Abstract
Peripheral arterial occlusive disease (PAOD) contributes to decreased exercise tolerance, poor balance, impaired proprioception, muscle atrophy and weakness, with advanced cases resulting in critical limb ischemia (CLI) where the viability of the limb is threatened. Patients with a diagnosis of CLI have a poor life expectancy due to concomitant cardio and cerebrovascular diseases. The current treatment options to avoid major amputation by re-establishing a blood supply to the limb generally have poor outcomes. Human skeletal muscle contains both multipotent stem cells and progenitor cells and thus has a capacity for regeneration. Phase I and II studies involving transplantation of bone marrow-derived progenitor cells into CLI limbs show positive effects on wound healing and angiogenesis; the increase in quiescent satellite cell numbers observed in CLI muscle may also provide a sufficient in vivo source of resident stem cells. These indigenous cells have been shown to be capable of forming multiple mesodermal cell lineages aiding the repair and regeneration of chronically ischemic muscle. They may also serve as a repository for autologous transplantation. The behavior and responses of the stem cell population in CLI is poorly understood and this review tries to elucidate the potential of these cells and their future role in the management of CLI.
Introduction
Peripheral arterial occlusive disease (PAOD) contributes to decreased exercise tolerance, poor balance, impaired proprioception, muscle atrophy and weakness, with advanced cases resulting in critical limb ischemia (CLI). Patients with CLI suffer from rest pain, ulceration and gangrene, which results in perigenicular amputation if severe or where revascularization fails. 1 The presence of CLI is such a poor prognostic indicator that within the first year of diagnosis around 25% of patients will die from a cardiac or cerebrovascular incident. In this time period around 30% of patients will undergo a perigenicular amputation and about 20% will endure unrelenting pain or tissue loss. 2,3
Generally, these patients are high-risk surgical candidates with multiple co-morbidities and poor mobility. 2,3 Current treatment options to avoid limb loss by re-establishing in line blood supply to the limb are limited, with poor outcomes predicted by multilevel arterial disease and a lack of sufficient runoff vessels supplying the distal limb. Percutaneous transluminal angioplasty (PTA) is less invasive than bypass surgery, but nevertheless has its risks and limitations and there is an overall peri-procedural morbidity rate of 5%. 4,5
In those patients in whom traditional surgery or PTA has failed or is inappropriate, one of the remaining options is therapeutic angiogenesis. The administration of angiogenic agents or stem cells directly into affected muscle has shown some early promise. 6 –12 Human skeletal muscle has an impressive capacity for regeneration due to the different stem cells and progenitor cells residing within it. A clear understanding of the behavior of these stem cells, their ability to repair, regenerate and self-renew in ischemic conditions may lead to potential therapeutic strategies involving these multipotent cells in preventing, treating or even reversing the damage caused by atherosclerosis.
Satellite cells
One of the most studied stem cells in skeletal muscle is the satellite cell, originally identified as a bipotent myogenic precursor cell, which was first recognized in the sartorius muscle of the frog and named by Alexander Mauro in 1961.
13
These cells were observed under electron microscope as a distinct population of cells, lying above the muscle fiber sarcolemma but beneath the fiber basal lamina, and accounting for up to between 3 and 6% of visible myonuclei seen,
14
see Figure 1. Their structure was found to be completely different to muscle cells, being mononucleate with large nuclei and a relatively small amount of independent cytoplasm. Under light microscopy, satellite cells may be distinguishable from myonuclei due to their location, smaller size and greater nuclear content.
15
The general dimensions of a quiescent satellite cell in mammalian skeletal muscle measure around 25 µm in length, 4 µm in height and 5 µm in width.
16
A quiescent satellite cell seen at ×22,200 magnification (scale bar in nanometers) with a transmission electron microscope. The skeletal muscle biopsy was known to be from critically ischaemic tissue. Of note, a single large nucleus is visible with a prominent but small nucleolus and little cytoplasm. The membrane of the satellite cell is smooth with no projections indicating quiescence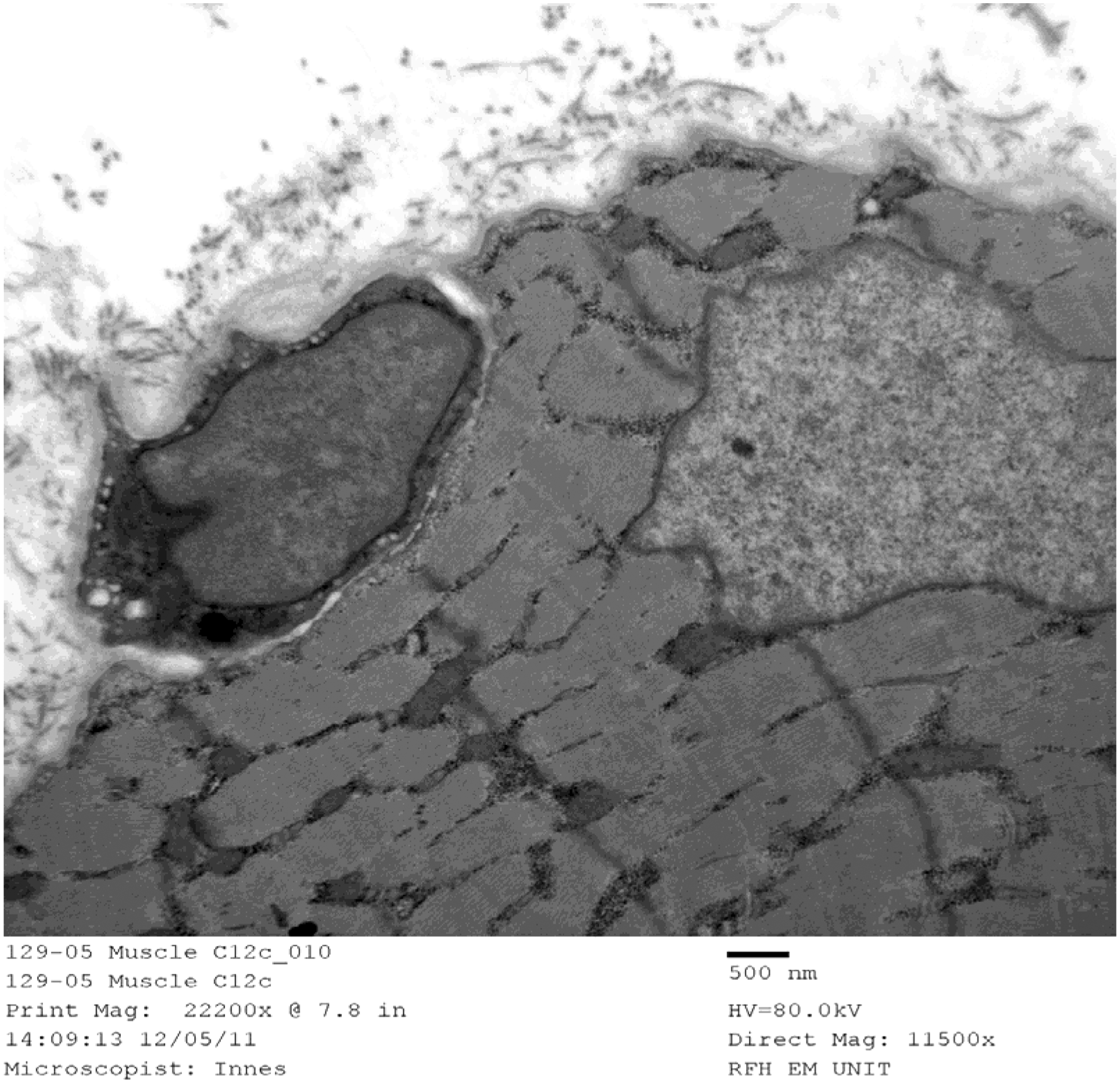
Destruction of satellite cells in adult small mammals via gamma irradiation leads to retarded muscle growth, demonstrating that they are required for normal skeletal muscle maintenance. 17 –19 Satellite cells therefore are the myogenic precursor cells of postnatal muscle and are responsible for the repair and regeneration of muscle fibers in adult tissue, either by fusing together and forming new fibers or incorporating themselves into damaged muscle cells and their myonuclei. 20 Plasticity of muscle fibers is influenced locally by satellite cells under the control of the muscle regulatory factors (MRFs – see Figure 2). 21 –23
These cells are possibly the only significant source of new myoblasts in the adult tissue, but the contribution of other cells such as bone-derived hematopoietic cells, vascular progenitor cells, or interstitial cells has yet to be fully elucidated. 24 The coordinated action of these cells is required for the process of plasticity in the form of pre-conditioning, remodeling of muscle through fiber shift and the efficient repair of damaged myofibers. This occurs in ischemic tissue and although studies vary in their findings regarding which fibers are the most resilient in the face of advancing ischemia, they generally concur that the regenerative capacity of muscle becomes exhausted with worsening disease. In younger adults, this regenerative activity is well regulated, but tight coordination breaks down in advancing disease and individual satellite cells may act independently of normal cues. 25
Markers of satellite cells at different stages in cell cycle: (45)

Quiescent satellite cells as seen in Figure 3, are activated by the presence of a damaged basal laminar structure, with the integrity of the basal lamina being more important than previously thought. In a variety of animal models where muscle is damaged by crushing,
29
freezing
30
or injection with toxins,
31
the satellite cell response appears to follow two paths; dependent not on the mode of injury but upon whether or not the basal lamina is intact. Figure 3 demonstrates those factors responsible for the activation of quiescent satellite cells. When the basal lamina is intact, activated satellite cells contained within this layer migrate to the site of injury in the same muscle fiber via chemotaxic stimuli.
17,31
Activation of cells has been reported at around 6 h post-injury and within 24 h the cells display significant mitosis.
29
The satellite cell then merges with the myofiber and helps repair or regenerate either myofiber material, contribute to existing myonuclei or replenish the satellite cell population
20
and this process is schematically represented in Figure 2. In injuries where there is disruption of the basal lamina, satellite cells are able to migrate from adjacent myofibers by projecting across tissue bridges initiated from an out-pouching process of the satellite cell itself.
17,32
This suggests that whatever the etiology of muscle damage, the processes of repair and regeneration are the same.
Schematic diagram demonstrating the role of the satellite cell, as a bipotent stem cell, contributing to myofibers, myonuclei or self renewal of the satellite cell population. In CLI, quiescent satellite cell numbers increase, demonstrating that proliferation occurs in abundance but maturation and terminal differentiation are inhibited Factors responsible for activation of satellite cells. A multitude of factors including cells, growth factors, hormones and neurotransmitters can activate resident satellite cells. In this figure, a satellite cell, recognizable from its anatomical position under the basal lamina of the sarcolemma of the muscle fiber, is stimulated by a wide variety of cytokines and growth factors such as the family of insulin-like growth factors, (IGFs) also including mechano growth factors (MGFs), as well as tissue growth factor (TGF), platelet derived growth factor, hepatocyte growth factor and fibroblast growth factor have been demonstrated in activating quiescent satellite cells. Hypoxia (and ischaemia) likely mediated by HIF-1 a (hypoxia inducible factor) and nitric oxide (NO) has also been shown to stimulate these cells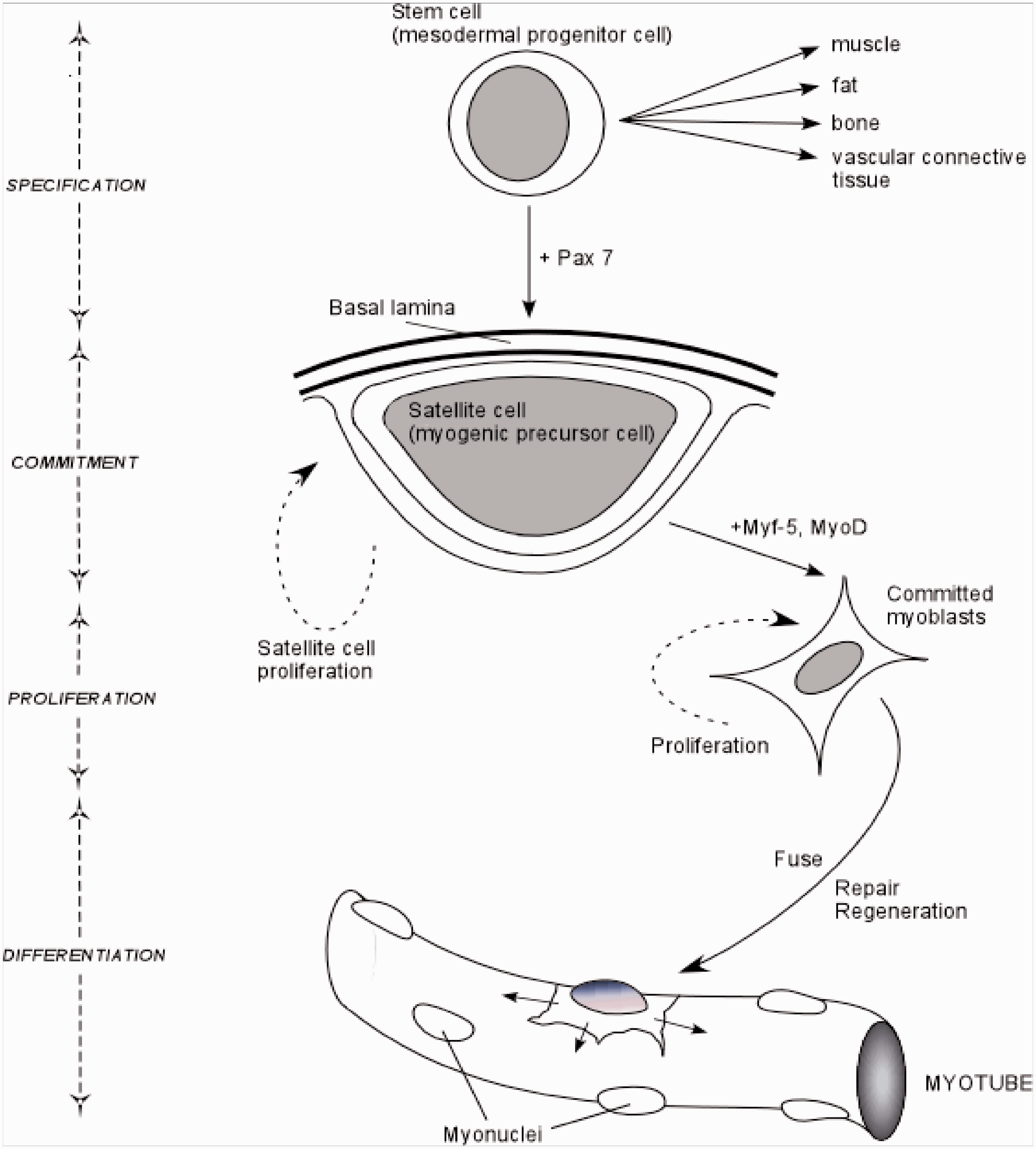
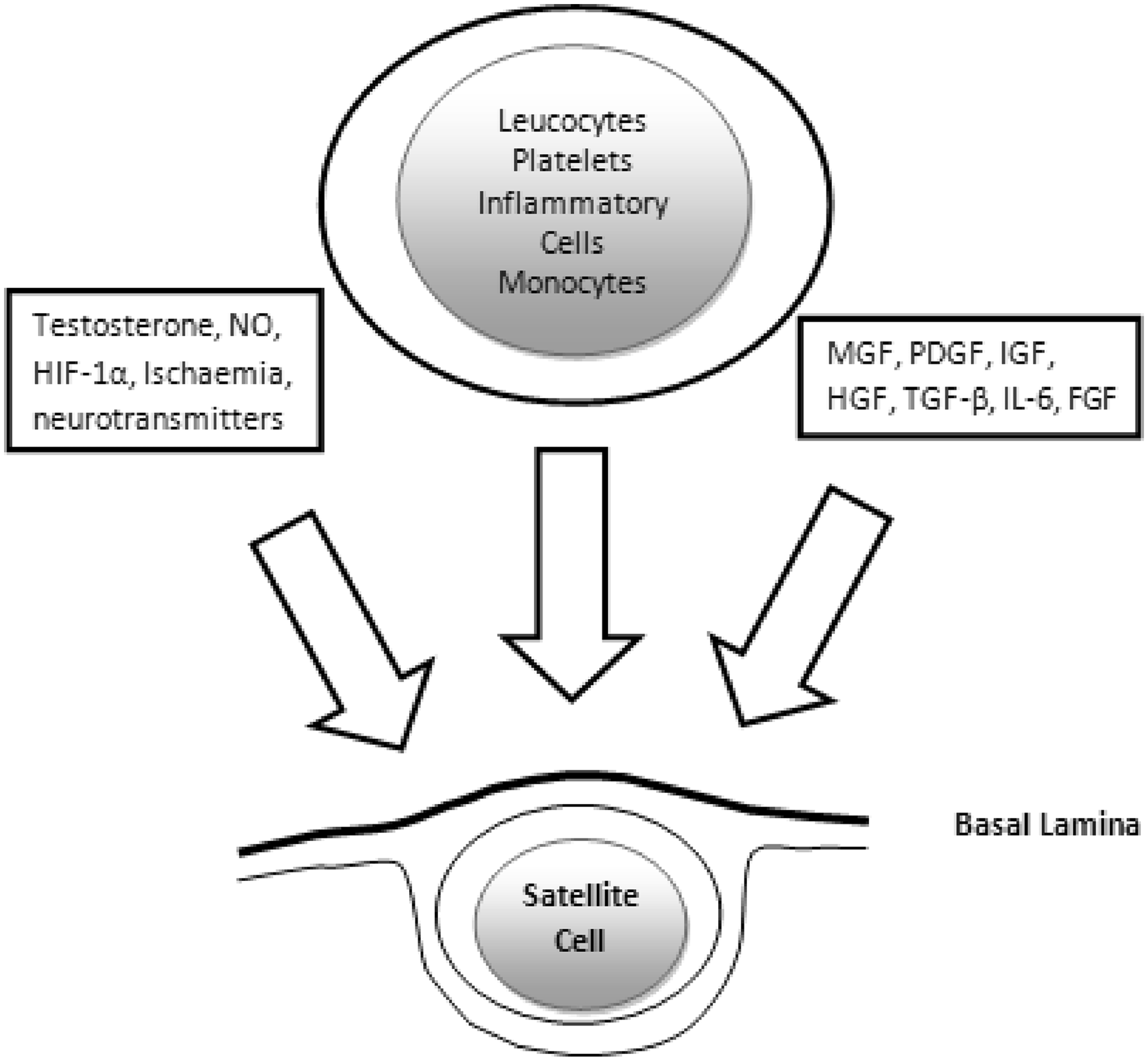
Aside from basal laminar disruption, other activators of satellite cells include migrating inflammatory cells, mediators such as nitric oxide (NO), the MRFs such as myf-5, specific extracellular signaling through Wnt, IL-6 and the insulin-like growth factors (IGFs) 33 as seen in Figure 3. These lead to myoblastic proliferation, but differentiation is retarded, due to the inhibition of the Notch system. It has been determined that the IGF-1 splice variant known as mechano growth factor (MGF) is expressed by damaged myoblasts and initiates the activation of satellite cells. 34 In healing tissue, expanding myoblasts closely interact and are under the control of nearby inflammatory and stromal cells. Active satellite cells or myogenic precursors contribute to mature post-mitotic myofibers after stimulation by IGF-1ea, another splice variant of IGF-1. 33 –36
Animal experiments have demonstrated that exercise-induced ischemia can increase the proportion of satellite cells and myonuclei. 37 The activation of satellite cells in response to such training ensures that the proportionate number of myonuclei per fiber remains constant, allowing continuing control despite the increase in fiber size and number. This is reflected in the increasing number of myonuclei seen in exercise-induced muscle hypertrophy, whilst a loss of myonuclei is observed occurring during atrophy. 38
The frequency of satellite cells is not known to differ according to their location within a muscle or around any specific fiber type. 39 Larger myofibers have more satellite cells suggesting each satellite cell has a “sphere of influence.” This becomes less organized with age as the nuclear domains governed by the myonuclei enlarge and satellite cell numbers decrease. 40 Satellite cell proliferation has also been reported as increasing due to the following: denervation, youth, endurance exercise, low-frequency electrical stimulation, stretch, testosterone and immobilization. 24,37,41 –45
In vivo studies demonstrate a decline in the satellite cell population with age. 25,46 Coupled with this reduced capacity in the older population, nerve degeneration and a reduction in myofiber cytoplasm to nucleus ratio intrinsic within the fibers (leading to volume loss), is also observed. 30,31 It is unclear whether the number of satellite cells or their regenerative potential is the greatest limit on their ability to repair and regenerate, although both are implicated. Some studies report a decrease in satellite cell numbers in skeletal muscle with advancing age 41,47 –49 while others also observed a decrease in their regenerative potential. 25,41,47,48 There have been few reports regarding satellite cell number and behavior in critically ischemic skeletal muscle, but it seems likely that the intrinsic activated stem cells are overwhelmed by the extent of advanced ischemic disease on a background of limited regenerative capacity due to age. Although originally identified as bipotent, i.e. capable of self renewal or forming myoblasts, there is increasing evidence that satellite cells, like bone marrow-derived mesenchymal stem cells, are capable of regenerating most other tissue types – see Figure 2 – given the right stimuli. 50 When transplanted from skeletal muscle into infarcted cardiac muscle, satellite cells can regenerate the cardiomyocytes adjacent to them 51 ; however, due to a decrease of the adhesion molecules N-cadherin and connexin 43, the regenerated cells do not perform entirely in concert with the rest of the myocardium 52,53 and may impair remodeling and cardiac function. 54 Pioneering studies transplanting cells into ischemic tissue, particularly cardiac muscle, have given us some insight into their behavior; but more in vitro work isolating different stem cells and observing them in chronically hypoxic conditions is required in order to understand the factors that may promote their activation. 10 Skeletal muscle itself has vast regenerative potential; yet whilst resident cells in skeletal muscle may proliferate, differentiation is inhibited and the regenerative potential of these cells seems to be overwhelmed by advancing disease or ischemia. The proliferation of satellite cells in ischemic tissue with disordered differentiation is similar to the response of angiogenic cells in advanced ischemia.
Alternate stem cells
HSCs, or side population cells (SPCs), may be capable of replenishing the population of resident stem cells by migrating beneath the basement membrane. Other sources of such mesenchymal stem cells, or multipotent adult progenitor cells (MAPCs) 55 can be identified using the nestin antigen and include osteoblasts, endothelial cells and mesoangioblasts. Nestin is an intermediate filament protein found in both nerve and muscle cells, but most importantly has been shown to be present in progenitor cells, such as muscle-derived satellite cells or HSCs and is therefore useful as a cell marker. 56 Establishing the precise role of HSCs is difficult and there are unanswered questions regarding the origin of these multipotent cells, which have been stimulated to differentiate not only into hematopoietic lineages but also hepatocytes, 57 cardiomyocytes, 51,58 renal tubules 59 and gastrointestinal epithelial and vascular endothelial cells. 60,61 Increased numbers of HSCs, identified by antigen markers such as CD34, have been observed in CLI muscle. 62,63 Despite the presence of more than one type of stem cells, repair of ischemic tissue is inadequate, suggesting that the cells identified are either incapable of repairing ischemic tissue or are unable to differentiate into mature and organized tissue types.
Other stem cells present include inflammatory monocytes and resident fibro-adipogenic progenitors (FAP), which have been demonstrated to assist in the myogenic response. 35,36 These cells may also modulate the behavior of satellite cells. Both FAPs and bone marrow-derived monocytes have been shown to inhibit normal haematopoietic stem and satellite cell differentiation and thus they inhibit normal function. 64 FAPs proliferate in skeletal muscle tissue when myogenic precursor cells fail to undergo normal myogenic differentiation due to disease processes or age-limited mitogenesis or lipofuscin accumulation. 64 Chronic damage such as that caused by ischemia may result in fatty degeneration when muscle atrophies and disorder are further complicated by the accumulation of white adipose cells and fibrocytes, reducing the ability of the affected tissue to restore its function.
It has been demonstrated that fibro-adipocytes form multi-mesenchymal lines, including HSCs, 65 neuronal cells 66 and myogenic precursors 67 as well as fibrocytes and adipocytes 35,36,64,67,68 represent an exciting source of multipotent cells. 69,70 They also have a regulatory effect on other stem cell types. Adipose-derived stem cells have been successfully and safely implanted in patients with CLI and evidence from a Korean trial (n = 15) published in 2012 showed that there was a significant clinical improvement in two-thirds of patients as measured by pain scores, amputation rates, claudication distance and appearance on angiography. 71
Future applications
The knowledge that stem cells may be safely harvested and preserved cryogenically 72,73 offers a method of storing multipotent cells to be administered as autologous transplants when required to treat diseases developed later in life. Stem cells have already been harvested and grown in vitro to be delivered back to the patient as intravenous or intramuscular infusions 7,74 The US National Institute of Health website details numerous phase I and II trials underway to assess the success of using differing stem and progenitor cell types. The results of studies assessing the performance of CD34+ cells (Losordo), CD133+ cells (SCRIPT-CLI Trial), bone marrow-derived mononuclear cells (JUVENTAS IIa), bone marrow concentrate, bone marrow-derived mesenchymal stem cells, angiogenic cell precursors and even mesenchymal-stem-cell-like endometrial cells in CLI are eagerly awaited.
Not only is there a wealth of evidence that stem cells are implantable, but this has been achieved intravenously, intramuscularly, via intra arterial (IA) catheters, engrafting or bone marrow transplantation. 7,53,74,75 Recent evidence suggests that regardless of the mode of transplantation, stem cells have been identified in all tissues of the donor body after just a few hours. Current evidence also suggests that IM or IA routes of administration are as effective as each other. 76
Similar to conditions such as Duchenne muscular dystrophy (DMD) where muscle wasting and atrophy are caused by a defective gene coding for the protein dystrophin (which is necessary for normal muscle function), CLI causes a similar loss of form and function and results in typical pathognomonic changes such as fiber atrophy, loss of polygonal structure, fiber clumping, fiber size variability, fiber loss and other structural abnormalities. In animal models of DMD, HSCs have been injected intravenously into mdx mice and the complete restoration of the hematopoietic system and the incorporation of donor-derived nuclei into myofibers have been observed with partial restoration of dystrophin expression in the affected muscle. 20
Phase I and II studies of stem cells harvested from disparate tissues and in turn administered to critically ischemic human skeletal muscle as a strategy to improve limb perfusion and tissue healing, have shown some early promise. Progenitor cells from such disparate tissues have similar characteristics and given the right stimuli are able to fulfill a multipotent fate. 50 Bone marrow-derived progenitor cells are found freely circulating and may be recruited to assist in the regeneration of tissue.
In a recent phase I clinical trial, combinations of bone marrow-derived stem cells containing endothelial progenitor cells and mesenchymal stem cells were administered intramuscularly to the gastrocnemius muscles of a small number of patients with CLI. 7 The results were an improvement in exercise tolerance (3.48 ± 1.72 fold increase in seven out of 10 patients, with the others limited by a non-treated leg in bilateral disease), improved Ankle Brachial Pressure Index (ABPI) within one month of therapy until cessation of follow-up at 10 ± 2 months (0.34 ± 0.19 initially to 0.69 ± 0.18 at (P < 0.002), increased transcutaneous oxygen levels (TcO2 33 ± 6 mmHg at baseline to 46 ± 10 mmHg) and all patients reported an improvement in Quality of Life scores. Angiograms (P = 0.002) and perfusion scans were also assessed and objective improvements were demonstrated. 7
Bone marrow-derived mononuclear cells have also been injected intramuscularly deep to the ulcers of type II diabetics with CLI; these cells shortened recovery times by up to a month, improving claudication symptoms, raising TcO2, normalizing ABPIs and demonstrating magnetic resonance angiography proven angiogenesis. 7,74 Transplantation of embryonic stem cells in this way has also demonstrated that they not only promote tissue repair but also replenish the resident satellite cell pool. 7,74,77
The recently published phase II randomized controlled trial “Use of Vascular Repair Cells (VRC) in Patients with Peripheral Arterial Disease to Treat Critical Limb Ischemia” (RESTORE-CLI; n = 72) has shown some encouraging signs that stem cells promote faster healing and improve clinical outcomes. 78 The trial did not demonstrate any adverse effects due to the intramuscular injection of autologous bone marrow-derived tissue repair cells. The study did show a significant lengthening in the “time to treatment-failure,” (determined as doubling of ulceration, major amputation or new gangrene; P = 0.0032) and although “amputation free survival” was reduced by 32% this was not found to be significant (P = 0.3880) for the relatively low number of patients recruited. 79
Although stem cells appear to be relatively safe in the majority of recipients, like many medical interventions there have been reported failings or adverse effects. A recent aborted Swedish trial of autologous peripheral blood mononuclear cells to treat CLI demonstrated that of the small number of participants included (n = 9); four patients suffered major morbidity or mortality as a direct consequence of bone marrow stimulation. Two suffered myocardial infarctions (one fatal), there was one death from heart failure and another from a massive mesenteric thrombosis, all forcing the researchers to terminate the study prematurely. 80 An increased frequency of ventricular arrhythmias has been previously been reported with the use of cardiac-implanted skeletal myoblasts 81 –84 suggesting that stem cell transplantation may not always be appropriate and until their safety and efficacy are better understood universal therapeutic application should be approached with caution.
Footnotes
Author contributions
This article was written by CH and in part rewritten by JT. Proof reading was performed by DMB and AK provided the diagrams. The paper was conceived by DJA who also provided the electron microscopic picture of a satellite cell.
ACKNOWLEDGEMENTS
The authors received no specific funding for this article.
