Abstract
Objective: Magnetofluorescent nanoparticles (MFNPs) offer the ability to image cellular inflammation in vivo. To better understand their cellular targeting and imaging capabilities in atherosclerosis, we investigated prototypical dextran-coated near-infrared fluorescent MFNPs in the apolipoprotein E-deficient (apo E−/−) mouse model.
Methods and Results: In vitro MFNP uptake was highest in activated murine macrophages (
Conclusions: Here we show that (1) the in vitro and in vivo cellular distribution of atherosclerosis-targeted MFNPs can be quantified by using fluorescence imaging methods; (2) in atherosclerosis, dextranated MFNPs preferentially target macrophages; and (3) MFNP deposition in murine atheroma can be noninvasively detected by in vivo MRI. This study thus provides a foundation for using MFNPs to image genetic and/or pharmacological perturbations of cellular inflammation in experimental atherosclerosis and for the future development of novel targeted nanomaterials for atherosclerosis.
Introduction
Nanoparticulate imaging agents are often of similar dimensions (1–100 nm) as molecules that interface with the surface or interior of cells, and therefore offer a unique window into the nanobiology of cells [1,2]. Indeed, a wave of novel nanomaterials have recently emerged that will ultimately enable cellular and molecular imaging in cardiovascular disease [3]. Within atherosclerosis, a variety of magnetic nanoparticles (MNPs) have been employed to report on various aspects of inflammation [3–5]. For example, up-regulation of endothelial adhesion molecules, proteases, and peroxidases play key roles in the inflammatory process and represent ideal imaging targets [6]. While MNPs specially targeted to these processes are currently being developed, a number of polymer-coated MNPs are in advanced clinical trials. MNP agents tested in atherosclerosis include carboxydextran-coated ferumoxtrans [7,8], short-circulating ferumoxides [9], and long-circulating dextran-coated ferumoxtrans [10–13]. Although some MNPs are detectable by in vivo high-resolution magnetic resonance imaging (MRI), their reporter capabilities, fate, and specific cellular distribution in atherosclerosis are not entirely understood.
To further extend the imaging capabilities of MNPs, we have developed fluorescent moieties of MNPs, or magnetofluorescent nanoparticles (MFNPs). In addition to conferring strong, reversible relaxation (R2 > R1) effects on MRI, these multimodality nanomaterials also possess a distinct fluorescence signature that permits detection in a variety of fluorescence imaging systems such as flow cytometry, fluorescence reflectance imaging (FRI), and fluorescence microscopy. These dextranated MFNPs modified with near-infrared or visible fluorochromes have been useful in imaging cellular inflammation in models of cancer [14] and diabetes [15].
To better understand the cellular targeting and imaging capabilities of MNPs in atherosclerosis, we characterized the in vitro and in vivo cellular distribution of prototypical dextran-coated MFNPs by capitalizing on their fluorescence properties. We next examined the ability of in vivo MRI to noninvasively image these atherosclerosis-targeted MFNPs and confirmed the results with ex vivo multimodality MRI and FRI.
Materials and Methods
Synthesis of the Near-Infrared Fluorescent Magnetofluorescent Nanoparticles
The imaging agent used was a monocrystalline iron oxide magnetic nanoparticle (MION-47) [16], consisting of a 3-nm core of (Fe2O3)
Quantification of MFNP Uptake by Murine Cells
MFNP uptake into activated macrophages, endothelial cells, and smooth muscle cells was quantified by using flow cytometry. Monocytes were isolated via density centrifugation and then plated onto cell-culture-treated dishes and cultured in complete monocyte medium containing human recombinant granulocyte–macrophage colony stimulating factor (GM-CSF, BD Biosciences, Rockville, MD) at a concentration of 12 ng/ml. The cells were maintained at this concentration of GM-CSF for 7 days before use in experiments. Murine heart endothelial cells were then isolated as previously described [21]. Activation was induced by incubation of 25 ng/ml of tumor necrosis factor alpha for 4 hr. For smooth muscle cell studies, NIH 3T3 cells were obtained from ATCC (Manassas, VA), passaged according to instructions, and activated with 50 μg/ml of lipopolysaccharide for 24 hr. Activated cells of each subtype were then incubated with the MFNP (0.1 mg iron/ml) for 4 hr and then analyzed via flow cytometry (10,000 cells per sample; FACSCalibur, Becton Dickinson, Franklin Lakes, NJ). The experiments were performed in duplicate or triplicate.
Apolipoprotein-E-Deficient Model of Atherosclerosis
We employed the apolipoprotein E gene deletion mouse (apo E−/−) as a model of atherosclerosis [22]. A total of 11 mice (average age 29 weeks) were used in the study. Mice received a high-cholesterol diet (TD 88137 adjusted-calories diet; Harlan Teklad, Indianapolis, IN) for an average of 19 weeks. Eight mice were injected with the MFNP (15 mg/kg iron), and 3 mice were injected with 0.9% saline. All animal studies were approved by the Subcommittee on Research Animal Care at Massachusetts General Hospital.
Magnetic Resonance Imaging
Imaging studies were performed 24 hr after MFNP or saline injection. A subset of mice (
All animals were then sacrificed and perfused with saline. For high-resolution ex vivo MRI (
NIR Fluorescence Reflectance Imaging
Resected aortae (
Cellular Assessment of MFNP Deposition in Atheroma
The resected proximal aorta was frozen in OCT compound (Sakura Finetek, CA). The well-validated Paigen's approach was used to analyze the aortic root [24]. Cryostat sections (6 m thickness) were stained with hematoxylin and eosin (H&E) for general morphology. Cells were identified with cell-specific antibodies: anti-Mac-3 for macrophages, anti-CD31 for endothelial cells (both from BD Biosciences Pharmingen), and anti-alpha smooth muscle actin for smooth muscle cells (Lab Vision, Fremont, CA). Immunofluorescent staining was performed on serial sections of the mouse aortic root. Briefly, sections treated with 0.3% hydrogen peroxide were incubated for 60 min with primary or matched control antibodies, followed by respective biotinylated secondary antibody, and finally streptavidin conjugated with Texas Red (Amersham). Nuclei were stained with 4,6-diamidino-2-phenylindole (DAPI).
Plaque sections were imaged using an upright epifluorescence microscope (Eclipse 80i, Nikon, Melville, NY) with a cooled CCD camera (Cascade, Photometrics, Tucson, AZ). Fluorescence images were obtained in four spectrally resolved channels: 630 nm (for immunofluorescent cell-specific antibodies), 710 nm (for MFNP), 400 nm (for nuclear DAPI staining), and 535 nm (for autofluorescence). Exposure times ranged from 100 to 1000 msec depending on the channel. Fluorescence images were acquired as digital 12-bit images and then fused to form multicolor fusion images (IP Lab Spectrum version 3.9.3, Scanalytics, Rockville, MD). For each of the cell types detected by immunofluorescence and DAPI staining, the number of MFNP-positive cells was counted on 10 high-power fields (hpf) for each aortic root, a common approach to quantify cells in histological sections [25].
Image Analysis
MRI and NIRF aortic images were analyzed with a shareware software package (OsiriX). Within the aorta, plaque, and adjacent tissue, signal intensities were measured by using a manual region-of-interest (ROI) tracing tool. For MRI, the blood–plaque contrast-to-noise ratio (CNR) was calculated as follows: CNR = (blood signal – plaque signal) / standard deviation of the noise. The plaque ROI in the aortic root was manually defined from the strong negative contrast in the MFNP group; a similarly sized and located ROI was used for the saline group analysis. For FRI, the plaque target-to-background ratio (TBR) was calculated as TBR = (ROI average plaque signal) / (ROI average aortic background signal adjacent to the plaque). The peak plaque TBR was reported for each animal. After image analysis, images were further windowed by using Photoshop 7.0 software (Adobe Systems, San Jose, CA).
Statistical Analyses
Data are presented as mean ±
Results
Activated Macrophages Preferentially Bind MFNPs in Vitro
To quantitatively assess the cellular distribution of MFNPs in cells found in atherosclerotic lesions, we performed flow cytometry on activated murine macrophages, endothelial cells, and smooth muscle cells incubated with the MFNPs (0.1 mg iron/ml for 4 hr; Figure 1). MFNP uptake was 7.9-fold greater in activated macrophages (mean fluorescence intensity [MFI] 108.5 ± 1.6 arbitrary units [AU]) compared to activated endothelial cells (MFI, 13.7 ± 0.8 AU;
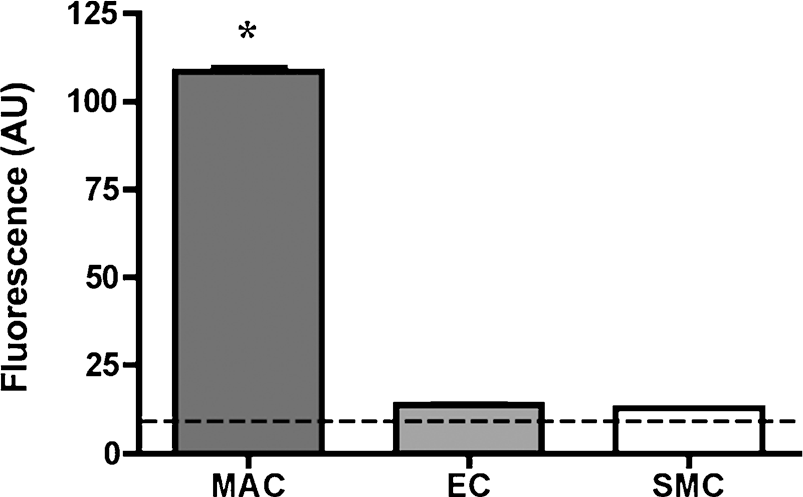
In vitro cell uptake of prototypical dextranated MFNPs in mouse atherosclerotic cells under stimulated conditions. Fluorescence intensity (arbitrary units [AU]) was measured by flow cytometry. Activated macrophages demonstrated significantly more MFNP uptake than activated endothelial cells or smooth muscle cells. The dashed line represents the autofluorescence level (10 AU) for all experiments. MAC = macrophages, EC = endothelial cells, SMC = smooth muscle cells. *p < .001, MAC versus EC, MAC versus SMC.
MFNP Deposition in Murine Atheroma Can Be Detected by in Vivo MRI
A total of seven apo E−/− mice underwent high-field MRI (

(A) In vivo bright-blood aortic MR images of representative apolipoprotein E-deficient (apo E−/−) mice. Twenty-four hours following either saline or MFNP 15 mg/kg iron oxide injection, T2*-weighted gradient-recalled echo images (TE 2.7 msec for all images) were obtained by ECG and respiratory gating by using a 9.4-T MR scanner. (Top) In apo E−/− mice that received saline, minimal signal loss was noted in the aorta (left, long axis view), aortic root (center, magnified view from dashed box in left image) and in the transverse aortic arch (right, magnified view from perpendicular slice defined by the dashed line in the left image). (Bottom) In contrast, apo E−/− mice that received MFNP demonstrated strong focal signal loss in the aortic root (left and middle, arrow demonstrating a “W” pattern of negative signal enhancement) and transverse aortic arch (right, arrow). In the MFNP group, the blood-to-plaque contrast-to-noise ratio (CNR) was 24.0 ± 0.7 (vs. 7.8 ± 0.7 in the saline group, p < .001). (B) Ex vivo 14-T high-resolution aortic MRI of representative apo E−/− mice that received MFNP. Focal MRI signal loss was noted in the aortic root and transverse arch, similar to the in vivo images (arrows). Furthermore, greater signal loss was noted with longer echo times (TE), consistent with a T2*-dependent relaxation effect from iron oxide. The cartoon below identifies the major cardiovascular structures of interest. AR = aortic root, LV = left ventricle, TA = transverse arch, CA = carotid artery.

Ex vivo macroscopic light (left) and near-infrared fluorescence (NIRF) reflectance images of representative apo E−/− mouse aortae. (Top) In control apo E−/− mice that received saline, minimal NIRF signal was noted within atherosclerotic plaque. (Bottom) In contrast, MFNP-injected mice demonstrated strong focal NIRF signal enhancement in plaques in the aortic root, transverse arch, and descending aorta, corresponding to visible plaques (arrows) on the light image (left). NIRF images were processed and windowed identically.
Ex vivo 14T confirmed the in vivo MRI findings by also demonstrating strong negative signal enhancement in aortic root and aortic arch plaques, as seen in vivo (see Figure 2). Furthermore, longer echo times substantially increased the MR signal loss in aortic plaques compared to the normal aorta, consistent with MNP deposition in plaque and a T2*-dependent effect [26].
MFNP Deposition in Atheroma Can Be Detected by Fluorescence Reflectance Imaging
Aortae from all 11 apo E−/− mice underwent macroscopic NIR FRI. In the 8 mice that received MFNPs, strong NIRF signal enhancement was noted in atherosclerotic plaques in the aortic root and aortic arch (Figure 3), confirming the results seen during in vivo and ex vivo MRI. There was good correspondence between visible aortic plaques seen on light images and NIRF signal. No significant NIRF signal enhancement was seen in the 3 control apo E−/− mice that received saline. The peak plaque-to-background ratio in the NIRF MNP group was 423% higher than in the saline group (5.9 ± 2.5 vs. 1.6 ± 0.4,
MFNPs Colocalize with Immunofluorescent Cells in Apo E−/− Atheroma
Focal NIRF signal was noted in aortic plaque sections of apo E−/− mice that received the MFNP imaging agent (Figure 4). Signal was detected in various cellular-rich areas of atherosclerotic lesions as well as the adventitia adjacent to atheroma. There was minimal NIRF signal in the cellular-poor areas, the normal aorta, or in aortic plaques of apo E−/− mice that received saline. On correlative fluorescence microscopy, MFNPs were readily identified within immunofluorescent cells in atherosclerotic lesions (Figure 5). Macrophages were the predominant MFNP-positive cell type (20.6 ± 7.3/hpf) compared to endothelial cells (6.6 ± 1.8/hpf) and smooth muscle cells (4.3 ± 1.8/hpf,

Representative microscopic NIRF and histopathological (H&E) images of adjacent sections of the aortic root of apo E−/− mice. (Top) Saline-injected mice did not demonstrate focal NIRF enhancement within atheroma (original magnification, ×25). (Middle) MFNP-injected mice, in contrast, demonstrated marked focal NIRF enhancement in atherosclerotic lesions (original magnification, ×25). (Bottom) Higher magnification image demonstrates MFNP deposition in areas of cellular infiltration (arrowheads) but not in relatively acellular areas (original magnification, ×100).

Fluorescence microscopy of MFNP distribution in atherosclerotic lesions. Cell types were immunofluorescently labeled in a distinct fluorescent channel from the NIR. (Top left) Quantitative analysis demonstrated significantly higher number of MFNP-positive macrophages per high-power field (hpf) compared to endothelial cells and smooth muscle cells (p < .0001). (Top right, bottom left, bottom right) Representative fused fluorescence microscopic images of the MFNP (red) and immunofluorescently labeled cell antigens (green). (Top right) Macrophages represented the majority of MNFP-positive cells and demonstrated yellow-orange NIRF and immunofluorescent signal colocalization (arrowheads). (Bottom left) Modest MFNP uptake into endothelial cells (arrowheads) was also seen adjacent to strongly MFNP-positive subendothelial cells, presumably macrophages (arrows). (Bottom right) Similarly, MFNP uptake by smooth muscle cells of the fibrous cap or media (arrowheads) was found adjacent to MFNP-positive subendothelial areas of atherosclerotic lesions (*, asterisk). Occasional uptake was also noted in the adventitial layer of the atherosclerotic aorta (arrow). Original magnification, ×200. MAC = macrophages, EC = endothelial cells, SMC = smooth muscle cells. *p < .001, MAC versus EC, MAC versus SMC.
Discussion
In this report, we determined the cellular targeting and multimodal imaging capabilities of clinical-type magnetofluorescent nanomaterials in experimental murine atherosclerosis. We found that both the in vitro and in vivo cellular uptake of MFNPs was significantly higher in macrophages compared to endothelial cells and smooth muscle cells. Second, we demonstrated that the MFNPs allow imaging of cellular inflammation in murine atherosclerosis by in vivo MRI, as well as ex vivo multimodality imaging. These results have implications for the development of novel atherosclerosis-targeted imaging nanomaterials and for the noninvasive assessment of inflammation in atherosclerosis.
A variety of nanosized materials has been used as imaging agents for in vivo MRI [3–5,27–29]. In the current investigation, we employed a dextran-coated MFNP with a hydrodynamic diameter of 38 nm. This size is similar to that of large molecular weight fluorescent dextrans [30] that bind to SIGNR1 and SIGNR3, recently described C-type lectins found on macrophages [31,32]. This finding provides a possible link between the MFNP's nanosize dimension, dextran coating, and cell-internalization properties. Another important aspect regarding the MFNP's size is that it can avoid detection by the reticuloendothelial system, resulting in a prolonged blood half-life and greater vascular bioavailability compared to larger sized MNP preparations [33]. Additional favorable features of the multimodal MFNP include its fluorescence readout capabilities, including FRI, fluorescence microscopy, and flow cytometry for quantitative studies.
In particular, the fluorescence properties of the MFNP allowed assessment of its cellular distribution in cell types found in atherosclerosis (Figures 1 and 5). We found that prototypical dextranated MFNPs accumulated predominantly in macrophages (65% of the MFNP-positive cells,
To first approximation, the developed MFNP uptake assay predicted the cellular distribution of the nanoparticles in vivo, as demonstrated in the current apo E−/− model. This assay can therefore provide an efficient method to characterize the cellular distribution and efficiency of novel atherosclerosis-targeted nanomaterials. What reasons might justify the development of new MFNP agents targeted to atheroma? Two observations may be relevant. First, although clinical-type MFNPs have a macrophage-dominant cellular distribution (as shown here and in other studies [7–13]), we found that the background MFNP uptake by endothelial cells and smooth muscle cells was 35% of the MFNP-positive cells. This feature may diminish the ability of clinical-type MFNP to specifically image macrophages, the key effector cell in atherogenesis and plaque rupture [6]. Second, MFNP delivery into macrophages could be augmented, in theory, by surface-modified MFNPs containing ligands that avidly bind to macrophages (e.g., to scavenger receptor A [6]). Higher concentrations of MNPs in atheroma could greatly improve the sensitivity of this approach for imaging cellular inflammation in atherosclerosis both experimentally and clinically [17].
Using high-field cardiac and respiratory-gated MRI, we demonstrated that prototypical MNPs could be imaged in murine atheroma in vivo (Figure 2). MFNP deposition in atheroma resulted in a significant tripling of the blood–plaque CNR compared to saline-injected apo E−/− mice. The blood SNR was 28% lower in MFNP-injected animals compared to saline-injected animals. This finding demonstrates that the high blood–plaque CNR was due to nanoparticle-conferred R2 relaxation effects in plaque, rather than R1 positive signal enhancement effects in blood. This feature of MNP is expected at higher B0 magnetic fields, where R1 relaxation rates fall significantly, resulting in net higher R2/R1 ratios [35]. MFNP-enhancement of atheroma was further confirmed on ex vivo MRI and NIRF imaging, demonstrating the multimodality imaging capability of the MFNP agent (Figures 2 and 3). Compared to the only other imaging study to demonstrate feasibility of MNP-enhanced MRI in murine atherosclerosis [34], there are several important differences in the present study. First, we investigated clinical-type (underivatized) MFNPs that primarily target macrophages in atherosclerosis, as opposed to a vascular cell adhesion molecule 1-targeted derivatized MFNPs. Second, we did not require the administration of an additional contrast agent (protected graft copolymer–gadolinium) to enhance the blood signal, likely because we employed powerful gradient coils, physiologic gating, and optimized MR pulse sequences. Third, we were able to image MFNP deposition specifically in the aortic root of the apo E−/− mouse, an area of particular biological interest.
There are limitations to the present study. Changes in the MFNP incubation time, iron concentration, and different cell types employed could modify the observed flow cytometry results. Similarly, the in vivo iron dose was relatively high (but less than in some other experimental studies [9,10]), which could influence its distribution in plaque. Time points other than 24 hr after MFNP injection were not investigated; other time points could result in a different cellular distribution of MFNP in apo E−/− atheroma. Lymphocyte and other minority cell populations in atheroma were not investigated in this report, but could also be part of the MFNP cellular profile. Finally, stereological or three-dimensional approaches (e.g., multiphoton fluorescence microscopy) to quantify MFNP-positive cells may improve accuracy compared to the employed high power field based method.
Collectively, these findings establish that prototypical MNFPs can report on and image cellular inflammation in murine atherosclerosis. The study provides a foundation for the noninvasive assessment of cellular inflammation in atherosclerosis, particularly in concert with structural MRI of atherosclerosis [4,5]. Potential applications in atherosclerosis include assessing the effects of genetic and/or pharmacological manipulations of the inflammatory process in vivo. In addition, the validated MFNP uptake assays are expected to be useful in defining the cellular distribution and imaging capabilities of novel atherosclerosis-targeted MNFPs.
Footnotes
Acknowledgments
The authors acknowledge Nikolai Sergeyev, PhD, for the synthesis of the magnetofluorescent nanoparticle, Karen Mendelson, BS, for histological assistance, and Mikael Pittet, PhD, for analysis of the flow cytometry data. This work was supported by NIH Grants P50-CA86355 (RW), R24-CA92782 (RW), UO1-HL080731 (RW, FJ) and by the Donald W. Reynolds Foundation (FJ, RW).
