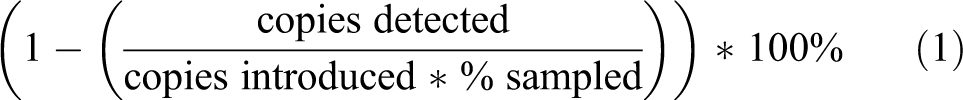Abstract
Most laboratory techniques are performed in biosafety cabinets (BSCs) to provide contamination control of the experiments. By using specifically directed airflow and high-efficiency particulate air (HEPA) filtration, potential contaminants are removed from the work area within the BSC. Aerosols may be created by common lab practices, such as centrifugation, pipetting, and opening tubes. HEPA filters are very effective at removing various-sized contaminants but do not prevent gases and vapors from penetrating through them. HEPA filters are not effective at all sizes of particles. Researchers have speculated that DNA may not be captured by HEPA filters, allowing for contamination of subsequent experiments by aerosolized DNA. Here we found that DNA is captured by a HEPA filter at the same efficiency as the filter is rated and discovered that DNA cannot be dislodged from a HEPA filter.
High-efficiency particulate air (HEPA) filters are the main line of defense against contamination used in biosafety cabinet (BSC) technology. HEPA filters remove particles by 3 different mechanisms: impaction, interception, and diffusion. As seen in Figure 1A, all 3 of these methods lead to very high particulate removal of a wide range of sizes with the most penetrating particle size of 0.21 μm. When tested at 0.3 μm, near its worst, the HEPA filter removes 99.97% or better of all particulates. National Sanitation Foundation (NSF) requires HEPA filters to be of Type C or better, removing a minimum of 99.99% of 0.3-μm particulates. 1
High-efficiency particulate air (HEPA) filter function and design. (A) The efficiency of a HEPA filter of removing particles of a particular size. (B) The folded structure of the borosilicate sheets of a HEPA filter cross section. These pleats help direct air through the filter and ensure a greater surface area for filtration.
HEPA filters are composed of 1 continuous pleated sheet of borosilicate fibers woven into a crosshatched design. This sheet has spacers in between the pleats to hold the shape and increase the filter surface area. Two-part polyurethane is used to create an airtight seal between the filter membrane and a sturdy frame, preventing any possible leakage and potential contamination risk. 1 This rigid shape and structure shown in Figure 1B is critical for directing the air through the filter material to ensure particulate removal.
It has long been speculated by researchers and laboratory personnel whether DNA can be captured in a HEPA filter when procedures are conducted within a BSC due to the unique properties and size of DNA. Plasmid DNA has been estimated to be 200 nm or 0.2 μm in diameter when supercoiled, 2 but the ability of DNA to exist in multiple conformations leads to uncertainty. In addition, DNA could exist within an aerosolized droplet of varying sizes, depending on its source of creation. As depicted in Figure 1A, the larger the droplet, the greater chance it has of being captured. However, as the droplets dry, its size returns to the most penetrating size of HEPA filters. Whether it will be captured in the filter media or not is uncertain.
Here we test the ability of DNA to be captured by a HEPA filter in 2 scenarios: (1) direct application to a HEPA filter and (2) circulating in the airstream of a Class II Type A2 BSC (a partially recirculating cabinet, the most common style found in laboratories). We then tested whether DNA can be dislodged from a HEPA filter after a physical shock or an electrical power failure, as well as if DNA is destroyed by a full cabinet decontamination with chlorine dioxide (CD).
Methods
Direct Application to the HEPA Filter
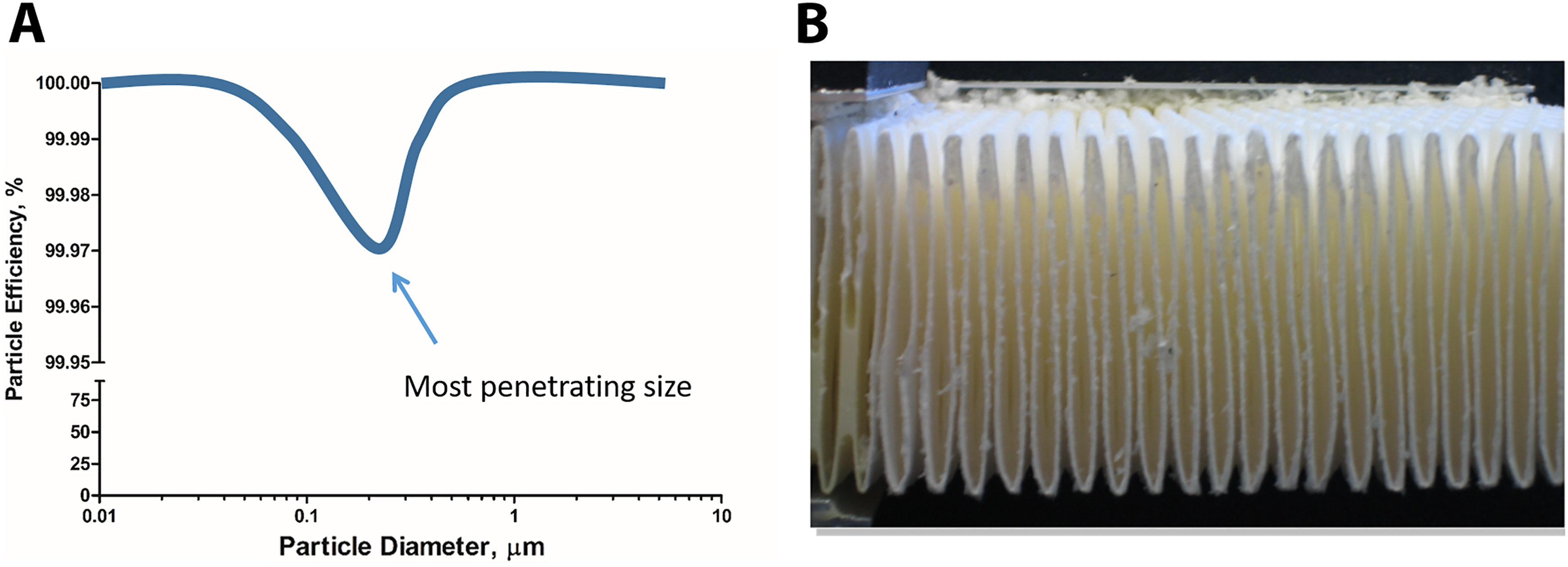
Plasmid DNA (60 mL, 1 × 109 copies/mL in Tris-EDTA (TE) buffer) was placed in a Collison nebulizer, which generates a defined droplet size of ∼1 μm. 3 A Class II Type B2 BSC (BioChemGARD; Baker, Sanford, Maine) was installed with new HEPA filters, adjusted to a setpoint of 105 fpm intake air with an 8-inch view screen opening by a qualified technician according to NSF standards, and cleaned with 10% bleach and 70% ethanol. The worksurface was removed to expose the 2 side-by-side exhaust HEPA filters. The nebulizer was secured to a ring stand and placed directly in front of either side of the HEPA filters at a height of 6, 9, and 12 inches (Figure 2). Preexperiment samples were collected for determination of background DNA content. Glass microscope slides and Whatman filter paper were placed above and below the HEPA filters within the airstream (Figure 2C). The nebulizer was then run for 1 minute on each side with and without the HEPA filters installed (Figure 2A and 2B, respectively) with samples being collected in between each experimental condition. Samples were collected by cotton swabs prewetted with TE buffer on glass slides or the direct metal surface and pieces of the Whatman filter paper. The use of air samplers was disregarded in this situation as they were determined to be to obtrusive to the airflow patterns given the choice of sample locations and would not mimic a BSC under normal operating conditions. The quantity of DNA introduced in each condition was determined by mass displacement of the solution within the nebulizer. The BSC was thoroughly cleaned with 10% bleach and 70% ethanol in between experiments and replicates. Electrical power failure on the BSC and exhaust blower system was simulated 3 successive times to test for unintentional DNA contamination. In both situations, samples were captured downstream of the HEPA filter.
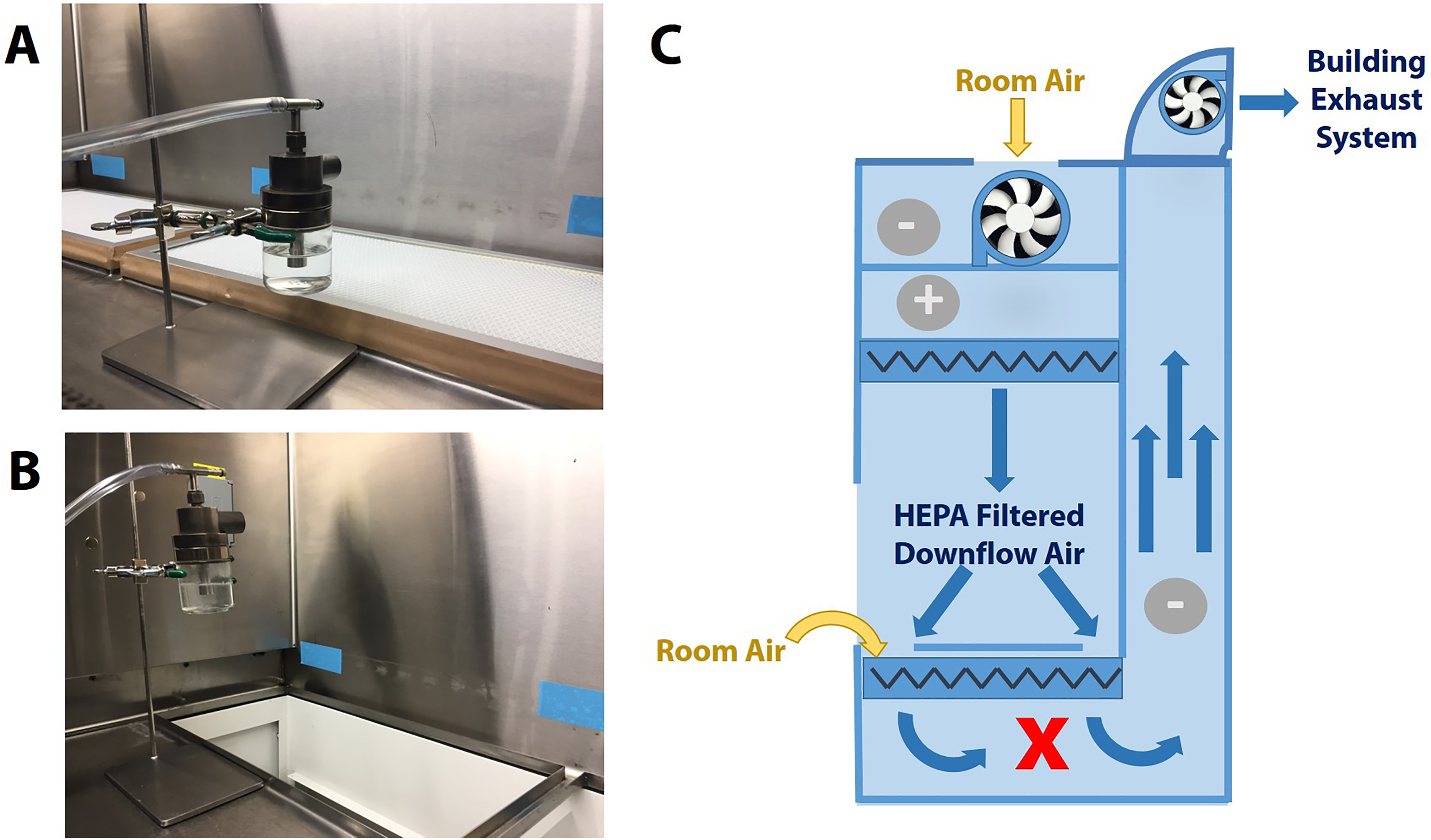
Experimental design for direct application of the aerosolized DNA to under the worksurface high-efficiency particulate air (HEPA) filters of a Class II Type B2 biosafety cabinet (BSC). Depicted are the filters in place (A) and removed (B). (C) Schematic of a Class II Type B2 BSC showing airflow arrows, positive and negative pressure areas (+ and –, respectively), HEPA filter locations (zigzag lines) including one below the worksurface, motor/blower, and the sampling location denoted by bold X.
Mechanical and Electrical Removal of DNA from HEPA Filters
HEPA filters were removed and reinstalled jarringly to try to remove any DNA within the HEPA filter. Electrical power failure on the BSC and exhaust blower system was simulated 3 successive times to test for unintentional DNA contamination. In both situations, samples were captured downstream of the HEPA filter.
DNA Collection in a BSC Airstream
A Class II Type A2 BSC (SterilGARD; Baker) was installed with new supply and exhaust HEPA filters, adjusted to a setpoint of 105 fpm intake air and 50 fpm downflow velocity with an 8-inch view screen opening by a qualified technician to NSF standards, and cleaned with 10% bleach and 70% ethanol. Glass microscope slides and Whatman filter paper were placed in the sample locations throughout the BSC: on the worksurface, below the worksurface, upstream from the supply HEPA filter, directly downstream from the supply HEPA filter, and directly downstream from the exhaust HEPA filter (Figure 3B). Plasmid DNA (60 mL, 1 × 109 copies/mL in TE buffer) in a Collision nebulizer was placed 8 inches away from the bottom of the view screen and run for 15 minutes (Figure 3A). Samples were collected by cotton swabs prewetted with TE buffer on the glass slides or directly on the metal worksurface or collecting the Whatman filter paper. The quantity of DNA introduced in each condition was determined by mass displacement of the solution within the nebulizer. The BSC was thoroughly cleaned with 10% bleach and 70% ethanol in between replicates and experiments. Capture efficiency was calculated by Equation 1.
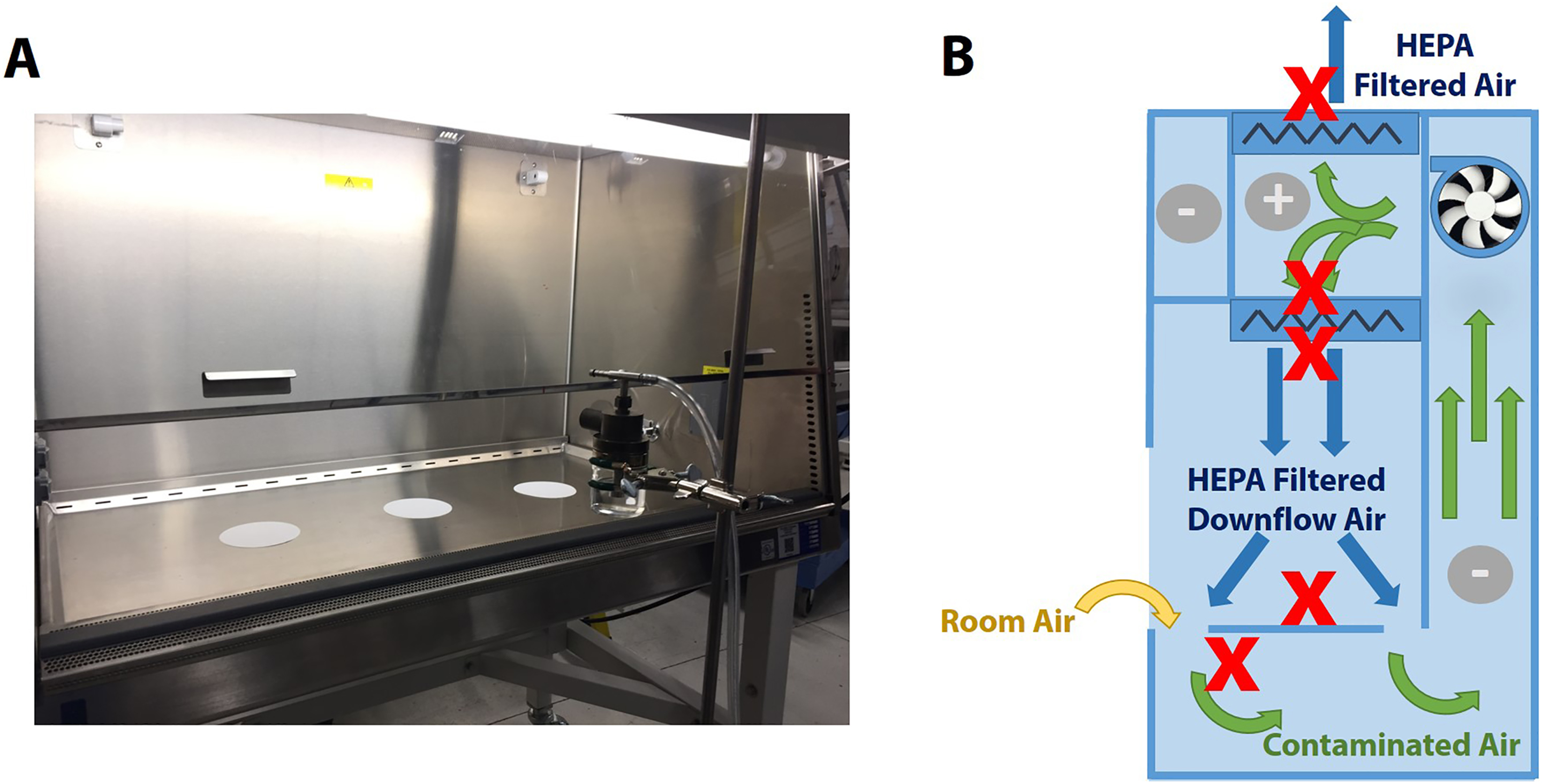
DNA in the biosafety cabinet (BSC) airstream. (A) The nebulizer layout in front of the BSC. (B) Diagram of a Class II Type A2 BSC showing airflow arrows, positive and negative pressure areas (+ and –, respectively), high-efficiency particulate air (HEPA) filter locations (zigzag lines), motor/blower, and the sampling locations denoted by bold Xs.
DNA Destruction by CD
After aerosolization of the plasmid DNA into the Class II Type A2 BSC (SterilGARD; Baker) in the previous experiment, the cabinet was prepared for a standard biodecontamination with CD using the Mini Chlorine Dioxide System (DRS Laboratories, Allentown, PA) per the manufacturer’s instructions.
DNA Quantification
Samples collected with cotton swabs were extracted by eluting in 150 μL of TE. Samples collected using filter papers were hole punched into 150 μL TE. Both types of samples were vortexed and centrifuged, and 5 μL of sample was added to quantitative polymerase chain reaction (qPCR). DNA quantification was performed by using qPCR with MMQCI’s (Saco, ME) internal assay and plasmid-based standards on Roche (Basel, Switzerland) LightCycler 2.0. Positive control samples were used to ensure the DNA had not been denatured or degraded during the aerosolization or drying processes.
Results
Direct application of the aerosolized DNA to the HEPA filter (Figure 2A) resulted in a complete capture of all DNA-containing particles (Table 1) as demonstrated by placing the nebulizers directly upstream of the HEPA filters located directly below the worksurface on the Class II Type B2 BSC and collecting the samples directly downstream of these exhaust HEPA filters (Figure 2C). After attempting to remove the DNA captured in the HEPA filter through mechanical and electrical means by simulating a power failure of the cabinet and external exhaust system, no DNA was detected (Table 1).
Direct Application of Aerosolized DNA to a HEPA Filter Using a Class II Type B2 Biosafety Cabinet.
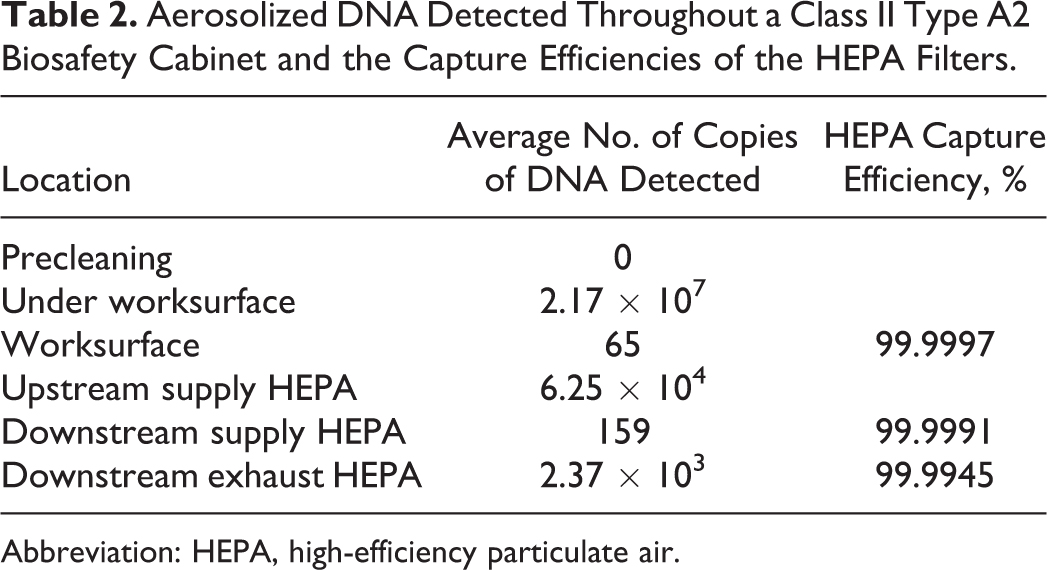
When DNA was aerosolized within a Class II Type A2 BSC as depicted in Figure 3A, DNA was detected at significant quantities throughout the cabinet as expected based on airflow: under the worksurface and upstream from the supply HEPA filter that provides clean air to the worksurface (Table 2, Figure 3B). However, unlike the direct application above, downstream from both the supply and exhaust HEPA filters, low quantities of plasmid DNA were detected. Using airflow calculations previously determined,4,5 it is known that the mass flow velocity of any compound added to the airstream by the intake air is equal to the concentration of compound in the downstream air and exhaust air. Combined with the known amount of DNA added to the airstream (volume displaced * DNA density = copies introduced) and the proportion of the surface area tested compared to the full HEPA face area, the capture efficiency can be calculated by Equation 1. All downstream HEPA samples exhibited better than 99.99% DNA removal efficiency (Table 3).
Aerosolized DNA Detected Throughout a Class II Type A2 Biosafety Cabinet and the Capture Efficiencies of the HEPA Filters.
Abbreviation: HEPA, high-efficiency particulate air.
Effectiveness of Decontamination on DNA Removal.a

Abbreviation: HEPA, high-efficiency particulate air.
DNA concentration was measured after nebulization (predecontamination), after a chlorine dioxide gaseous decontamination (postdecontamination), and after cleaning the surface with 10% bleach and 70% isopropanol sequentially (postcleaning).
CD was applied to determine if a gaseous biodecontamination protocol would remove DNA from the metal surfaces within the BSC. As shown in Table 3, CD resulted in the removal of 99.7% of the contaminated surface. After further cleaning with bleach and isopropanol, all traces of plasmid DNA were removed.
Conclusion
Upon direct application of DNA to the HEPA filter by the nebulizer inside the Class II Type B2 BSC, it was observed that all DNA aerosols were captured with no variation observed when the nebulizer was placed 6, 9, or 12 inches above the exhaust HEPA filters (Table 1, Figure 2A–C). However, when the DNA was introduced to an entire BSC as simulated in the Class II Type A2 BSC (Figure 3A), DNA was detected both upstream and, to a lesser extent, downstream of both the supply and exhaust HEPA filters (Table 2). DNA was expected to be detected upstream of the HEPA filter (under the worksurface and pre-HEPA samples; Figure 3B), as these samples were exposed to the DNA-contaminated room air introduced by the nebulizer. Controlling for the area sampled compared to the total amount of DNA introduced into the airstream (Equation 1) allows for the determination of the efficiency of DNA removal by the HEPA filter. All 3 areas where DNA was detected post-HEPA had efficiencies greater than 99.99%, as specified by NSF Standard 49 (Table 2). 1 The disparity between the results from the Class II Type B2 and Type A2 BSCs may be explained by particle size. Direct aerosolization produces droplets ∼1 μm/1000 nm in size. These particles are an ideal size to be captured by a HEPA filter. However, as the droplets move through the airstream, they will dry, as a droplet has a “wet time” of 0.6 seconds, 3 and this will decrease the DNA aerosol size to the most penetrating size of the HEPA filter, ∼0.2 μm or 200 nm (Figure 1).
CD was able to remove 99.7% of the surface-contaminated DNA but not all (Table 3). The most thorough cleaning option was bleach and isopropanol, but this solution only pertains to easily reachable surfaces.
Recommendations
Here we have shown that direct application of DNA aerosols will be captured completely by a HEPA filter, but as the droplets move through the BSC, there can be up to 0.01% contamination. To eliminate this potential contamination, under worksurface or redundant HEPA filters, a 100% exhausted BSC design could be employed depending on where the concern for DNA contamination is (eg, the samples, the worker, the lab). It could also be helpful to use a more stringent filter, such as an ultra-low particulate air (ULPA) filter with 99.999% particulate removal efficiency or a super ultra-low particulate air (SULPA) filter with 99.9999% particulate removal efficiency.
Footnotes
Declaration of Conflicting Interests
The authors disclosed the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors are employed by the Baker Company in Sanford, ME, or Maine Molecular Quality Controls, Saco, ME, as indicated on the title page.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.