Abstract
Introduction and Purpose
Radiopharmaceutical therapy (RPT) dosimetry can be challenging to perform due to sparse data measurements and variations in how the time activity curve (TAC) is determined. In this work, a single system of equations was theoretically derived to estimate the TAC.
Methods
A pharmacokinetic (PK) model was developed to estimate patient specific rate constants for a given set of body compartments. The PK model and an optimizer were numerically implemented to determine the rate constants and, using these physiologic data, to generate TACs and time integrated activities (TIAs) for 3 tissue systems from clinical data gathered in 5 patients. A fourth (aggregate) tissue compartment is added using conservation of activity considerations.
Results
Feasibility of the PK model was demonstrated by successfully generating TACs and TIAs for all patients in a manner comparable to existing methods in the literature. The data are compared to smaller sampling regimes. Differences between the 3- and 4-compartment models show that conservation of activity considerations should be part of TAC estimations.
Conclusion
The results here suggest a new paradigm in RPT in using the rate constants so identified as a diagnostic tool and as a vehicle to achieving individualized tumorcidal dose and/or the maximum tolerable dose to normal tissues.
Introduction
Radiopharmaceutical therapy (RPT) is a rapidly evolving modality that uses internally committed radiopharmaceuticals to treat disease through delivery of radiation dose. 1 The benefit of RPT is that for some disease sites (eg, oncologic), the radiopharmaceutical has a high target to non-target ratio (also known as the tumor-to-normal ratio or TNR) that spares normal tissue while delivering tumorcidal dose to malignant lesions. 2
Despite high TNRs for many RPTs, normal tissues often still receive substantial dose. One currently accepted paradigm for RPT with substantial normal tissue dose is to deliver a total activity that will result in a dose: (1) known to be tumorcidal to a given tumor; and/or (2) that does not exceed the maximally tolerable dose (MTD) for any normal tissue structure. 3 Normal tissue structures that receive relatively high dose during RPT are known as organs-at-risk (OAR) and the MTD is the dose threshold below which deterministic effects are not expected to be present in a given OAR.
The dose to the tumor(s) and/or OAR(s) can be estimated through sparse measurements of activity using serial imaging and/or blood draws at known time points following a given RPT cycle.4–9 The activity contained within each tumor and/or OAR is plotted as a function of time and is fit with a known function to create a time activity curve (TAC); the TAC can be integrated to determine the time integrated activity (TIA), which can be used to estimate tissue dose using a variety of formalisms and softwares.8,10
The TAC fit can be performed using any user defined function, but is most often performed using standard functions such as combinations of exponential functions; 11 these methods have been widely implemented. 10 These fits are typically performed for each tissue of interest independent of other tissues (eg, the tumor TAC is created independent of the kidney TAC for a given patient). Variability in activity measurement, TAC generation, and TIA calculation has demonstrably large variations in estimated radiation dose to tumors and/or normal tissue. 12 This approach can be considered as a “top down” approach to modeling the TACs and, while useful, do not provide additional information that may be of significant clinical relevance.
The objective of this article is to develop and demonstrate feasibility of a novel pharmacokinetic (PK) model that uses a single system of equations to simultaneously determine the rate constants for all compartments as a function of one another for a given patient, and produce the TACs of interest. As the patient by definition is a single system with all compartments interconnected, this PK model may provide an initial step in advancing the determination of the TAC in patient specific RPT dosimetry. This is a “bottom up” model, where the rate constants, which are related to many physiologic parameters, are determined and they are used to determine the TACs. The determined rate constants may provide additional diagnostic/disease stage classifications that have not been pursued at this point. PK models have been widely used in pharmacology,13,14 but their use in RPT has thus far been relatively limited.15–17
To accomplish this objective, the PK model was theoretically developed in the “Theoretical considerations: PK Model” and “Conservation of Activity and a 4-Compartment” sections to provide the TAC for typical RPT infusion regimens. An optimization algorithm based on a hybrid gradient descent method is presented over the rate constant parameter space in the “Rate Constants: Optimization” section and its numerical implementation is subsequently described in the “Numerical Methods Implementation: Optimization” section. This optimization allowed for rapid convergence of the PK model to the known patient data. The patient data used in this study consisted of activity in 3 organ systems (blood, tumor, and kidney) measured at multiple time points using the clinical data provided by the publicly available American Association of Physicists in Medicine (AAPM): Understanding
Methods
Theoretical Considerations: PK Model
Let 

The 3 compartment PK model.
In typical scenarios and as is the case here (the patient data provided by the TACTIC challenge), specification of activities can be somewhat vague. The theoretical results are normalized to incorporate the corrections made in the TACTIC data, which was provided as % activities within each organ system at each time point. Rate constants will have units of
The problem presented in Figure 1 can be described mathematically as


Solving for the eigenvalues of the matrix 
Denoting the eigenvalues of 

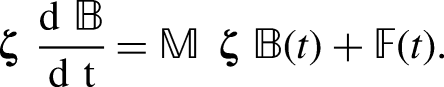



Three special cases are worthy of consideration to flesh out the computations in the implementation section, compare the evolution of the activities and to partially (and implicitly) determine the rate constants on infusion models. Each of these cases has been and/or is clinically used to administer RPT (noting that very short injection times can be considered a delta function).
Case 1: Suppose that the isotope is introduced intravenously as a delta function at time 
Case 2: If the isotope is introduced intravenously more slowly but at a steady (constant) rate, then 


Case 3: For gravity infusion, saline is added via gravity to vial of isotope that is intravenously connected to the patient. The saline drip increases the volume of the vial and results in the liquid vial contents entering the patient at a constant rate (mL/s) with an activity concentration that decreases exponentially as saline replaces the isotope. The amount of isotope infused can be described by an exponential
Putting this infusion regimen into (4) and (5) and integrating, similarly to Case 2, the following result is arrived at
Before moving forward, there is a subtle point that requires re-visiting and will be important for the determination of the rate constants. There are 2 ways to introduce the initial activity in (1). The initial activity can be inserted as a delta (forcing) function (vector) 

Conservation of Activity and a 4-th Compartment
The 3-compartment model shown in Figure 1 was necessitated as the activities in only those compartment were measured clinically. However, using a conservation scheme, a 4-th compartment may be introduced to account for significant communication between blood and all other organs throughout the body, and possible elimination from those areas. While the measured activity in the 4-th compartment is not available, theoretical activity in the 4-th compartment and the resulting eliminated activities can be calculated. As will be seen, this will allow significant improvement in the fitting of the theoretical results to the clinically measured data.
To maintain brevity and as the solution here follows the 3-compartment model closely, only the changes will be highlighted here. Let 






The 4-compartment PK model.
Rate Constants: Optimization
The above compartmental PK model provides an exact solution of the activities as a function of time in each compartment. For specificity, we shall describe our numerical simulations for the 3-compartment model; the 4-compartment model descriptions are exactly analogous, with 2 additional parameters
To provide agreement with experimentally obtained data, the following steps were performed: The error is minimized between the experimentally determined activities 
Numerical Methods Implementation: Optimization
Implementation of this PK model can be described methodically as follows, with code snippets performing the most relevant of these operations provided in Appendix A. This numerical solution was tested using the data available from the TACTIC Challenge data, which is reproduced in Appendix B. The data provided in Appendix B was described by the TACTIC challenge as [verbatim] “25 synthetic datasets of peri-therapeutic time-activity curves in kidneys, blood, and tumor after [Lu-177] PSMA therapy. These data are generated based on randomly selected high-quality clinical datasets, with realistic uncertainties added. While a sample of data for 2 patients is shown throughout the results section, in Appendix C the corresponding data for entire five patient cohort are shown for comparison sake. Since the clinically provided data here relies on the AAPM data, a full discussion of the statistical uncertainties inherent in the data due to photon counting, finite time interval data acquisition, patient variations and other factors is not feasible. Also, no additional information such as body weight, patient age and other parameters were available to quantify variations between the different patients.
The implementation follows the following steps:
As each set of calculations is patient specific, the following calculations will be done for each patient separately and the patient specific indices will be omitted. Define the 6-dimensional parameter orthant Construct the Construct the Using Equation 5, compute the components of the vector The Find the Partitioning a bounded subset of the parameter orthant, say A random starting point and random step sizes for each parameter are generated, then the above step is repeated until the process converges at a certain number of significant digits. The number of significant digits can be increased by increasing the number of iterations while decreasing the step sizes whenever Since the matrix Finally, the rate constants are used to estimate activity in each organ at time increments of


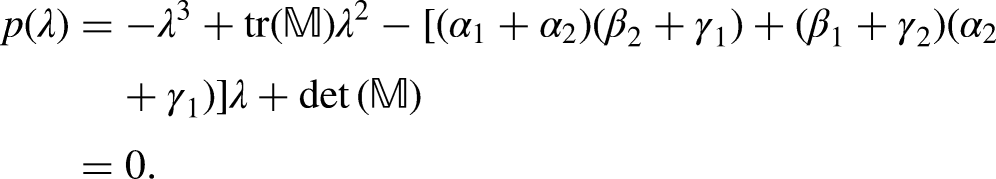
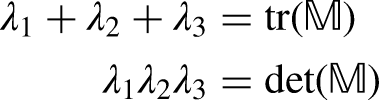
Results
Figures 3 to 5 show the experimental patient data and the resultant PK model predictions of activity with rate constants optimized to minimize the LSE for the 3-compartment model. These 3 figures each show the kidney, blood, and tumor logarithm of % activities, respectively, for Patient 10 for the 3 infusion methods described above. Patient 10 was chosen as a representative example of the data set; all other patients demonstrated similar results. Note that to retain the same patient descriptors used in the AAPM TACTIC data, the 5 patients are numbered 6 to 10.
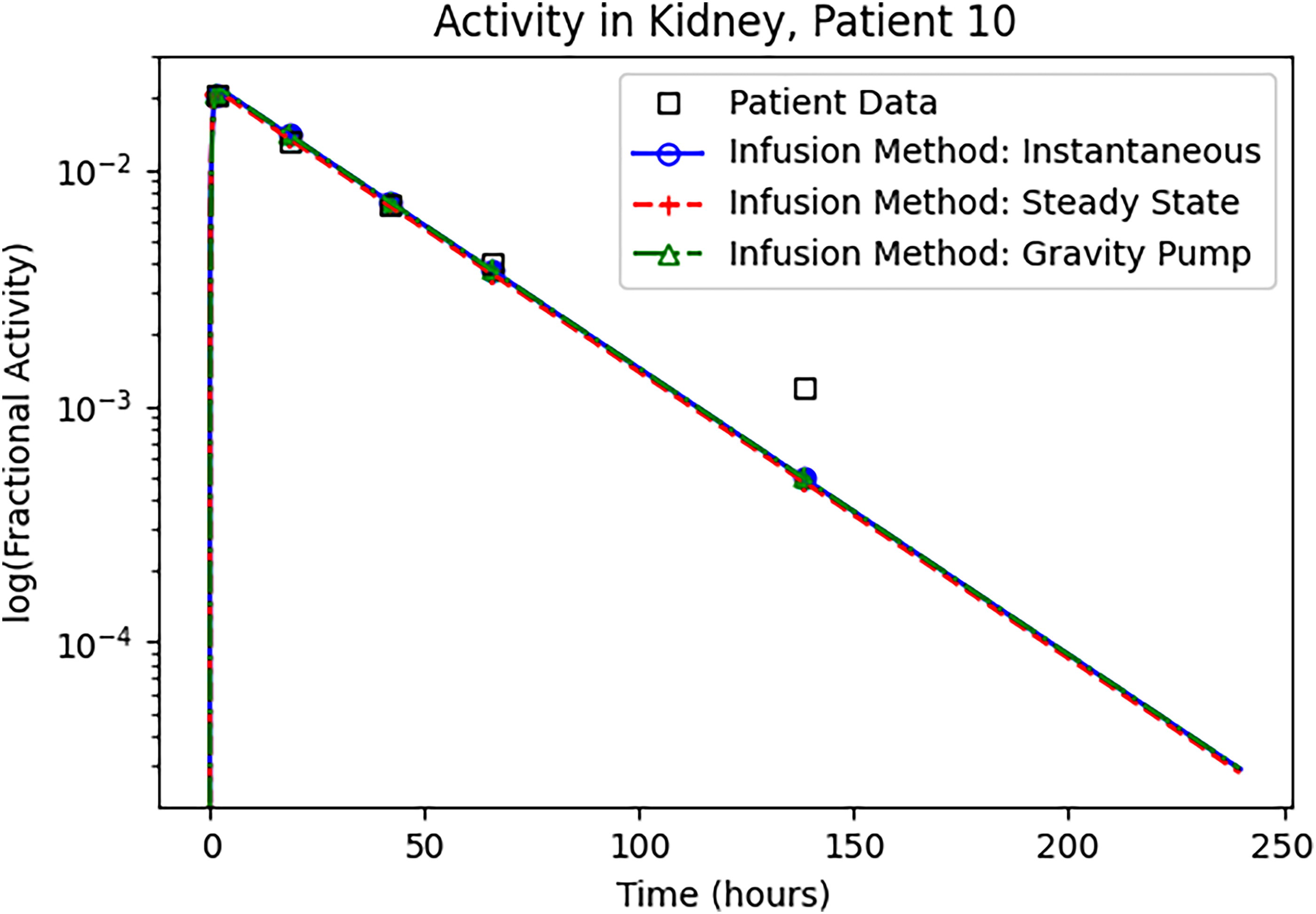
Comparison of PK model prediction to known patient data for the kidney in Patient 10 for all 3 infusion methods using the 3-compartment model. The rate constants, estimated time integrated activity, and LSE are provided in Table 1.

Comparison of PK model prediction to known patient data for the blood in Patient 10 for all 3 infusion methods using the 3-compartment model. The rate constants, estimated time integrated activity, and LSE are provided in Table 1.

Comparison of PK model prediction to known patient data for the tumor in Patient 10 for all 3 infusion methods using the 3-compartment model. The rate constants, estimated time integrated activity, and LSE are provided in Table 1.
The optimized rate constants for the 3 infusion regimens for Patient 10 using the 3-compartment model are shown in Table 1 as a representative example. A comparison of the optimized rate constants for steady-state infusion using the 3-compartment model for all 5 patients is shown in Table 2. The TIA and LSE for each organ system are also shown in these tables. It is important to point out that this model is not a fit to the activities. Rather, the rate constants between the different compartments are determined, and those are used to infer the activities in the different compartments.
Comparison of the Rate Constants, Time Integrated Activities, and LSEs for Different Infusion Models in Patient 10 Using the 3-Compartment Model.

Abbreviations: LSEs, least square errors; TIA, time integrated activity.
Comparison of the Rate Constants, Time Integrated Activities, and LSEs for the Steady-State Infusion Method in All 5 Patients Using the 3-Compartment Model.

Abbreviations: LSEs, least square errors; TIA, time integrated activity.
As a comparative measure, Figure 6 was generated with the 5 data points from Patient 6 using the steady-state PK 3-compartment model and also by fitting the known patient data to:
a single exponential model with 2 parameters of the form a bi-exponential model with 3 parameters of the form a bi-exponential model with 4 parameters of the form
These functions were fit to the data using the Solver add-on (minimization of objective function) in Microsoft Excel (Microsoft Corporation, Redmond WA). The LSE found using the Excel solver addon by minimizing the objective function was comparable to the LSE presented in Tables 1 and 2 (

Comparison of pharmacokinetic (PK) model prediction (steady state) using the 3-compartment model and best-fit lines with error minimized using the Microsoft Excel Solver add-on to known data (blue circles).
Due to various clinical limitations, often it is hard to obtain measurements at 5 different time points. The fit here has also been carried out using 2 time-point and 3 time-point to compare to the given 5 time-point measured data. Figures 7 to 10 show these results, where different combination of 2-points and 3-data points are used for the optimization process. The optimized rate constants and blood–kidney–tumor TIAs and the LSE are shown on each of these figures for comparison.

Comparison of pharmacokinetic (PK) model prediction with 2 time-points and measured data using the 4-compartment model. The PK model was optimized using only with the patient data acquired at 1.4 and 18.5 h; the fit only utilizes these 2 data points and is compared to all clinically measured data.

Comparison of pharmacokinetic (PK) model prediction with 2 time-point measured data using the 4-compartment model. The PK model was optimized using only with the patient data acquired at 18.5 and 42.2 h; the fit only utilizes these 2 data points and is compared to all clinically measured data.

Comparison of pharmacokinetic (PK) model prediction with 2 time-point measured data using the 4-compartment model. The PK model was optimized using only with the patient data acquired at 65.8 and 138.4 h; the fit only utilizes these 2 data points and is compared to all clinically measured data.

Comparison of pharmacokinetic (PK) model prediction with 3 time-point measured data using the 4-compartment model. The PK model was optimized using only with the patient data acquired at 1.4, 18.5, and 42.2 h; the fit only utilizes these 3 data points and is compared to all clinically measured data.
Using the conservation scheme developed in the Conservation of Activity and a 4-Compartment section, Figures 11 and 12 were generated. Figure 11 shows the activities in the blood, kidney, and tumor in Patient 10 using the 4-compartment model, the corresponding 8-parameter rate constants, the different organ TIAs and the LSE. Figure 12 is to be compared to Figure 13, the 3-compartment model results initially presented in Figures 3 to 5, but here put in a similar format as in Figure 12. The rate constants, TIAs, and LSEs for these 2 plots are summarized in Tables 1 and 3. Finally, Figure 14, to be compared to Figure 6, compares the 4 compartment model activities to the exponential and biexponentials fits of those data.

Comparison of pharmacokinetic (PK) model prediction where conservation is used to provide a complete 4-compartment model.

Complete 4-compartment model with different infusion regimens.


Comparison of pharmacokinetic (PK) model prediction (steady state) using the 4-compartment model and best-fit lines with error minimized using the Microsoft Excel Solver add-on to known data (blue circles).
Comparison of the Rate Constants, Time Integrated Activities, and LSEs for Different Infusion Models in Patient 10 Using the 4-Compartment Model.
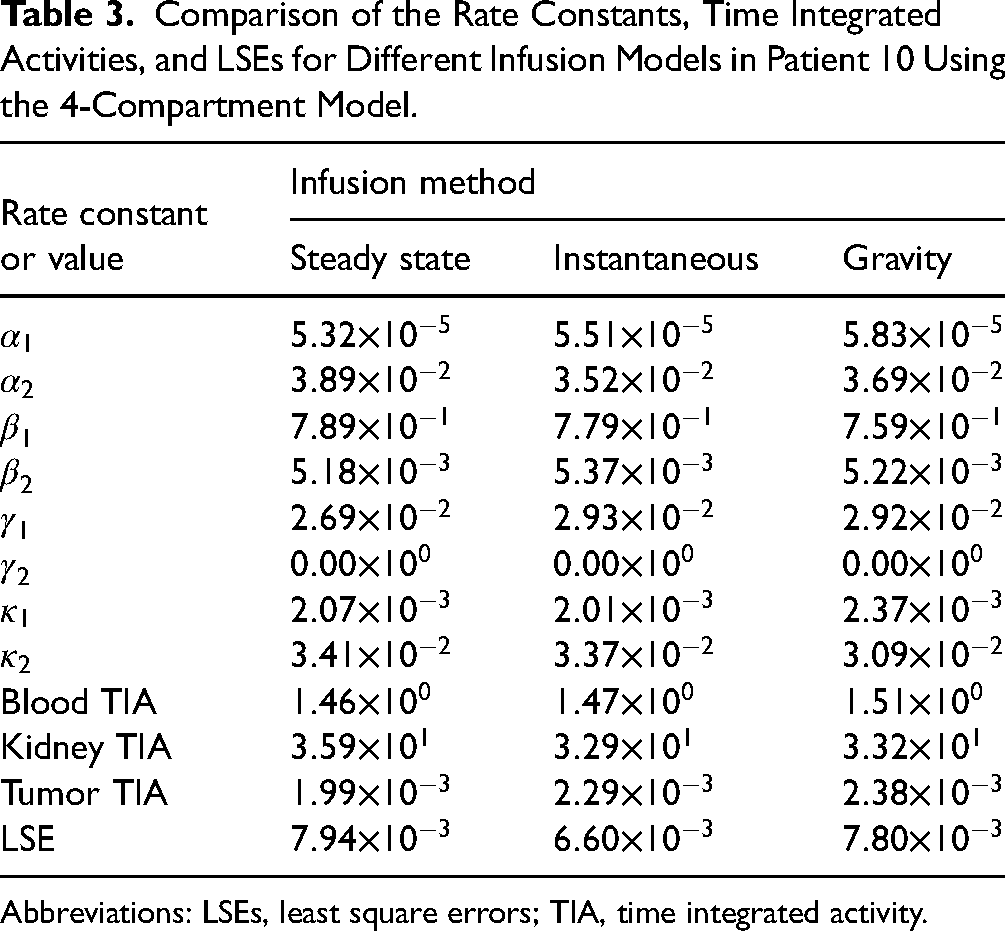
Abbreviations: LSEs, least square errors; TIA, time integrated activity.
Discussion
In RPT, patient-specific dosimetry is the calculation of radiation dose delivered to malignant lesions, tissue systems, and/or the whole body of individual patients. Often patient-specific dosimetry can be challenging. This is due to several factors. One factor is the sparse collection of data following RPT; the number of data points collected is generally clinically limited to a maximum of 5 post-RPT imaging and/or blood draw data points over a period of 5 to 10 days, with fewer data points (eg, 1 or 2) being most preferable for clinical workflow and patient convenience. 8 Another factor is that every patient is heterogeneous; patient anatomy (eg, given tissue size) and physiology (eg, uptake, distribution, metabolism, and excretion of the radioactive drug by a given tissue) can very greatly per patient. 1
Because of these factors, historically patient-specific dosimetry has been performed by fitting simple functions such as exponentials or bi-exponentials to the collected data. These functions are individually fit to data from each tissue of interest; the parameters of the functions and thus the final TIA for each organ are independent of all other organs.
Physiologically, the patient is a single system; the rate constants between various tissues and thus the resultant TIA are interconnected. In this article, a PK model was developed to simultaneously determine the rate constants (which can be used to determine the TAC and TIA) for all organ systems of interest in a given patient. Extraction of the rate constants using the PK model may provide a new diagnostic tool that would have considerable clinical applications. One potential application may be the estimation of activity in organ systems where the activity is not measured through a priori knowledge and sparse measurements of other patient organs. An additional application may be in the development of new radiopharmaceuticals with improved rate constants that would target specific sites.
However, the main purpose of this article was to demonstrate feasibility of the PK model using clinical patient data. Figures 3 to 5 demonstrate the PK model developed in this article can be utilized to model the TAC in several organ systems using sparse clinical measurements of activity in patients. Importantly, these figures have scales on the
In semi-log plots, while the fit for the blood compartment may not appear as good as the fit for the tumor and kidney compartments, the calculated percent errors for each of the organs (blood, kidney, and tumor) were 0.13%, 0.29%, and 6.06% for the blood, kidney, and tumor, respectively. The different order of magnitude change in scales for the blood compartment compared to the other organs is the cause of this apparent dispersion.
Figure 6 demonstrates the results of the PK model are comparable to existing single exponential and bi-exponential models (3 and 4 parameters) described in the literature.8,19–21 This comparison is significantly improved in the 4 compartment case (to be discussed shortly) and it further validates the feasibility of the PK model compared to other existing methods. While the superiority of the model described was not assessed other than through analysis of LSE, comparability of the described PK model to existing models shows promise. Intuitively utilizing a PK model provides a co-dependency of the different compartments and this would be superior to evaluating each compartment as a separate system.
Figure 6 shows the PK model provides excellent agreement with the single exponential model and the 2 bi-exponential models for the kidney. In the tumor, there was less agreement, but this plot allows for some interesting interpretations. The first is that the single exponential model, while reasonably fitting the data, is not valid from a biological sense for an organ system such as the tumor, because the single exponential model assumes the maximum activity concentration in each organ occurs at
When reviewing the activity in blood as shown in Figure 6, the single exponential model and the 2 bi-exponential models cannot be differentiated from one another and fit the known data very well. The PK model shows a rapid increase in blood activity due to the infusion, which is what occurs physically (particularly when considering the delta forcing function). While the PK model appears to model the late washout phase of blood well, the initial washout phase appears relatively poorly modeled with the blood activity concentration decreasing much faster than demonstrated by the patient data. This is most likely due to the use of 3 compartments and with the excretion pathway
Additionally, the challenge of fitting all compartments with a PK model were not unexpected, as shown in a prior publication 15 . PK models often have difficulty in fitting all data sets. Thus, while the results of this article demonstrate feasibility of using a PK model, it is clear the model can be further refined and calibrated for each patient, even if relatively few data points are collected (< 5) or if data is not collected for all organ systems. This PK model may be utilized to fill in missing gaps and provide a more accurate patient dose calculation. It is hoped that as this PK model matures, future studies will demonstrate improvements of the PK models over existing methods for producing TACs and TIAs.
The PK model additionally demonstrated some interesting trends as shown in Table 1, which, while somewhat illustrative in nature (since the optimization is done relative to the given data set), shows close agreement between the estimated TIA and rate constants in the blood, kidney and tumor for all infusion methods. The variations in the TIA were within 1% of each other, and the variability in the extracted rate constants were about 5%.
Interestingly, Table 2 demonstrates the variability in the TIA and rate constants among the Patients 6 to 10 can be by as much as a factor of 4. The tumor TIA in Patient 10 was
As one final point of discussion, the PK model allows for modeling of the actual administration of the radiopharmaceutical used in RPT (instant bolus injection, steady-state infusion over a known time period, and gravity infusion). To the knowledge of the authors, the currently utilized curve fitting methods (exponential and bi-exponential) do not model the time dependent administration. For some organs, such as blood (and thus bone marrow), that may result in underestimation of dose. Of course, the PK model as demonstrated in the above results also appears to underestimate the activity in blood; this will be investigated through the implementation of more compartments and also the incorporation of more physiological data.
The fitting problem has also been carried out using 2 time-points and 3 time-points to compare to the given 5 time-point measured data. Figures 7 to 10 show these results. The rate constants, TIAs, and the LSEs are also shown in these figures. Interestingly and quite surprisingly, optimization with smaller data points leads to quite small LSEs, almost comparable to the 5-data point results. This demonstration may serve as a possible validation of using very small data point sets and having confidence that the optimized results for the rate constants and activities are relatively close to those obtained with more data points. Figure 7 to 9 also vary the 2 time points chosen and show comparable results.
Brosch-Lenz et al22,23 compare single-time point (STP) to MTP imaging protocols for Lu-177 PSMA therapy and show data for a single photon computed tomography (SPECT) scan at 48 or 72 h scans. While their results are comparable to MTP data, their methods provide
Returning to the analytical modeling of the input function, if a therapy is designed to provide certain tumorcidal and MTD doses, the administered activity cannot be performed retrospectively and changing future therapies may not be effective due to tissue stunning or other similar effects. Thus, utilization of a PK model may allow for more patient-specific dosing using optimized input functions to achieve the rate constants and dose levels that will result in the greatest likelihood of normal tissue sparing and disease control. This is feasible since the PK model can provide the rate constants for a given tissue system from activity measurements obtained by tracer studies and sampling at only a few distinct time points. The resulting rate constants can then be used to create the entire TAC and thus calculate the activity needed for the desired outcome (tumorcidal dose or reaching a given MTD). This method is analogous to standard practice in both radiation oncology (eg, intensity modulated radiation therapy to provide a certain dose distribution) and radiology (eg, automatic exposure control to provide a certain image quality). This can be inferred from Figures 3 to 5 and is also discussed in the literature. 11 As there is an expected significant variation across patients, this model can lead to developing individualized therapies and better long-term outcomes for patients.
Conclusion
In this article, a theoretical PK model framework was developed to estimate rate constants and TIAs from post-therapy activity measurements in patients undergoing RPT. This PK model used one system of equations to describe the flow of the radiopharmaceutical in the patients body through various compartments. The PK model determined the rate constants and used these physiologic parameters to determine the TACs. The fit to clinical data provided validation, and comparison with existing methodologies demonstrated feasibility.
This PK model based determination of the rate constants provides a diagnostic tool that deserves a closer look to categorize and classify normal tissues, tumors, and patients. Looking at populations of patients and establishing statistics for these parameters would allow physicians to determine if individual patients are significantly different from the population. In addition, and much more importantly, this or similar type PK models may be used to create steady-state activities and TIA in the tumor, by prescribing appropriate infusion regimens to achieve those patterns. This inverse problem is the subject of the immediate next research effort aimed to refine the above findings.
Finally, a thorough analysis of the dependence and variation of the rate constants on the counting statistics, regional nature of measured TACs, and dosimetry using Monte Carlo simulations is the subject of ongoing research.
Footnotes
Abbreviations
Acknowledgments
We gratefully thank the expert (anonymous) referees of this article who pointed out several very important comments and allowed us to refine and highlight the presentation of these results.
Data Availability
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix A. Numerical Solutions for the PK Model
This appendix provides the functions written in Python to perform the numerical solutions for the 3-compartment model (the 4-compartment model can be similarly numerically solved). In the first code block, a function is defined to use the 6 rate constants to form matrix
In the second, third, and fourth code snippets, the result of the first code block is used to determine the activity
Appendix B. TACTIC Challenge Data Set
The patient data used in this study was provided by the American Association of Physicists in Medicine TACTIC Challenge and is reproduced in Table B1. The measurements are from synthetic data sets that originated from high quality data sets. All relevant corrections applied (eg, recovery coefficient) were applied and realistic uncertainty was added. The time points are given in hours after start of RPT infusion and the activities for each organ are given as % activities. The infusion method was not provided with the AAPM TACTIC challenge data.
Appendix C. 4-Compartment Fit of Other Patients Data
Below for comparison, we provide 15 plots that show the 3-organ fit to the 5-point data sets for Patients 6 to 10 (patient data from the AAPM TACTIC challenge) using the 4-compartment model. These plots, presented in semilog graphs, are to be compared with Figure 12 and its associated Table 3 of rate constants, TIAs and LSE for the steady-state infusion method. These plots and the associated parameter values show the variability in the different patients.
