Abstract
Biosafety and biorisk management are emerging areas of safety that are not well understood by the general public. With recent headlines questioning the safety practices and procedures at federal agencies, educating the public on what steps are taken to encourage a safe and transparent working environment in high-containment laboratories is important. This article describes for the ABSA International biosafety community, as well as for the general public, the biorisk program at the National Institutes of Health and discusses the steps taken to train researchers, report injuries, and encourage a culture of safety within the laboratories, rather than a culture of fear. It attempts to dispel the provocative headlines used by the media, through education on the process and procedures used to protect the researchers, as well as the public, when handling agents whose manipulation and propagation are restricted to high-containment laboratories. In doing so, the authors seek to better educate the public and help to create better awareness of the work that occurs in these facilities.
Recent reports1,2 of safety lapses at federal laboratories and other research institutions have brought biosafety to the forefront as a national issue. Articles describing releases of air from laboratories working with high-risk agents, as well as reports of failures to properly inactivate contaminated material, have generated intense media focus that has led to increased public concern over the risks of potential exposures to laboratory workers, such as accidental punctures from contaminated needles or contaminated broken glass. In addition, these articles have heightened concerns about the possibility of releases that could affect the residents of the communities near high- and maximum-containment laboratories. Biosafety concepts are not well known or understood by the general public, and this heightened scrutiny is requiring institutions and organizations to defend procedures and policies that are commonplace in the biosafety industry. With such incendiary headlines as “Universities, Feds Fight to Keep Lab Failings Secret,” 2 the media’s attempt to inform the public can actually serve to incite fear and generate misconceptions regarding the need for and intent of high-containment research. As biosafety professionals, our responsibility is to inform the public about the need to study potentially dangerous pathogens and to educate it on the measures that are taken every day to safeguard the laboratory staff, the community, and the nation. Along with a better appreciation of these safety measures comes an understanding of how biosafety professionals can advance their research mission and still maintain a safe working environment and community.
Laboratories in general contain a wide assortment of hazards that, without proper staff training and control measures, can present an environment for potential injury from more than just biological hazards. Chemical, radiologic, and physical hazards are all causes for concern when working in the laboratory. A fundamental step in mitigating these hazards is conducting a proper risk assessment of the agents, chemicals, and procedures involved in the work being planned. Having a biosafety professional and/or other safety and health professionals meet with the principal investigator (PI) to discuss the materials and practices of the proposed work is essential. These discussions take into consideration the techniques to be performed, the level of training required, as well as additional engineering controls, equipment, or supplies necessary to perform the work safely. This article provides an overview of the National Institutes of Health’s (NIH’s) approach to managing risk in laboratories working with biological materials. The authors hope that this information will assist in providing not only the biosafety community, but also the public, with a more realistic perspective on the work performed and will remove the need for the dramatic verbiage that is sometimes used to characterize the activities associated with these high- and maximum-containment laboratories.
Protocol Registration and Review
At the NIH, work with human, animal, and plant pathogens, as well as biological toxins and recombinant nucleic acids, is registered by the PI with the Division of Occupational Health and Safety (DOHS) and the Institutional Biosafety Committee (IBC). This is done through an electronic registration and workflow management system. All institutes or centers within the NIH performing research adhere to the following procedures. The submitted protocols are routed to the NIH Biosafety officer (BSO) for review to determine that all required information is correct and complete, that animal study proposals and vector maps are attached, and that the first level of dual-use research of concern (DURC) screening is performed. This information is reviewed by the NIH BSO, who conducts a preliminary risk assessment based on the information submitted. BSO comments are captured in the system and are available to other reviewers (eg, IBC reviewers and safety specialists). If the BSO determines that additional information or clarification is required, the electronic submission is routed back to the PI. Upon receiving all required information and electronic signatures, the BSO forwards the submission to all IBC members for review and discussion at a monthly IBC meeting. An electronic signature is required from the PI to complete submission. This e-signature affirms that all research personnel participating in the research have been apprised of the hazards that may be associated with the protocol and have received the required safety training. Also, each individual participating in the protocol must e-sign an affirmation that he or she has been apprised of the hazards and has taken the appropriate training. Submissions are amended, as changes to protocols occur and are reviewed at least annually.
Integration of Occupational Medical Services
Occupational medical services (OMS) must be involved in the preliminary preparations for infectious disease research. This involvement is important in 2 ways. First, OMS can develop agent-specific surveillance monitoring and postexposure medical response plans and provide one-on-one counseling and training to employees. Second, the culture of “accident and injury prevention” is reinforced within the NIH research community. Operationally, NIH uses the aforementioned electronic registration and workflow management system to facilitate this. A secure interface exists between the electronic registration system and the OMS (part of DOHS) electronic records system to obtain information regarding which personnel are working with human pathogens or other potentially hazardous biological materials. Using this information, OMS enrolls personnel in the appropriate medical response or surveillance program, conducts a targeted medical history and evaluation, ensures that appropriate immunizations are available, and reinforces exposure response protocols among other medical services. Should a potential exposure occur, the OMS health care providers and the DOHS safety specialist work together to assess the actual risk to the employee from the potential or real exposure and ensure that prophylactic measures are taken immediately, if necessary. During the past 23 years, the median time from injury/exposure to the delivery of postexposure prophylaxis has been 90 minutes. OMS health care providers are available to respond 24 hours, 7 days per week, 365 days a year.
IBC’s Role and Need for Transparency
The NIH IBC meets monthly to review recombinant nucleic acid protocols, as well as other nonrecombinant material, that have been submitted by researchers the previous month. In addition to the review of nonexempt nucleic acid research, the IBC is responsible for the review of all protocols involving human pathogens (those organisms requiring containment at biosafety level 2 [BSL-2] or above). All work with organisms performed at BSL-3 and above is reviewed in full committee by the IBC. Due to the number of infectious disease agents used at the NIH and the number of protocols requiring review, the review and preliminary approval of those protocols utilizing agents handled at BSL-2 or below have been delegated to the NIH BSO. The full committee provides final review and approval at the next IBC meeting. Additionally, the NIH IBC provides advice and guidance to the DOHS and the NIH Office of Intramural Research regarding NIH-wide biosafety policies and procedures.
As mandated by the NIH guidelines, 3 the IBC is composed of members who represent the various research interests across the NIH with the expertise needed to review the types of proposals that come before the committee. Senior scientists, nondoctoral scientists, veterinarians, senior research clinicians, and administrators serve the NIH on this committee. The IBC also includes community members who represent the interests of the general public and communities that surround the NIH. The IBC community members participate in all matters before the IBC. IBC meetings are not held unless at least 1 of 3 community members is present. All meetings are open to the general public. In the event of the need to discuss protected patient information or other proprietary/confidential matters during a meeting, the IBC holds an executive session after other matters before the committee have been concluded. Noncommittee attendees are then excused from the meeting. All matters discussed and decisions rendered in the executive session are recorded in the meeting minutes protecting sensitive, patient, or proprietary information, and general meeting minutes are made available to the public. Note that in the past 30 years, executive sessions have been rarely held, demonstrating the NIH’s commitment to transparency in the review of all publically funded research.
Multitiered Dual-Use Review
The NIH has incorporated a DURC screening process based on a multitiered approach. DURC is any “life science research that … can be reasonably anticipated to provide knowledge, information, products or technologies that could be directly misapplied to pose a significant threat with broad potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, material or national security.” 4 This screening is performed for all intramural research involving infectious agents and for recombinant or synthetic nucleic acid research.
This article provides a brief overview of the NIH screening process. Initial screening begins with the PI and is performed as part of the electronic registration approval process by the IBC. Answers to a series of dual-use screening questions are provided by the PI and reviewed by the NIH BSO and the IBC. Should it be determined that the proposed research may result in dual-use potential, the IBC requests a mitigation plan from the PI. If, upon review of the mitigation plan, all potential concerns are adequately addressed, the IBC will approve the plan, and the proposed work may proceed provided that all other requirements are met. Each registration is rescreened annually for dual-use potential. This is to ensure that changes in protocols and/or preliminary findings or data collected do not change the dual-use potential or require updating of a mitigation plan. Rescreening also occurs when amendments to protocols are electronically submitted. Electronic reminders to rescreen protocols are sent to PIs annually. If research is suspected of being DURC, the work is reviewed by the Dual Use Committee, composed of NIH senior managers and scientists to determine what if any action is required. The IBC, PIs, and institute scientific directors may also refer research protocols to the Dual Use Committee for final dual-use determination. Review of positive answers to screening questions or elevated review of a mitigation plan does not imply that the work is DURC but rather that potential dual use–related issues have been considered and addressed. In addition, NIH policy is that all manuscripts be screened for dual-use potential during the publication clearance process. These procedures are intended to ensure that dual-use potential is monitored over the life of the research project.
Communications Between the IBC and the Animal Care and Use Committees
Protocols involving the use of animals are also reviewed by the institute’s Animal Care and Use Committee (ACUC). The NIH currently has 18 ACUCs that collectively perform the duties of the Institutional Animal Care and Use Committee. Each institute’s ACUC is composed of staff from within the institute, including nonscientific personnel. Each committee also has 1 safety specialist, who is a voting member, and 1 community member, who represents the interests of the general public. Each ACUC meets monthly and provides an open forum to raise concerns and discuss questions about the research. In addition to animal welfare matters, the ACUCs review new research proposals and the renewal of current proposals to ensure that infectious agents and radiologic and chemical hazards involved in the work are identified and properly assessed from a safety and health perspective. ACUC approval is required before beginning research. Review of these protocols is another avenue for capturing the use of agents that PIs may not realize require registration. ACUCs generally require that IBC registration documents accompany animal study proposal submissions. Identification of potential hazards prior to the start of work helps to protect employees from hazards of which they may not be aware and ensures that proper safeguards have been identified and are in place.
Accident/Incident Reporting and Investigation
Even with the preventive measures taken to mitigate hazards, no environment is free of risk; accidents happen. It is crucial that staff—whether injured, exposed, or potentially exposed—promptly receive the appropriate medical treatment and that these incidents are prevented from reoccurring. To help reduce the risk of reoccurrence, the NIH has worked to establish a culture among federal and nonfederal staff that encourages them to report all injuries, potential exposures, and illnesses to the OMS.
In all cases, injuries, incidents, and potential exposures reported to OMS are entered into the electronic health record system. This system generates a case report that details (1) the nature and location of the injury or incident that may have resulted in a real or potential exposure and (2) the demographic information about the involved employee, including the institute where he or she is employed. The case report also provides a brief description of the incident as described to the health care provider by the patient. Once entered into the system, a notification is sent to the safety specialist assigned to the institute of the patient.
The DOHS safety specialist follows up with an accident/injury investigation focused on root cause analysis and includes interviews with all involved parties, supervisors, and witnesses, if any. In some instances, this may involve contact studies, depending on circumstances and the agent involved. Details—such as the type of personal protective equipment worn; the use of infectious, chemical, or radiological materials; and the types of laboratory equipment or techniques employed at the time of the incident—can aid in determining the cause of the incident and in turn prevent future incidents. In particular, the safety specialist assesses the level of training that the individual received on specific laboratory practices prior to the incident. Accident and injury statistics are reviewed on a quarterly basis to allow for reallocation of resources to address emerging or emergent risks. Such resources include the development of hands-on training classes, as well as safety awareness campaigns and quick-reference safety guides, in addition to research into safer products and to distribution of these findings to the NIH community at large. Figure 1 shows the types of accidents/injuries that occurred in research laboratories or animal care facilities from October 1, 2013, through September 30, 2014. Less than 4% (28 of 769) of these incidents involved potential exposure to a biological agent. A closer review of the data showed that no laboratory-acquired infections occurred during this period.
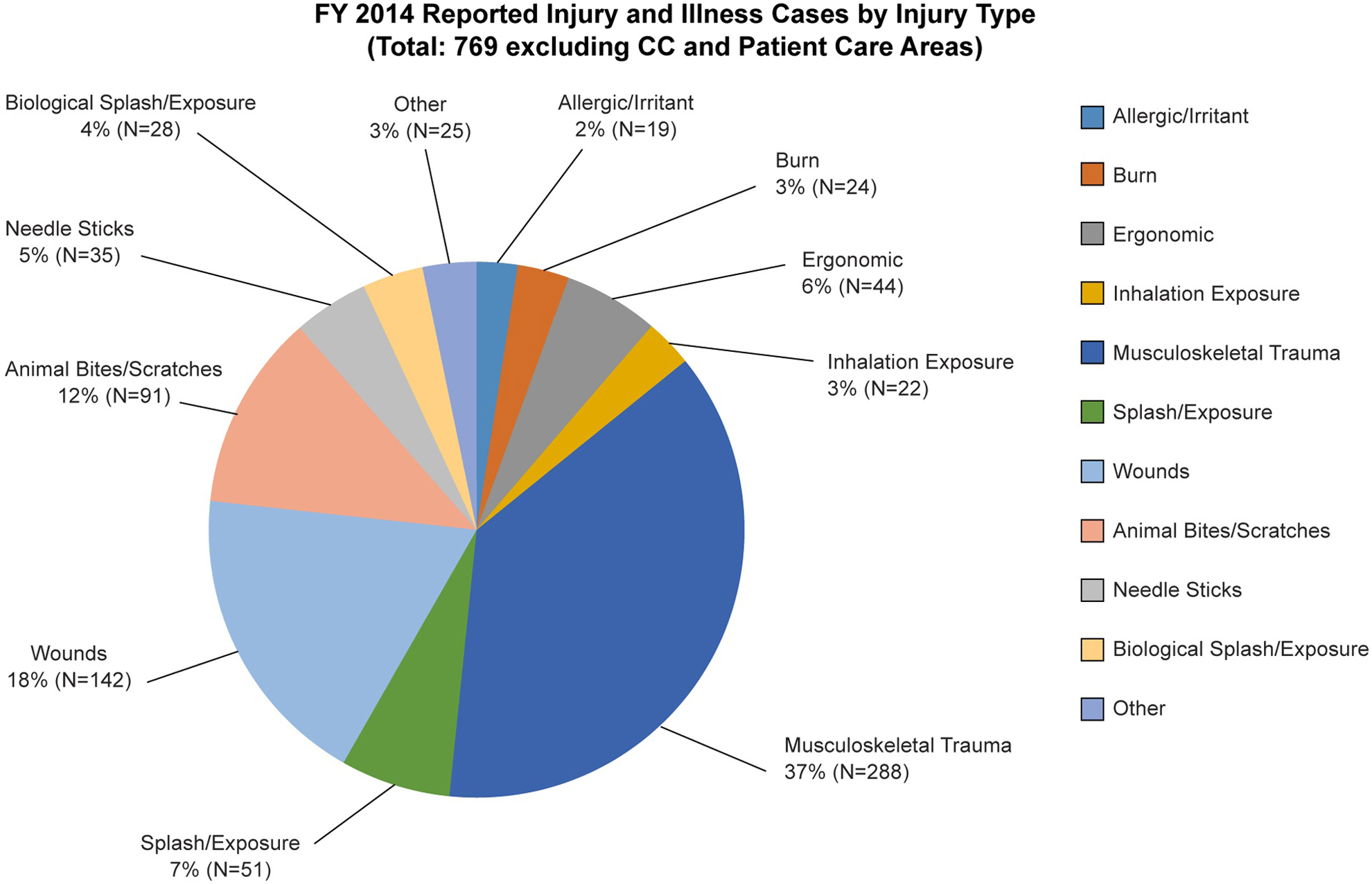
Review of fiscal year 2014 accident/injury statistics and trends reveals that <4% of the total incidents recorded involved exposure to biological agents. Other injuries or incidents involving concomitant exposures to biological agents are captured as biological splashes and exposures. Percentages are rounded to nearest whole number for ease of display. Total reported incidents (N = 769) exclude incidents from the National Institutes of Health (NIH) Clinical Center (CC) and other patient care areas. Data were obtained from the NIH accident/injury recording system as of October 28, 2014.
Safety and Health Training and Information
Literature searches into laboratory-acquired parasitic infections that have occurred at other research locations have shown that “some of the accidents that resulted in laboratory-acquired infections were directly linked to poor laboratory practices.” 5 Examples of poor laboratory practices included not adhering to laboratory standard operating procedures, inappropriate or insufficient personal protective equipment for laboratory practices, and lack of proper training for new employees. To promote better laboratory practices, the NIH requires that employees take initial laboratory safety training and annual refresher training. The training courses are role based and are associated with the type of hazards that may be encountered in the laboratory. To accommodate a variety of learning styles and course content, the NIH offers lecture, online, and hands-on laboratory classroom training courses. NIH policy places the responsibility for ensuring completion of mandatory laboratory safety training with the supervisor. In a large research institution such as the NIH—which has approximately 1200 PIs as well as thousands of postdoctoral fellows, students, and visiting scientists—tracking training compliance can be problematic. To address this issue, a safety training registration and tracking tool is used to assign role-based training to each laboratory member, track training completion, and send reminders to complete refresher training. This enables supervisors, safety committees, and safety and health staff to track an employee’s training compliance or that of a work group. Figure 2 shows the increased number of personnel completing required safety training since implementation of the safety training registration and tracking tool.
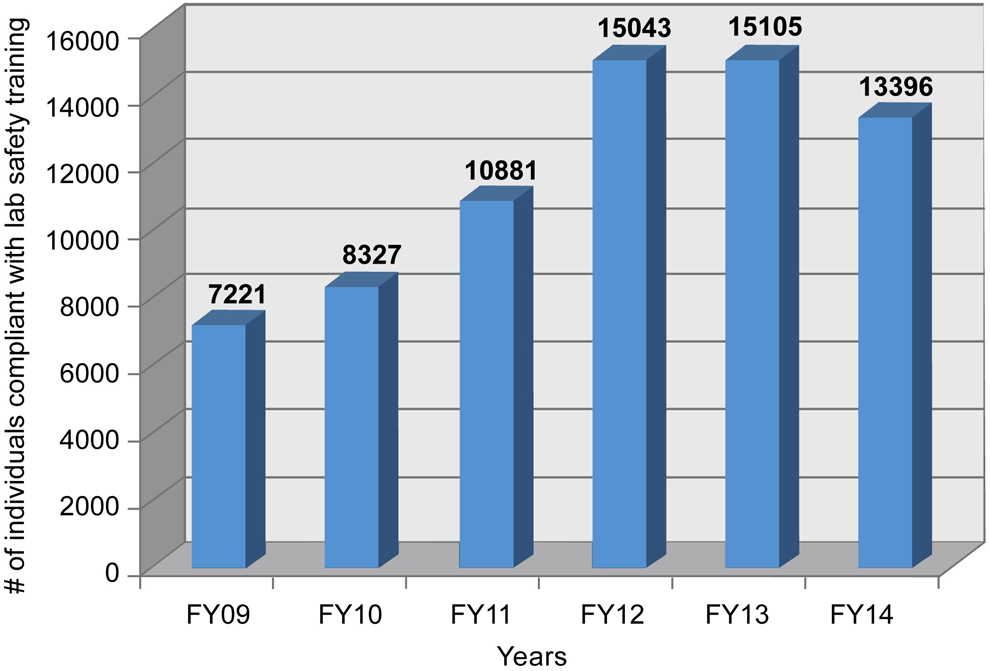
Since instituting the National Institutes of Health Training Registration and Tracking System in 2009, compliance with mandatory laboratory safety training requirements has risen dramatically. FY, fiscal year.
Information on safe work practices for handling chemical or biological materials is also available in easy-to-use wall calendars that are displayed in laboratories across the NIH. The NIH “1, 2, 3” poster, illustrating procedures to be followed should an employee sustain a laboratory exposure, is posted in all laboratories where infectious agents are used (Figure 3). By providing these training and informational resources, the NIH is constantly reinforcing the idea that “safe science is good science.”

Emergency response posters for exposure to infectious materials are posted in all laboratories where infectious materials are handled.
Focused and specialized one-on-one training is provided in response to incidents or accidents. Once the cause of an incident or injury has been determined, the safety and health professional will engage the involved party in identifying additional training, tools, or equipment that could prevent future reoccurrences. While additional tools and/or equipment can be helpful, retraining on laboratory practices and procedures is the most helpful way to prevent reoccurrence. Depending on the type of injury or incident, retraining may be focused on a general overview of laboratory practices or a mentored retraining in laboratory procedures and proficiency practical exercises to demonstrate competency. The level of retraining is determined through open discussions among the laboratory manager, staff scientist or PI, the injured individual, and the safety specialist. Various factors are considered, such as the individual’s educational level and laboratory experience, the complexity of laboratory procedures, and the biological or chemical agents in use. Mentored training may last for several weeks to a month depending on the extent of retraining required. Including the affected individual when developing a corrective training plan is important to promote its effectiveness and to dispel any notion that retraining is a punishment. By dispelling the perception that reporting accidents, injuries, or potential exposures will result in punitive action, the inclusion of the affected individual in the discussion promotes reporting incidents to management. This increases awareness of laboratory procedures or agents that are associated with a higher risk. Success of corrective actions can be measured by the number of reoccurring or similar accidents that occur within a given quarter or year.
The NIH provides the community with a number of ways to report unsafe work conditions, practices, or events. Whether through direct reporting to a supervisor or a DOHS safety specialist, a call to the main DOHS office, or an entry on the anonymous reporting website, employees and community members can voice their concerns without fear of reprisal. As such, a safety culture is embraced rather than feared as a way of punishing employees; as reported, “staff have concerns about experiencing negative repercussions, either personally or more generally to the Agency, as a result of reporting incidents.” 1 Multiple avenues of reporting demonstrate a willingness by management to hear community concerns while alleviating fears associated with reporting.
Biological Laboratory Audits
Laboratories are audited routinely where research is conducted with potentially hazardous biological materials (infectious agents, nonregulated toxins, poisons, venoms, select agents and toxins, etc.). These audits, based on the BSL of the work, address >100 items covering >20 safety topic areas. Figure 4 provides an example of the laboratory safety audit tool used at the NIH and the areas covered. Survey findings are recorded electronically, and imminent hazards must be corrected immediately. Reports emailed to the PI identify items to be corrected or hazards that must be remediated. PIs are responsible for correcting deficiencies and recording corrective actions in the electronic laboratory survey system. Resolutions of all deficiencies are tracked by the safety specialist. BSL-2 laboratories are audited annually. BSL-3 laboratories are audited twice per year: one visit is announced, and one visit is unannounced. Figure 5 shows audit questions that identified the top 10 safety deficiencies found in NIH biological laboratories from October 1, 2013, through September 30, 2014. These data are from 1250 laboratory surveys conducted in 45 research buildings on the NIH Bethesda campus.

Example of National Institutes of Health biosafety level 2 safety audit survey instrument.

The top 10 safety infractions (deficiencies) found in National Institutes of Health (NIH) laboratories conducting biological research during fiscal year 2014. BBP, Bloodborne Pathogen; CHP, Chemical Hygiene Plan; ECP, Exposure Control Plan; SOP, standard operating procedure.
Summary
This review of the DOHS presents some of the strategies used by the NIH to identify hazards and mitigate risks associated with biological and infectious disease research. Available data are presented to show the effectiveness of these strategies. Given the number of laboratories conducting potentially hazardous biological research at the NIH, the percentage of potential biological exposure events is small: 3.64% of the total number of injuries, illnesses, and exposures reported. Note also, as stated earlier, that no laboratory-acquired infections were reported during the period from October 1, 2013, to September 30, 2014.
The public and the research community must be informed of risks associated with biomedical research, and the magnitude of those risks must be contextualized and viewed in a balanced way. This discussion must include the laboratory safety program in all its facets, as well as its effectiveness in proactively managing risks through sound strategies and responsible oversight.
Footnotes
Acknowledgments
Special thanks to Deborah E. Wilson, John Veitch, Lydia Kibiuk and Larry Johnson of the National Institutes of Health’s Division of Occupational Health and Safety for their gracious help in data collection and presentation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
