Abstract
Mycobacterium bovis (M. bovis) is the etiologic agent of tuberculosis in mammals, including humans. The seriousness of disease and low infective dose require that the agent be handled under biosafety level 3 conditions. Many experimental protocols include histologic examination of infected tissues, which are often performed in biosafety level 2/1 laboratories, thus requiring inactivation of M. bovis within the tissues, preferably during tissue fixation. Fixation with a 10% neutral buffered formalin (3.7% formaldehyde) solution for 24 hours inactivated M. bovis in situ. In contrast, zinc fixative did not inactivate M. bovis in situ. Fixation with 10% neutral buffered formalin rendered tissues safe to transition to biosafety level 2/1 laboratories for histopathologic examination.
Mycobacterium bovis is a zoonotic infectious agent that causes tuberculosis in animals and humans. Viable M. bovis is handled under biosafety level 3 (BSL-3) conditions, in a manner similar to Mycobacterium tuberculosis (M. tuberculosis). For convenience and to facilitate a higher throughput, many histologic and/or pathologic procedures are performed under BSL-2/BSL-1 laboratories. Transfer of biological material from a BSL-3 to lower containment levels requires that M. bovis be rendered noninfectious.
The scientific literature has reported inconsistent results of mycobacteria inactivation by formalin and similar agents, such as embalming fluid. Comparison between studies is complicated by the fact that the researchers used solutions that contained varying formulations of formalin, different inactivation/fixation times, and inactivation in different tissues.
One of the first studies that attempted to culture M. tuberculosis from formalin-fixed lung tissue reported that M. tuberculosis could not be cultured from tissues that had been fixed in formalin for between 72 hours and 9 years. 1 No growth of M. tuberculosis occurred in the 5 cases that were examined. A later study investigated 138 cases of tuberculosis in South African miners. 2 Entire lungs were gravity inflated with 10% formalin, immersed in 10% formalin, and stored for 2 weeks to several months. Upon receipt at the laboratory, lungs were immersed in 10 L of 10% formalin for 10 days. Viable mycobacteria were cultured from 12 lungs (9%). Note that fixation of the entire lung, as done in this study by Gerston et al, leaves doubts about the degree of fixation of deeper areas of the lung from which samples were obtained. In contrast to the lung fixation reported by Gerston et al, Bartos et al reported that M. tuberculosis could not be cultured from human cadavers fixed with formalin following “standard” embalming practice. 3
In a more recent study based on experimentally infected guinea pigs, a multidrug-resistant strain of M. tuberculosis resisted inactivation by 10% formalin fixation for 6 months while multidrug-susceptible strains were completely inactivated. Pretreatment of tissues for 2 hours in 75% ethanol prior to formalin fixation resulted in inactivation of resistant and susceptible strains. 4
A widely held belief is that 10% formalin, the most commonly used tissue fixative, inactivates microorganisms. The Clinical and Laboratory Standards Institute guidelines state, “Ten percent formalin (3.7% formaldehyde) present in at least ten times the volume of tissues, which has been properly sectioned and adequately permeated, will inactivate all important infectious agents except the agent of Creutzfeldt-Jakob disease.” 5 The caveats mentioned are notable. The tissue was “properly sectioned and adequately permeated” by the formalin. Despite the commonly held belief, there is some ambiguity in the scientific literature regarding the tuberculocidal properties of formalin in tissues infected with members of the M. tuberculosis complex.
Correct preservation of the tissue structure as well as the inactivation of M. bovis is critical for downstream histopathologic applications. Formalin-based fixatives are known to preserve the morphologic features of the tissues; however, they destroy some surface epitopes needed for immunohistochemical analysis. Zinc-based fixatives have been reported to be superior to formalin-based fixatives at preserving morphological as well as epitope structures.6,7 Inactivation of M. bovis nor M. tuberculosis by zinc fixatives was not reported.
The vast array of tissue size, fixation protocols, and length of fixation renders published results difficult to extrapolate to routine laboratory procedures used in veterinary mycobacterial laboratories. We sought to determine if formalin and zinc fixatives will inactivate M. bovis under routine laboratory conditions.
Methods
Formalin Fixation of Fresh Tissues
Holstein calves were infected with M. bovis via the aerosol method. 8 At necropsy, tissue was collected from the medial retropharyngeal and mediastinal lymph nodes and lung. Each tissue was cut into slices no more than 5 mm thick. Three adjacent slices were treated as follows. One slice was cultured for the presence of M. bovis as described here. Two slices were submerged in fresh 10% neutral buffered formalin (CB Formalin, Anatech LTD, Battle Creek, MI; prepared according to the manufacturer’s directions) at a ratio of at least 10:1 (formalin:tissue by volume) for 24 hours at room temperature on a rocking platform. At the conclusion of the 24 hours, 1 slice was removed and processed for culture of M. bovis. The second slice was placed in 70% ethanol for an additional 24 hours at room temperature. At the conclusion of the ethanol treatment, the slice was cultured for M. bovis as described.
Zinc Fixation of Fresh Tissues
Two strains of M. bovis were used to challenge Holstein steers with 1 × 104 colony-forming units via the aerosol method. 9 Calves were euthanized 4 months postinfection. Adjacent tissues slices approximately 5 mm thick were cut from various affected lymph nodes and lung in such a way as to ensure that each slice contained gross lesions. Following slicing, tissue slices were immediately immersed in freshly made zinc fixative (0.1M Tris buffer with 0.05% w/v calcium acetate, 0.5% w/v zinc acetate, and 0.5% w/v zinc chloride [pH 7-7.4]; as described by González et al 10 ) at a ratio of at least 10:1 (zinc fixative:tissue by volume) for 24, 48, or 72 hours. At the completion of the fixation period, tissue samples were homogenized and cultured as described.
Culture of M. bovis from Fixed and Unfixed Tissues
Tissue pieces were cultured with a standard laboratory procedure. 11 Briefly, pieces were independently added to blender jars with 25 mL of phenol red broth and blended for 30 seconds. Homogenized tissues were transferred to 50-mL centrifuge tubes and centrifuged for 15 minutes at 3300 × g. The supernatant was discarded. With a cotton-tipped swab, the solution was streaked onto 2 plates with Middlebrooks 7H11 plus antibiotics. M. bovis BCG (1/20th volume) was added to the remaining solution, mixed, and then streaked onto 2 additional plates with a cotton-tipped swab. BCG was added to the fixed tissue samples to ensure that the inactivation reagent had been properly removed from the tissue samples prior to plating. Plates were incubated at 37°C for 8 weeks. Colonies were counted and confirmed to be M. bovis by polymerase chain reaction. 12
Results
Formalin Fixation
Tissues containing gross lesions were used in this analysis. Adjacent tissue slices were cut to ensure that each slice contained gross lesions and were approximately 5 mm thick. Each was randomly assigned to 1 of 3 groups; unfixed, formalin fixed, and formalin plus ethanol fixation.
M. bovis was not cultured from any tissues that were fixed in neutral buffered formalin for 24 hours, nor was M. bovis cultured in tissue that spent an additional 24-hour incubation in 70% ethanol (Table 1). To confirm that the tissues contained M. bovis, unfixed tissue samples were cultured. The majority of the unfixed tissue samples yielded between 11 and >200 (to numerous to count) colony-forming units, thus confirming that the viable M. bovis was present in the tissue. The process was repeated 2 additional times with animals necropsied on different days, with similar results. Addition of a small volume of M. bovis BCG to the homogenized tissues confirmed that active formalin had been removed from the tissue (Table 1), further confirming that the negative growth result was a consequence of inactivation rather than the continued presence of formalin in the homogenized tissue sample. Contamination of the cultures by nonmycobacteria did not occur.
Inactivation of Mycobacterium bovis by 10% Neutral Buffered Formalin and Ethanol.
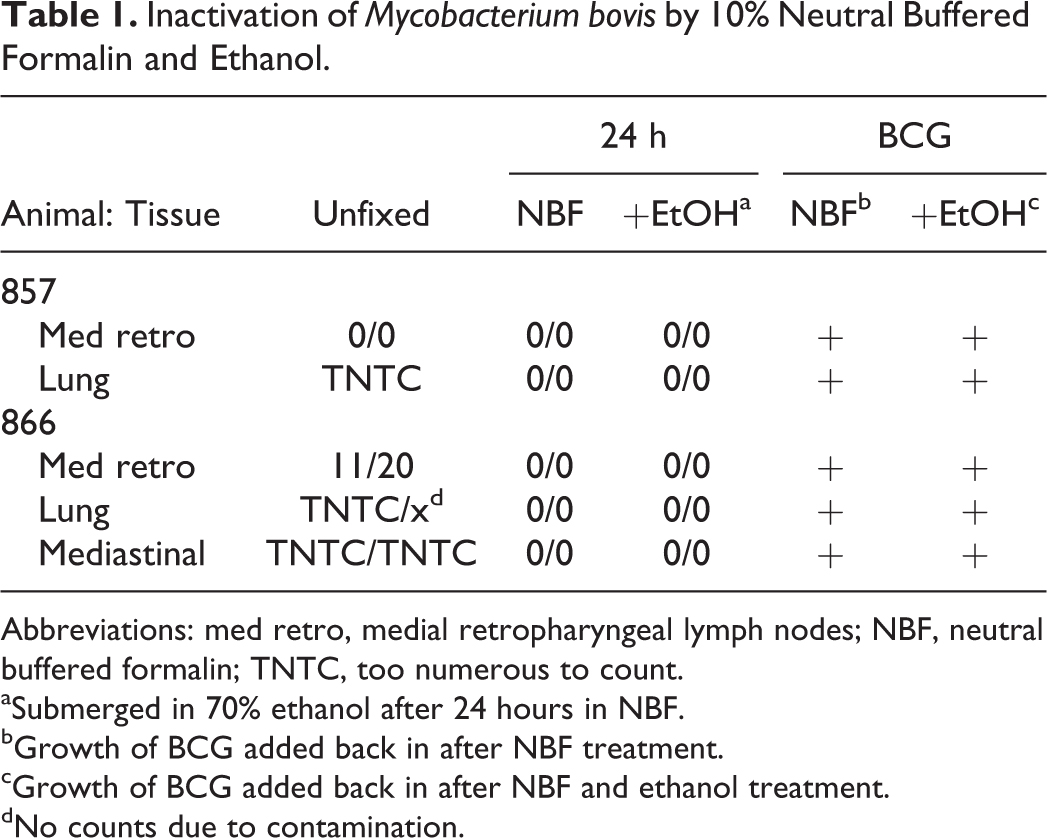
Abbreviations: med retro, medial retropharyngeal lymph nodes; NBF, neutral buffered formalin; TNTC, too numerous to count.
Submerged in 70% ethanol after 24 hours in NBF.
Growth of BCG added back in after NBF treatment.
Growth of BCG added back in after NBF and ethanol treatment.
No counts due to contamination.
Zinc Fixation
Tissues containing lesions sliced approximately 5 mm thick and immediately fixed with freshly prepared zinc fixative were evaluated. Tissue was fixed for 24 hours and, when sufficient tissue was available, 48 and 72 hours. Following fixation, tissues were homogenized and cultured as described in the Methods section. Zinc fixative did not inactivate M. bovis in situ even after 72-hour fixation (Table 2). Cultures were also contaminated with nonmycobacteria, suggesting that the zinc fixative tested here is not bactericidal.
Inactivation of Mycobacterium bovis by Zinc Fixative.
Abbreviation: ND, not done.
Bacterial growth other than M. bovis on cultures without antibiotics (relative number of contaminating colonies: +, 1 or 2; ++, few; +++ heavy).
Discussion
Tissues from M. bovis-infected animals must be handled under BSL-3 conditions. The primary purpose of fixing tissues is to preserve the tissue structure and to enable histopathologic examination of the tissues. However, fixation that kills the M. bovis in situ has a definitive advantage because the tissues can enter the normal histologic preparation stream at most institutions that operate under BSL-2/1 conditions. Historical data had reported inactivation and failure of inactivation of M. tuberculosis complex organisms by formaldehyde-containing solutions. The diversity of tissue type, size, and length of fixation makes comparing these studies difficult. Here we sought to determine if M. bovis is inactivated in situ under the routine fixation protocols in the modern histopathology laboratory.
Fixation of M. bovis-containing tissues with zinc fixative did not render the M. bovis inactivated. The failure to inactivate M. bovis requires that these tissues continue to be handled under BSL-3 conditions. Any benefit to using zinc fixation will need to be balanced by the need to perform standard cutting, fixing, and staining procedures under BSL-3 conditions.
Inactivation of M. bovis in tissues occurred when treated with neutral buffered formalin for at least 24 hours. The second incubation in ethanol for 24 hours was performed to aid in the fixation and preservation of the tissues; however, it likely aided the inactivation of M. bovis within tissues. Note that tissues used in this study were approximately 0.5 cm thick. This was done to ensure that formalin could completely permeate the tissue and affect the M. bovis present. 13 Attempting to fix tissue thicker than 0.5 cm may produce more variable results because complete penetration of the formalin is required. Second, the tissues were fixed in at least 10 volumes of fixative to 1 volume of tissue, again, to ensure that reactive formalin was in excess, thus further ensuring that the M. bovis present could be inactivated. These data confirm that tissues fixed with 10% neutral buffered formalin can be moved to the BSL-2/1 laboratory space for continued processing.
Conclusion
A 10% neutral buffered formalin solution will inactivate M. bovis within infected tissues within 24 hours when the tissue slices are no more than 0.5 cm thick. In contrast to the formalin data, zinc-based fixatives did not inactivate M. bovis even after 72 hours.
Footnotes
Authors’ Note
All animal studies were conducted after receiving approval by the Institutional Animal Care and Use Committee at the National Animal Disease Center. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendations or endorsement by the US Department of Agriculture, which is an equal opportunity provider and employer.
Acknowledgments
We thank Shelly Zimmerman and Jayne Wiarda for their expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by the US Department of Agriculture CRIS Project 5030-32000-222.
