Abstract
Introduction:
Thermosynechococcus elongatus BP1 is a thermophilic strain of cyanobacteria that has an optimum growth at 57°C, and according to previous analysis by Yamaoka et al, T elongatus BP1 cannot survive at a temperature below 30°C. This suggests that the thermophilic property of this strain may be used as a natural biosafety feature to limit the spread of genetically engineered (GE) organisms in the environment if physical containment fails.
Objective:
To further explore the growth and survivability range of T elongatus BP1, we report a growth and survivability assay of wild-type and GE T elongatus BP1 strains under different conditions.
Methods:
Wild-type and GE T elongatus BP1 cultures were prepared and incubated in the laboratory (high temperatures and constant light source) and greenhouse conditions (lower/varied temperatures and sunlight) for 4 weeks. The cell density was monitored weekly by measuring the optical density at 730 nm (OD730). To assess the survivability, a sample of each culture was added to fresh media, placed in laboratory conditions (42.2°C and 30 µE m–2 s–1) in multi-well plates and observed for growth for up to three weeks. Lastly, the number of viable cells were determined by plating a diluted sample of the culture on solid media and counting colony-forming units (CFU) after 1 day, 2 weeks and 4 weeks of incubation in laboratory or greenhouse conditions.
Results:
Our experimental results demonstrated that growth was hindered but that the cells did not entirely die within 2 to 4 weeks at warm temperatures (31.42°C-36.27°C). The study also showed that 2 weeks of exposure to cool temperature conditions (15.44°C-25.30°C) was enough to cause complete death of GE T elongatus BP1. However, it took 2 to 4 weeks for the wild-type T elongatus BP1 cells to die.
Conclusion:
This study revealed that the thermophilic feature of the T elongatus BP1 may be used as an effective biosafety mechanism at a cool temperature between 15.44°C and 25.30°C but may not be able to serve as a biosafety mechanism at warmer temperatures.
Keywords
Introduction
Genetically engineered (GE) cyanobacteria may be useful to serve as a fourth-generation bioenergy production platform that enables the conversion of sunlight, CO2, and water to biofuels and/or bioproducts by inserting a series of transgenic genes through the techniques of synthetic biology. Photosynthetic production of biofuels and/or bioproducts in this manner would represent a new type of agriculture for bioenergy, which has shown many advantages, such as the decreased competition for arable land and its eco-friendly nature.1 -7 In addition, the metabolic engineering design of the microalgae can be used to render the process of fuel production more efficient for a variety of biofuels in different species.8 -15 Although insertion of biofuel-producing transgenes into wild-type cyanobacteria is a promising technique, GE cyanobacteria may pose certain potential risks if they were to break physical containment and escape into the environment, such as horizontal gene transfer,16 -19 persistence in the environment, and potential risk to aquatic populations.20,21 Biocontainment to limit the potential spread of genetically modified organisms is an important consideration if they are to be used for commercial purposes. Approaches for biocontainment of GE cyanobacteria include additional genetic manipulation to nucleases, CO2-concentrating genes, and toxin-antitoxin systems to produce “kill switches.”22 -27 It is also important to study the growth and survivability of the GE cyanobacteria in natural environments to determine how such species would behave if exposed to the environment. We propose that biocontainment may be contained in the organism itself, by using its natural properties as a growth barrier such as producing alcohol in thermophilic cyanobacterium. This approach also reduces the need for further genetic manipulation for biocontainment.
Thermosynechococcus elongatus BP1 is a thermophilic strain of cyanobacteria that was discovered in a hot spring of Japan and thrives at an optimal temperature of 57°C.28,29 It has exhibited an inability to survive in temperatures below 30°C. 28 Upon further study to assess the survivability of wild-type as well as GE forms at select lower temperatures, it was found that its thermophilic nature may serve as a biosafety mechanism that could limit the spread of GE cyanobacteria exposed to the environment. Currently, there appears to be a lack of available literature investigating the use of thermophilic cyanobacterium as a natural biocontainment approach. In addition, with genetic modification, the behavior of the T elongatus BP1 transformants containing a cassette of foreign genes may be different from that of the wild-type BP1. This further supports the need for additional temperature studies. In the present work, the growth and survivability of a wild-type and GE T elongatus BP1 was monitored in the greenhouse of Old Dominion University in Norfolk, Virginia during 2 different periods of the year with different temperature ranges: cool temperatures (15.44-25.30°C) from March 4, 2019 to April 1, 2019, and warm temperatures (31.42-36.27°C) from July 1, 2019 to July 29, 2019.
Materials and Methods
Strains and Growth Conditions
T elongatus BP1 (BA000039.2) is a thermophilic cyanobacteria strain isolated from the Beppu hot spring in Japan and used in several research laboratories, including the Lee Laboratory at Old Dominion University. T elongatus BP1 cells were grown under a constant light source (30 μE m−2 s−1) at 42.2°C (temperature measured using a mercury glass thermometer) in a Percival incubator/environmental control room (I66LLVLXC9) with 50 to 60% relative humidity. Synechocystis PCC6803 is a freshwater strain of cyanobacteria that is used as the mesophilic control in this study. The Synechocystis PCC6803 cells were grown in a 25°C shaking incubator. All strains of wild-type and GE cyanobacteria cells were grown in liquid BG-11 medium for this study. For GE T elongatus BP1 cultures, selective pressure was added to cells by supplementing media with kanamycin (40 μg/mL) and tetracycline (10 μg/mL) for BP1-pKA and BP1-BY20, respectively.
Genetic Transformation of T elongatus BP1
Wild-type T elongatus BP1 cells were genetically transformed with the pKA (Supplementary Figure S1) or YFP-ST-R-Lipase (Supplementary Figure S2) vectors by electroporation. Wild-type T elongatus BP1 were grown in liquid BG-11 media, and the initial optical density (λ = 730 nm) was determined using a BioTek Synergy HT multimode microplate reader. To concentrate the cell liquid culture, the T elongatus BP1 cells were collected by centrifugation and resuspended in a volume of water to give an estimated OD730 of 20 (obtained by calculation). Four hundred microliters of the sample of the concentrated (OD730 of 20 obtained by calculation) T elongatus BP1 was mixed with 1 μg of designer construct DNA and electroporated using the BioRad PowerPac at 5 kV/cm, 25 μF, and 200 Ω. After electroporation, the T elongatus cells were transferred to 5 mL of warmed BG-110SA media immediately and incubated at 42.2°C (temperature measured using a mercury glass thermometer) in the Percival environmental chamber for 24 h. BG-110SA is a modified BG-11 medium in which 4 mM of (NH4)2SO4 was used as a source of nitrogen instead of KNO3. 19,33 The replacement of nitrate by ammonium in the culture medium is because the nirA promoter used in the DNA cassettes is inducible to nitrate. The cells were spread on agar BG-110SA media containing kanamycin (40 μg/mL) and tetracycline (10 μg/mL) for the BP1-pKA and BP1-BY20 transformants, respectively. After 11 to 13 days, transformant colonies that appeared were restreaked on fresh agar plates and inoculated into liquid media with their respective antibiotics. In this article, the GE T elongatus BP1 with the pKA designer cassette is referred to as BP1-pKA, 19 and the GE T elongatus BP1 with the YFP-R-Lipase designer cassette is referred to as BP1-BY20. GE T elongatus BP1-pKA is an integrative transformant that comprises the 2-keto acid decarboxylase gene, NADH-dependent ADH gene, and the kanamycin resistance gene (Supplementary Figure S1). 19 GE T elongatus BP1-BY20 is an integrative transformant containing the transgene DNA construct encoding for the yellow fluorescent protein, Rhizopus oryazae lipase gene, and the tetracycline resistance selectable marker (Supplementary Figure S2). The pKA and YFP-ST-R-Lipase constructs were introduced by homologous recombination into the genome of T elongatus BP1 at the sequence positions 1926542-1928706 and 363134-363135, respectively.
Growth Study of Wild-Type and GE T elongatus BP1 and Synechocystis PCC6803 in the Greenhouse and Laboratory Incubators
Cultures of wild-type T elongatus BP1, wild-type Synechocystis PCC 6803, and GE T elongatus BP1-(pKA/BY20) were freshly prepared for the assays in the greenhouse and incubators in the laboratory. The incubators mimic those of a laboratory setting, in which the light source is constant and temperature is controlled. For this environment, cultures were placed in the Percival environmental chamber (for the wild-type and GE T elongatus BP1) at ∼42.2°C (temperature measured using a mercury glass thermometer) and IKA KS 4000i control incubator (for the Synechocystis PCC 6803) at 25°C. Greenhouse conditions are more similar to a natural environment, in that there is a dark/light cycle and varied temperatures. Cultures were placed in the greenhouse at Old Dominion University in Norfolk, Virginia. The climate-controlled parameters (temperature and humidity, which is not relevant here since the cells are in liquid culture) of the greenhouse were deactivated during the time of the study so that the temperature and lighting vary with the natural environment observed outside. The greenhouse is a standard A-shape greenhouse, and the roofing is made of clear glass and metal. A photograph of the flasks in the greenhouse is shown in Supplementary Figure S3. Wild-type Synechocystis PCC 6803 was used as a mesophilic control culture.
The study was conducted in the greenhouse during 2 different periods of the year where temperatures and day length varied. A study was conducted in the greenhouse at cool temperatures (March 4 to April 1, 2019) and warm temperatures (July 1 to July 29, 2019). The temperature of the greenhouse was monitored using the digital temperature sensor connected to the LinkConn 1000 v 4.0 software. The temperature was recorded daily at noon (12:00
Survivability Test of Wild-Type and GE T elongatus BP1 and Wild-Type Synechocystis PCC6803 in the Greenhouse
Survivability is the ability of cells to recover from the exposure to greenhouse conditions when placed back in laboratory conditions. The survivability study was conducted following the cool and warm incubation assays. To prepare the survivability assay, a 0.5-mL sample of wild-type and GE cyanobacteria cultures in greenhouse or laboratory conditions was added to 2.5 mL of fresh BG-11 media in each well of a 24-well Costar plate in duplicate and placed in the Percival environmental incubator (∼42.2°C). After inoculation in the Percival environmental chamber, the multiwell plates were observed and photographed every week for a 3-week period for evidence of growth. The optical density at 730 nm was also monitored weekly during incubation in the Percival environmental chamber. Supplementary Figures S6 to S17 and S22 to S33 present the survivability of the greenhouse liquid cultures with an initial cell population density of 5 × 106 cells/mL and the control (laboratory-incubated) liquid cultures with an initial cell population density of 5 × 106 and 1 × 107 cells/mL.
In addition, to quantify the number of viable cells, the CFU were measured for the GE T elongatus BP1 liquid cultures; after day 1, week 2, and week 4 of greenhouse exposure, a diluted liquid culture sample was plated on BG-11 agar plates (with and without kanamycin 40 μg/mL or tetracycline 10 μg/mL). The plates were then incubated in the Percival environmental chamber (∼42.2°C) for 2 weeks. The colonies were then counted on each BG-11 agar plate, and the CFU were calculated as the viable cells/mL. Supplementary Figures S18, S19, S34, and S35 present the CFU of the control (laboratory-incubated) liquid cultures with an initial cell population density of 5 × 106 and 1 × 107 cells/mL.
Results and Discussion
Growth and Survivability of Wild-Type and GE T elongatus BP1 at Cool Temperatures (15.44°C to 25.30°C)
Cultures for wild-type and GE T elongatus BP1 were prepared and placed in a greenhouse located at Old Dominion University in Norfolk, Virginia, for a 28-day period from March 4 to April 1, 2019, during which the conditions exhibited variability in light cycle and temperature. The temperature recorded daily during the duration of the experiment at noon (12
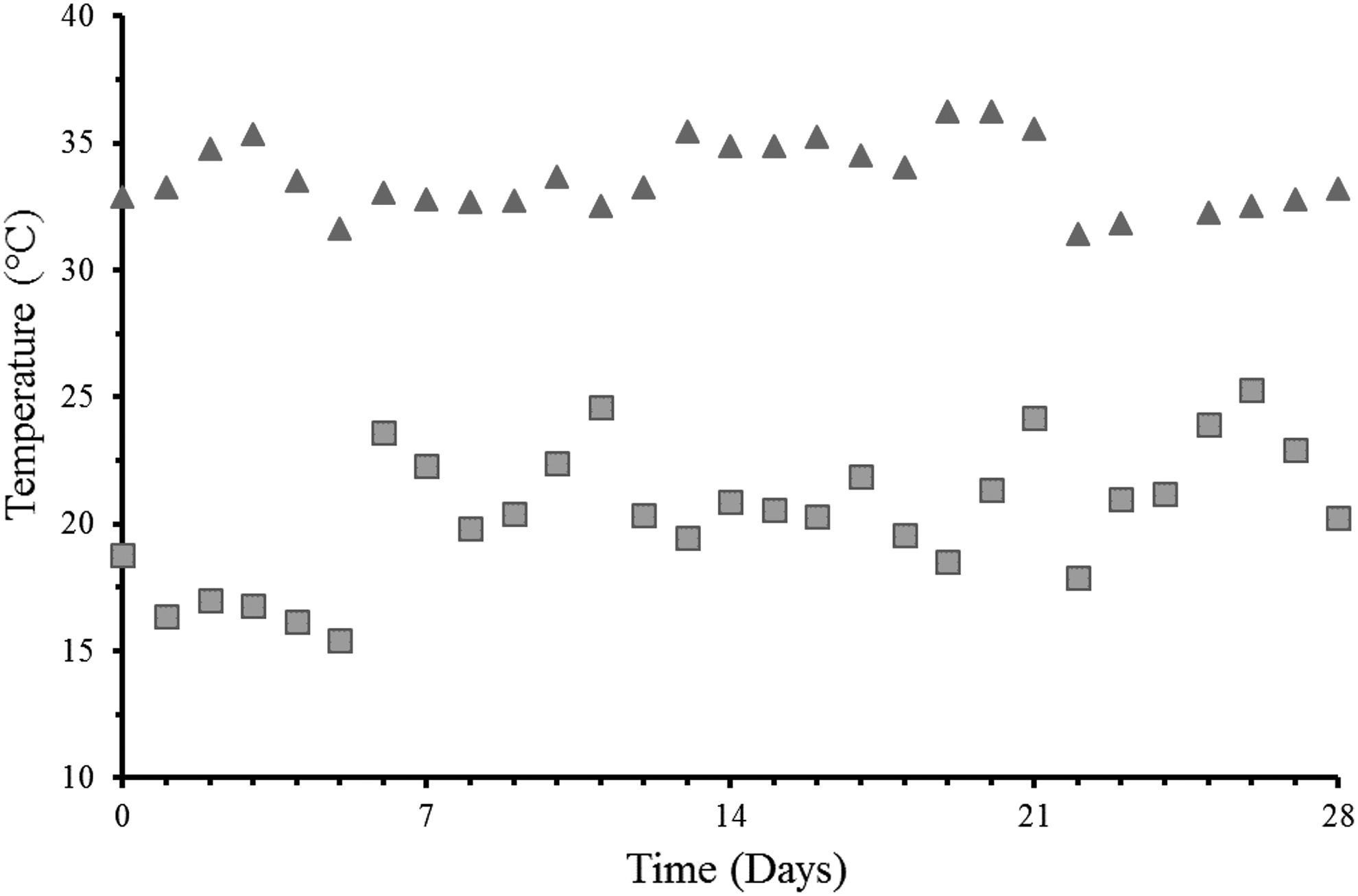
The greenhouse temperatures as recorded for the growth study during a period with cool temperatures ranging from 15.44°C to 25.30°C (represented with ▪) and warm temperatures ranging from 31.42°C to 36.27°C (represented with ▴). The temperatures were recorded daily at noon (12:00 PM) during the duration of the study. The time frames during which the temperatures were recorded were March 4, 2019 to April 1, 2019 (for the cool temperatures), and July 1, 2019 to July 29, 2019 (for the warm temperatures).
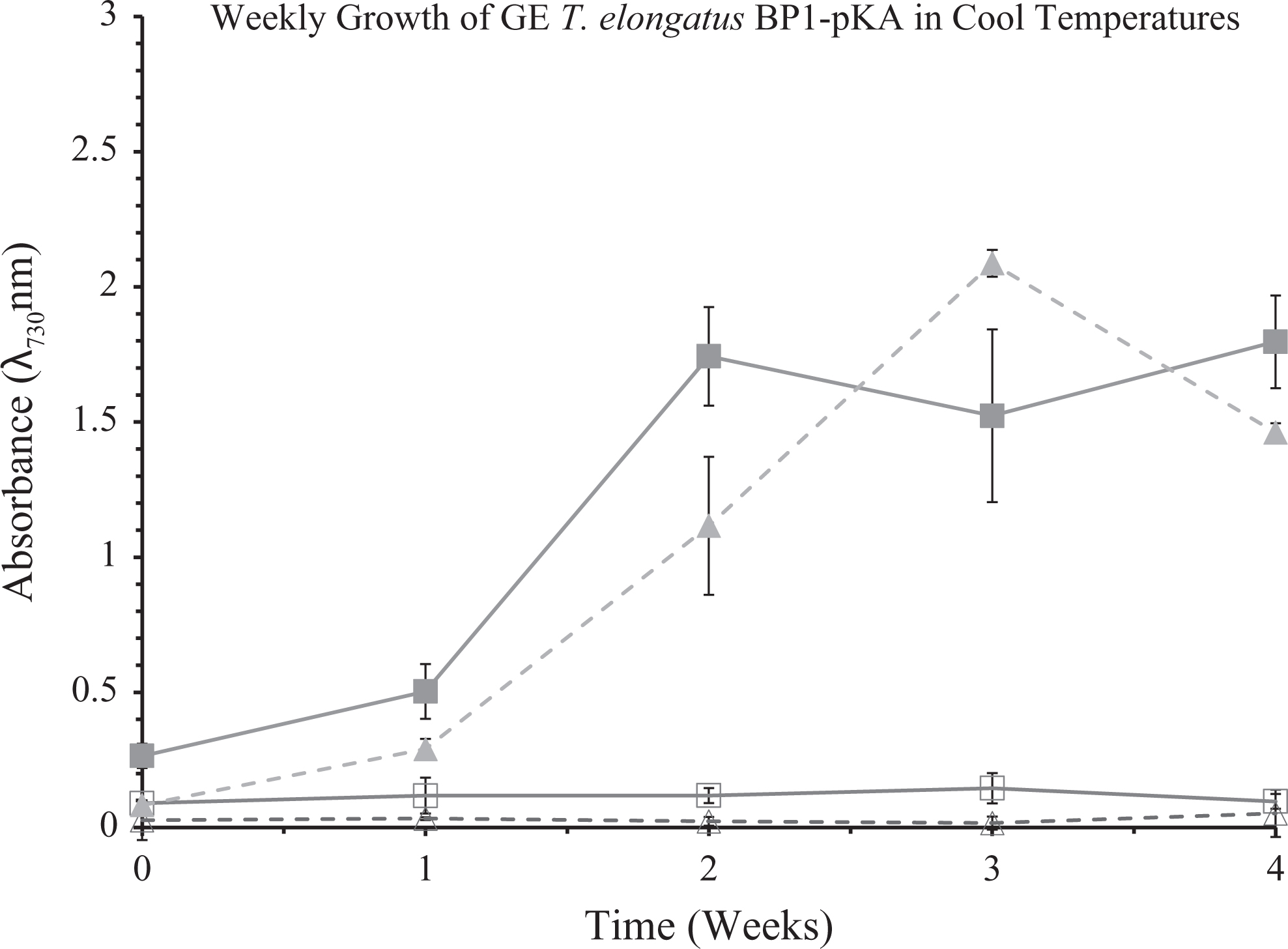
Optical density of wild-type and genetically engineered (GE) T elongatus BP1 (BP1-pKA) cultures at 1 × 107 cells/mL during cold temperatures (15.44°C to 25.30°C) were monitored weekly during a 4-week period in the greenhouse. Control cultures were grown in a Percival environmental chamber (∼42.2°C) in a Percival incubator. The results shown here are liquid culture absorbance measurements at 730 nm in duplicate for each replicate (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: (▪) is wild-type T elongatus BP1in Percival, (□) is wild-type T elongatus BP1 in the greenhouse, (▴) is the GE T elongatus BP1-pKA in Percival, and (▵) is the GE T elongatus BP1-pKA in the greenhouse. Wild-type T elongatus BP1 = solid lines, BP1-pKA = dashed lines, closed symbols = control set, open symbols = greenhouse set.
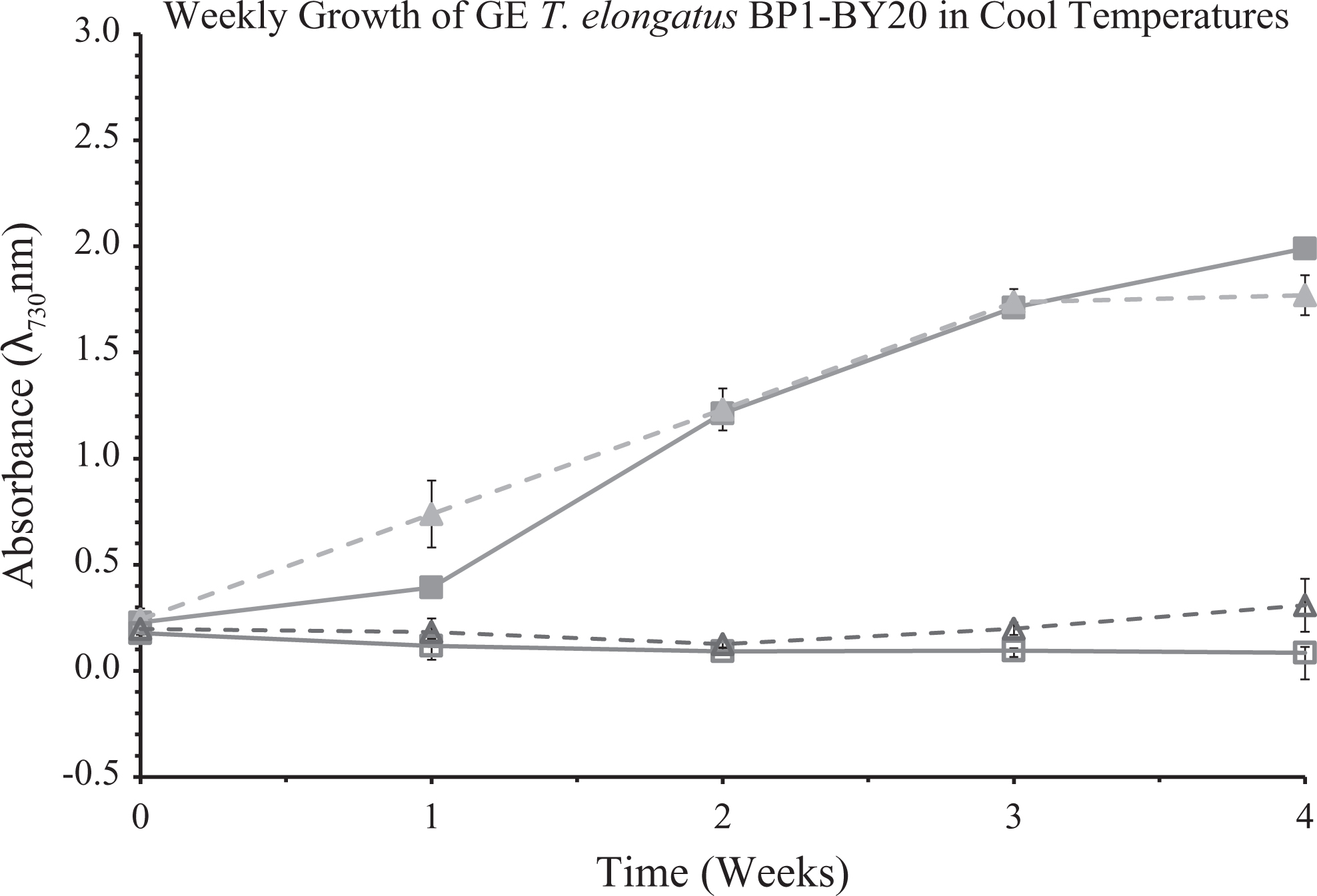
Optical density of wild-type and genetically engineered (GE) T elongatus BP1 (BP1-BY20) cultures at 1 × 107 cells/mL during cool temperatures (15.44°C to 25.30°C) were monitored weekly during a 4-week period in the greenhouse. Control cultures were grown in the Percival environmental chamber (∼42.2°C). The results shown here are liquid culture absorbance measurements at 730 nm in duplicate for each replicate (n = 6). The graph is displayed as follows: (▪) is wild-type T elongatus BP1in Percival, (□) is wild-type T elongatus BP1 in the greenhouse, (▴) is the GE T elongatus BP1-BY20 in Percival, and (▵) is the GE T elongatus BP1-BY20 in the greenhouse. Wild-type T. elongatus BP1 = solid lines, BP1-BY20 = dashed lines, closed symbols = control set, open symbols = greenhouse set.
Day Length in Norfolk, Virginia During the Different Time Frames of the Growth Study.a

The day length was retrieved from the website of Time and Date during the period of March 4, 2019, to April 1, 2019, and July 1, 2019, to July 29, 2019. 30
The optical density (OD730) measurement as shown in Figures 2 and 3 is a convenient way to monitor the growth of the liquid cultures. However, the optical density (OD730) measurement also has its limitations, as reported previously. 31 Particularly, although the observation of a flat growth curve for each of the wild-type and GE T elongatus (BP1-pKA and BP1-BY20) liquid cultures showed nearly zero growth under the greenhouse conditions, it did not necessarily indicate the complete death of the thermophilic organisms (Figures 2, 3, S4 and S5). Therefore, it is important to verify whether the thermophilic cyanobacterial cells are still viable or not after the greenhouse temperature treatment. To test the survivability of wild-type and GE T elongatus BP1 that had been exposed to the cool temperatures in the greenhouse (15.44°C to 25.30°C) for 1 week, 2 weeks, 3 weeks, and 4 weeks, a sample of the liquid cultures was taken and introduced into the wells of 24-well CoStar plates with fresh BG-11 media, subsequently placed into the Percival environmental chamber (∼42.2°C), and observed over a 21-day period for signs of photoautotrophic growth under a constant light source (30 μE m−2 s−1). As part of the control experiment with the BP1-pKA set, the wild-type T elongatus BP1 cultures (1 × 107 cells/mL) that were previously exposed to the greenhouse conditions for 1, 2, and 3 weeks showed signs of recuperation after returning to the Percival environmental chamber conditions (∼42.2°C). On the other hand, the GE T elongatus BP1-pKA sample that was previously exposed to the greenhouse conditions for only 1 week was able to grow after returning to the Percival conditions (∼42.2°C), as shown by visual observation and optical density measurement (Figures 4 and 5). The survivability study with the BP1-BY20 set showed similar results in that the GE T elongatus BP1-BY20 sample that was previously exposed to the greenhouse conditions for only 1 week was able to recuperate upon returning to the Percival (∼42.2°C) conditions (Figures 6 and 7). The wild-type T. elongatus BP1 used in that set was also able to grow back after 1 week of exposure to the greenhouse conditions; after 2 weeks of the greenhouse treatment, the cells were not able to grow back, possibly indicating complete death of the cells. Surprisingly, the BP1-BY20 that had an initial lower cell density (5 × 106 cells/mL) was not able to recuperate even after 1 week of greenhouse exposure (Supplementary Figures S8 and S9). This observation indicated that the survivability of the cells is also dependent on the initial concentration of the cell liquid cultures. This is also consistent with a previous study on the effects of initial cell density on cyanobacterial growth and proliferation. 32
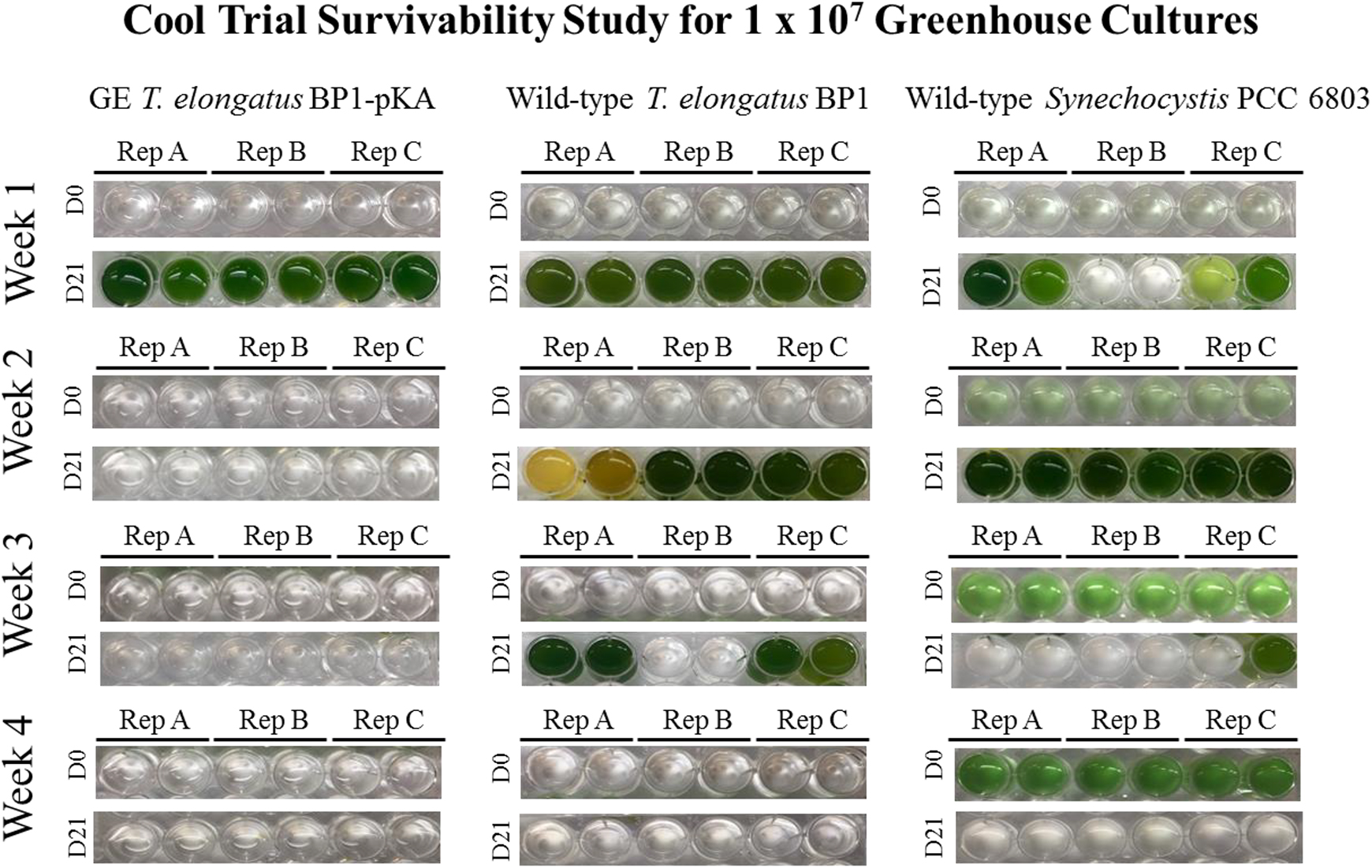
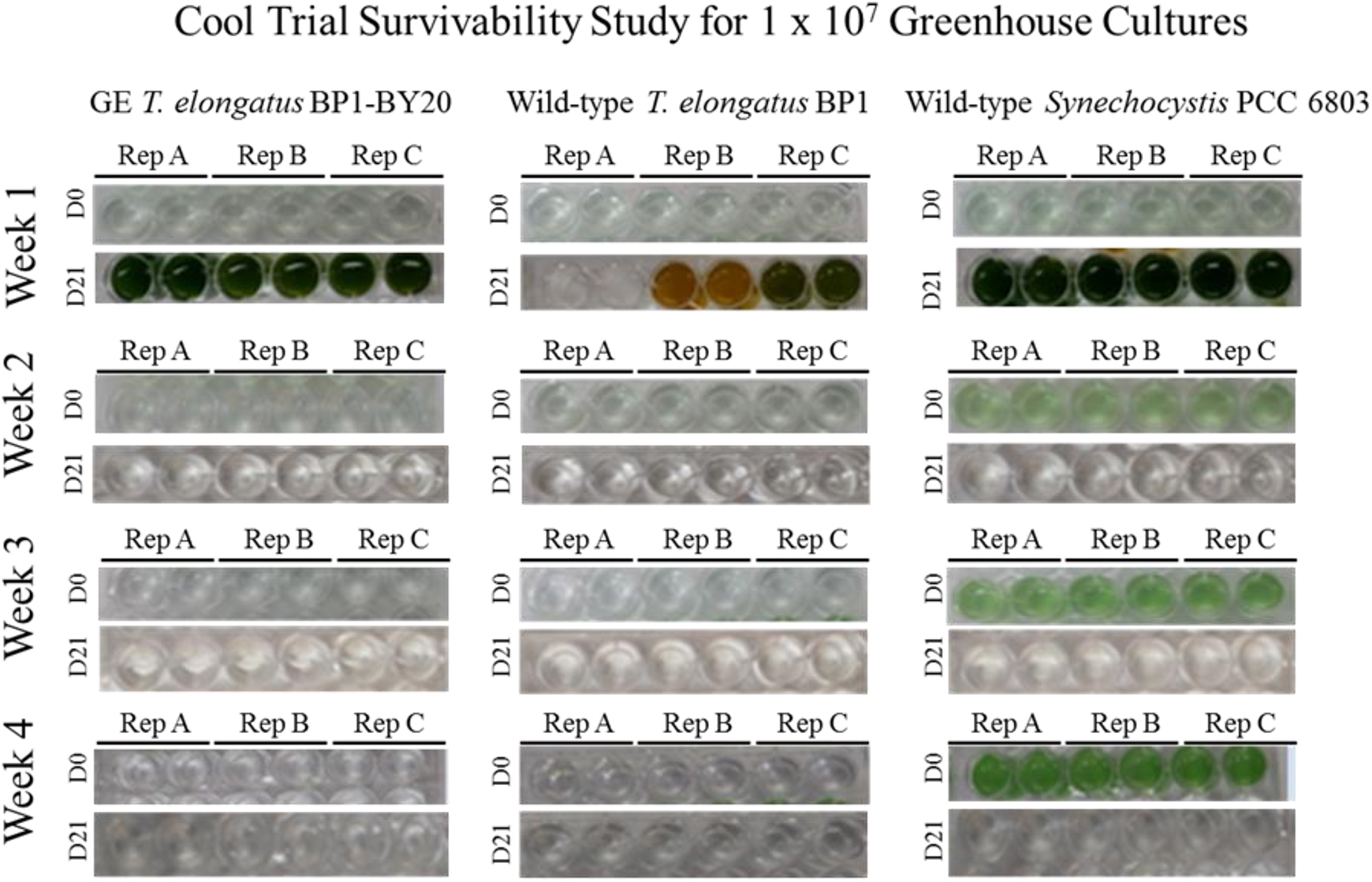
Survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC 6803, and genetically engineered (GE) T elongatus BP1-pKA cultures at 1 × 107 cells/mL during cool temperatures (15.44°C to 25.30°C) in the greenhouse. After 1, 2, 3, and 4 weeks in the greenhouse, a sample of each liquid culture was inoculated into fresh BG-11 media in duplicate and placed into the Percival environmental chamber (∼42.2°C) for 21 days. The pictures of the multiwell plates were taken on day 0 (the day of inoculation) and after 21 days in the Percival environmental chamber (∼42.2°C). Please refer to the online version of this article for color representation: https://journals.sagepub.com/doi/figure/10.1177/1535676019896640
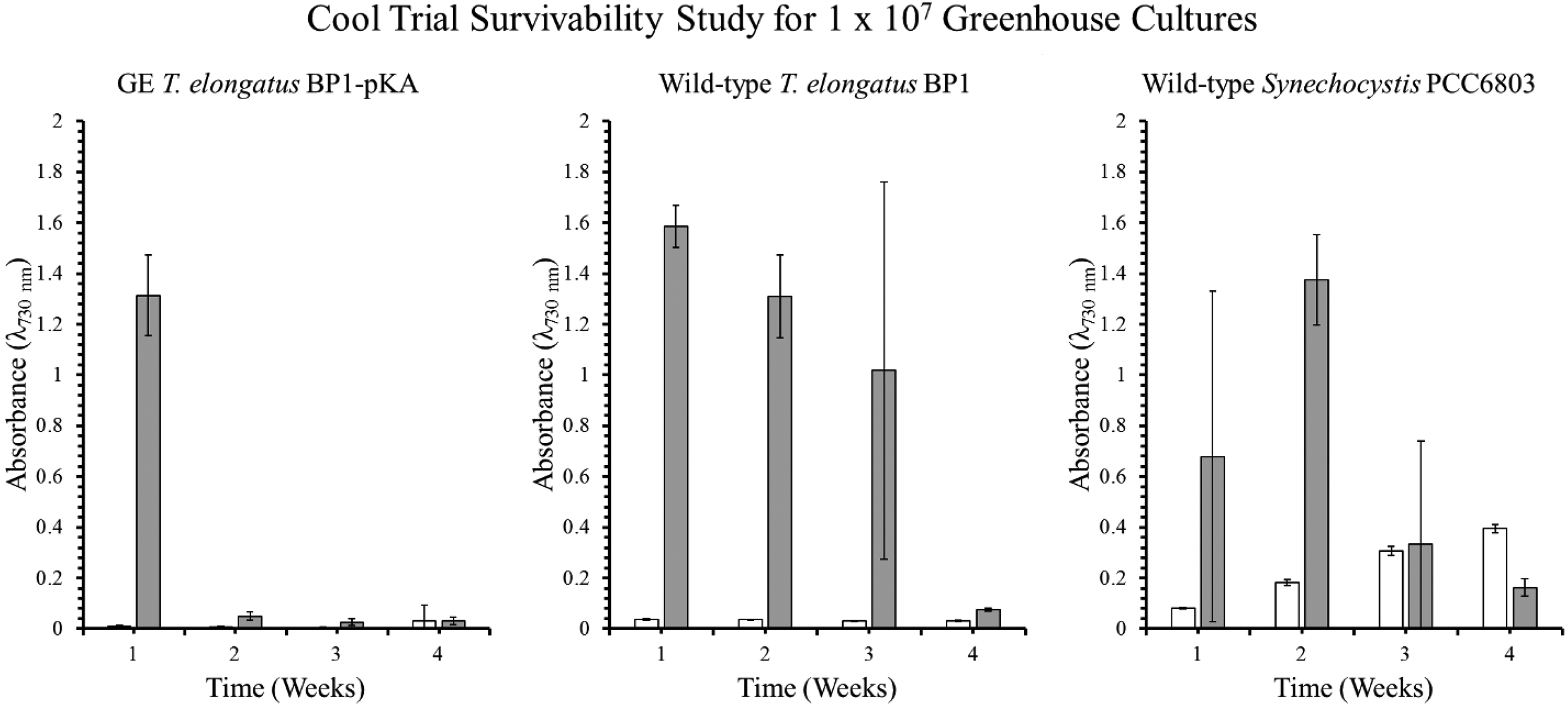
Optical density (λ = 730 nm) of the survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC6803, and genetically engineered (GE) T elongatus BP1-pKA cultures at 1 × 107 cells/mL during cold temperatures (15.44°C to 25.30°C) in greenhouse conditions. A sample of culture after 1, 2, 3, and 4 weeks in the greenhouse was inoculated in fresh BG-11 media and placed into the Percival environmental chamber (∼42.2°C) for 21 days. The growth was monitored by measuring the optical density (λ = 730 nm) weekly. The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = OD730 before being placed in the Percival chamber, and gray bar (▪) = OD730 after 21 days in the Percival chamber.
Survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC 6803, and genetically engineered (GE) T elongatus BP1-BY20 cultures at 1 × 107 cells/mL during cool temperatures (15.44°C to 25.30°C) in the greenhouse. A sample of each replicate was inoculated in fresh BG-11 media after 1, 2, 3, and 4 weeks in the greenhouse in duplicate and placed in the Percival environmental chamber (∼42.2°C) for 21 days. The pictures of the multiwell plates were taken on day 0 and after 21 days in the Percival environmental chamber (∼42.2°C). Please refer to the online version of this article for color representation: https://journals.sagepub.com/doi/figure/10.1177/1535676019896640
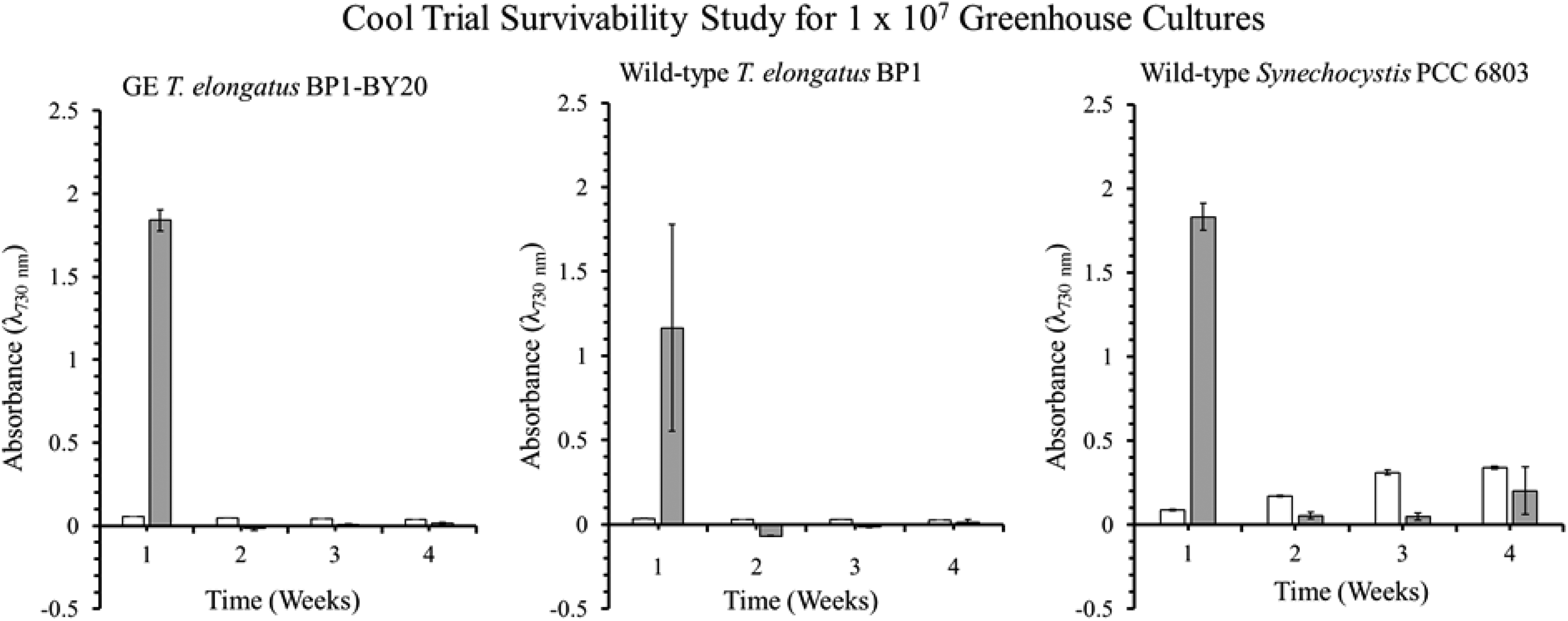
Optical density (λ = 730 nm) of the survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC6803, and GE T elongatus BP1-BY20 cultures at 1 × 107 cells/mL during cool temperatures (15.44°C to 25.30°C) in greenhouse conditions. A sample of culture after 1, 2, 3, and 4 weeks in the greenhouse was inoculated in fresh BG-11 media and placed in the Percival environmental chamber (∼42.2°C) for 21 days. The growth was monitored by measuring the optical density (λ = 730 nm) weekly. The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = OD730 before being placed in Percival chamber and gray bar (▪) = OD730 after 21 days in the Percival chamber.
To determine if the cyanobacteria cells were viable after 1 day and 28 days of incubation in greenhouse conditions, a diluted sample taken from each greenhouse-incubated cyanobacterial liquid culture was plated on BG-11 agar plates with and without antibiotic, and colony-forming units (CFU) were counted after 2 weeks under constant light source (30 μE m−2 s−1) at ∼42.2°C in the Percival environmental chamber. The GE T elongatus BP1-pKA sample from the 28-day greenhouse-temperature incubation treatment showed no formation of colonies on the BG-11 agar plates after returning to the Percival (∼42.2°C) temperature and lighting conditions, indicating that the cells within the culture had died and were no longer viable (Figure 8). The wild-type and GE T elongatus BP1-pKA cultures that were previously in laboratory conditions for 28 days showed significant growth and were all able to show signs of growth during the survivability assays under the Percival (∼42.2°C) temperature and lighting conditions as expected (Supplementary Figures S12, S13, and S18). Similar results were observed with the GE T elongatus BP1 (BP1-BY20), in that the sample that was previously exposed to the greenhouse conditions for 28 days did not develop any CFU on the BG-11 agar plates under the Percival (∼42.2°C) temperature and lighting conditions, whether the initial cell concentration was high (1 × 107 cells/mL) or low (5 × 106 cells/mL), as shown in Figure 9. On the other hand, the control set associated with the GE BP1-BY20 that was incubated in the Percival environmental chamber at ∼42°C for 28 days showed growth, as expected, after 1 week, 2 weeks, 3 weeks, and 4 weeks under the Percival (∼42.2°C) temperature and lighting conditions, as seen in the survivability experiments in the multiwell plates (Supplementary Figures S16 and S17). The control set (Percival-incubated BP1-BY20 liquid culture) also showed CFU on the control BG-11 agar plates as well as the tetracycline-containing BG-11 agar plates; however, the CFU for BP1-BY20 on the tetracycline plate were lower than that on the control BG-11 agar plates, which may be due to slower growth in the presence of the antibiotic (Supplementary Figure S19).
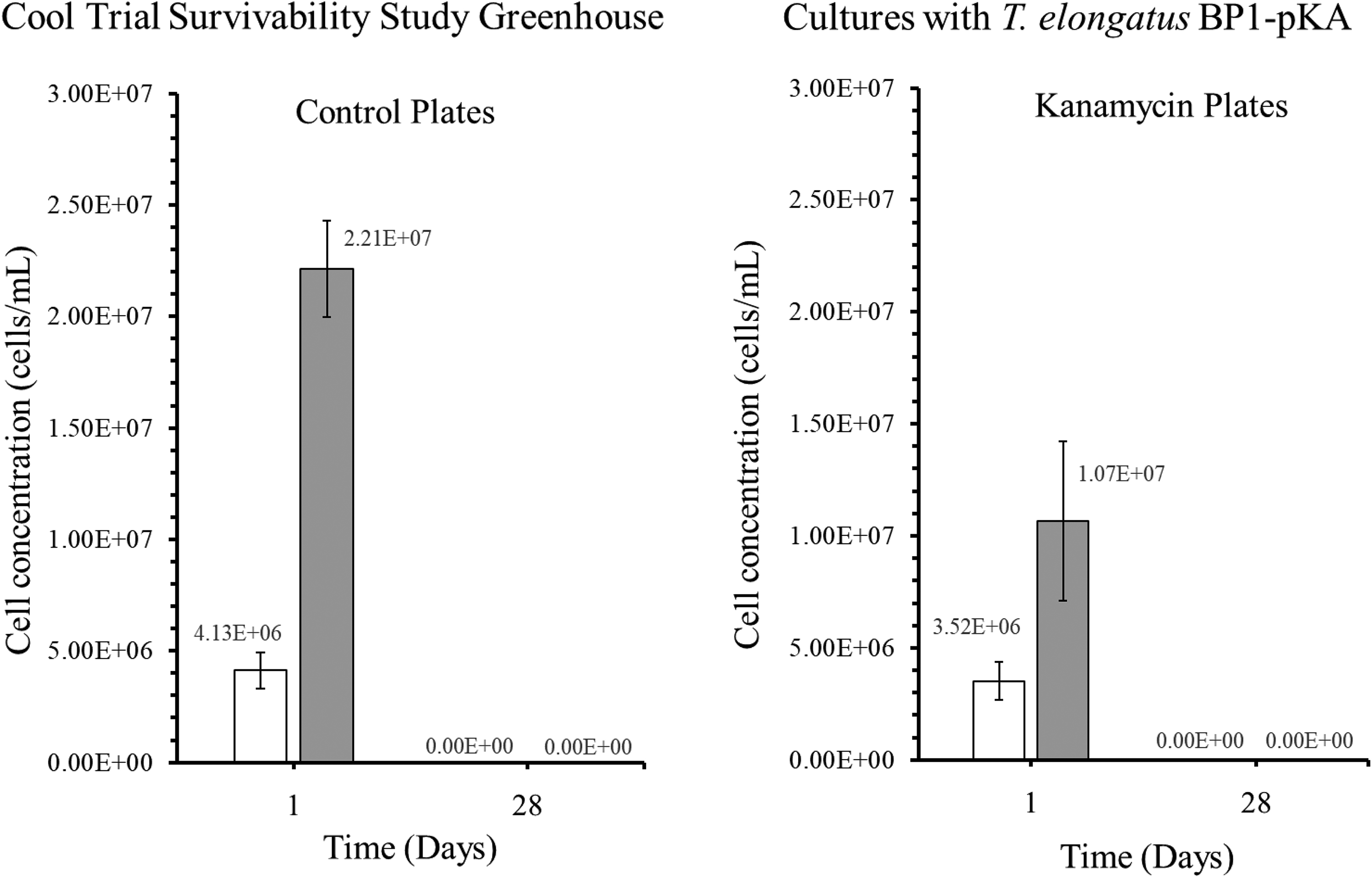
Colony-forming units (CFU) in cells/mL for the genetically engineered (GE) T elongatus BP1-pKA cultures during cool temperatures (15.44°C to 25.30°C) in greenhouse conditions. A sample from the greenhouse cultures was plated on BG-11 agar plates after 1 and 28 days and placed in the Percival environmental chamber (∼42.2°C) for 2 weeks. The colonies were counted, and the dilution factor was applied to obtain the CFU for each culture, which is also known as the viable cell concentration (cells/mL). The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = cultures at 5 × 106 cells/mL, and gray bar (▪) = cultures at 1 × 107 cells/mL.
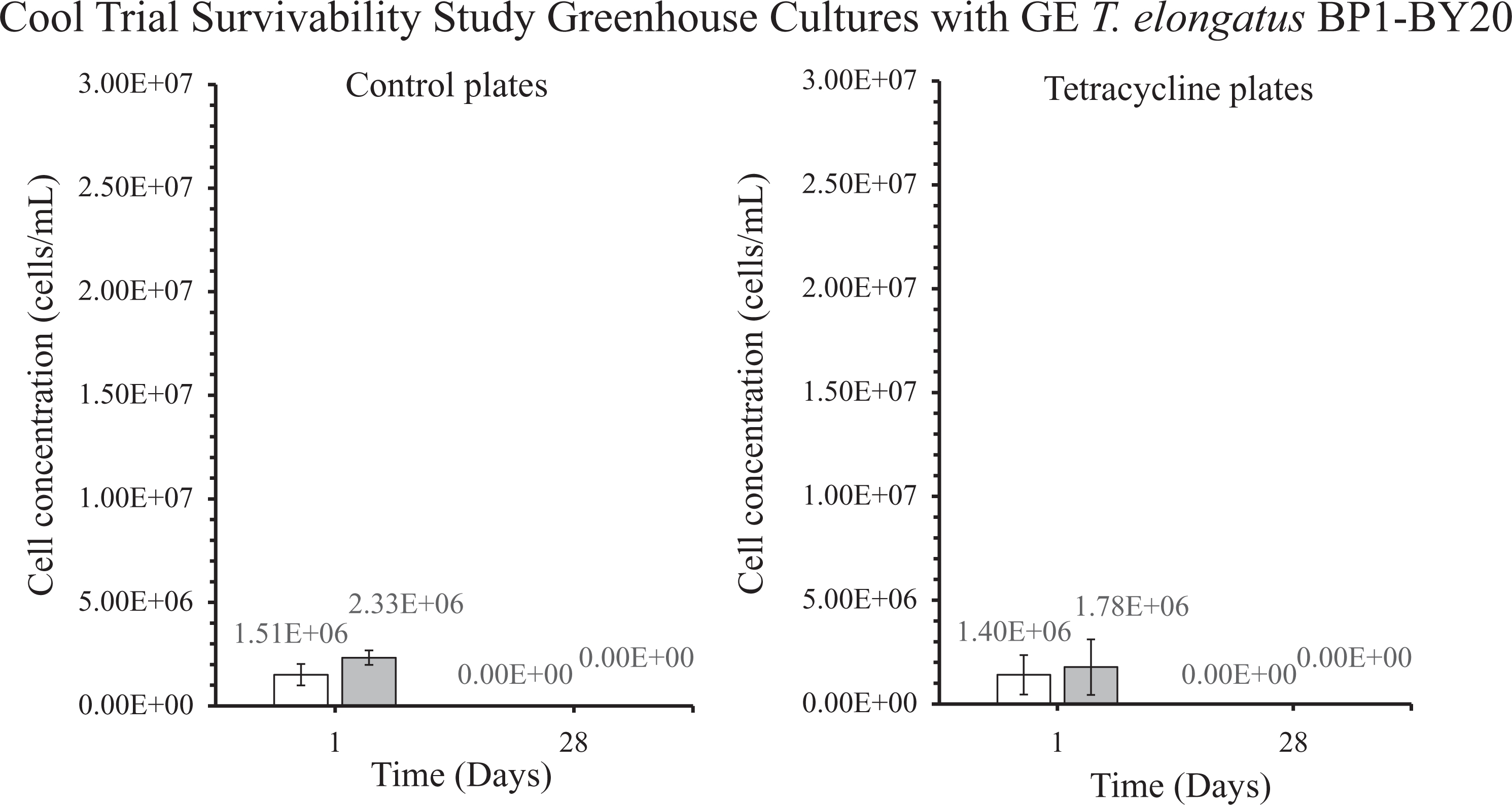
Colony-forming units (CFU) in cells/mL for the genetically engineered (GE) T elongatus BP1-BY20 cultures during cool temperatures (15.44οC to 25.30οC) in greenhouse conditions. A sample from the greenhouse cultures was plated on BG-11 agar plates after 1 and 28 days and placed in the Percival environmental chamber (∼42.2οC) for 2 weeks. The colonies were counted, and the dilution factor was applied to obtain the CFU for each culture, which is also known as the viable cell concentration (cells/mL). The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = at 5 × 106 cells/mL, and gray bar (▪) = 1 × 107 cells/mL.
The control experiments using the Percival (∼42.2°C) temperature-incubated cultures of wild-type T elongatus BP1, GE T elongatus BP1-BY20, and GE T elongatus BP1-pKA showed that after inoculation in fresh BG-11 medium and incubation in the Percival incubator at ∼42.2°C, all of the cells were able to recuperate (Supplementary Figures S12, S13, S16, S17). This showed that the death of the wild-type T elongatus BP1, GE T elongatus BP1-BY20, and GE T elongatus BP1-pKA cells incubated in greenhouse conditions was most likely due to the low temperature and light conditions of the greenhouse.
Another set of control liquid culture flasks containing the mesophilic wild-type Synechocystis PCC 6803 was also incubated in the greenhouse during cool temperatures (15.44°C to 25.30°C). The purpose was to test the effect of the cool temperature on a nonthermophilic strain as a control. The survivability study conducted on the mesophilic strain showed that the wild-type Synechocystis PCC 6803 was actively growing in the greenhouse during these cool temperatures, as shown on the visual photographs and the OD730 measurements (Figures 4 –7); there is an increase in optical density of the source liquid culture from the 1-week, 2-week, 3-week, and 4-week greenhouse-incubated cultures at the time of inoculation before they were placed into the Percival environmental chamber (∼42.2°C). However, since the Percival environmental chamber (∼42.2°C) is not ideal for the growth of the mesophilic wild-type Synechocystis PCC 6803, their growth varies in the Percival environmental chamber; for example, when inoculated from greenhouse cultures, they were not able to grow after 2 to 3 weeks of greenhouse incubation (Figures 4 –7), whereas the cultures inoculated from room temperature showed growth after 4 weeks (Supplementary Figure S10-S17).
The growth study, survivability multiwell plates, and BG-11 agar plate results suggest that wild-type and GE T elongatus BP1 cells do not survive well in cool temperatures in comparison with the Synechocystis PCC 6803, which was as expected since this strain of cyanobacteria thrives at 40°C to 57°C and the greenhouse conditions exhibited temperatures that are lower than what is optimal for T elongatus BP1 growth. This also supports the initial finding of Yamaoka et al. 28
Growth and Survivability of Wild-Type and GE T elongatus BP1 at Warm Temperatures (31.42°C to 36.27°C)
To see the effect of warmer temperatures in the greenhouse, a similar study was conducted in the greenhouse from July 1 to July 29, 2019. The greenhouse temperatures during the time of the study ranged from 31.42°C to 36.27°C (Figure 1), and the day length varied from 14 hours 38 minutes 40 seconds to 14 hours 6 minutes 32 seconds (Table 1). The growth study in warm temperatures showed a slight decrease in optical density (OD730) during the 28-day period in greenhouse conditions for both wild-type and the 2 GE T elongatus BP1 (BP1-pkA and BP1-BY20) cultures (Figures 10, 11, S20 and S21). Again, the growth study data indicated that T elongatus BP1 did not actively grow, although the temperature range is 10°C to 15°C higher than that of the cool temperature study.
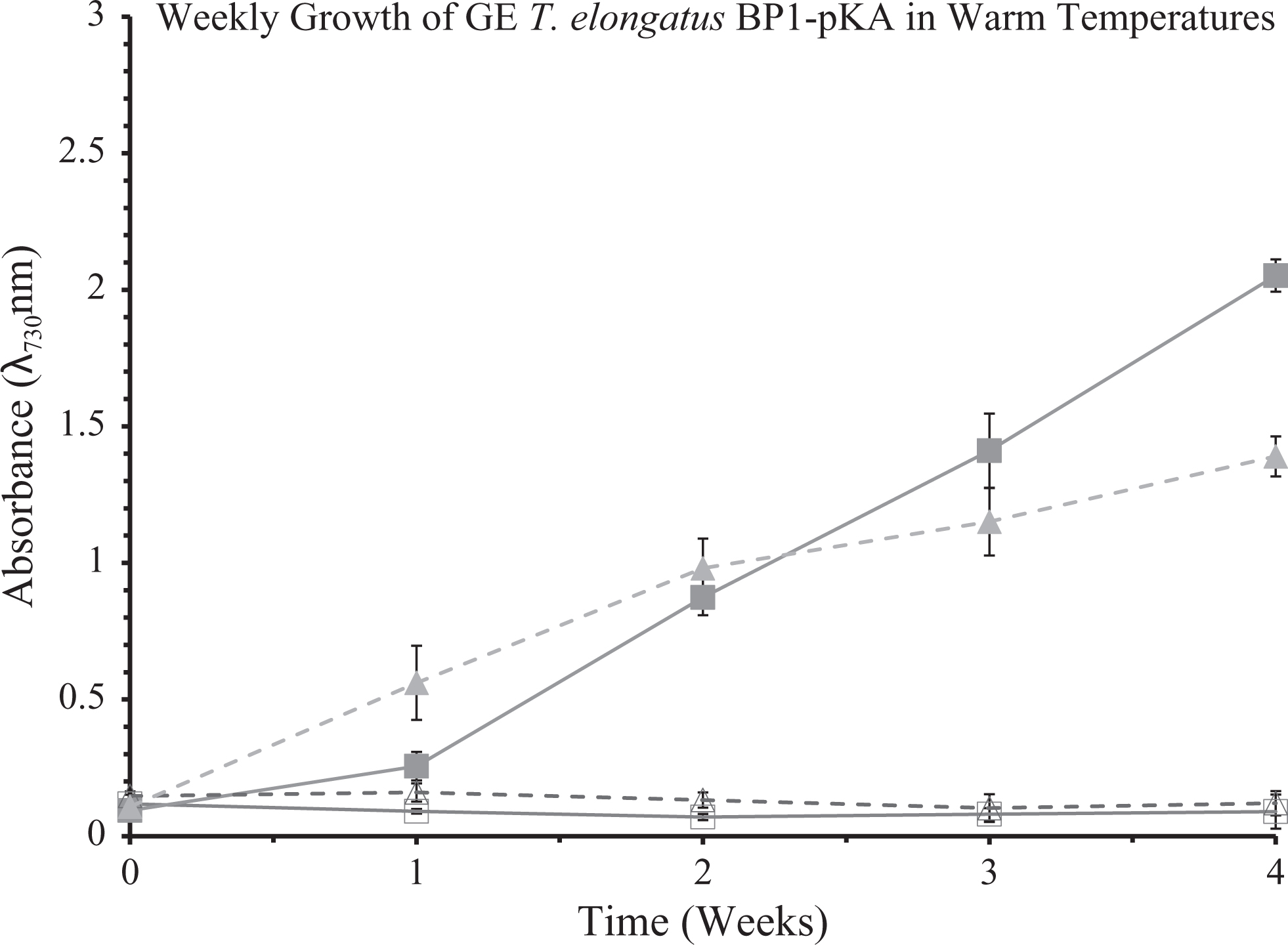
Optical density of wild-type and genetically engineered (GE) T elongatus BP1 (BP1-pKA) cultures at 1 × 107 cells/mL during warm temperatures (31.42οC to 36.27οC) were monitored weekly during a 4-week period in the greenhouse. Control cultures were grown in the Percival environmental chamber (∼42.2οC) in a Percival incubator. The results shown here are liquid culture absorbance measurements at 730 nm in duplicates for each replicate (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: (▪) is wild-type T elongatus BP1 in Percival, (□) is wild-type T elongatus BP1 in the greenhouse, (▴) is the GE T elongatus BP1-pKA in Percival, and (▵) is the GE T elongatus BP1-pKA in the greenhouse. Wild-type T elongatus BP1 = solid lines, BP1-pKA = dashed lines, closed symbols = control set, open symbols = greenhouse set.
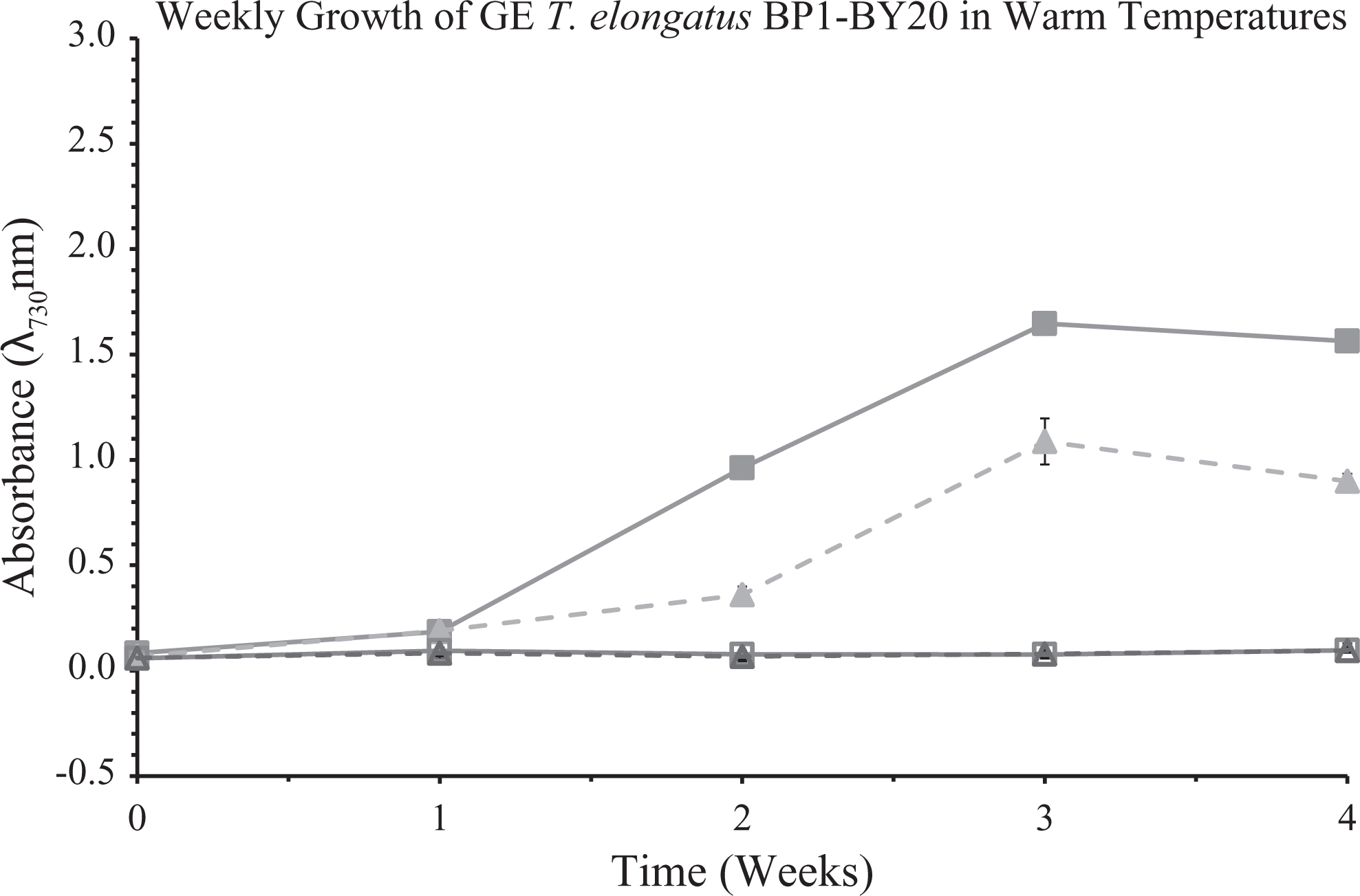
Optical density of wild-type and genetically engineered (GE) T elongatus BP1 (BP1- BY20) cultures at 1 × 107 cells/mL during warm temperatures (31.42οC to 36.27οC) were monitored weekly during a 4-week period in the greenhouse. Control cultures were grown in a Percival environmental chamber (∼42.2οC) in a Percival incubator. The results shown here are liquid culture absorbance measurements at 730 nm in duplicates for each replicate (n = 6). The graph is displayed as follows: (▪) is wild-type T elongatus BP1 in Percival, (□) is wild-type T elongatus BP1 in the greenhouse, (▴) is the GE T elongatus BP1- BY20 in Percival, and (▵) is the GE T elongatus BP1-BY20 in the greenhouse. Wild-type T elongatus BP1 = solid lines, BP1- BY20 = dashed lines, closed symbols = control set, and open symbols = greenhouse set.
In contrast to the survivability assay conducted with cool temperatures, visible observation and optical density measurements showed that wild-type T elongatus BP1 was able to recuperate after 4 weeks of exposure to greenhouse conditions (Figures 12 –15). The GE T elongatus BP1-pKA after 4 weeks in greenhouse conditions was still able to recuperate (Figures 12 and 13). This observation is supported by counting CFU on BG-11 agar plates to determine the number of viable cells in the greenhouse-temperature–treated cultures, which revealed only a small percentage of cells were still viable after 28 days of exposure to the greenhouse conditions (Figure 16). However, the survivability multiwell assay performed in the Percival environmental chamber (∼42.2°C) under a constant light source (30 μE m−2 s−1) over a 3-week period showed that only the 1-, 2-, and 3-week (not 4-week) greenhouse-incubated GE T elongatus BP1-BY20 samples were able to recuperate (Figures 14 and 15). To quantify the number of viable cells over time, the cell liquid cultures were plated on BG-11 Agar plates, and the CFU were measured after returning to the Percival (∼42.2°C) temperature and lighting conditions. The GE T elongatus BP1-BY20 sample that was previously exposed to the greenhouse conditions for 28 days (4 weeks) showed a small number of CFU. Based on the liquid culture volume plated on BG-11 agar plates, the number of viable cells in the GE T elongatus BP1-BY20 sample that was previously exposed to the greenhouse conditions for 28 days was determined to be 5.78 × 105 and 2.34 × 105 viable cells/mL as calculated from the number of colonies observed on the control BG-11 agar plates and on the tetracycline BG-11 agar plates, respectively. This result indicates that only about 2% to 5% of the initial cell population (1 × 107 cells/mL) remained viable after the 28 days of greenhouse exposure. In addition, the low population of viable cells may explain why the multiwell survivability assay did not show recuperation for the BP1-BY20 sample that had been exposed to the greenhouse condition for 4 weeks (Figures 14 and 15).
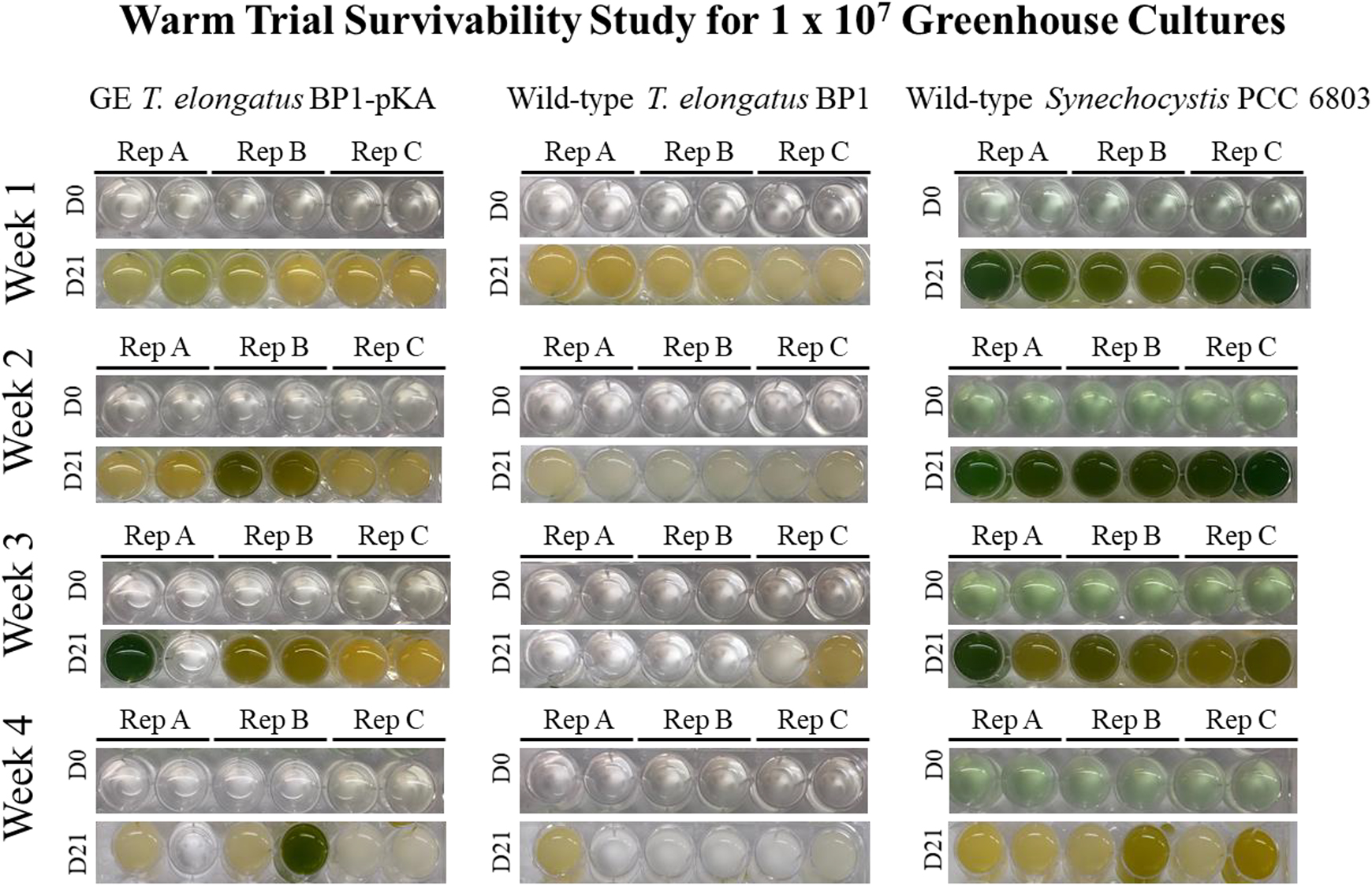
Survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC 6803, and engineered (GE) T elongatus BP1-pKA cultures at 1 × 107 cells/mL during warm temperatures (31.42οC to 36.27οC) in the greenhouse. A sample of each replicate was inoculated in fresh BG-11 media after 1, 2, 3, and 4 weeks in the greenhouse in duplicate and placed in the Percival environmental chamber (∼42.2οC) for 21 days. The pictures of the multiwell plates were taken on day 0 and after 21 days in the Percival environmental chamber (∼42.2ο). Please refer to the online version of this article for color representation: https://journals.sagepub.com/doi/figure/10.1177/1535676019896640
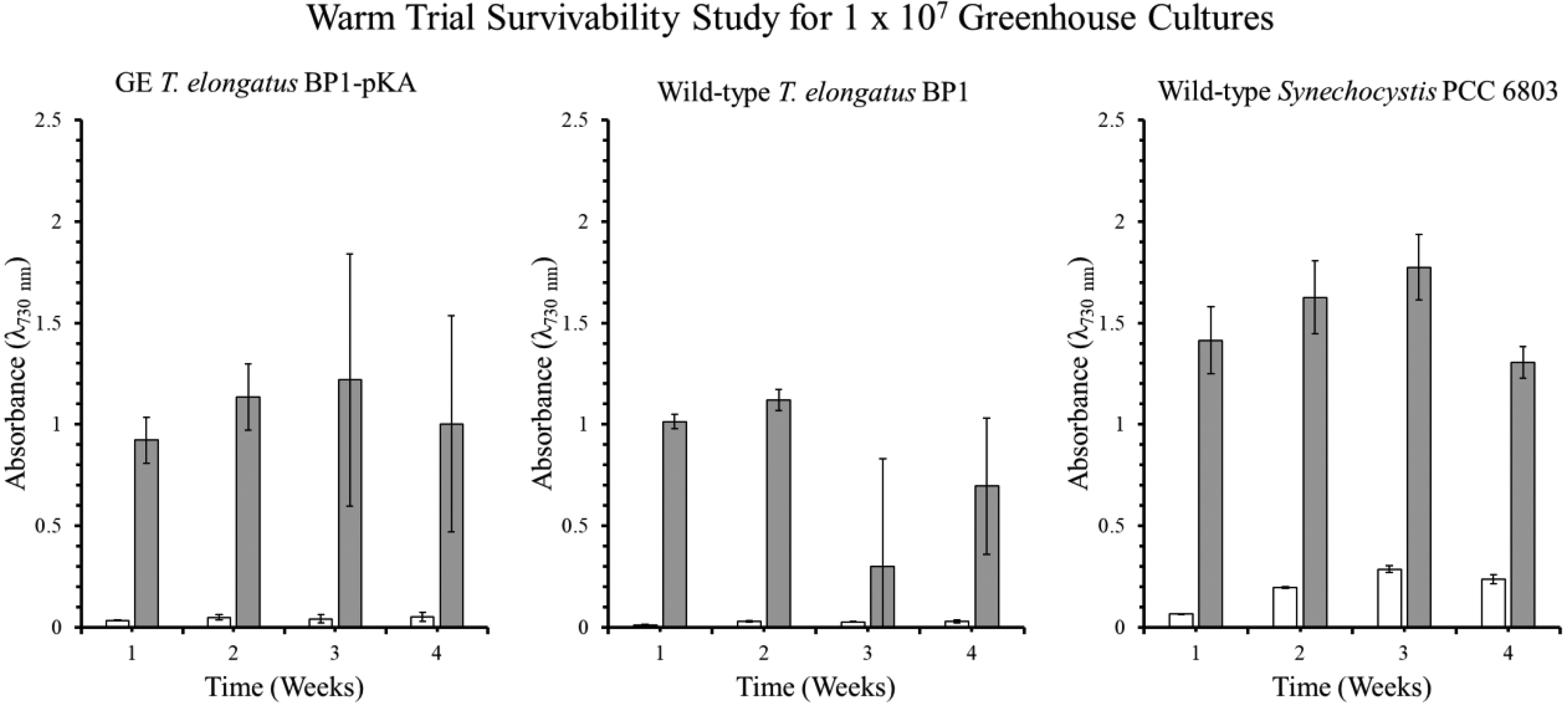
Optical density (λ = 730 nm) of the survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC6803, and genetically engineered (GE) T elongatus BP1-pKA cultures at 1 × 107 cells/mL during warm temperatures (31.42°C to 36.27°C) in greenhouse conditions. A sample of culture after 1, 2, 3, and 4 weeks in the greenhouse were inoculated in fresh BG-11 media and placed in the Percival environmental chamber (∼42.2°C) for 21 days. The growth was monitored by measuring the optical density (λ = 730 nm) weekly. The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = OD730 before being placed in the Percival chamber, and gray bar (▪) = OD730 after 21 days in the Percival chamber.
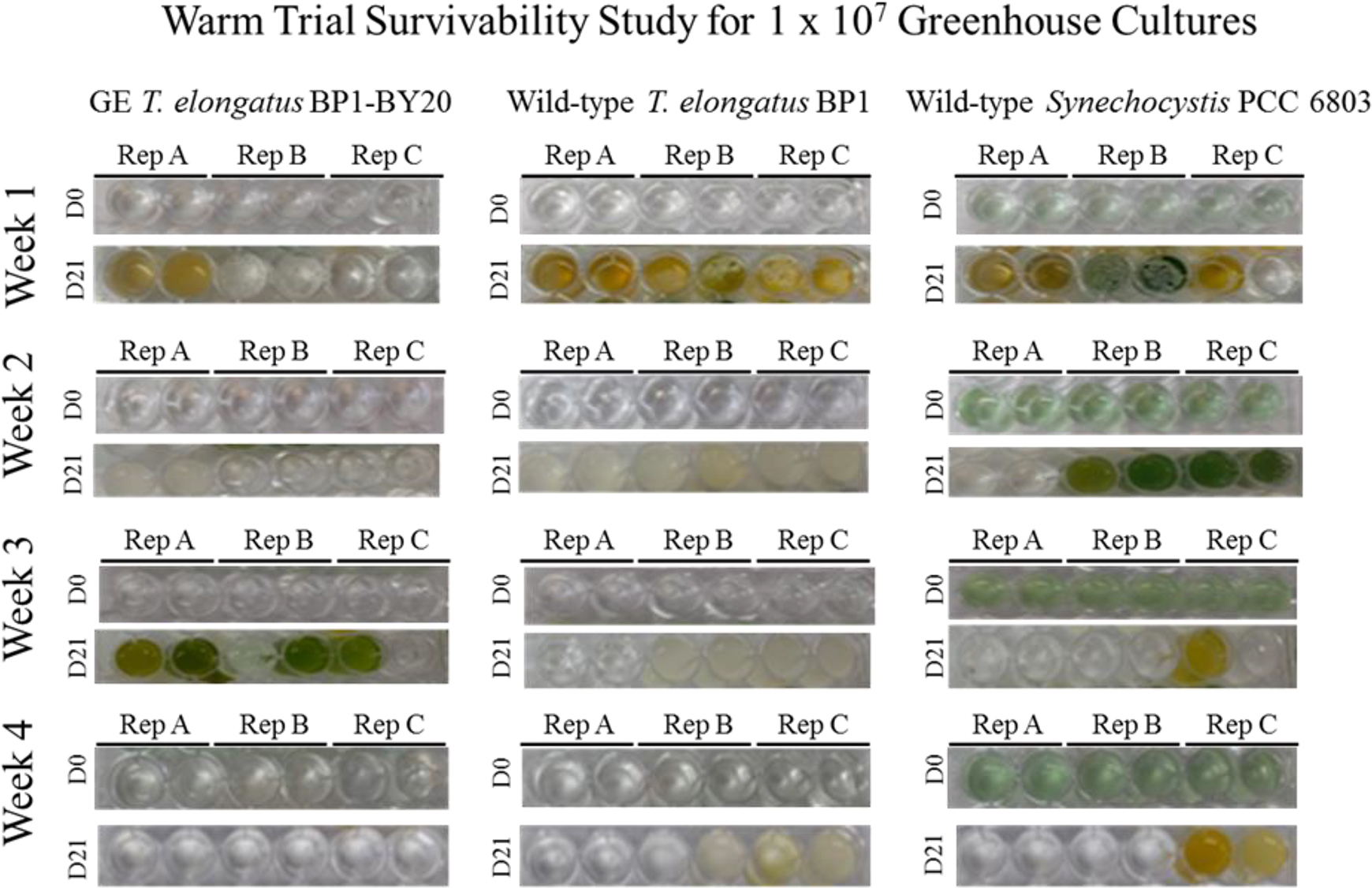
Survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC 6803, and genetically engineered (GE) T elongatus BP1- BY20 cultures at 1 × 107 cells/mL during warm temperatures (31.42°C to 36.27°C) in the greenhouse. A sample of each replicate was inoculated in fresh BG-11 media after 1, 2, 3, and 4 weeks in the greenhouse in duplicate and placed in the Percival environmental chamber (∼42.2°C) for 21 days. The pictures of the multiwell plates were taken on day 0 and after 21 days in the Percival environmental chamber (∼42.2°C). Please refer to the online version of this article for color representation: https://journals.sagepub.com/doi/figure/10.1177/1535676019896640
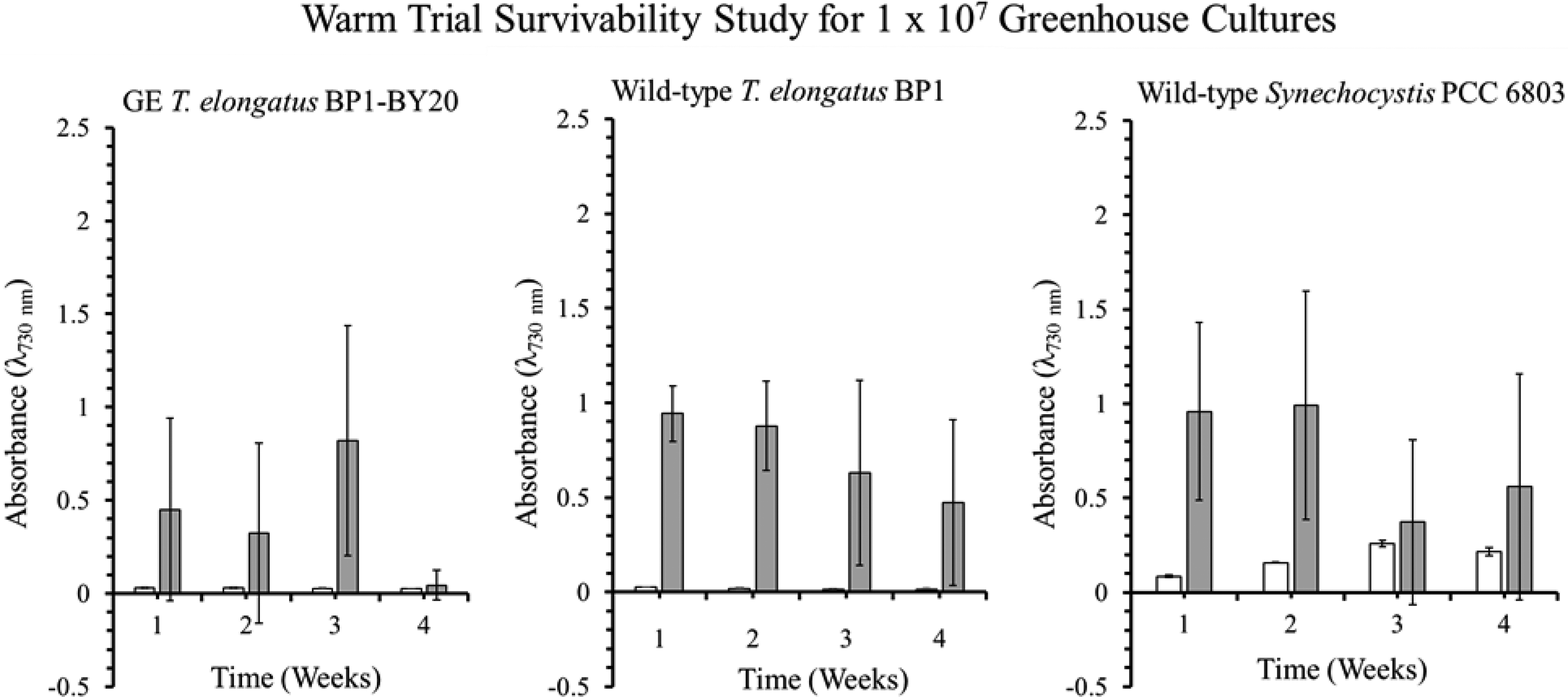
Optical density (λ = 730 nm) of the survivability assay for wild-type T elongatus BP1, wild-type Synechocystis PCC 6803, and genetically engineered (GE) T elongatus BP1- BY20 cultures at 1 × 107 cells/mL during warm temperatures (31.42°C to 36.27°C) in greenhouse conditions. A sample of culture after 1, 2, 3, and 4 weeks in the greenhouse was inoculated in fresh BG-11 media and placed in the Percival environmental chamber (∼42.2°C) for 21 days. The growth was monitored by measuring the optical density (λ = 730 nm) weekly. The results are shown as the mean value of the duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = OD730 before being placed in the Percival chamber, and gray bar (▪) = OD730 after 21 days in the Percival chamber.
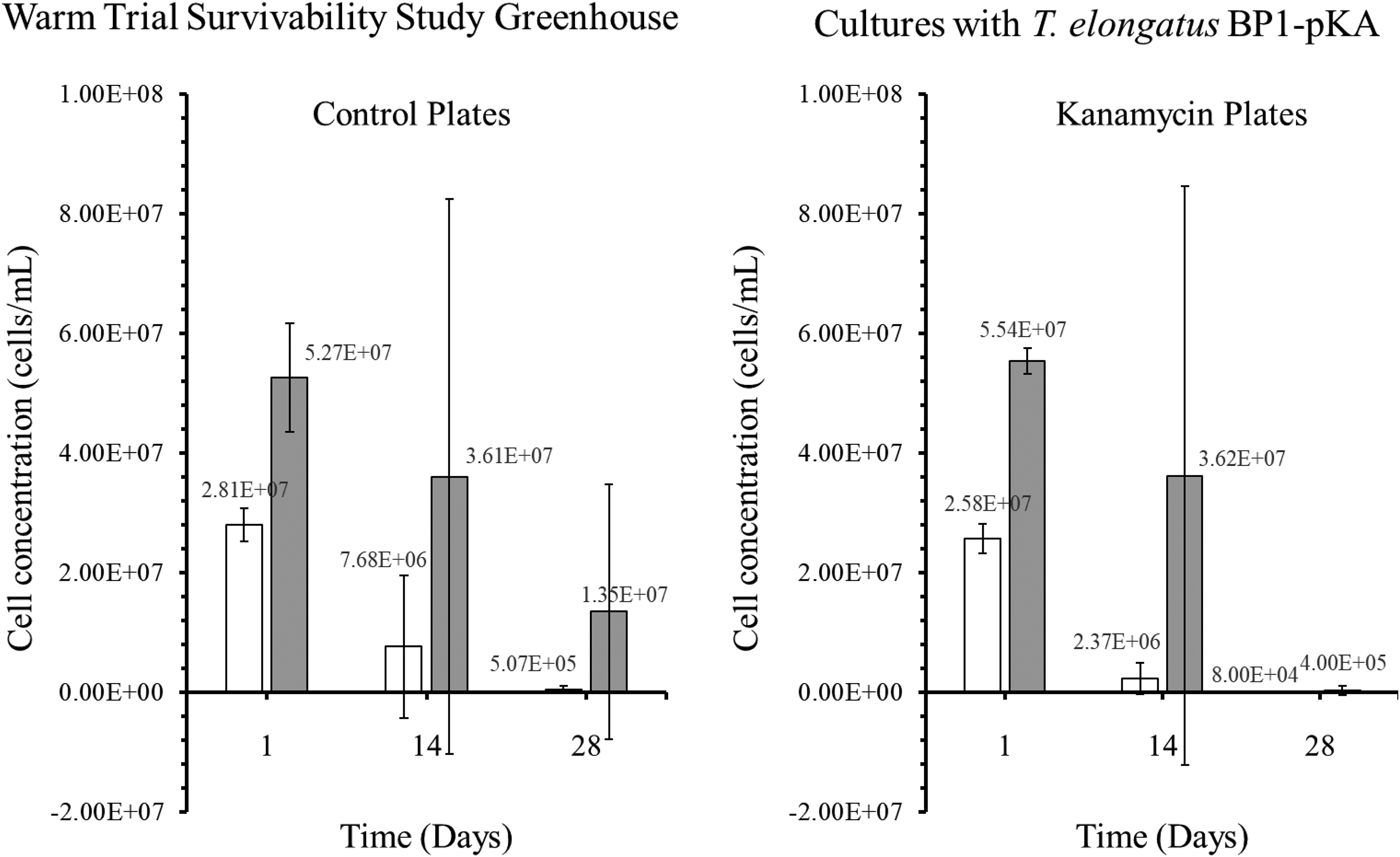
Colony-forming units (CFU) in cells/mL for the genetically engineered (GE) T elongatus BP1-pKA cultures during warm temperatures (31.42°C to 36.27°C) in greenhouse conditions. A sample from the greenhouse cultures was plated on BG-11 agar plates after 1, 14, and 28 days and placed in the Percival environmental chamber (∼42.2°C) for 2 weeks. The colonies were counted, and the dilution factor was applied to obtain the CFU for each culture, which is also known as the viable cell concentration (cells/mL). The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = cultures at 5 × 106 cells/mL, and gray bar (▪) = cultures at 1 × 107 cells/mL.
Although the weekly monitoring of flask liquid cultures (Figures 10 and 11) did not show T elongatus BP1 growth, they seem to be able to remain viable for a longer period of time in temperatures ranging from 31.42°C to 36.27°C allowing them to recuperate after 1, 2, 3, and 4 weeks of incubation in this warm temperature range. The results of CFU on antibiotic (kanamycin and tetracycline) BG-11 agar plates (Figures 16 and 17) also suggested that the GE BP1-pKA and BP1-BY20 retained the transgene DNA throughout the 4 weeks of incubation in the greenhouse during the warm season experiment.
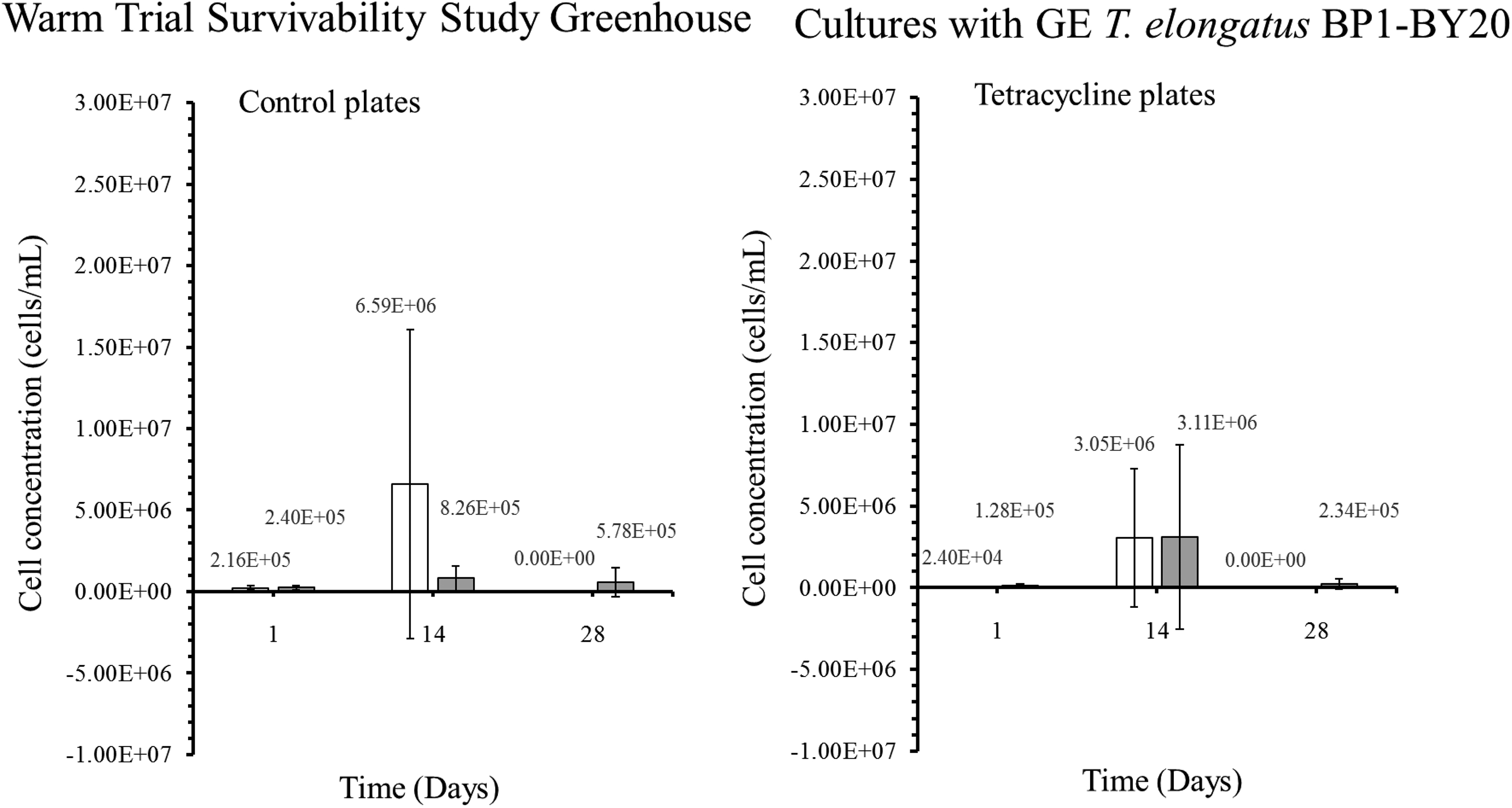
Colony-forming units (CFU) in cells/mL for the genetically engineered (GE) T elongatus BP1-BY20 cultures during warm temperatures (31.42°C to 36.27°C) in greenhouse conditions. A sample from the greenhouse cultures was plated on BG-11 agar plates after 1, 14, and 28 days and placed in the Percival environmental chamber (∼42.2°C) for 2 weeks. The colonies were counted, and the diluted factor was applied to obtain the CFU for each culture, which is also known as the viable cell concentration (cells/mL). The results are shown as the mean value of duplicate measurements of each replicate culture (n = 6). The error bars denote the standard deviation (n = 6). The graph is displayed as follows: white bar (□) = at 5 × 106 cells/mL, and gray bar (▪) = 1 × 107 cells/mL.
Overall, the experimental results from the multiwell survivability assay suggest that upon being genetically modified with the designer DNA constructs of pKA and YFP-ST-R-lipase (Supplementary Figures S1 and S2), the viability of the GE T elongatus BP1 was slightly (but not very significantly) different from that of the wild-type T elongatus BP1 at both cool temperatures (15.44°C to 25.30°C) and warm temperatures (31.42°C to 36.27°C). Another experiment (data not shown) was conducted in April, when the greenhouse temperatures were moderate (24.5°C to 31.5°C) using GE T elongatus BP1-BY20. The results were similar to that of the warm temperature study, in that the GE T elongatus BP1-BY20 and wild-type T elongatus BP1 were not actively growing but remained viable after 1, 2, 3, and 4 weeks under the greenhouse conditions. While the results may be empirical evidence for the total death or partial death of wild-type and GE T elongatus BP1 under greenhouse conditions depending on the temperature ranges tested, the thermophilic nature of T elongatus BP1 may be considered to serve as a biosafety-guarded mechanism in case the thermophilic organisms somehow escape from their hot temperature bioreactors.
Conclusion
T elongatus BP1 is a thermophilic strain of cyanobacteria with optimum growth at 57°C. 28 In this study, we observed that the wild-type and GE T elongatus BP1 were not able to actively replicate under greenhouse conditions during cool (15.44°C-25.30°C) and warm (31.42°C-36.27°C) temperatures. However, the survivability assay demonstrated that wild-type and GE T elongatus BP1 under some greenhouse temperature conditions did not entirely die and were able to recuperate after reinoculation in fresh media and placed back in laboratory growth chamber (∼42.2°C) conditions. The assay with liquid cultures incubated in the greenhouse during cool temperatures (15.44°C-25.30°C) in March showed that there were still some live wild-type T elongatus BP1 cells (within 1 to 3 weeks of cool temperature exposure) and GE T elongatus BP1 cells (within 1 week of cool temperature exposure). However, after 2 weeks (for the GE T elongatus BP1-pKA and GE T elongatus BP1-BY20) and 2 to 4 weeks (for the wild-type T elongatus BP1) exposure to the cool temperatures in the greenhouse, the wild-type and GE T elongatus BP1 cells were completely dead; there was no evidence of recuperation after they were placed back into the Percival environmental chamber. The study in July under greenhouse warm temperature (31.42°C-36.27°C) conditions showed that the wild-type and GE T elongatus BP1 cells were able to grow back in the Percival environmental chamber (∼42.2°C) under constant lighting (30 μE m−2 s−1) conditions after 1 day and 28 days of greenhouse exposure, respectively. This indicated that the cells were able to survive through the duration of the study, although they were not shown to be actively growing. Overall, the results suggest that the thermophilic nature of T elongatus BP1 could serve as a natural biosafety guard during cool temperature season to limit their spread if they were to break containment and escape to the outside environment.
Footnotes
Acknowledgments
The authors would like to thank Thu Nguyen, Bryan Koury, An Ha, and Kevin Nguyen for their active participation during discussions and valuable insights. The authors would also like to thank the Vice President of Research, Office of Research, the Dean of the College of Sciences at Old Dominion University, for use of the Arthur and Phyllis Kaplan Orchid Conservatory for greenhouse space.
Author Contributions
O.S. and C.L.B. transformed and verified YFP-ST-R-lipase and pKA constructs in T elongatus BP1, respectively. O.S. performed the growth and survivability study for the cool (15.44°C-25.30°C) and warm (31.42°C-36.27°C) temperatures with the GE T elongatus BP1-BY20. C.L.B. performed the growth and survivability study for the cool (15.44°C-25.30°C) and warm (31.42°C-36.27°C) temperatures with the GE T elongatus BP1-pKA. O.S. and C.L.B. contributed to the experimental design, analyzed the data, and cowrote the article. L.H.G. contributed to the data analysis, co-supervised the experiments, and critically read and edited the article. J.W.L. designed and supervised the experiments, inspected all experimental results, and cowrote and finalized the article.
Ethical Approval Statement
All research was conducted with prior approval from ODU Institutional Biosafety Committee. All authors listed have made significant contribution to the reported study.
Statement of Human and Animal Rights
Not applicable to this study.
Statement of Informed Consent
Not applicable to this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Biotechnology Risk Assessment Grants (BRAG) program award no. 2016-33522-25624 from the US Department of Agriculture to J.W.L. and L.H.G.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
