg/mL streptomycin at 37°C in a 95/5% air/C02 atmosphere. Cells were harvested and resuspended for tumor implantation at a concentration of 105 cells/5 μL.
Animal model. Intracerebral 9L tumors were induced in male Fischer 344 rats weighing between 125 and 150 g. Briefly, 9L cells (105) were implanted in the right forebrain at a depth of 3 mm through a 1-mm burr hole. The surgical field was cleaned with 70% ethanol and the burr hole was filled with bone wax to prevent extracerebral extension of the tumor. Animals were imaged using MRI beginning at 12 days postcell implantation to select tumors between 60 and 80 μL in volume for in vivo nanoparticle studies.
Nanoparticle administration. PAMs were administered as a suspension (approx. 40 mg/mL) in normal saline by tail-vein injection at a dose of 200 mg PAM/kg body weight (approx. 7 mg Fe/kg). Briefly, an Angiocath was placed in the tail vein of the animal flushed with 10 units/mL of heparin and a pre-prime infusion line of PAM were connected to the Angiocath. PAMs were injected over 30 sec during single-slice dynamic MR scanning (see below).
MRI in vitro. MRI was performed using a 12-cm bore, 7 Tesla Varian animal imaging system (Varian, Palo Alto, CA). For in vitro relaxivity and contrast testing, PAMs (Table 1) were diluted to 8 mg/mL in deionized water, and placed in separate tubes to a volume of 2 mL. A saline sample was also made up in the same volume, and all tubes were imaged within a single field of view (FOV) using a 38-mm quadrature volume coil.
R1, R2, and R2* Measured using a 7-T MRI System (25°C) for each PAM Preparation

All values expressed as mean ± SD.
All images were acquired with a FOV of 35 × 35 mm over a 128 × 128 matrix and using a slice thickness of 2 mm. The number of averages was varied depending on the repetition time used. T1- and T2-weighted images were acquired using a spin-echo sequence. For Ti-weighted images, the echo time (TE) was set to 9 msec and the repetition time (TR) was varied between 100 msec and 10 sec. Intensities within manually drawn regions of interest (ROIs) were used to calculate R1 for each sample. In T2-weighted images, TR was fixed to 2 sec and TE was varied between 9 and 100 msec, enabling a measurement of R2. For measurement of R2* and analysis of T2*-weighted contrast, gradient-echo images were acquired with TR fixed at 1 sec, and TE varied between 2 and 100 msec.
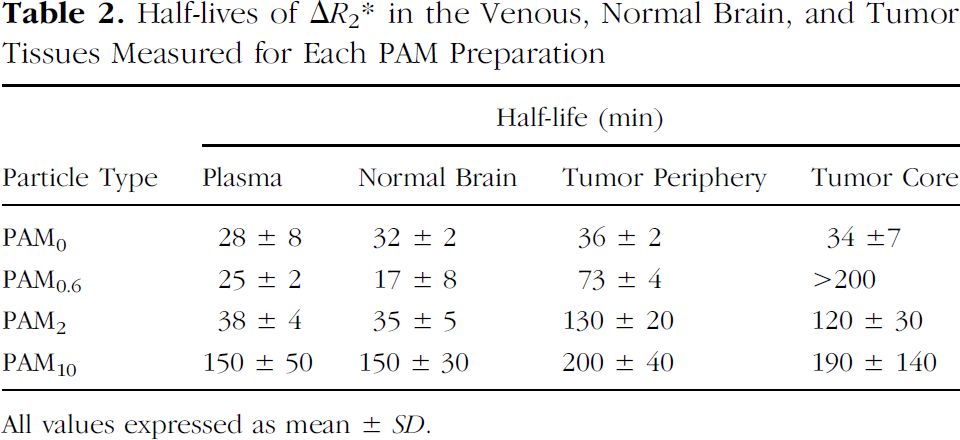
MRI in vivo. To determine the distribution and preliminary pharmacokinetic behavior, MR images were obtained using dynamic T2*-weighted gradient-echo MRI. After animal preparation as described above, axial slices of the rat brain were acquired using a 30 × 30 mm FOV over a 128 × 128 matrix, a 1-mm-thick slice, TR = 10 msec and TE = 5 msec. Pre-scans were acquired before intravenous injection of PAMs and immediately following the injection, the dynamic course of signal decrease was followed by continuous acquisition of 20 images of duration 2.5 sec each, to allow observation of the rapid changes in contrast within the vasculature as well as normal and tumor tissue. After this, images were acquired at approximately 8-min intervals over a 2-hr time period. Images were also later acquired at long time-points (12+ hr). Images were analyzed by measuring signal intensity time courses within manually drawn ROIs in the vein, normal brain, tumor periphery, and tumor core. Relative concentration of the nanoparticle contrast agents was derived from the signal intensity:
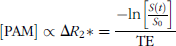
Results
In Vitro Characterization of Nanoparticle Preparations
Modification of the surface by attachment of various PEG sizes did not result in any significant change in the mean hydrodynamic radii (63 ± 6 nm). Measurement of the MR relaxivity of PAM preparations by T2*-weighted imaging (Figure 2) revealed that addition of PEG moieties of varying size did not significantly alter the relaxivity (Table 1). More importantly, data presented in Table 1 reveal R2 and R2* relaxivity in the range 620–1140 sec−1 mM−1, which is approximately fivefold greater than that which has been measured in other superparamagnetic iron oxides (SPIOs) [21]. The R1 values were in the range of 1.7 to 2.4 sec−1 mM−1, which is significantly lower than R1 values measured in other SPIO preparations [21].

T2*-weighted (TR = 2 sec, TE = 7 msec) gradient-echo MRIs of tubes containing 2 mL (clockwise from top left) of PAM0, PAM10, PAM0.6, and PAM2 nanoparticles (0.8 mg/mL) and a separate tube containing saline (center).
In Vivo Characterization of Nanoparticle Preparations
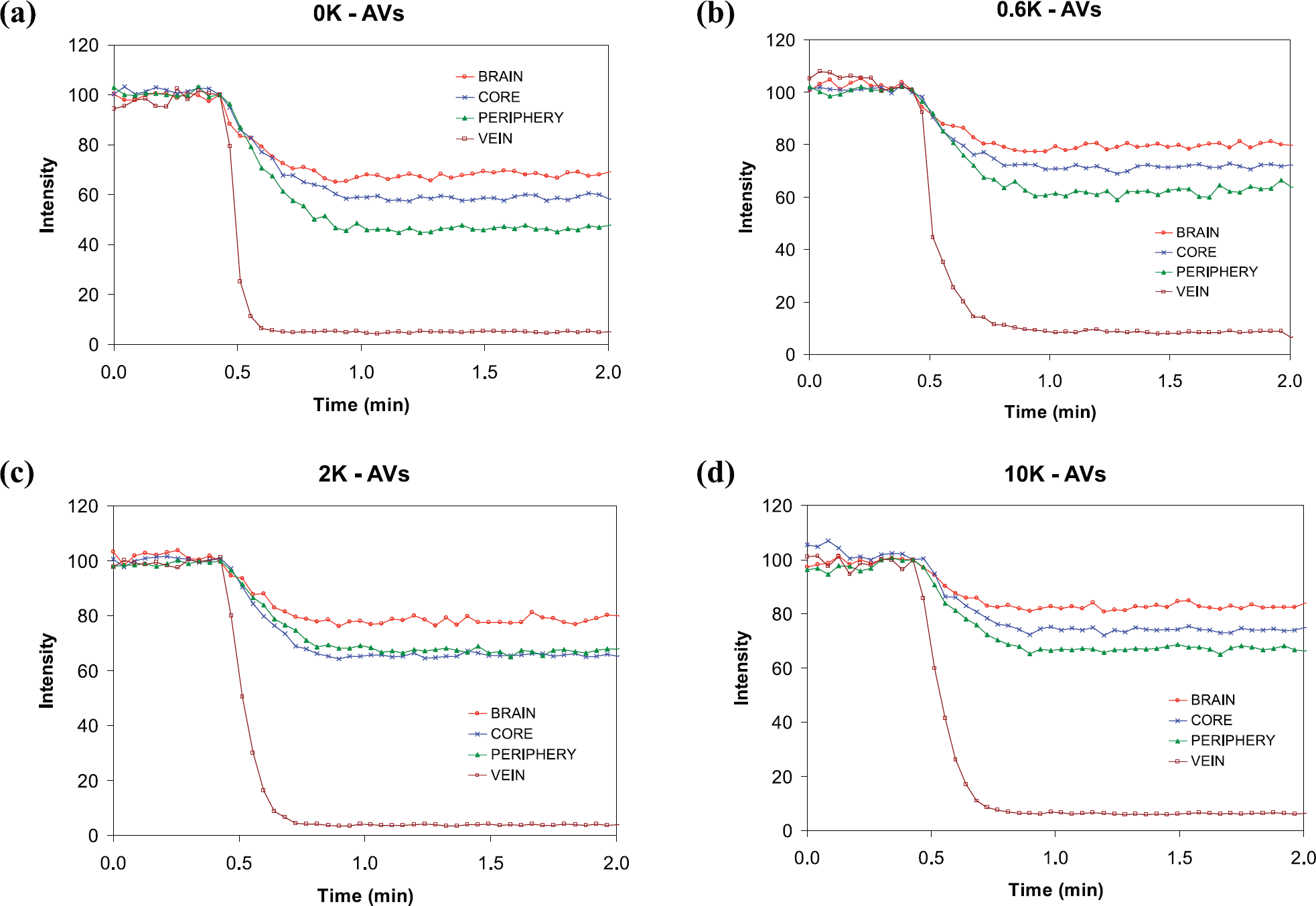
The usefulness of SPIO nanoparticles depends on the ability of the nanoparticles to produce significant contrast enhancement in the tissue or tumor of interest. In order to assess the viability of PAMs for in vivo applications, dynamic changes in tissue contrast were studied in blood, brain, and tumor tissues. All nanoparticle preparations were evaluated following intravenous administration in intracerebral 9L tumor-bearing rats (n = 3 animals/PAM preparation). The precontrast agent injection scans (0 sec) revealed that the vessels corresponding to the jugular vein and the carotid artery were hyperintense and the tumor tissue was isointense relative to the contralateral normal brain. Intravenous injection of 1 mL nanoparticle (42.5 mg/mL) resulted in a rapid decrease in vascular MR signal intensity as observed in the images shown in Figure 3, over 30 sec. A decrease in signal intensity within the brain, muscle, and tumor was also observed due to the presence of PAMs within the vasculature of these tissues (Figure 3). For all nanoparticle preparations, the magnitude of the signal decrease was shown to be significantly greater in the tumor mass as compared to contralateral normal brain (Figure 4). Heterogeneity in tumor contrast was observed in the images, which most likely reflects the variations in blood vessel density throughout the tumor. As shown in Figure 4, the venous MR signal decreased dramatically by the end of the 30-sec injection period. Decreases in MR signal intensity for normal brain (approx. 20%), tumor core (approx. 30%), and tumor periphery (approx. 40–50%) occurred over 60 sec. Using Equation 1 with the pre and post (2 min) contrast images, ΔR2* maps were calculated (Figure 5). These maps can be considered as relative cerebral blood volume (rCBV) maps [19,22]. Figure 5 demonstrates the heterogeneity in ΔR2* (and therefore rCBV) in the periphery of the tumor, compared with the tumor core.

Gradient-echo MRI of the rat brain containing a 91 glioma, at 0, 30 sec, 2 min, 15 min, and 1 hr following intravenous administration of (a) PAM0, (b) PAM0.6, (c) PAM2, (d) PAM10 nanoparticles. Each image was acquired over 25 sec with TR = 10 msec, TE = 5 msec, FOV = 30 × 30 mm, and slice thickness = 1 mm. In all cases, significant time-dependent signal reduction was observed in the tumor, normal brain, muscle, and vasculature.
Mean dynamic signal time courses (n = 3 in each case) over the first 2 min following intravenous injection of (a) PAM0, (b) PAM0.6, (c) PAM2, (d) PAM10 nanoparticles in the rat for ROIs in the vasculature, normal brain, tumor periphery, and tumor core.
Representative ΔR2* maps (sec−1) of the tumor tissue (color) overlaid onto a gradient-echo image for the four different formulations of PAM nanoparticles in four different 9L tumors of similar size, (a) PAM0, (h) PAM0.6, (c) PAM2, (d) PAM10. ΔR2* was calculated using Equation 1, where t = 2 min. Therefore, the color overlays can he viewed as rCBV maps.
Partial recovery of contrast changes was observed by 1 hr post nanoparticle administration as shown in Figure 3 for PAM0, PAM0.6, and PAM2 preparations. However, for the PAM10 preparation, retention within the vasculature was observed to be much longer than in the other formulations, with significant contrast observed at 1 hr postinjection. Changes in tissue MR signal intensities (Figure 6a) were converted to relative concentrations using Equation 1 and were fitted to a monoexponential decay (Figure 6b). The exponential decay rates calculated from these nonlinear least-squared fits for each of the tissue types are displayed in Table 2.

(a) Scatter plot (n = 3) for signal intensity time courses in the vasculature shown over 80 min following intravenous injection of PAM0.6, PAM0.6, PAM2, PAM10 nanoparticles in the rat, plotted as % initial signal (S0) with fits to the data, (b) Nonlinear least square fits to relative concentration time courses derived from the intensity data.
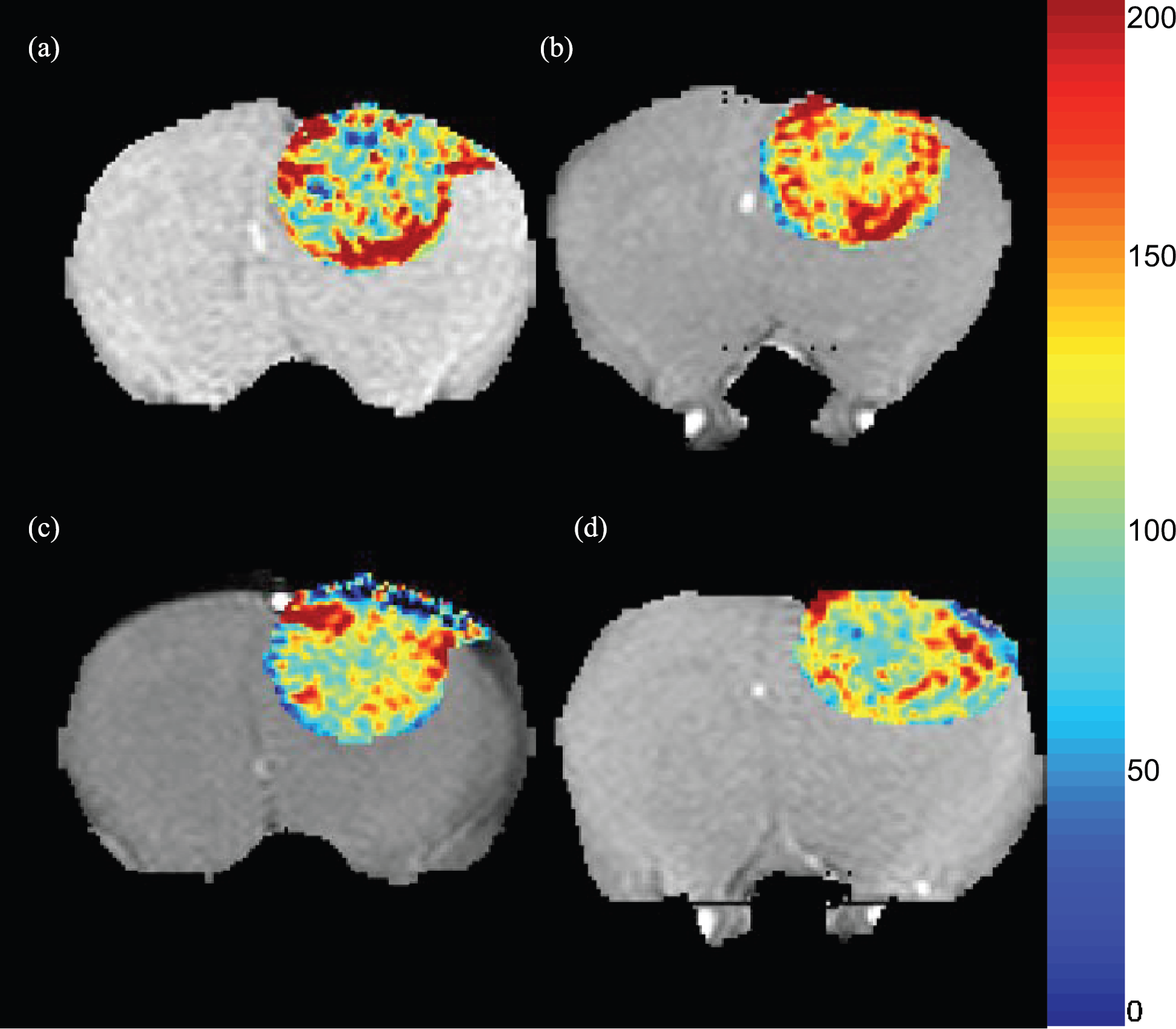
Half-lives of ΔR2* in the Venous, Normal Brain, and Tumor Tissues Measured for Each PAM Preparation
All values expressed as mean ± SD.
The calculated plasma half-life values for each of the nanoparticle preparations revealed how addition of PEG related to uptake and clearance of PAMs in the plasma, normal brain, and tumor periphery (Table 2). PAM0 and PAM0.6 particles were found to have the fastest plasma clearance times (half-life in the range of 25–30 min), while the PAM2 preparations were found to have a plasma half-life of 38 min. The PAM10 particles were found to have the slowest clearance, with a half-life of about 150 min.
For all PAM preparations, the clearance half-life from the normal brain occurred at a similar rate to the plasma. However, clearance from the tumor was significantly (outside the 95% confidence interval) retarded for PAM0.6 and PAM2 relative to plasma levels. For the PAM0.6 preparation, clearance from the tumor core was significantly slower than that in the tumor periphery, occurring over many hours, and could not be measured accurately over the time scale of our measurements.
Discussion
Recent developments in cancer therapeutics has suggested that molecular targeted therapeutics may be superior to nonspecific cytotoxins due to a larger therapeutic index [23]. These developments have resulted in a need for the ability to image molecular targets and events noninvasively over time [24,25]. Within the field of molecular imaging, radiotracer technology currently dominates due to its advantages in sensitivity. One of the disadvantages of radiotracer imaging is the lack of high spatial resolution, an aspect at which MRI excels. In an effort to overcome the limitations in MR sensitivity, SPIO technology is an area of active investigation [13–16,22,26] driven by the extremely high MR relaxivity characteristics of these contrast agents [21]. A key structural feature of the PAM nanoparticles described in this report distinguishes them from previously reported SPIOs. In contrast to a single crystal of iron oxide coated by dextran or starch, these PAM nanoparticles may contain up to 10–15 crystals of iron oxide which are incorporated into the PA matrix. The matrix provides an additional favorable property as encapsulation of iron oxide helps to shield it from interacting with vascular components thus separating the chemistry from the biology.
This study has shown that incorporation of iron oxide nanoparticles within a PA matrix during synthesis is feasible and results in a stable nanoparticle with significantly enhanced R2 and R2* relaxivity. Detection of molecular events, mediated for example by cell surface receptors within a tissue, is limited by the finite number of accessible targets. An agent with increased sensitivity due to high relaxivity, such as the PAM, is highly desirable.
The ability to modify the PA matrix to include amine-functionalized groups allows incorporation of an array of targeting materials (including proteins, peptides, and small molecules). These amine-functionalized preparations were utilized in the current study to incorporate PEG moieties of varying sizes. This resulted in an enhanced plasma half-life which increased with the size (>0.6 kDa) of the PEG subunits. The ability to alter the plasma half-life of the PAM nanoparticles by chemical modification demonstrates the versatility of the platform. For example, the use of a nanoparticle as a blood pool contrast agent requires a prolonged plasma half-life. Application as a targeted contrast agent would require a plasma half-life which allows for sufficient time to enable binding to the target followed by clearance to enable discrimination of targeted signal from background.
Addition of PEG moieties above 0.6 kDa increased the plasma half-life. The use of hydrophilic polymers such as PEG to coat the surface of nanoparticles is well known to increase plasma half-life [1]. An unanticipated finding was that the PAM0.6 particles were found to have the longest half-life within the tumor core (>200 min). The basis for this observation warrants further investigation as the phenomenon may be useful in certain applications. The 9L tumor is known to have a disrupted blood–brain barrier allowing entry of contrast agents such as GdDTPA and MION into the extravascular space [27]. However, the degree of contrast enhancement and ΔR2* was significantly higher in the tumor periphery compared with the tumor core (Figures 4 and 5), consistent with the higher vascular density characteristic of the periphery.
In summary, we have described a novel type of SPIO nanoparticle (PAM) having a high MR relaxivity. This platform echology has potential for use in a wide variety of applications, such as molecular targeting of contrast agents, delivery of therapeutic agents, and as blood pool agents for studying tumor vasculature [19,22,28]. Although iron oxide was used in the current study as the MR relaxing agent, future studies could include the incorporation of paramagnetic agents such as gadolinium chelates, which may be advantageous for certain applications.
Footnotes
Acknowledgments
This work was supported in part by the following NIH/NCI grants: P01CA85878, R24CA83099, N01CO07013, and N01CO37123. We thank Maria Moreno and Gail Roberts for helpful discussions.
