Abstract
The emergence of highly infectious diseases such as Hepatitis and HIV increasingly drives the importance of health and safety in medical laboratories. This study compared the biosafety and biosecurity concerns between public and private medical diagnostic laboratories in the western part of Kenya. A total of 35 public and 78 private medical diagnostic laboratories were assessed for compliance to biorisk measures using the Kenyan biosafety and biosecurity checklist. Among the surveyed laboratories, the majority (68.1%) were located in the rural areas. Biosafety and biosecurity concerns were observed as follows: nonslip floors and stairs (95.6% of all laboratories surveyed), phlebotomy room (20.4%), hand-washing sinks (71.7%), laboratory access controlled (24.7%), inventory for valuable biological materials (16.8%), portable fire extinguishers (23%), emergency exits (15.9%), issuance of appropriate vaccination (60.2%), presence of a trained biosafety officer (60.2%), availability of equipment SOPs (86.7%), biosafety cabinets (48.7%), color-coded waste disposal bins (15.9%), laboratory waste segregation (50.4%). Private laboratories were more likely than their public counterparts to have premises meeting the building standard requirement (
Keywords
In the recent past, because of disease outbreaks such as Zika, Marburg, and Ebola viruses and the growing fear of bioterrorism, strengthening biosecurity and biosafety issues has become an urgent global goal. 1 Laboratory biosafety (defined as the containment principles, technologies, and practices that are implemented to prevent unintentional exposure to pathogens and toxins or their accidental release) and biosecurity (institutional and personal security measures designed to prevent the loss, theft, misuse, diversion, or intentional release of pathogens and toxins) 2 are increasingly becoming an integral part of Kenyan medical and diagnostics laboratories. 3 The safety of laboratory personnel and the larger environment is highly correlated with knowledge of biosafety and biosecurity concepts, as well as adherence to good laboratory practices. 4 Laboratories, especially biosafety level 3 and above, should deploy strategies both physical and technical that permit safe manipulation of highly infectious biological materials while preventing their accidental release. 5 The majority of laboratory accidents are dominated by human error, poor laboratory techniques, and technical faults.2,6 Granting that it is not entirely possible to eliminate such errors and other potential risks, proper training on laboratory biosafety and biosecurity has been shown to significantly minimize these errors. 7 In the past 5 years, Kenya has seen a remarkable increase in the development and training of laboratory personnel on biosafety and biosecurity based on WHO guidelines. 2
The Kenyan public medical diagnostic laboratory system is largely affiliated with health facilities that range from level 1 (local community-based services with very limited diagnostic capacity) to level 6 (national referral services with specialized health care services, including hospitals, laboratories, blood banks, and research institutions). 8 The level 3 to 6 laboratories that handle infectious microorganisms and biological agents should be in the frontline in promoting biosafety/biosecurity practices and prudent waste management. 9 Studies, however, show that in developing countries, the majority of the laboratories are marked by undeveloped infection control programs. 10 Lack of awareness, lack of trained personnel in infection control practices, substandard and inadequate infrastructure, and procurement challenges have been cited as some of the determinants resulting in poor infection control programs in Africa.11,12
As in other countries, the medical diagnostic laboratories in Kenya play a pivotal role in disease detection and eventual management. These laboratories are anticipated to have the capacity to detect and handle known or unknown (novel) biological agents with minimal risk of exposure both to staff and the environment. 10 It has been shown that biosafety and biosecurity training and eventual compliance to the safety measures reduce the risk of infections.3,7,13 Therefore, regular monitoring and assessment of diagnostic laboratories for the presence of biosafety devices and compliance with standard biosafety measures not only will promote a safer working environment, but also could impact greatly on maintaining qualitative laboratory service delivery. In Kenya, the legislative guideline for enforcing biosafety in clinical laboratories was developed in 2012, “The Kenyan Biosafety and Biosecurity Policy Guidelines.” 14 We sought to compare the compliance rate to biosafety and biosecurity guidelines between public and private medical diagnostic laboratories in Trans Nzoia County in western Kenya.
Methods
Study Setting and Design
This cross-sectional study was conducted January to July 2017 among public and private medical diagnostic laboratories classified as biosafety level 2 and below. A total of 35 public and 78 private medical diagnostic laboratories in Trans Nzoia County in western Kenya were approached and agreed to participate in the study. Consent was obtained from managers and/or heads of all laboratories. This study was approved by the ethical review committee of the National Commission for Science, Technology and Innovation. Additional approvals were obtained from the Ministry of Interior and Coordination of National Government, Ministry of Education Science and Technology State Department of Education and from County Government of Trans Nzoia, State Department of Health. Confidentiality about the health workers and the facilities visited was maintained.
Interviews
A detailed questionnaire and checklist adopted from the Ministry of Health, Kenyan Biosafety and Biosecurity Guidelines (http://www.mbdsnet.org/wp-content/uploads/2014/06/Kenyan-Biosafety-and-Biosecurity-Guidelines.pdf) was used to obtain relevant information related to biosafety and biosecurity from the heads and/or owner of each laboratory.
Statistical Analysis
Descriptive and inferential statistics were then performed on the data. Fisher’s exact test and odds ratio (OR) analysis was used to test the distribution differences in variables between public over private medical diagnostic laboratories at the significance level of
Results
Response Rate
Of the 114 BSL-2 laboratories located in Trans Nzoia County, 113 consented to have the survey conducted on their premises, yielding a response rate of 99.1%.
Demographic Characteristics
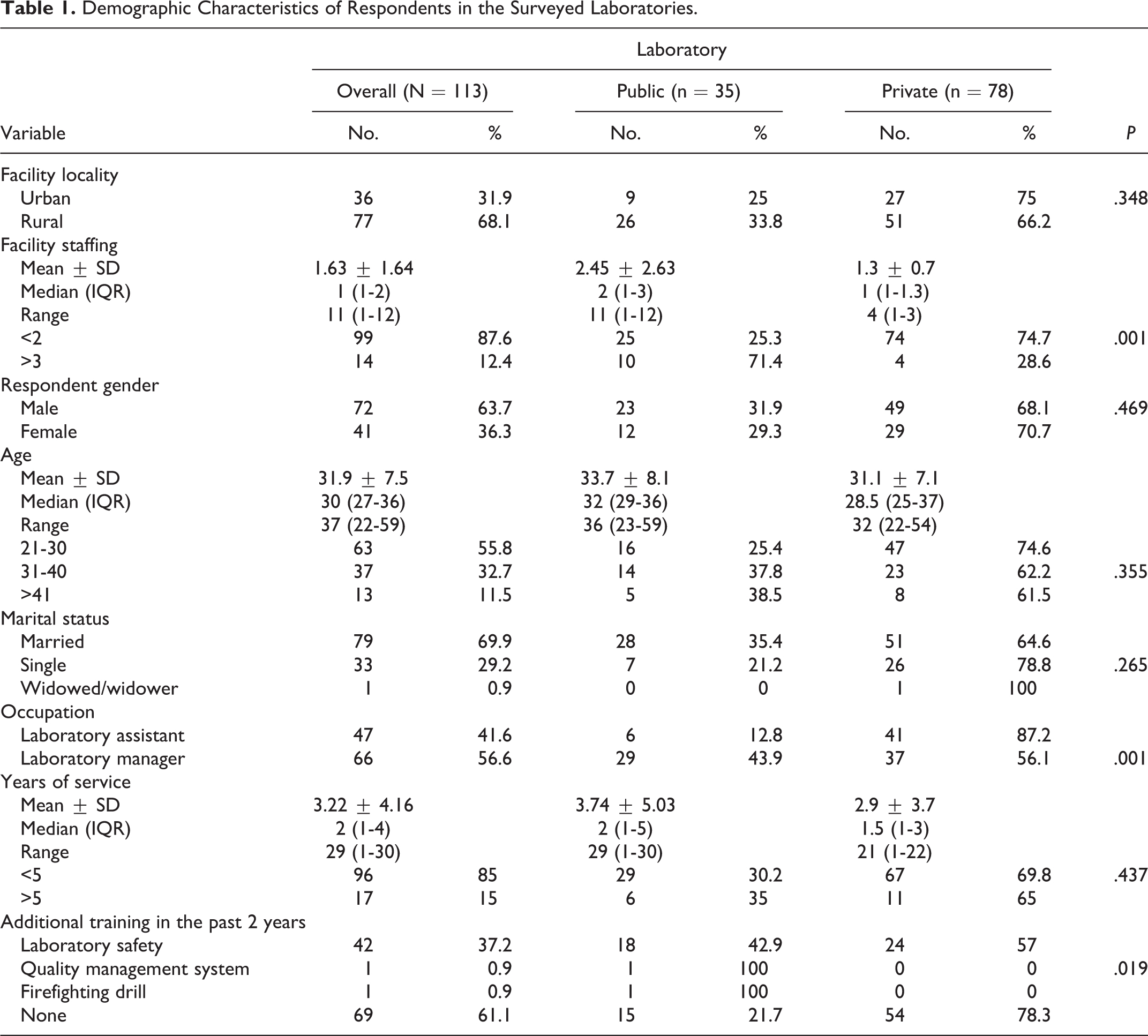
Table 1 summarizes the demographic information about the respondents and laboratories surveyed. The majority (61.1%) of the laboratories were located in rural areas (66.2% private versus 32.8% public laboratories). The public laboratories had more personnel, ranging from 1 to 13 (mean 2.45 ± 2.3), versus private laboratories, ranging from 1 to 3 (mean 1.3 ± 0.7). Respondents were predominantly male (63.7%) (68.1% private versus 31.9% public laboratories) and had a mean age of 33.7 ± 8.1 years public compared to 31.1 ± 7.1 private laboratories. Overall, the majority (55.8%) were between 21 and 30 years of age. Respondents’ occupations included all the 2 occupational categories in the laboratory with management/quality assurance composing the greatest proportion (56.6%) (56.1% private versus 43.9% pubic laboratories). The majority of respondents (85%) had <5 years of experience in a medical diagnostic laboratory (69.8% private versus 30.2% pubic laboratories). For those respondents who attended additional training in the past 2 years, the majority (37.2%) attended laboratory safety training, 0.9% on Quality management systems, and 0.9% on firefighting drills.
Demographic Characteristics of Respondents in the Surveyed Laboratories.
Laboratory Handling of Infectious Materials and Personal Protective Devices
Table 2 describes the practices related to laboratory handling of infectious materials and the use of personal protective devices. In all the laboratories examined, 59.3% handled valuable biological material (“valuable biological materials” are microorganisms natural or laboratory modified with intrinsic histological, medical, epidemiological, commercial or scientific value), 16.8% had an inventory for these biological materials, and 53.1% had records of all incoming infectious materials. Only 15.1% of the laboratories unpacked specimens in a biosafety cabinet with care and attention to possible breakage and leakage. In about 50.4% of the laboratories personnel wore gloves and other protective clothing during the unpacking of specimens. The presence of color-coded waste disposal bins, color-coded bin liners, waste segregation, waste treatment prior to disposal, proper waste collection and disposal, use of appropriate disinfectants, and wearing proper PPE at the time of the survey visit were observed in 55.8%, 54.9%, 53.1%, 37.2%, 66.4%, and 60.2%, respectively, of all laboratories examined.
Laboratory Handling of Infectious Materials and Personal Protective Devices.
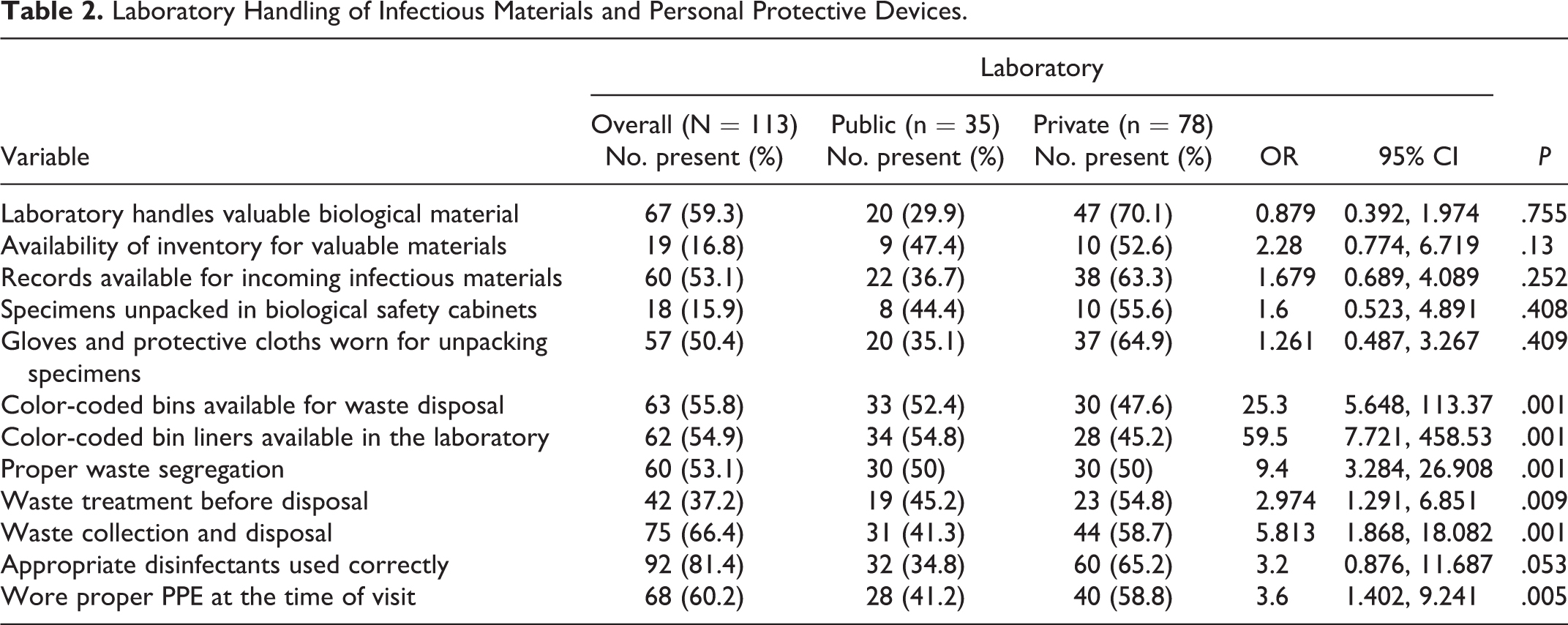
Compared to private, public laboratories were more likely to have color-coded waste disposal bins (OR 25.3, 95% confidence interval [CI] 5.648, 113.337,
Biosecurity and Biosafety Engineered Devices
Table 3 summarizes the overall, public and private medical laboratory safety and storage conditions in Trans Nzoia. Based on the results, premises meeting the building standard requirement, nonslip floors and stairs, adequate working space, separate phlebotomy room, hand-washing sinks, chemical-resistant working benches, washable working benches, and reliable power back-up were observed in 71.7%, 95.6%, 72.6%, 20.4%, 71.7%, 59.3%, 81.4%, and 22.1%, respectively, of all laboratories surveyed. Furthermore, labeled shelves, lockable freezer and storage rooms, the presence of room-monitoring thermometers, adequate ventilation, adequate laboratory illumination, availability of water/soap and paper/dryer towels, a fire alarm system, portable fire extinguishers placed in designated places, and exits marked using illuminated signs were observed in 23%, 56.6%, 14.2%, 73.5%, 84.9%, 54.9%, 95.6%, 10.6%, 23%, and 15.9%, respectively, of all laboratories assessed.
Laboratory Biosecurity in Engineered Devices.
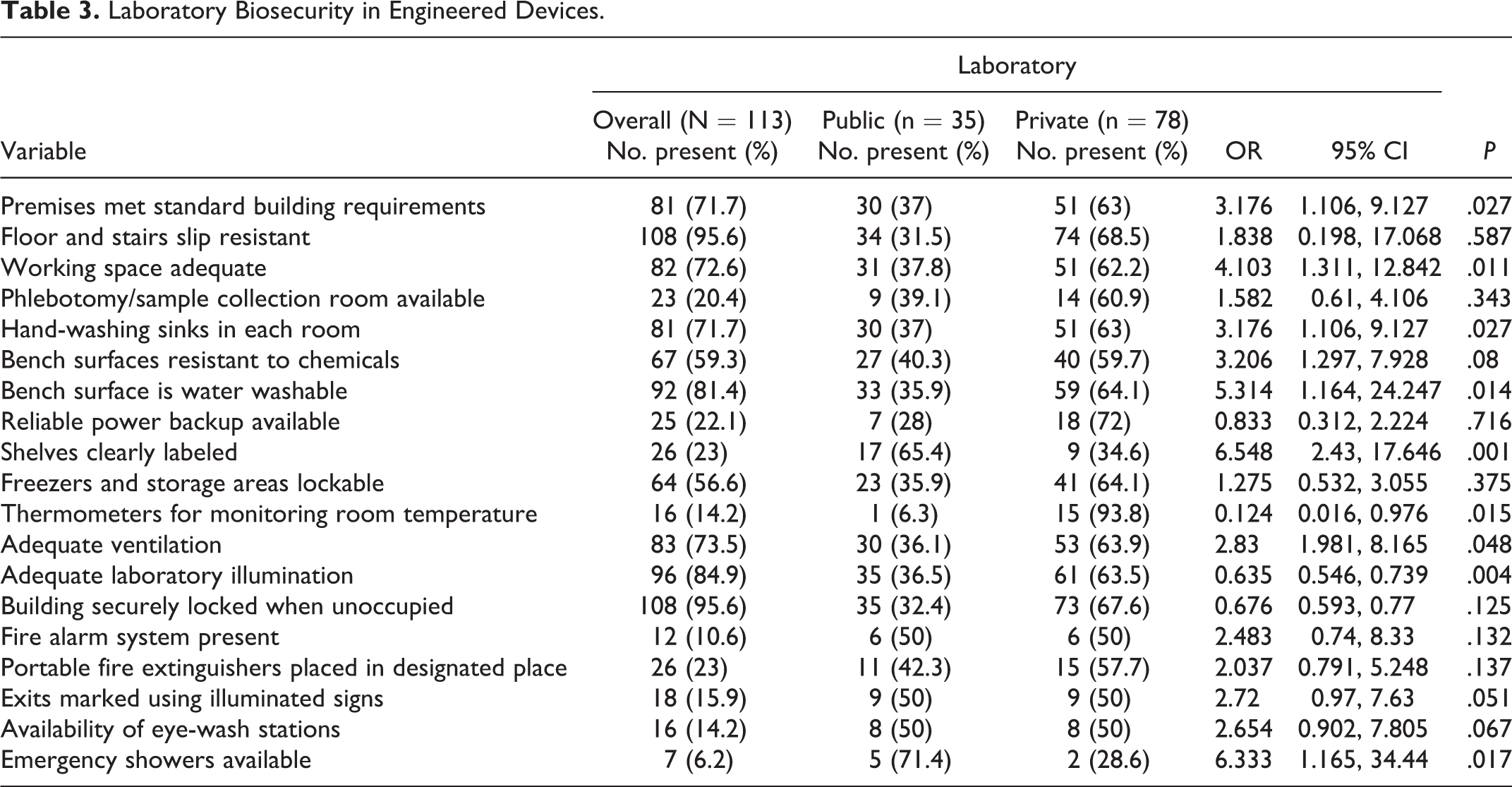
Comparing public and private medical laboratories, private laboratories were more likely than their public counterparts to have premises meeting the building standard requirement (OR 3.2, 95% CI [1.1, 9.127],
Administrative Control of Biosafety
As described in Table 4, the issuance of appropriate vaccination to staff was observed in 60.2% of all medical laboratories surveyed, and 86.7% had a biosafety officer. Availability and training of staff on equipment SOPs was seen in 48.7% of the laboratories surveyed, while availability and training of staff on Laboratory Information Management Systems was seen in 18.6% of the laboratories.
Laboratory Administrative Control of Biosafety.
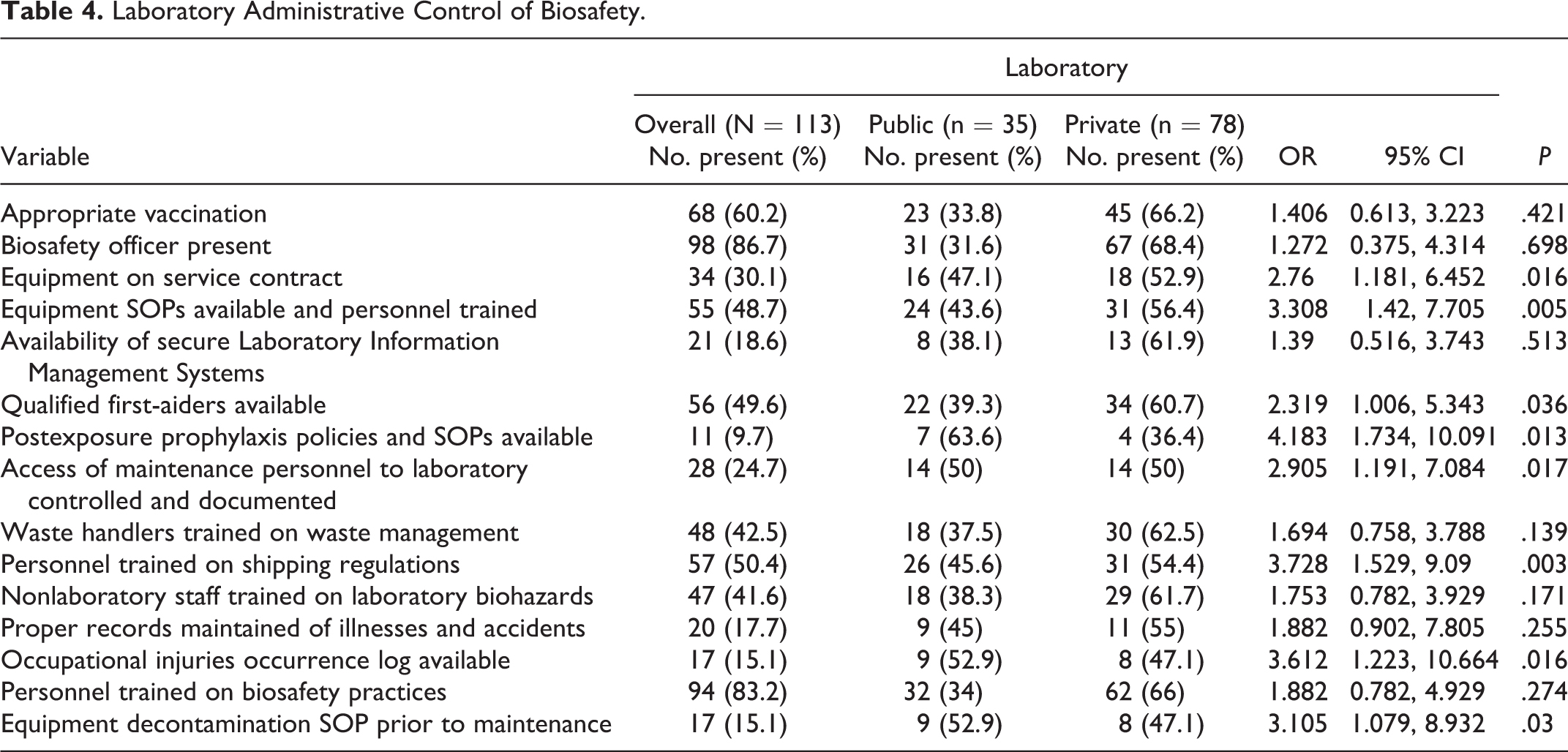
The presence of qualified first-aiders trained in laboratory emergencies such as accidental ingestion of poisons and infectious materials, the existence of postexposure prophylaxis policies SOPs posted in areas accessible to all staffs, documented access control to rooms with valuable material/equipment, waste handlers trained on waste management, personnel trained on shipping regulations, nonlaboratory staff trained on laboratory biohazards, maintenance and availability of proper records of illnesses and accidents, availability of an occupational injuries occurrence log, personnel trained on biosafety practices, and the presence of equipment decontamination SOPs prior to maintenance were found in 49.6%, 9.7%, 24.7%, 42.5%, 50.4%, 41.6%, 17.7%, 15.1%, 83.2%, and 15.1%, respectively, of all laboratories surveyed (Table 4).
Public laboratories were more likely than their private counterparts to have SOPs on PEP policies (OR 4.183, 95% CI [1.734, 10.091],
On the other hand, the private laboratories were more likely than their public counterparts (Table 4) to have equipment on service contract (OR 2.76, 95% CI [1.181, 6.452],
Discussion
The medical diagnostic laboratory plays an integral role in disease diagnosis and scientific research, both of which are the foundation for disease control and prevention. 2 These facilities handle biological materials for numerous justifiable reasons including educational, scientific, medicinal, and health-related. 15 Although laboratory personnel are expected to act responsibly by avoiding exposing the community to biorisk, to follow safe working practices (biosafety) associated with practices to keep their work and materials safe and secure (biosecurity), and to follow an ethical code of conduct (bioethics), 2 the laboratory environment should also be conducive for efficient operations and must not compromise the safety of the staff or the reputation of the services rendered. 15 This necessitated Kenya, among other global nations, to develop legislative guidelines for enforcing biosafety in biological laboratories. 14 Many laboratory personnel intermittently have been exposed the Kenyan guideline—with those from private sectors trained in additional other guidelines. This study compared the compliance to biosafety and biosecurity guidelines between public and private medical diagnostic laboratories in Trans Nzoia County in western Kenya.
The survey showed that the majority of the private laboratories, apart from being manned by fewer personnel who were younger and had less than 5 years of experience in the medical diagnostic laboratory compared to public laboratories, were located in rural areas. This could raise eyebrows in the following areas: First, the rural settings are often hard to reach and may be out of the reach of the regulatory authority (Kenya Medical Laboratory Technicians and Technologists Board) charged with monitoring them for compliance to biosafety regulations in Kenya. This was affirmed in a study in Nigeria by Abdulraheem et al. 16 Second, the employment of younger and fewer personnel and relatively inexperienced personnel often implies lack of allocated time for proper training in the biosafety and biosecurity issues; this was evidenced by the fact that only 37.2% who attended additional training on laboratory safety issues in the past 2 years. Third, even if the laboratories met the biosafety and biosecurity aspects, compliance by personnel could be a challenge. The employment of younger and fewer laboratory personnel and the concomitant lack of an appointed biosafety officer have been reported in Nigeria 10 and Sudan, 17 as well as in Bangkok. 18
Slightly over half (59.3%) of the laboratories examined handled valuable biological material, 53.1% had records of all incoming infectious materials, and only 15.1% of them unpacked specimens in a biosafety cabinet with care and attention to possible breakage and leakage. Personnel wearing gloves and other protective clothing during the unpacking of specimens was seen in 50.4% of the laboratories. Generally poor to average waste segregation and proper waste collection and disposal were evident in this survey. Should our observation be a course for worry? It is generally recognized that any clinical specimen may contain infectious agents regardless of its source; therefore, the potential for worker exposure may actually be greater from the more numerous routine specimens, which would not be identified with a hazard warning label.19,20 Universal precautions must be followed at all times when handling all clinical specimens whether labeled or unlabeled. 20 This poor to average compliance rate with personal protective behaviors among personnel handling clinical samples has also been reported in Sudan 17 and Canada. 21 In this survey, more than three-quarters of all the laboratories attained compliance for biosecurity and biosafety engineered devices with regards to the following attributes: premises meeting building standard requirement, availability of nonslip floors and stairs, adequate working space, availability of a separate phlebotomy room, hand-washing sinks, chemical-resistant working benches, and washable working benches. Comparing public and private medical laboratories, private laboratories scored higher with regards to having premises meeting the building standard requirement and having adequate working space, hand-washing sinks, chemical-resistant working benches, and water-washable working benches. The laboratories scored poorly regarding availability of lockable freezer and storage rooms, adequate ventilation, availability of fire alarm systems, and the presence of illuminated and marked exits. Even lower levels of biosafety precautions were reported in Sudan. 17 On the contrary, in Nigeria Oladeinde et al 10 reported that public diagnostic laboratories were more likely to have separate and restricted microbiological isolation units (equivalent to the phlebotomy room in our setup) and a hand-washing sink. Compliance to biosecurity and biosafety aspects in the laboratories is fundamental to prevent unintentional exposure and the accidental release or loss of pathogens and toxins.2,5
Working in environments with pathogens is a potential hazard for scientists and health care workers. 22 Infection with hepatitis B virus (HBV) has been generally recognized as key among health and laboratory personnel; however, this is currently on the decline among this population largely due to widespread immunization with the hepatitis B vaccine. 23 In this study, issuance of appropriate vaccination was observed in 60.2% of all medical laboratories surveyed. In Nigeria only 10% of the laboratories had an HBV vaccination program for all laboratory workers. 10 In the same country, another study showed more than 90% of laboratory health workers were not immunized against HBV. 24 This trend of a poor HBV vaccination program is an indication that the laboratory workers in this region are at increased risk of acquiring occupation-related HBV infection. The very low presence of qualified first-aiders trained on laboratory emergencies, the near nonexistence of postexposure prophylaxis policy SOPs, the lack of waste handlers trained on waste management, and the low number of nonlaboratory staff trained on laboratory biohazards in all laboratories surveyed further raise biosafety and biosecurity issues in this region of Kenya. Kaufman et al 25 and Kotwal and Taneja 13 noted the importance of documented SOPs in not only ensuring the consistency of test performance and results but also in following up SOPs with a complete strategy for increasing and sustaining safe laboratory behavior.
Although 86.7% of all the laboratories reported having a biosafety officer, it was unclear if these were biosafety officers per se and if they had undergone appropriate training because of the poor biosafety outcomes. Biosafety officers are expected to develop safety policies and procedures and orientation and training programs for all laboratory staff in biosafety and to ensure safety policies compliance among others. 25 Kaufman et al 25 reported no biosafety officer was found in the Nigerian laboratories surveyed, which was linked to the many administrative lapses on biosafety issues observed and reported.
One of the major strengths of this study was the ability to contribute to the wealth of knowledge by showing the compliance rates to biosafety and biosecurity in the entire county of western Kenya. However, some of the limitations to our assessment need to be pointed out, too. First, the cross-sectional nature of our study allowed us to describe only the compliance rates to biosafety and not a causal conclusion. Such outcomes can be confirmed in a longitudinal study. Second, this study was confined to the use of the Kenyan biosafety and biosecurity policy questionnaire and checklist report, which could be different from those of WHO and the CDC. 26 Because WHO and the CDC questionnaire and checklist could have been used during the initial training in Kenya, we may not have captured the true picture of biosafety and biosecurity compliance. These limitations notwithstanding, our findings indicate an overall poor biorisk score attributed to fire prevention and protection, administrative control, and personal protective devices. Enhanced training on biosafety and biosecurity is likely to improve compliance with standard biorisk codes in western Kenya.
Footnotes
Acknowledgments
We would like to thank the study participants and all the heads and/or owners of medical diagnostic laboratories surveyed. We also acknowledge Kitale County Hospital Medical Superintendent Emmanuel Wanjala and Medical Laboratory Officers Godfrey Sande and Everlyne Kweyu.
Author Contributions
HMO, CK, and SM conceived the study. HMO and MON supervised interviews, analyzed the data, and prepared the manuscript. CK, SM, and MON provided guidance and mentorship during the implementation of the study. All authors reviewed and approved the final manuscript.
Approvals
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
