Abstract
Introduction:
Many emerging and reemerging pathogens have been identified as major public health threats in Bangladesh. Collection, transportation, and storage of infectious materials and management of generated waste from diagnosing those diseases require strict adherence to biosafety and biosecurity practices. Such activities in Bangladesh need substantial development.
Methods:
A novel multipronged approach was followed to create awareness and provide resources to strengthen nationwide biosafety and biosecurity status. The approach included, but was not limited to, developing resource persons (RPs), developing laboratories’ baseline assessment tools, training assessors, conducting assessments, organizing awareness and training programs, identifying laboratories dealing with biohazards, developing a biosafety cabinet certification program, developing a Web site, and developing customized biosafety and biosecurity guidelines.
Results:
Currently, 133 RPs and 29 assessors are available in Bangladesh. The RPs organized 8 divisional awareness programs and trained about 3,000 professionals. Assessors conducted baseline assessments of 18 key laboratories, and RPs identified 127 laboratories in Bangladesh dealing with biohazards. NSF-accredited certifiers are now certifying biosafety cabinets in Bangladesh. Guidelines were developed and disseminated to the members. Those RPs who were organizing activities under the program are now organizing biosafety and biosecurity training sessions as academic activities.
Conclusions:
There is a shift from no biosafety and biosecurity practice toward a growing culture of biosafety and biosecurity practices in research and diagnostics in Bangladesh. To sustain the momentum of this development and to further strengthen the program, allocation of necessary resources and strong leadership support from the government of Bangladesh and donor groups are indispensable.
Keywords
Introduction
Bangladesh is a developing country in southern Asia, in the Ganges River delta on the Bay of Bengal, with a population of approximately 170 million by the end of 2019. 1 The country is divided administratively into 8 divisions and 64 districts. On average, 3,000 people live in 1 mi2 in a total area of 55,598 mi2. The top 5 major leading causes of death are the infectious diseases tuberculosis, influenza and pneumonia, diarrheal diseases, hepatitis B, and encephalitis. 2 Outbreaks and epidemics of emerging and reemerging pathogens, including multi-drug-resistant and extensively drug resistant pathogens, have been identified as major public health threats. 3 -5
Specimen management and diagnosis of highly contagious diseases require adequate biosafety and biosecurity practices. Biosafety involves the safe handling and containment of infectious microorganisms and hazardous biological materials on the basis of risk assessment and mitigation, and biosecurity involves mechanisms to establish and maintain security and oversight of pathogenic microorganisms, toxins, and relevant resources from theft and misuse. 6,7 In other words, biosafety protects people from biohazardous materials, whereas biosecurity protects biohazardous materials from people. 8 In the field of biotechnology, in which biosafety and biosecurity are not clearly defined, working and manipulating genetic materials for the expression and invention of new methods and techniques for the development of vaccines, diagnostics, and improvement of crops require stricter control to avoid misuse of these techniques. 6,7 All personnel must follow applicable policies and procedures while handling and performing activities involving biohazards. 6,7 However, the current life science infrastructure in Bangladesh needs significant improvement to create an efficient and sustainable culture of biosafety and biosecurity, ensuring protection of workers, the public, and the environment from biohazards.
Bangladesh has many government hospitals, private hospitals, and diagnostic centers. 9 In the animal sector, there are 11 disease investigation laboratories, 2 livestock research institutes, and many universities and institutes conducting research and diagnostic activities. Biosafety officers (BSOs) and suitable biosafety guidelines 10 are not available for those facilities. The International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b), has appointed the first full-time BSO for 14 laboratories and field activities. icddr,b’s BSO, the solitary biosafety and biosecurity trainer in the country, organized the first formal training in April 2010, inviting laboratory personnel from icddr,b and the neighboring institutes.
In 2011, the Bangladesh Biosafety and Biosecurity Society (BBBS) was formed of members from the Director General of Health Services, the Department of Livestock Services, the Bangladesh Livestock Research Institute, Bangabandhu Sheikh Mujib Medical University, Bangladesh Agricultural University, the University of Dhaka, the Institute of Epidemiology Disease Control and Research, icddr,b, and other government medical colleges, universities, and research organizations. Initially, the Virtual Biosecurity Center (http://www.virtualbiosecuritycenter.org) and Biosafety and Biosecurity International (http://www.biosafetybiosecurityinternational.com) provided substantial support for initiating the BBBS and designing its bylaws and terms of references. Starting in 2013, the Centers for Disease Control and Prevention provided funding and technical expertise for a biosafety and biosecurity program enriching and using the BBBS as platform to ensure sustainability. The main objectives of this program are to develop resource persons (RPs), conduct a baseline assessment of laboratories, develop training modules, organize an awareness program, organize a training program, develop NSF-accredited biosafety cabinet (BSC) certifiers, identify laboratories dealing with biohazards, and develop and disseminate guidelines. The program was initiated to systematically strengthen biosafety and biosecurity status and to contribute to controlling the spread of infectious diseases in the country, leading to sustainable development in health and agriculture. Here we present our approach to strengthening biosafety and biosecurity in Bangladesh, which is inclusive of all sectors, top down, comprehensive, and sustainable. The overall goal is establishing a sustainable culture of biosafety and biosecurity in the country.
Method
Our multipronged method included the development of an adequate number of biosafety professionals, conducting assessments using standardized tools, fostering awareness among top- and middle-level management and bench-level professionals, conducting training sessions using a standardized training curriculum, performing laboratory inventories and identification, establishing a BSC certification program, developing a Web site, strengthening the BBBS, and developing and disseminating national guidelines.
Development of RPs
RPs were developed to support diagnostic, basic, and applied research laboratories in the country. It was planned to develop at least 5 RPs for each of the 8 divisions in Bangladesh. Participants were selected from different areas of biological sciences at the level of assistant professor or higher, and preference was given to individuals working in medical microbiology, biochemistry, molecular biology, biotechnology, zoology, botany, agricultural sciences, veterinary and animal sciences, and environmental sciences. A minimum of 30 and no more than 40 individuals were registered for a 2-week “training of the trainers” (ToT) program. The ToT program was based on the second edition of the Instructor’s Guide for Biosafety Training developed by the Division of Occupational Health and Safety, National Institutes of Health, and the World Health Organization’s Collaborating Centre for Applied Biosafety Programmes and Training. Training included theoretical, practical, and situation-based exercises. The ToT program was organized from 2015 to 2018, once in a year, in April.
Baseline Assessments
Two assessment tools for conducting baseline assessments of human and animal laboratories were developed. 11 Both tools constituted modules addressing the following topics: national biosafety and biosecurity information, laboratory premises, biosafety procedures, biosafety personnel, sample collection practices, transportation, laboratory biosafety practices, equipment and BSCs, laboratory waste disposal, Stocks of personal protective equipment, safety training, laboratory incidents, prophylaxis, microbial cultures and strains, and storage of specimens and cultures. An additional anthrax module was included for assessing animal laboratories.
Assessors from various backgrounds were selected and preference was given to RPs. Assessors were subjected to theoretical training on the assessment tools; types of assessments; roles and responsibilities of assessors; actions before, during, and after assessment engagements; interview skills; finding and following leads; report writing; and presentation skills. After the daylong theoretical training, participants were trained hands-on in conduct assessments under the supervision of lead assessors. Successful candidates were certified and allowed to conduct assessments independently.
Developing Training Modules
A daylong seminar session was designed to create awareness among top management, midlevel staff members, and bench workers involved in research, diagnostic laboratories, and in clinical care. Tailored modules on the principles and practices of biosafety and biosecurity were developed. These training modules covered safe laboratory design, general laboratory safety issues, chemical safety, biosafety and biosecurity, and biomedical waste management. Another tailored training module for bench workers was developed and translated into Bengali for their easy understanding. 12
Awareness and Training Program
Our experience and subsequent assessments confirmed that there is a lack of understanding of biosafety and biosecurity at every level in the hierarchy. Thus, we divided the employees into 3 different levels: top management (including vice chancellors, principals, professors, laboratory directors, divisional directors of health, department heads, and similar positions), midlevel (including associate professors, assistant professors, lecturers, and technical officers), and bench-level employees (including technicians and technologists). Awareness sessions for top-level management were usually presided over by the university vice chancellor or medical college principal. These presentations addressed policy, regulations, and resources required for a successful biosafety and biosecurity program. The midlevel and bench-level groups were provided with additional process and procedural details and requirements. On average, 30 individuals from different areas of biological sciences attended the 1-day program.
After the awareness sessions for all divisions, a 2-day training program was initiated in different venues at various universities, medical colleges, or institutes at the district level. Scientists, faculty members, and research students participated in these training sessions. Participants were provided didactic and practical experience with safety modules at a laboratory operations level and challenged to identify missing safety components. 12
Laboratories Identification
Although a large number of laboratories exist in Bangladesh, only a few were subjected to baseline assessments to determine biosafety and biosecurity status. However, to bring laboratories in Bangladesh under a common umbrella of biosafety and biosecurity program, a laboratory identification program was initiated to identify laboratories’ biosafety levels (BSLs). A standard operating procedure and form were developed to identify laboratories in the country. 13 The RPs under different divisions were tasked to identify laboratories in their vicinities. 13 In addition to the tests performed, particular focus was given to the presence of a functioning autoclave, a BSC, a fire extinguisher and fire blanket, and an eyewash and a safety shower at those facilities.
BSC Certification Program
BSCs were used without calibration and certification by accredited certifiers after their initial installation. Beginning in 2010, BSCs at selected institutes are certified by NSF-accredited field certifiers from foreign countries. 6 However, the travel costs of certifiers from foreign countries to certify a single BSC are higher than the actual certification cost, which is prohibitive for smaller institutes and laboratories that have 1 or few BSCs. Therefore, since 2011, BSCs at a few facilities—icddr,b, the Institute of Epidemiology Disease Control and Research, the Bangladesh Livestock Research Institute, and the Central Disease Investigation Laboratory—have been certified collectively to minimize travel costs. For a sustainable solution, it was planned to develop an in-country BSC certification program by procuring certifying equipment and getting at least 1 NSF-accredited certifier to certify BSCs anytime it is required, following the guidelines. 6,7,14
Strengthening the Society
Since its inception, the BBBS has worked to disseminate knowledge and science regarding biosafety and biosecurity and has organized annual preconference workshops and conferences, along with periodic membership drives. A goal was set to increase the number of knowledgeable members of the BBBS and to improve communication of biosafety and biosecurity messages. Accordingly, all RPs were given the opportunity to become lifetime members of the BBBS, to continue working under its banner and with the Biosafety Office of icddr,b. This activity also supported the development of a Web site 15 for the BBBS, to establish a network among researchers to disseminate knowledge and science.
Develop and Disseminate National Guidelines
Under the purview of the BBBS, a core committee was formed to review and identify the content of the current guidelines. 6,7,10,16 -22 The committee included relevant experts from the Director General of Health Services, the National Institute of Biotechnology, the University of Dhaka, and icddr,b. 23 The committee finalized content to develop the first draft, which was presented to a wider body of professionals and the RPs for input. Following that, the draft was presented to faculty members from various universities, medical colleges, and research organizations for review and recommendations. Subsequently, the draft document was presented and finalized in the following workshop. Hard copies of the guidelines were distributed to society members, and a soft copy is available on the BBBS Web site. 23
Results
RP Development
The 4 annual training sessions produced a cadre of 133 RPs, among whom 54 are from agricultural, veterinary, and animal sciences; 42 are medical doctors; and 37 are from other disciplines of biological sciences (Figure 1A). There are at least 5 RPs in each division in Bangladesh (Figure 1B); however, the majority are in Dhaka. This is because most of the major institutes and health facilities are in Dhaka division or near the capital city.
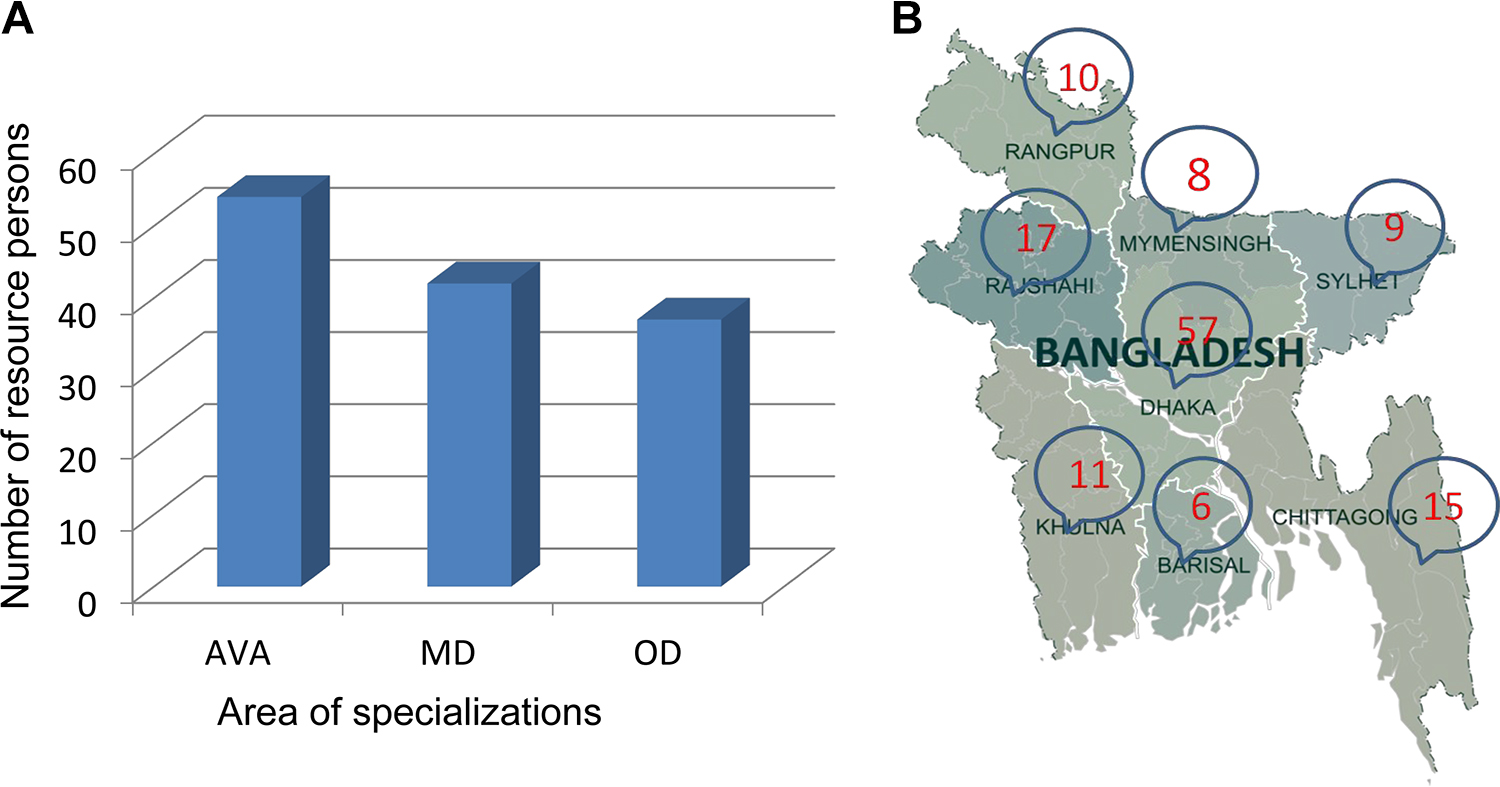
(A) Development and distribution of biosafety and biosecurity resource persons in Bangladesh. In total, 133 resource persons are distributed over 8 divisions in Bangladesh. Fifty-four are from agricultural, veterinary, and animal sciences (AVA); 42 are medical doctors (MD); and 37 represent other disciplines of the biological sciences (OD). (B) Distribution of biosafety and biosecurity resource persons in Bangladesh. There are 10 resource persons in Rangpur, 8 in Mymensingh, 17 in Rajshahi, 9 in Sylhet, 57 in Dhaka, 11 in Khulna, 6 in Barisal, and 15 in Chating.
Baseline Assessment of Research and Diagnostic Laboratories
The 2 assessment tools developed were created and deployed with Bangladeshi human and animal laboratories. 11 A cadre of 29 assessors (9 from human sciences, 7 from veterinary and animal sciences, and 12 from different areas of biological sciences in academia) were trained to conduct assessments. Accordingly, assessments were conducted at 18 laboratories (Figure 2), and reports were submitted to the respective authorities. Figure 2 shows that 6 clinical laboratories scored between 12% and 40% (an acceptable score is ≥80%). In the animal sector, the scores of field disease investigation laboratories are comparable with those of clinical laboratories; however, the animal sciences research and diagnostic laboratories achieved relatively high scores (Figure 2). Many gaps and opportunities were identified and provided to each laboratory in the written report. Recommendations were provided to address existing gaps in short-term, midterm, and long-term plans. Following are a few examples of recommendations.
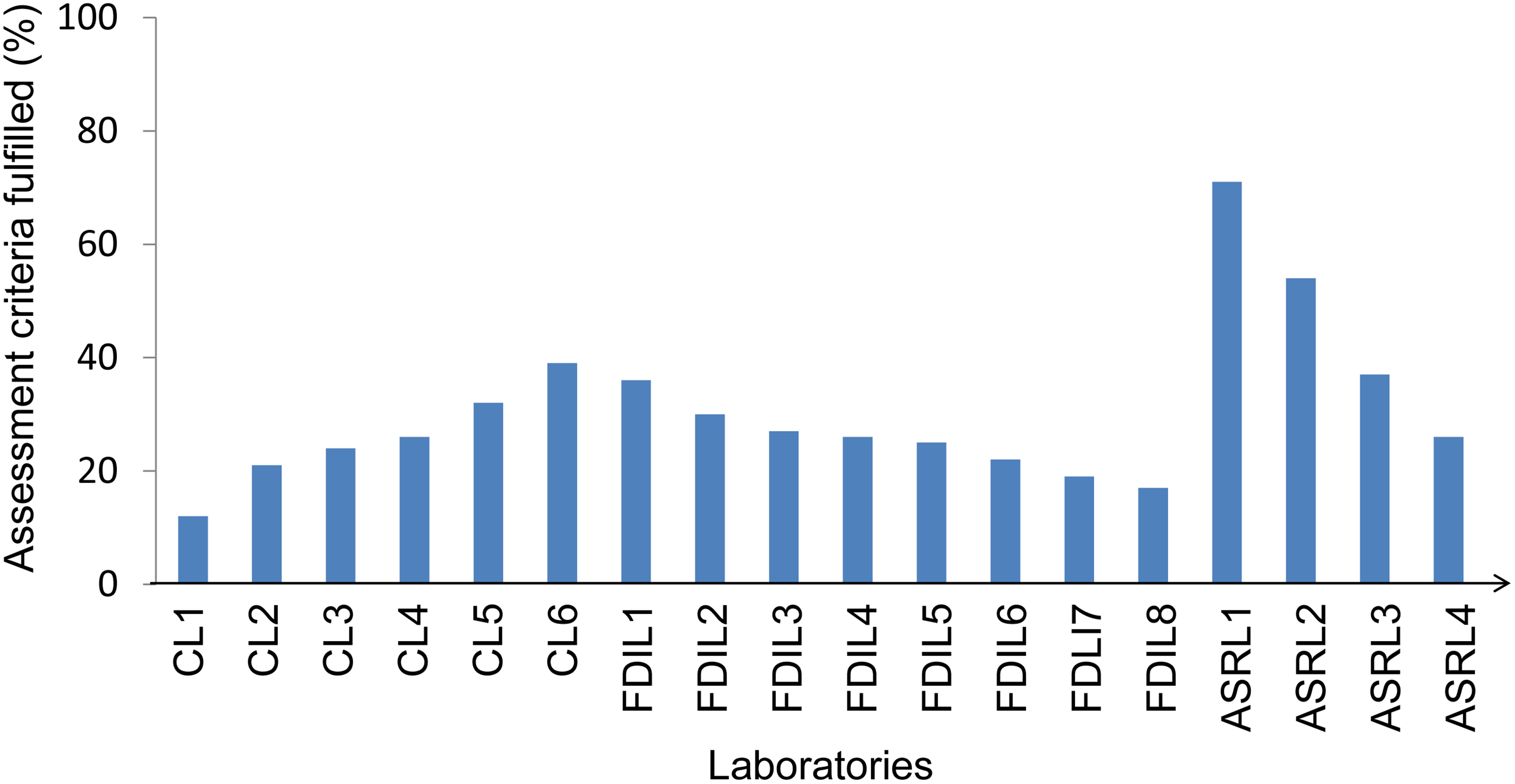
Baseline assessment of 6 clinical laboratories (CLs) under medical colleges, 8 field disease investigation laboratories (FDILs) under the Department of Livestock Services, and 4 veterinary and animal research laboratories (ASRLs) under different universities and research centers in Bangladesh.
Short-term plans include creating awareness among top management, training midlevel staff members, training bench workers, maintenance of standard practice for sample collection, proper transportation of clinical specimens, adherence to personal safety procedures and periodic training, maintenance of safety standards for sample processing, and proper waste segregation, handling, and disposal.
Midterm plans include development of a safety manual, monitoring the status of immunization, use of personal protective equipment (shoes, gown, and laboratory coat), and accident and incident reporting.
Long-term plans include developing biosafety and biosecurity guidelines, developing laboratory facilities according to the guidelines, and developing a biosafety program following the guidelines.
Awareness and Training Program
RPs organized awareness and training programs in different divisions (Figure 3). Through this comprehensive approach, a total of 1,126 individuals inclusive of 372 from top management, 413 midlevel staff members, and 341 bench workers participated (Figure 3). The experience gained from the divisional awareness program led to the development of a 2-day institutional biosafety and biosecurity training session including modules focused on design aspects to confer anticipated safety to laboratorians, general laboratory safety issues, occupational exposure to chemicals and biological agents, chemical safety on the basis of a material safety data sheet, biosafety and biosecurity, and biomedical waste management. A tailored version of these training modules was also translated into Bengali for laboratory workers. More than 2,000 laboratory personnel were trained using these modules from 2015 through 2019 (Tables 1 and 2). In addition, a 1-day biorisk management workshop was organized in 4 districts (Table 1 and 2). Most of the programs (26 of 29) were organized by the trained RPs at their institutes independently, to create a culture of biosafety and biosecurity practices (Table 2).
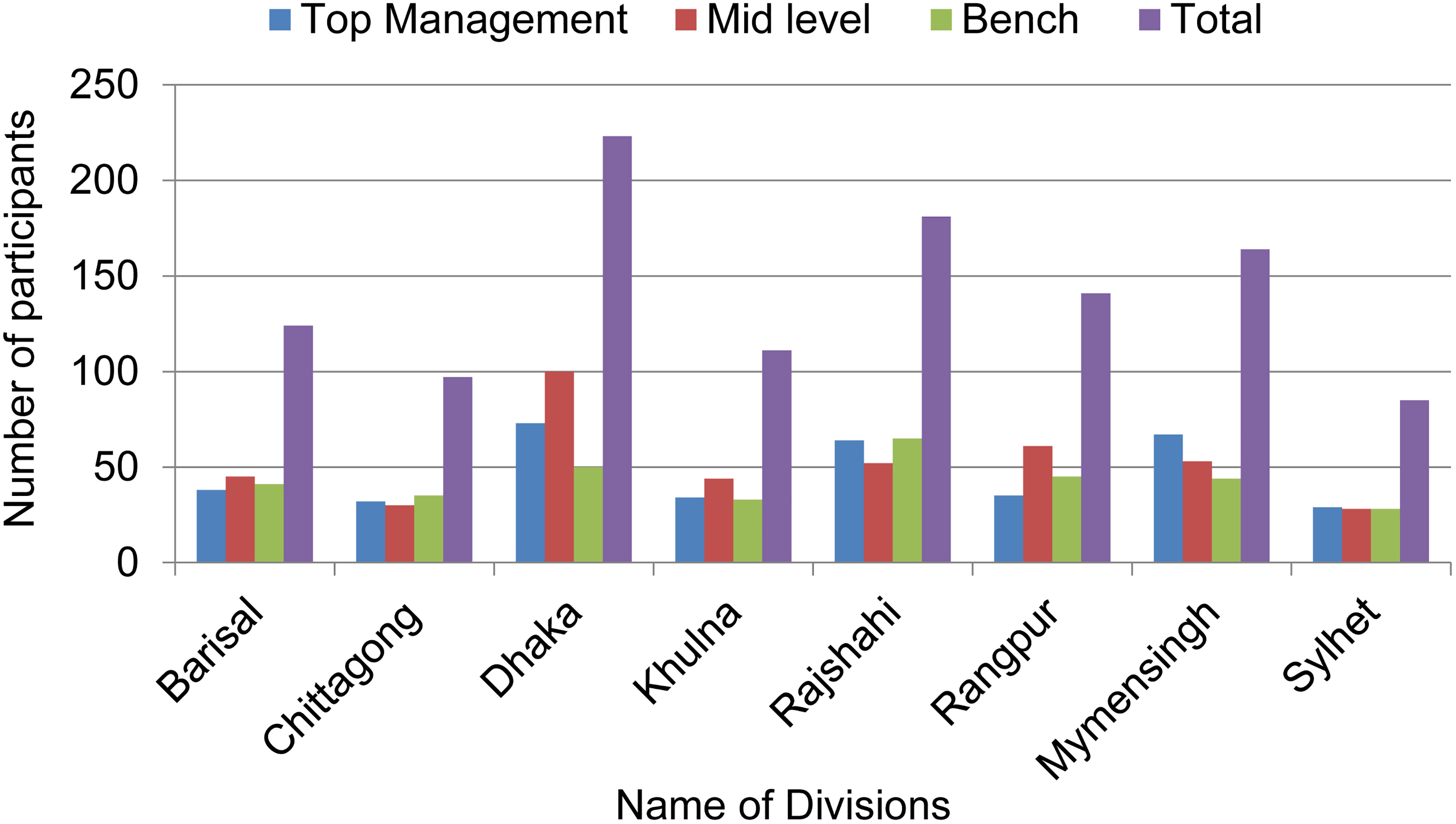
Divisional awareness program in 8 divisions at the major universities, medical colleges, and government institutions. These programs were organized in Barisal division in August 2015 at Patuakhali Science and Technology University, in Chittagong division in June 2015 at the Bangladesh Institute of Tropical and Infectious Diseases, in Dhaka division in December 2015 at the Directorate General of Health Services, in Khulna division in August 2015 at Khulna University, in Mymensingh division in November 2015 at Bangladesh Agricultural University, in Rajshahi division in August 2015 at the University of Rajshahi, in Rangpur division in September 2015 at Rangpur Medical College Hospital, and in Sylhet division in June 2015 at Shahjalal University of Science and Technology. The divisional awareness program was organized for top management, midlevel staff members, and bench workers, respectively, in Barisal for 38, 45, and 41 participants; in Chittagong for 32, 30, and 35 participants; in Dhaka for 73, 100, and 50 participants; in Khulna for 34, 44, and 33 participants; in Rajshahi for 64, 52, and 65 participants; in Rangpur for 35, 61, and 45 participants; in Mymensingh for 67, 53, and 44 participants; and in Sylhet for 29, 28, and 28 participants.
Training Programs Organized in Different Districts at Different Universities, Institutions, and Venues.a
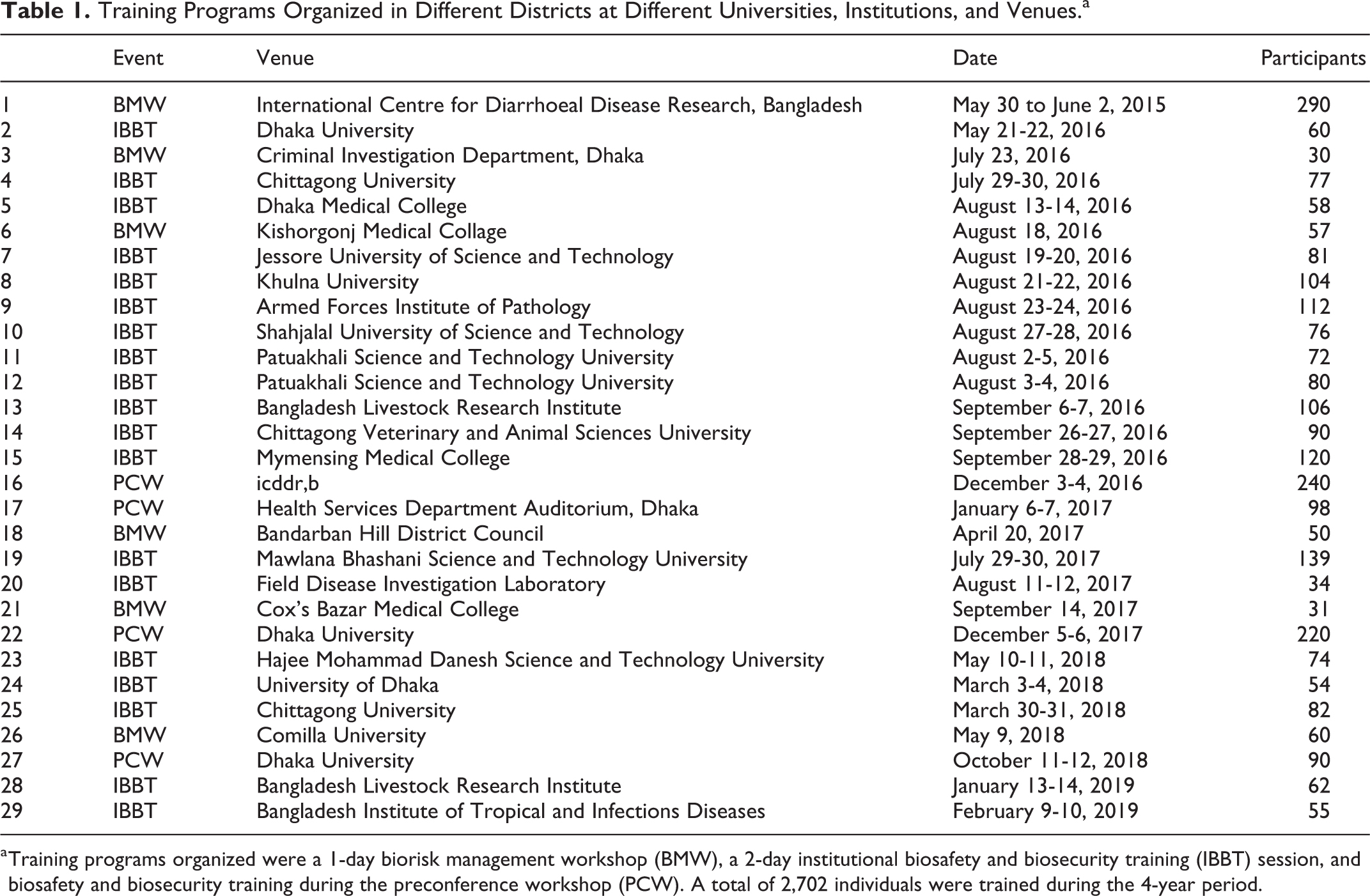
a Training programs organized were a 1-day biorisk management workshop (BMW), a 2-day institutional biosafety and biosecurity training (IBBT) session, and biosafety and biosecurity training during the preconference workshop (PCW). A total of 2,702 individuals were trained during the 4-year period.
Characteristics of Training Programs.

a Programs organized by resource persons (RPs) were supported by the Biosafety Office (BSO) of the International Centre for Diarrhoeal Disease Research, Bangladesh, to allocate funds received from Centers for Disease Control and Prevention (CDC)/Biosecurity Engagement Program (BEP) or other sources. The annual conference (AC) and preconference workshop (PCW) were organized by the RPs independently using the platform of the society. Twenty-six of the 29 programs was organized by the RPs, which clearly indicates the success of the RP development program. A total of 4,157 individuals participated in the various programs.
Abbreviations: BMW, biorisk management workshop; IBBT, institutional biosafety and biosecurity training.
Identification of Research and Diagnostic Laboratories
The baseline assessments results from 18 laboratories (Figure 2) were considered as representative of biosafety and biosecurity practices for other laboratories in the government sector. Thus, to reach out to as many laboratories as possible within a short period of time to plan and extend support, this activity was undertaken with the assistance of the RPs. 13 One hundred nine laboratories were physically identified (Figure 4) during July and August 2016. We combined the relevant data from baseline assessments of the 18 previously assessed laboratories (Figure 2) with the data from laboratory identification and estimated the total number of BSL-1 and BSL-2 laboratory facilities 6,7,23 (Figure 4). Among the 127 laboratories identified, 72% of the laboratories (n = 92) are considered BSL-2, and the rest are considered BSL-1. 6,7,23 Among the BSL-2 laboratories, 28% conducted activities without any safety equipment (autoclave, BSC, fire extinguisher or fire blanket, and eyewash or safety shower), 13% had at least 1 autoclave, 20% had at least 1 autoclave and a BSC, and 10% had autoclaves, BSCs, and even laminar flow cabinets. Most alarmingly, 2% of the laboratories were found to be using laminar flow cabinets to culture infectious microbes, even though these laboratories had BSCs. Functional and inspected fire extinguishers or fire blankets and eyewash stations or safety showers were not observed during the assessments (Figure 4). Dedicated sinks for handwashing were not found in any of the laboratories; 24 however at least 1 sink was available at all the facilities.
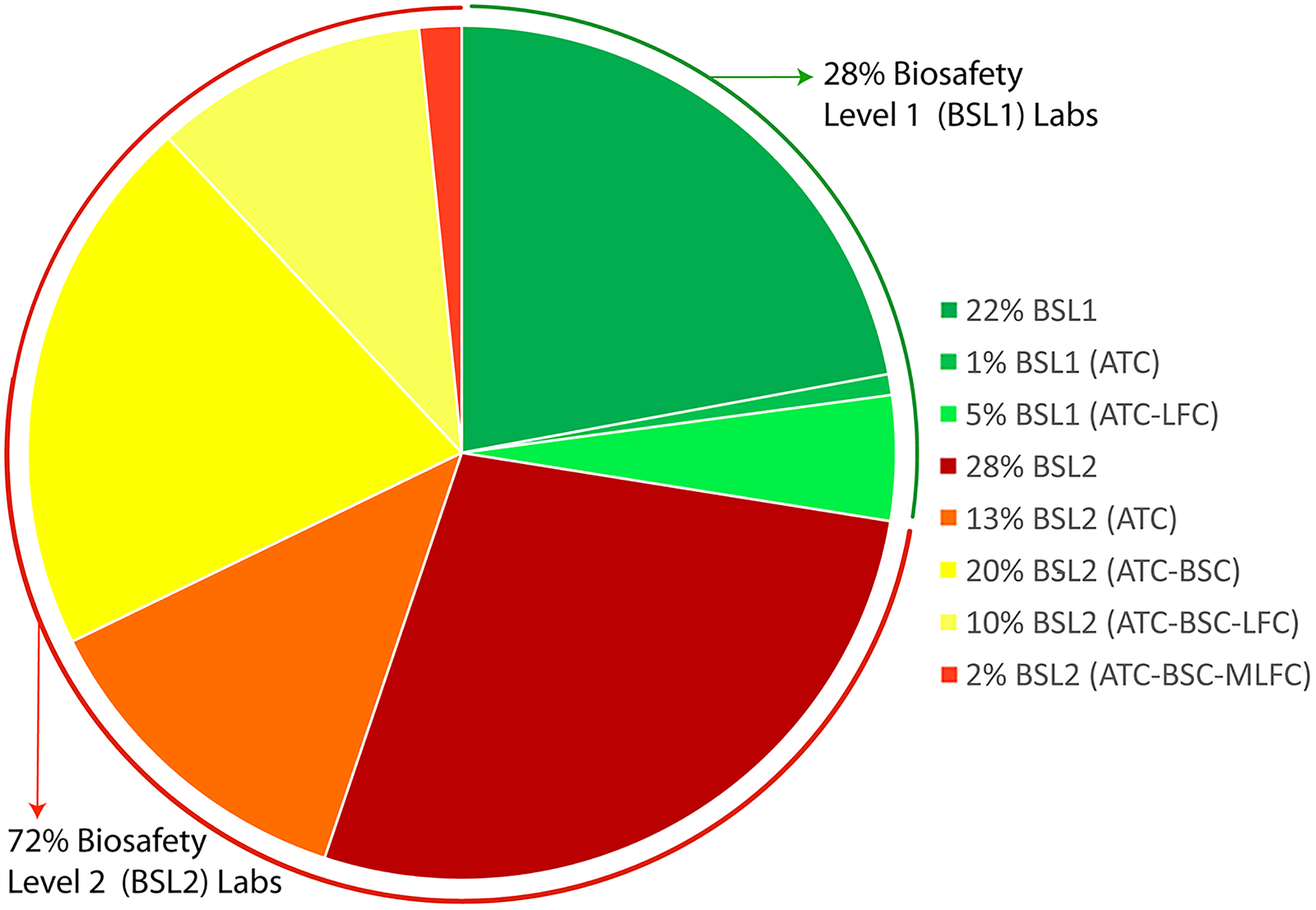
Identification of laboratories dealing with biological hazards in Bangladesh. Including the laboratories subjected to baseline assessment, 127 laboratories (100%) are included. The figure shows the percentage of laboratories considered biosafety level (BSL) 1 and BSL-2 on the basis of the activities performed within BSL-1 laboratories with only autoclave (ATC) and both an ATC and a laminar air flow cabinet (LFC), BSL-2 laboratories without any safety equipment, BSL-2 laboratories with an ATC only, BSL-2 laboratories with an ATC and a biosafety cabinet (BSC), BSL-2 laboratories with an ATC, a BSC, and an LFC, and BSL-2 laboratories with an ATC and a BSC using an LFC for dealing with or culturing infectious agents (MLFC).
BSC Certification
Currently, 3 NSF-accredited BSC certifiers are available to certify BSCs throughout the country. 14,15 An engineer from icddr,b successfully obtained NSF accreditation and in his first year certified more than 60 BSCs in the reference laboratories in Bangladesh. Besides BSC certification, these certifiers also provide training in the use and maintenance of BSCs. This has resulted in the technical advantage of having in-country BSCs certifier for (1) certification of BSCs at installation; (2) annual maintenance by accredited certifiers; (3) recertification of BSCs when moved; and (4) on-time annual recertification. Financial advantages include (1) costs are comparatively low, (2) there are no overseas travel costs, and (3) BSC certification is not considered extravagant, a total cultural shift.
Strengthening the BBBS
The BBBS provides the platform for the program Strengthening Biosecurity/Biosafety Capacity in Bangladesh (SBBCB). The society was initiated in November 2011; however, because of lack of support, it was not possible to organize biosafety and biosecurity activities in a robust manner. The achievement of the SBBCB program is that all the RPs originating from the ToT program between 2015 and 2018 were absorbed in the society as lifetime members. A team of divisional coordinators was formed from these RPs to coordinate activities in 8 divisions in Bangladesh (Figure 1). 15 Additionally, a Web site was developed to establish a network among the researchers and to disseminate knowledge and science regarding biosafety and biosecurity. Ultimately, the BBBS’s affiliation with the International Federation of Biosafety Associations and with the Centers for Disease Control and Prevention has been the key strength in organizing seminars, workshops, and conferences.
The dynamic website 15 provides guidance documents, information, and resources to its members. All laboratorians are encouraged to become society members. The BBBS is a unique overarching and inclusive platform in promoting biosafety and biosecurity program across all scientific disciplines and institutes in Bangladesh.
Development and Dissemination of National Guidelines
The core committee coherently developed guidelines to ease the burden of regulatory compliance and to nationally streamline biosafety and biosecurity requirements and practices across various sectors. The single reference guidelines provide guidance for facilities handling human, animal, and plant pathogens for research and diagnostic purposes.
23
The guidelines include the following topics: Section 1.0: Research and Diagnostics with Biohazardous Materials Chapter 1.1: Introduction Chapter 1.2: Purview—Extent and Exemptions Chapter 1.3: Summary of Procedures Chapter 1.4: Experiments Covered by the Guidelines Chapter 1.5: Import, Export and Transport of Biohazardous Materials Section 2.0: Construction of Biomedical Research and Diagnostic Laboratories Chapter 2.1: General Requirement Chapter 2.2: Requirements for Microbiological and Biomedical Laboratories Chapter 2.3: Commissioning and Certification Section 3.0: Biological Waste Management Section 4.0: Appendix
This document will complement the existing biosafety guideline for genetically modified organisms published in 2008. Streamlining of regulatory requirements will help in establishing and monitoring biosafety and biosecurity practices across the country.
Discussion
The laboratory workforce requires adequate knowledge, training, and experience to perform operations safely and securely. Most important, relevant educational background with in-depth knowledge in the field of biosafety and biosecurity and theoretical and hands-on training in this area are considered the basic qualifications for working in a microbiological or biomedical research and/or diagnostic laboratory. One of the most pressing global public health challenges is the ongoing threat of emerging and infectious pathogens. Strengthening the biosafety and biosecurity status of public and veterinary laboratories to safely detect and report is important in the control of outbreaks. This should be a top priority for ensuring a healthy nation. Thus, the SBBCB program was initiated to develop RPs and through them promote biosafety and biosecurity, creating a sustainable culture of practices, contributing to sustainable development in health and agriculture in Bangladesh. The program involved a multipronged approach, and activities were either organized for the RPs as part of their basic and continuous development or by the RPs to promote and establish biosafety and biosecurity in the country. Activities organized for the RPs included (1) the ToT program, (2) training of assessors, (3) annual refresher training, (4) training in writing standard operating procedures, and (5) training in developing biosafety manuals. Activities organized by the RPs included (1) the divisional awareness workshop, (2) institutional training, (3) baseline assessments of laboratories, (4) the laboratory identification program, (5) visiting and auditing of laboratories, (6) contributing to guideline development, (7) organizing preconference workshops and annual conferences, (8) incorporating biosafety and biosecurity in academia, and (9) initiating institutional biosafety committees.
The vital accomplishment of the SBBCB program was the development of 133 RPs (Figure 1). RPs were further tasked to organize all the programs in a more systematic manner, promoting knowledge and science regarding biosafety and biosecurity (Tables 1 and 2). RPs are the backbone of this program. However, the number of active RPs is decreasing because of (1) retirement, (2) promotion, and (3) transfer. Although there are at least 5 RPs per division (Figure 1), this number will decrease in the future. This is due to a lack of funding support to continue the program. Therefore, continuation of this program is essential to maintain the minimum number of RPs.
The assessment of 18 laboratories can be considered an overall reflection of the status of laboratories in Bangladesh (Figure 2). All 18 laboratories are considered top-level institutions. Thus, through these assessments, the necessity of promoting biosafety and biosecurity in Bangladesh was undeniably comprehended. The situation of 6 clinical laboratories, inside and outside the capital city, offered a clear picture of the situation in the health sector. On the other hand, the situation of the field disease investigation laboratories, located in different parts of the country, and the other animal sciences research and diagnostic laboratories visited, presented a comprehensive image of the agricultural sector. As a result, the assessments helped us finalize the needs and types of training sessions; identify gaps in safety and security systems; and recognize the need to develop policies, procedures, and guidelines. Laboratory identification activity proved to be a successful attempt to reach out to the other laboratories in the shortest possible time to extend support and disseminate knowledge and science regarding biosafety and biosecurity. It was observed that 28% of BSL-2 laboratories were operating without functional autoclaves for sterilization (Figure 3). This finding is alarming considering the impact on the biosphere of the disposal of untreated laboratory waste.
Awareness workshops organized in 8 divisions (Figure 3) strongly suggest that biosafety and biosecurity should be a priority for all individuals working with biohazardous materials and should be the primary consideration when undertaking diagnostic and research activities. Universities should start teaching these 2 disciplines to students under the biological sciences faculty as a separate course or as part of the practical course curriculum. Accordingly, a proposed course curriculum has been uploaded on the Web site for the educators. 25 It was suggested that the words “biosafety” and “biosecurity” be translated into Bengali for clear and in-depth understanding of the subject matter and to develop an actual sense of the issues for growing responsiveness. Accordingly, the 2 words were translated, and customized training modules were also translated into Bengali. 12
It was also clear from the baseline assessments, awareness seminars, training sessions, and laboratory identification program that basic understanding of safety equipment and its need was vague among the laboratorians. Thus, emergency and training kits were developed, including all required safety equipment, and distributed to laboratories to initiate a culture of safety practices (Figure 5A). Throughout, it was understood that hand hygiene is inadequately practiced by laboratory technicians and health care providers. 24,26 For professionals dealing with hazards either biological or chemical, the main component of hand hygiene is using the appropriate type and size of gloves to avoid contact with hazardous materials and proper handwashing after use of gloves. However, we observed that there is a prominent lack of understanding of the significance of using gloves and the importance of handwashing. Thus, it was explained to all the trainees the significance of using gloves in avoiding contact and that handwashing works by dilution of the microbial count. As a result, if the infectious dose of an organism is low and handwashing fails to reduce the count lower than the infectious dose, there is a chance of infection even after washing hands with soap and water. Emphasis was given to train all participants in hand hygiene practices. This was further highlighted by demonstrating proper doffing of gloves and importance of handwashing by using Glo Germ (Figure 5B).
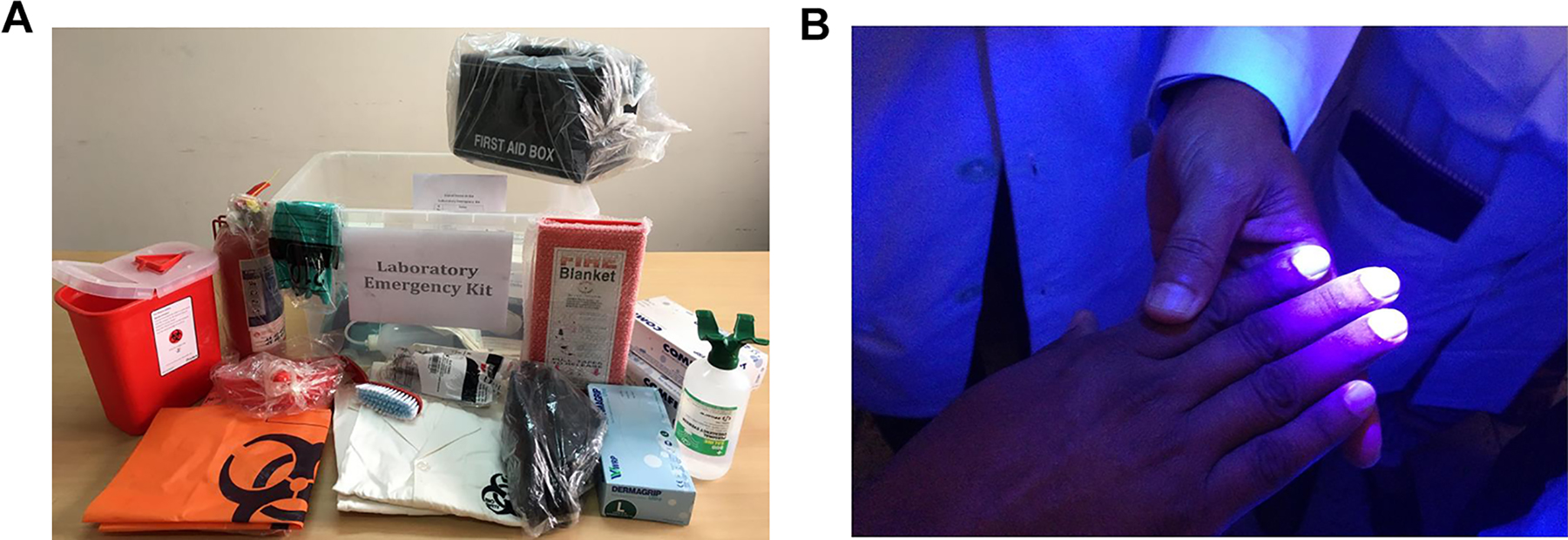
(A) A laboratory emergency and training kit for biosafety and biosecurity programs was distributed to 13 facilities. Distribution may be continued depending on the availability of funding. The kit contained 5 sample laboratory coats, 5 pairs of laboratory shoes, 3 pairs of goggles, 3 boxes of different sizes of gloves, 2 boxes of N-95 masks, 3 pairs of chemical-resistant gloves, 1 eye wash bottle, 2 pairs of forceps, 1 wash bottle, 1 dust scooper, 1 pack of absorbent tissues, 1 first-aid box, 1 fire blanket, 1 fire extinguisher, 100 biohazard bags, and 1 sharps container. (B) Demonstration of handwashing and its importance using Glo Germ (http://www.glogerm.com), a visual tool for teaching proper handwashing, aseptic techniques, and general infection control.
Bangladesh has now 3 NSF-accredited BSC certifiers in the country. According to the NSF Web site, among neighboring countries, India has 1, Malaysia has 3, and Indonesia has 2, and there are no certifiers in Pakistan, Nepal, Bhutan, and Sri Lanka. 27 In this respect, Bangladesh has had remarkable success in this area. A concrete network must be established in the country among BSC users, which is possible through the BBBS platform. The BBBS Web site describes the importance of the BSC certification program and the link of NSF-accredited BSC certifiers. The cabinets identified under the baseline assessments and the laboratory identification program will gradually be assigned under the annual BSC certification program. Thus, after initial certification, they will be oriented toward the availability of certification support in future. Information about BSC certification also has been posted on the Web site 14 so that users can directly communicate with certifiers in future.
Conclusions
Biosafety and biosecurity are areas in which the developing world requires substantial progress for sustainable development in health and agriculture. These 2 measures are designed to mitigate biological risks that pose potentially catastrophic consequences to a country’s health system, security, and political and economic stability. Unfortunately, these are often underprioritized nationally, regionally, and globally. 28 New infectious agents such as severe acute respiratory syndrome coronavirus, Middle East respiratory syndrome coronavirus, Zika virus, and severe acute respiratory syndrome coronavirus-2 have emerged. 29,30 Work with infectious agents in public and private research, public health, clinical and diagnostic laboratories, and animal care facilities has recently been expanded in Bangladesh. Thus, an active network working on biosafety and biosecurity in health and agriculture is indeed a timely indication of sustainable development in the country.
Since the inception of the biosafety and biosecurity program in September 2009 at icddr,b, the organization is trying to comply with all applicable international guidelines and standards. At that time, it was clearly understood that focusing solitarily on biosafety and biosecurity may not help the nation at large. Consequently, the SBBCB program with its multipronged approach was designed to support the entire nation. Undoubtedly, the knowledge and scientific information disseminated through the program’s activities will significantly help the nation in controlling the of spread of biohazards and infectious disease from health care and agricultural activities.
The biosafety and biosecurity program initiated in 2011 by the society has received substantial support through different activities from different donors and the government since 2013. Nationwide introduction has been successful; however, the expected initiation of a sustainable culture of practices is not yet in place. In the initial stage of the initiative, laboratory personnel considered maintaining biosafety and biosecurity as an ornamental component of laboratory management for a developing nation. The tiered nature of training programs has not only provided an opportunity for learning but also a platform to communicate among the various levels of laboratory technicians and administrators. The importance of biosafety and biosecurity in keeping the health care workforce safe, secure, and available at all times was very well understood. Throughout the journey, from the first ToT program in 2015 to institutional biosafety and biosecurity training in 2019 (Figure 6), it was observed that the mind-set of laboratory personnel has been transformed, and most technicians and administrators now consider biosafety and biosecurity as integral elements of laboratory activities, which is related to sustainability. Thus, the BBBS is now working to introduce the role of biosafety and biosecurity in sustainable development in health and agriculture. Gradually the program is shifting from a supported to a sustainable model, with the enthusiasm of the RPs and the induction of biosafety and biosecurity in educational curriculum. It has been well established that the major challenges are harmonization and enforcement of established policies nationwide and consistent support from senior management levels, budget constraints resulting in insufficient maintenance of safety equipment, and compromised safety features and insufficient staff awareness and engagement on biosafety and biosecurity measures, especially at the operational levels. 31 As a result, further support from the government and donor agencies is required to continue to grow the program and to take it to the desired level of sustainability.
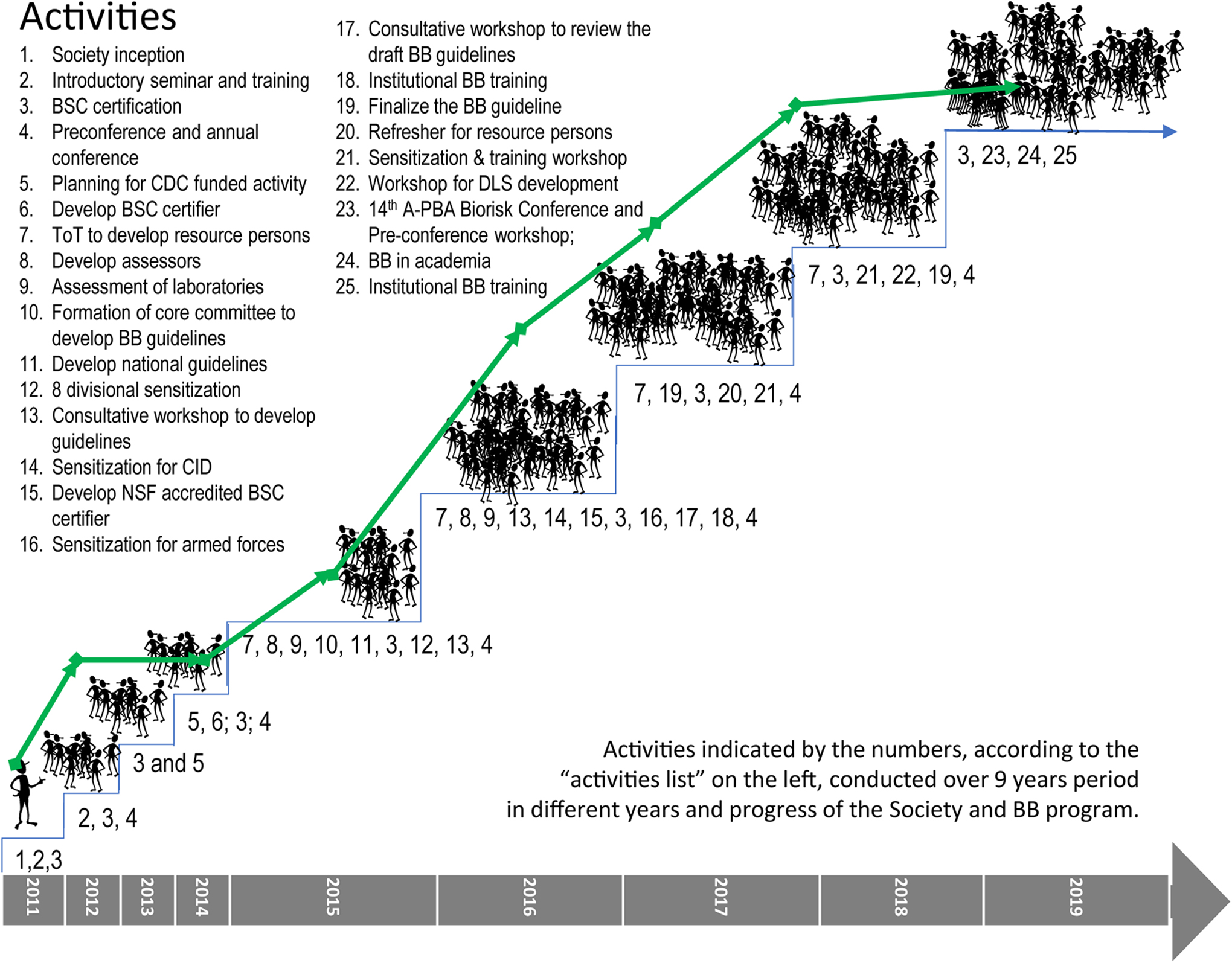
Evolution of the biosafety and biosecurity program in Bangladesh. The biosafety and biosecurity program in Bangladesh was formally introduced to the nation with the inception of the Bangladesh Biosafety and Biosecurity Society (BBBS) in 2011, and since then, little by little, different activities and components have been added. The program attained its full-fledged structure with an increase in the number of program participants as members of the society. The curve on the figure shows the progress of BBBS membership, indicating that the Strengthening Biosecurity/Biosafety Capacity in Bangladesh program substantially contributed to the increase in membership. Currently the society has 184 lifetime members, 162 general members, and 218 student members registered. A-PBA = Asia-Pacific Biosafety Association; BB, biosafety and biosecurity; BSC, biosafety cabinet; CDC, Centers for Disease Control and Prevention; CID =The Criminal Investigation Department; DLS = Department of Livestock Services.
Footnotes
Acknowledgments
We thank the director of disease control and the line director of communicable disease control, Directorate General of Health Services, for continuous guidance. We thank the Executive Director of icddr,b for continuous support. We also thank the Senior Director of the Laboratory Sciences and Services Division of icddr,b for continuous guidance. We thank Dr Barbara Johnson, Biosafety and Biosecurity International, for substantially supporting the initiation of the biosafety and biosecurity program in Bangladesh. We thank Dr Katherine Sturm-Ramirez, Dr Kevin L. Russell, and the Centers for Disease Control and Prevention for their support of the program. We thank Dr Natasha K. Griffith for her enormous support to initiate a structured biorisk management program in Bangladesh. We also thank Probal C. Rozario, Syeda Mah-E-Muneer, and Partha Sarathi Gope for their continuous managerial support. We thank all the divisional coordinators of the BBBS for their enormous support. We thank ESCO Micro Pte Ltd for continuously supporting the annual program of the society. We thank the Asia-Pacific Biosafety Association and the International Federation of Biosafety Associations for continuous mentoring support. We thank Dr Hubert P. Endtz, Professor of Tropical Bacteriology, Department of Medical Microbiology & Infectious Diseases, Erasmus Medical College, for implanting the concept of biosafety and biosecurity in Bangladesh.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Statement
Not applicable to this study.
Statement of Human and Animal Rights
Not applicable to this study.
Statement of Informed Consent
Not applicable to this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This activity was funded by icddr,b’s core donors and in part by the Virtual Biosecurity Center administered by the Federation of American Scientists for the activity “A Multi-Pronged Approach to Developing a Sustainable Means to Strengthen Biosecurity in Bangladesh” for 2011. The work was also funded by Centers for Disease Control and Prevention for the project SBBCB for 2013 to 2015. The continuation of SBBCB was further funded by the Biosecurity Engagement Program of the US Department of State for 2015 to 2018. icddr,b also gratefully acknowledges the following donors who provide unrestricted support: the governments of Bangladesh, Canada, Sweden, and the United Kingdom.
Prior Presentation
An abstract on this work was presented at the 14th Asia-Pacific Biosafety Association Annual Biorisk Conference, Dhaka, Bangladesh, September 19-20, 2019.
