Abstract
Visualization and quantification of inflammatory processes is of high importance for early diagnosis of a multitude of diseases. Magnetic resonance imaging (MRI) using iron oxide (FeO) nanoparticles as contrast agents allows the study of macrophage infiltration during inflammation in a variety of tissues. Macrophages are effectors of the immune response, their appearance being orchestrated by activated T lymphocytes. Therefore, tracking of labeled T lymphocytes, which initiate the immune process, should enable earlier detection of tissue inflammation. In this study, we investigate the feasibility of specifically labeling harvested T cells by using dextran-coated FeO nanoparticles and commonly available transfection agents (TAs). Physicochemical properties of the newly formed FeO/TA vesicles were determined as well as their cell toxicity and their T cell activation potential. The labeling efficiency of each FeO/TA combination was evaluated by measuring the transverse MRI relaxation rate
Introduction
The migration of immune-competent cells to sites of tissue inflammation is a key element in the immune response of the body. Visualization and quantification of these processes is critical from a diagnostic point of view for the understanding of inflammatory disease and the evaluation of potential therapeutic interventions. A number of imaging approaches has been proposed to monitor cell migration in vivo [1,2]. All of them involve specific labeling of the cells/cell types to be studied using either exogenous labels with radionuclides, magnetic resonance imaging (MRI) contrast agents, or fluorescent dyes as reporter groups, or by genetically modifying cells to constitutively express a reporter gene [3]. Among the different imaging approaches evaluated for studying cell migration, MRI is particularly attractive due to its superior spatial resolution, which is at least one order of magnitude higher than, for example, for nuclear and optical imaging.
Inflammation involves a massive accumulation of immune-competent cells, in particular macrophages, in the affected tissue. The infiltration of labeled macrophages can be monitored noninvasively by MRI following systemic administration of ferumoxides (FeOs), ultrasmall superparamagnetic iron oxide (USPIO) particles coated with dextran, as demonstrated, for example, in the rat experimental autoimmune encephalomyelitis (EAE) model [4]. Due to their high phagocytotic activity, blood-borne monocytes efficiently internalize nanoparticulate matter; hence, a substantial fraction of the macrophages that enter the inflamed tissue are labeled. In the rat EAE model, the use of USPIOs, initially developed for imaging of the liver lesions [5], provided an almost 100% sensitivity for lesion detection [6].
Macrophages are effectors of the immune-mediated injury in inflammation [7]; thus, visualization and quantification of the migration of these labeled inflammatory cells to specific brain areas is of high diagnostic relevance due to the enhanced probability of detecting active demyelinating disease [8]. Macrophage infiltration is orchestrated by other inflammatory mediators such as T lymphocytes, which attract circulating monocytes by secretion of cytokines. Correspondingly, labeling T cells [9] might provide a tool to detect inflammatory events at an even earlier time point, potentially prior to the occurrence of significant tissue damage. The use of USPIOs for MRI [10], their ability to tag T cells [11], and the possibility to track iron-tagged T cells in vivo by using MRI [12] have been demonstrated. The sensitivity of the technique allows detection of single T cells labeled with ferumoxides in vitro [13]. In contrast to macrophages, T cells cannot be labeled in situ by systemic administration of contrast agents due to lack of phagocytotic activity. Instead, they have to be harvested and labeled in cell culture. Several methods to increase the efficiency of cell labeling by using dextran-coated FeOs have been reported. One approach uses the transativator of transcription (Tat) peptide of the human immune deficiency virus 1 (HIV-1) to introduce tagged nanoparticles into the cells [14,15]. A more frequently used method is based on complexing FeOs with polycationic transfection agents (TAs) to form complexes that should enter cells [16]. TAs are positively charged macromolecules commonly used to shuttle foreign DNA into mammalian cells by formation of TA/DNA complexes [17]. There are two principal classes of TAs available: lipid-based agents such as lipofectamine, Lipofectamine2000, and FuGENE6, and polyamine polymers such as poly-
The aim of the current study was to determine whether, by combining commonly available TAs and FeOs approved for clinical use, freshly extracted and primary cultured T cells can be efficiently and reliably labeled to an extent sufficient for the in vivo MRI detection. The efficiency of this labeling technique and the labeling yield was assessed by using different readouts including the measurement of the transverse relaxation rate
Materials and Methods
Ferumoxide Particles
Labeling experiments were carried out with two kinds of negatively charged FeO particles: Endorem particles (Guerbet®, Rueil-Malmaison, France) are approximately 140 nm in size according to the supplier, and Sinerem (Guerbet®) particles have the same shape but are smaller in size (40 nm in diameter). A stock solution of each compound was stored at 11.2 mg iron/mL. In order to determine the optimal FeO/TA ratio to form stable FeO/TA complexes, different concentrations of FeOs (in micrograms per milliliter) were tested.
Transfection Agents
FuGene6® (Roche, Basel, Switzerland) is a lipidic multicomponent transfection agent designed to shuttle DNA inside eukaryotic cells by forming vesicles. The concentration of the active transfecting substance in FuGene6 is not disclosed by the manufacturer. Based on the transfection rate (1 μg/μL of DNA to 2–3 μL of FuGene6) suggested in the user manual, we assumed that the stock solution contains at least 1 mg/mL of active reagent.
Poly-
Lipofectamine™ 2000 (Invitrogen, Basel, Switzerland) is a lipid-based TA widely used to transfect eukaryotic cells. The stock solution is supplied at a concentration of 1 mg/mL.
PEI (Sigma-Aldrich, Buchs, Switzerland) was supplied at 1 mg/mL. The polymer molecular weight was between 25 and 75 kDa.
FeO/TA Complex Preparation
FeOs and TAs were always mixed apart from cell medium to allow complex formation. FeO/TA mixes were prepared in advance by mixing FeOs and TAs in RPMI medium without serum. For all labeling experiments, the volume of Endorem or Sinerem stock solution was calculated to have a final concentration of 22.5 μg iron/mL in FeO/TA mix.
Different concentrations for each TA were tested. The final volume of the FeO/TA mixture was always 100 μL. The FeO/TA mixture remained under gentle agitation for 60 min before being added to cell culture complete medium.
Cell Culture and Labeling Conditions
T lymphocytes were extracted from spleens of young Lewis rats a few days before labeling. Organs were homogenized by using a Potter homogenizer and the solution was filtered several times through cell strainers with pores of 100, 70, and 40 μm in diameter. After the last wash (centrifugation with phosphate buffer), rat pan T cells were selected by means of Miltenyi magnetic beads (Miltenyi Biotech, Bergisch Gladbach, Germany) associated with antibodies against cells expressing α/β as well as
The
T cells were cultured in RPMI medium plus HEPES containing 1 mM sodium pyruvate, 1 × MEM nonessential amino acids, 50 μM 2-mercaptoethanol, 50 μM gentamicin antibiotic, 1 × MEM vitamins, 2 mM
To label T cells, 100 μL FeO/TA mixture was added to 106 T cells to have a final labeling medium of 2 mL. Cells were allowed to rest in a humidified atmosphere at 37°C in 95%/5% air/CO2 during the labeling process.
Determination of Physicochemical Properties of FeO/TA Complexes
The size and the zeta potential (ZP) of pure FeO particles and pure TAs as well as FeO/TA complexes were measured with a Zetasizer® (Malvern Instruments Ltd., Malvern, UK). Ferumoxide solutions at 22.5 μg iron/mL and TA at 5 μg/mL were diluted in filtered distilled water. The size and ZP of FeO/TA complexes were also measured in nanopure water at a final volume of 1 mL. Although the FeO concentration remained the same in all the measurements (22.5 μg/mL), different concentrations of TAs were tested, that is, 0.5, 1, 2.5, and 5 μg/mL. All measurements were repeated at least three times.
FeO/TA Toxicity and T Cell Activation
After a certain labeling period and before each application, the number of living cells was estimated under the microscope by fluorescein diacetate (FDA) exclusion. To have a quantitative confirmation of cell viability, the proliferate activity of FeO/TA-labeled T cells was evaluated using the 3-[4,5-2-yl]-2,5-diphenyl tetrazolium bromide (MTT) assay (Roche).
To determine the toxicity of FeOs or TAs separately, T cells were seeded onto 96-well plates and then incubated at 37°C in 95%/5% air/CO2 with different doses of FeOs or TAs. MTT reagent was added 1 hr after mixing TAs and FeOs and the whole mixture was incubated for 24 or 48 hr. Thereafter, the detergent reagent supplied within the MTT assay kit was added to each well and the solution was incubated in the dark at room temperature overnight. All measures were repeated three times and the mean value was used for statistical studies.
T cell activation was evaluated based on their level of interleukin-2 (IL-2) secretion. The Elispot™ assay is designed for the detection of IL-2-secreting cells at the single-cell level. We have used this method to monitor cellular responses in the presence of either FeOs and TAs separately or FeO/TA complexes.
Estimation of the Fraction of Cells Containing Iron Oxide Nanoparticles
Cells labeled with superparamagnetic FeOs were sorted by exploiting their magnetic properties (magnetic selection). After labeling, T cells were centrifuged and washed three times to remove the excess of free FeO particles in solution. After the last centrifugation, the pellet was resuspended in 2 mL of phosphate-buffered saline (PBS). Cells were counted and poured onto a magnetic column. Due to the magnetic field applied, FeO-labeled cells were retained in the column. The column was washed three times with PBS to remove unattached material. Thereafter, the magnetic field was switched off by removing the magnet surrounding the column and the remaining cells were eluted by adding 2 mL of PBS and then counted by FDA staining.
Measurement of Iron Content
X-ray fluorescence spectroscopy by wavelength dispersion was used to determine traces of heavy metals like iron within the cells. After labeling, washing, and counting, cells were suspended in distilled water with 0.1% Tris buffer. Under osmotic pressure and detergent activity, cells were lysed and the iron was released into the solution. The mass absorption coefficient of the pellet was calculated by measuring the scattered X-ray radiation (Compton or Rayleigh scattering) experimentally. The trace content of the analyte was determined from the net impulse rate of its appropriate fluorescence line and from the intensity of the background line(s) by using a matrix normalized curve.
MRI Estimation of Labeling Efficiency
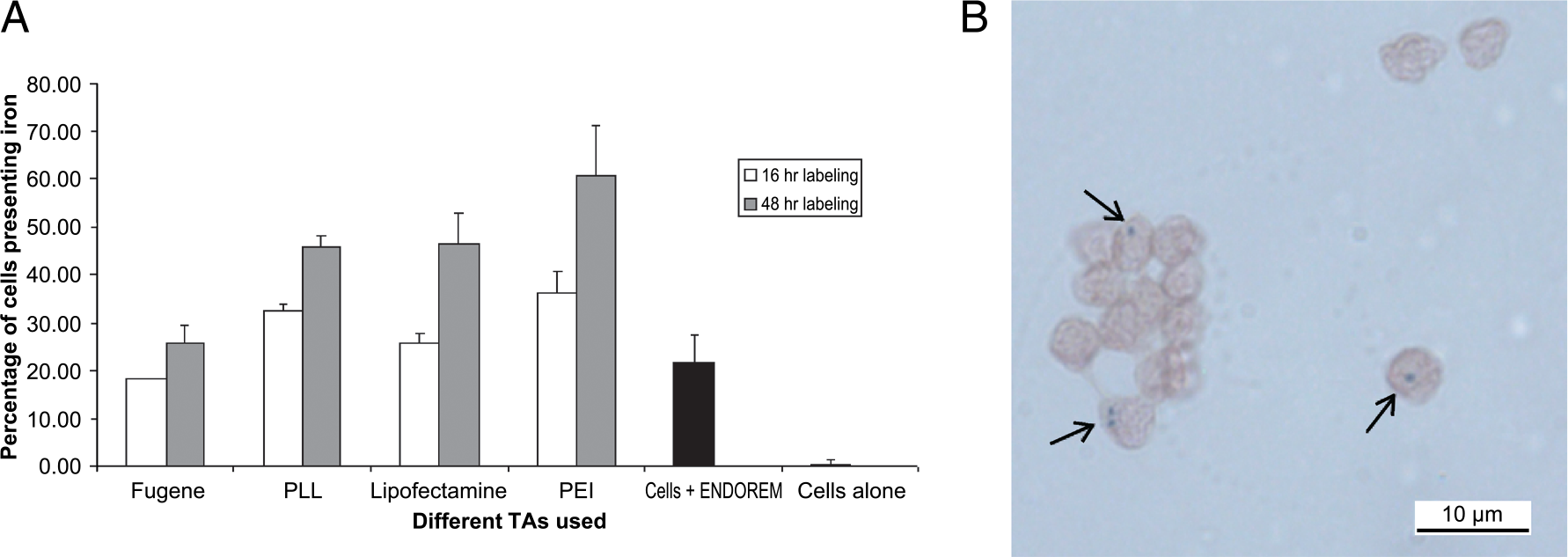
After each labeling procedures, cells were washed three times with PBS and resuspended in 1 mL of PBS containing 3% gelatin at a concentration of 106 cells/mL. Superparamagnetic nanoparticles such as FeOs lead to a significant increase of the transverse relaxation rate 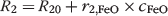

MRI experiments were carried out with a horizontal bore Pharmascan 70/16 spectrometer (Bruker Biospin GmbH, Karlsruhe, Germany) equipped with a self-shielded gradient insert of 90-mm inner diameter capable of switching 300 mT/m in 150 μsec. A 35-mm inner-diameter birdcage coil was used as transmitter/receiver device. The phantom consisted of a Plexiglas support containing Eppendorf vials filled with the corresponding cell suspensions.
Results
Physicochemical Properties of FeO and FeO/TA Complexes
Two important characteristics for nanoparticulate materials are the average size and the average ZP. Almost all particles in contact with liquid acquire a surface charge. The ZP is an indicator of this charge that predicts the stability of a colloidal suspension. The greater the ZP, the more stable the suspension due to repelling forces in between particles overcoming their tendency to form aggregates. Average particle size and ZP for filtered FeO nanoparticles, Endorem and Sinerem, are given in Table 1. We determined an average diameter of Endorem nanoparticles of 95 nm rather than the 140 nm as indicated by the manufacturer. Negative ZPs have been determined for iron-based TAs (e.g., −15 and −19 mV for Endorem and Sinerem, respectively) in agreement with results from Kalish et al. [19], who found ZPs of −41.3 and −2.03 mV for Feridex and monocrystalline iron oxide nanoparticles (MIONs) particles in water. Discrepancies between ZP values are probably linked to differences in size, coating, and concentrations of tested particles.
Average and Standard Deviation Values Calculated from a Solution of Iron Particles (Endorem or Sinerem at 22.5 μg iron/mL) Diluted in Filtered, Deionized, Distilled Water at pH 7

Concerning the particle size, average and standard deviation calculations are weighted with particle occurrence in solution. In this case, the standard deviation represents the breadth of the particle-size distribution curve.
When Endorem iron beads are mixed with TAs (Figure 1A), the average size of resulting particles is around 114 nm. However, certain mixing conditions involving lipidic TAs (e.g., FuGene and Lipofectamine) at low concentration (0.5mg/mL) induce the clustering of Endorem particles together, resulting in the formation of larger agglomerated iron clusters. In a similar manner, aggregation of Sinerem particles was observed at low concentration of TAs when using polymeric TAs such as 75-kDa PEI25 and 250-kDa PLL125 (Figure 1B). For all other conditions, an average diameter of Sinerem/TA complexes of 64 ± 20 nm has been determined.
The ZP of Endorem/TA complexes was found to be almost always positive except at low concentration of lipid TAs (e.g., FuGene6 and Lipofectamine). The average ZP obtained is 23.2 mV, and 250-kDa PLL125 forms the highest positively charged particles (Figure 1C). Concerning Sinerem/TA particles, the ZP is negative when lipid TAs are used except for an excess concentration of Fugene (Figure 1D). Using polyamine TAs, ZPs reach values of about 17 mV. Again, 250-kDa PLL125 led to the formation of the highest positively charged particles.
FeO Labeling Studies of T Cells without Using TAs
Previous studies have shown that larger iron particles (e.g., Endorem) led to better labeling efficiency [20]. To evaluate the effect of FeO nanoparticles on rat T cells, we carried out a series of experiments using Endorem as labeling substrate without adding TAs to enhance labeling efficiency.
Our results indicated that primary cultured T cells can be labeled by just adding FeO nanoparticles to the culture medium. Based on previous studies that demonstrate that primary hematopoietic cells incorporate iron particles after 2 hr incubation without TA [21], we decided to evaluate labeling efficiency for a time interval between 2 and 48 hr. Figure 2A shows that the

Average size of (A) Endorem (SPIO) and (B) Sinerem (USPIO) particles, respectively, when mixed with different TAs. Zeta potential for the various particle–TA clusters are shown in (C) and (D). The concentrations for TAs tested were 0.5, 1, 2.5, and 5 μg/mL.
The cell concentration in the labeling medium had an influence on the labeling efficiency: The higher the cell concentration in the solution, the lesser the

Optimization of T cell labeling with SPIO alone. Influence of incubation time (A), SPIO concentration (B), and cell concentration (C) on FeO uptake by primary cultured T cells as reflected by the transverse relaxation rate. The control value shown in (A) and (B) is the value for the T cells in the absence of any FeO nanoparticles (black bars). The Y axis shows the relaxivity per cell r2,cell (in sec−1) in the cell suspension. The number of cells were 106 cells/mL for (A) and (B).
Toxicity and Cell Activation
An important aspect in cell labeling studies is that the label or any of the co-added substances do not affect the normal cell physiology. Therefore, potential toxic effects of the labeling procedures have to be carefully evaluated. A commonly used toxicological readout is the cell growth rate. First, we evaluated the influence of FeO concentration on T cell growth rate. An increase in the concentration of FeO particles decreased the number of cells in the culture dish, indicating a cell toxic effect at high FeO concentrations (Figure 3A). In comparison to smaller FeO particles (Sinerem), the negative effect of Endorem particles on T cell growth was more pronounced at concentrations exceeding 25 μg iron/mL. This concentration probably allows the most efficient labeling of T cells without significantly affecting the cell growth rate. Based on these results and in agreement with published data [23], we decided to use nanoparticles at an iron concentration of 22.5 μg/mL for further labeling experiments.
In a second study, potential toxic effects caused by TAs were investigated (Figure 3B). After 48 hr of culturing in the presence of different TA concentrations, the MTT test showed that all TAs seemed to impair T cell growth and cell survival in vitro. FuGene6 and different-sized PLL appeared less toxic than Lipofectamine and PEI. With the exception of FuGENE6, a TA dose greater than 2 μg/mL dramatically impaired T cell survival for all TAs. For FuGENE6, the corresponding “toxic” level was reached at concentrations higher than 5 μg/mL. Toxicity tended to increase with higher molecular weight of PLL at concentrations between 1 and 2 μg/mL. Analogous results were observed when labeling dendritic cells (antigen-presenting cells: LADMAC cells) with PLL and FeO [18]. Lipofectamine seemed to be the more toxic compared to the other TAs. In particular, PEI had less toxic effects than Lipofectamine at all concentrations tested.
Third, as the purpose of current work was to evaluate the feasibility of labeling rat T cells for imaging studies in vivo, it was primordial to test whether T cell reactivity and activation were altered by the presence of FeOs, TAs, or both. IL-2 is well known to play a critical role in T cell expansion [24,25] and to participate in natural killer (NK) cell activation [26–28]. Figure 4 shows the effects of FeOs and various TAs on the degree of IL-2 secretion by rat T lymphocytes. Compared to the native T cells, administration of Endorem alone to the medium did not affect IL-2 secretion. Similarly, Lipofectamine had no significant effect on T cell activation as concluded from the measured IL-2 levels. Significantly increased IL-2 secretion was observed when T cells were exposed to polycationic agents such as PLL (

T cell toxicity of FeO nanoparticles and TAs. Assessment of cell toxic effects as a function of the FeO concentration (A) and the TA used (B). In the MTT assay, the absorption intensity at 560 nm is proportional to the cell concentration in the suspension. All these experiments were carried out at the same time following incubation with FeO particles and TAs under the same conditions. The control value for cell cultures not exposed to TA or FeO particles is indicated by the dashed line.
Estimation of Cell-Labeling Efficiency
To estimate the efficiency of cell labeling, FeO-labeled T cells were collected by use of a magnetic sorting device (MiniMACS™ Separator, Miltenyi Biotec) exploiting the superparamagnetic properties of FeO nanoparticles. Based on the assumption that FeO-labeled cells should be retained in the column by the applied magnetic field, this approach is commonly used to select immune cells presenting FeO beads at their surface [29]. FACS analysis of MACS isolated cells demonstrated a clear predominance of T cells expressing only the CD3 receptor (CD3+) as well as cytotoxic T cells (CD3+/CD8+). As proven by the content of the negative MACS selection, the majority of monocytes, B cells, and NK cells had been removed from our experimental sample (Table 2).
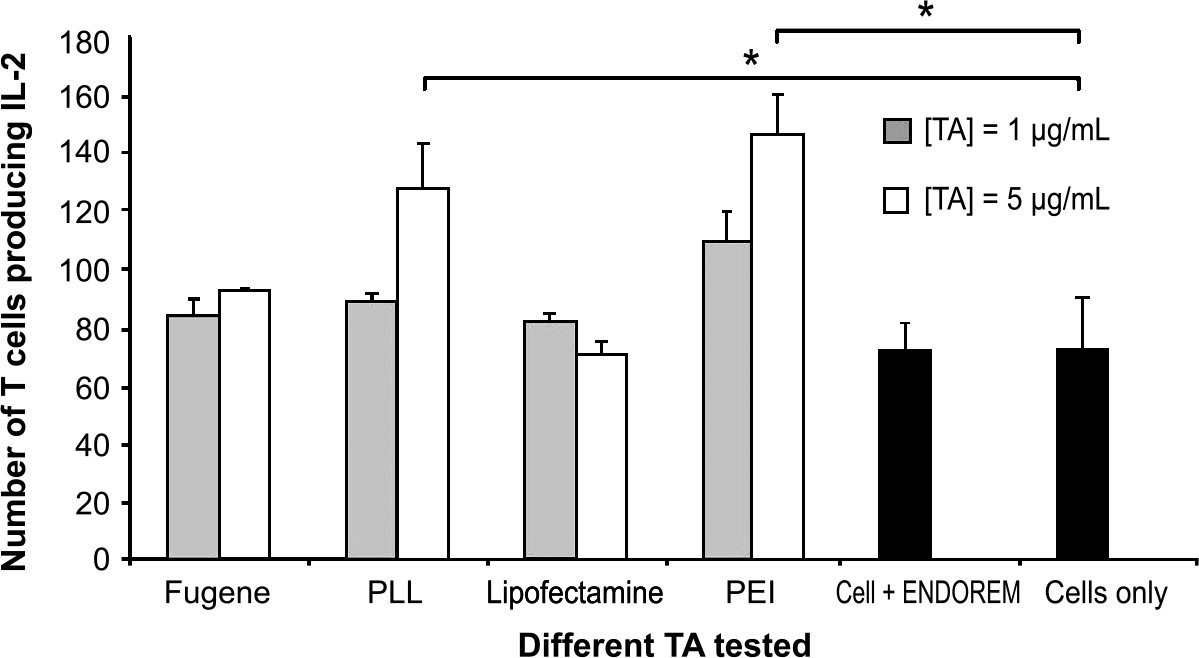
In our case, the fact that the labeling FeO particles are supposed to be internalized by the lymphocytes should not affect the interaction between the magnetic field column and FeO-tagged cells. As negative control, unlabeled cells were run over the column, and no retention of cells was observed. Hence, retention of cells is indicative of the presence of FeO labeling. Figure 5A shows the percentage of retained cells after 16 and 48 hr exposure to Endorem and different TAs. PEI yielded the highest labeling efficiency followed by PLL (with up to 60% of the cells carrying FeO labels after 48 hr) and Lipofectamine. In contrast, FuGene6 did not increase the uptake of FeO nanoparticles compared to FeO alone. Although other studies demonstrated no cell labeling in the presence of FeOs without TAs after 24 hr incubation [20], we found approximately 24% of rat T cells to be labeled with iron beads even in the absence of TAs. Optical microscopy observation of cells stained with Prussian blue confirmed that a fraction of cells carried FeO particles (Figure 5B, arrows).
FACS Phenotyping of Spleen and MACS Isolated Cells Was Performed by Using Fluorescent Antibodies Directed Against Receptors of CD3, CD4, CD8a, CD45RA, and CD161a.

Cell populations are given in percentages for 100% of living cells present in the sample before (first column) and after MACS selection (second and third column).
Experiments using fluorescence spectroscopy of X-rays confirmed the efficiency of PLL and PEI for labeling rat primary cultured T cells (Figure 6). Quantification of iron trace in the pellet of lysed T cells revealed almost five times more iron when PLL was used as TA compared to the non-TA enhanced uptake. This experiment demonstrated that polyamine TAs (PLL and PEI) led to significantly higher FeO loading to rat T cells than other TAs (lipofectamine and FuGene6).
T cell activation by FeO nanoparticles and TAs. IL-2 production of rat T cells in the presence of different TAs at concentrations of 1 and 5 μg/mL. The IL-2 production is proportional to T cell immune activation state. Black bars are the control values for IL-2 synthesis by T cells with and without SPIO. IL-2 secretion of T cells increases significantly following administration of PLL (p = .011) and PEI (p = .025) at a concentration of 5 μg/mL.
Efficiency of T cell labeling. (A) Percentage of cells containing FeO nanoparticles after labeling for 16 and 48 hr, respectively, for the different TAs. A magnetic selection of labeled cells has been made to select only cells doped with FeO. Black bars show control values for T cells exposed to SPIOs only. Minimal retention was observed for cells not exposed to iron (cells only). (B) Microscopic image of T cells with 100 × magnification after 16 hr labeling with SPIO in the absence of TA. FeO nanoparticles (see arrows) appear as blue dots (Prussian blue staining) in neutral red-colored T cells.
MRI Labeling
The decrease of the echo amplitudes in the CPMG experiment followed single exponential characteristics described by the transverse relaxation rate.

Assuming that all FeO particles within the sampling volume are sequestered in the cell, the total amount of FeOs per cell,

Figure 7 represents the average

X-ray spectroscopic evaluation of the cellular iron content after 48 hr of labeling for the various TAs. The black bar indicates the amount of iron in T cells labeled without addition of TA.
Discussion
The objective of this study was to compare the efficiency of the magnetic labeling of rat primary cultured T cells combining commonly available TAs and FeO nanoparticles. As magnetic tag, we used superparamagnetic dextran-coated Endorem, which exerts its effect on surrounding water molecules by increasing

Efficiency of T cell labeling assessed via transverse relaxation rates: Measurement of the r2,cell relaxation rate in test vials containing a 1-mL suspension of 106 cells labeled with 25 μg FeO particles/mL and 2 and 8 μL TAs. Values are given as relaxivity per cell and unit volume according to Equation 1b. The relaxation rates reflect the amount of iron in the sample and allows estimation the labeling efficiency (Equation 3). Results have been obtained as the average values of 10 measurements per TA either with SPIO or with USPIO. The control value is for cell labeled with SPIO only without TA.
Based on literature data [16,30] and our own in vitro results, we chose a concentration of about 25 μg iron/mL mixed with 1 to 2 μg/mL of TA and an incubation time of 48 hr to achieve high labeling of primary cultured T cells. An Endorem concentration corresponding to about 25 μg iron/mL SPIO and 2 μg/mL TA (TA/SPIO ratio = 2:25) was found to constitute thresholds for toxic effects, higher values leading to significant T cell toxicity as reflected by decreased growth rates, and the occurrence of cell death. A cell loss of more than 35% has been observed when using TA doses exceeding 2 μg/mL, confirming results obtained by Arbab et al. on TAs toxicity [18].
Large FeO particles (Endorem) turned out to be more toxic than smaller ones (Sinerem); however, they also led to better direct labeling of T cells in the absence of TA. As our culture conditions were the same in both cases, we can assume that the size of particles is the only factor affecting both the cell mortality and the enhanced labeling efficiency. Provided that particles are taken up into the cytoplasm, bigger particle sizes would imply more efficient endocytosis. The calculated size of the SPIO/TA particle at a ratio of 2:25 was of the order of 100 to 120 nm in diameter irrespective of the TA used (Figure 1A). However, the ZPs of these particles clearly depended on the TA mixed with FeO, whereas PEI and different-sized PLL led to a positively charged particle with a ZP of approximately +40 mV. The ZP of FeO adducts with lipophilic TAs such as FuGene6 and Lipofectamine was considerably smaller (ZP ≈+10 mV) or, as in the case of Lipofectamine, at concentrations less than 2 μg/mL even slightly negative (Figure 1C). Two features of the FeO particles seem to be relevant for high uptake of FeO/TA complexes: a particle size of the order of 100 μm (and potentially more) and reasonably high positive charge [32–34]. The latter is in line with the observation that membrane-penetrating peptides comprise a large number of positively charged amino acids such as arginine [35].
Although a previous study did not observe SPIO uptake in the absence of TA [36], we found that approximately 24% of the primary cultured rat T cells were loaded with FeO nanoparticles after 48 hr exposure to a culture medium containing only SPIO but no TA. This is probably due to the natural capacity of living cells to take up foreign complexes by endocytosis. Moreover, this capacity should be enhanced by the dextran coating of iron particles. Indeed, DEAE–dextran (diethylaminoethyl dextran) is widely used to transfect mammalian cells [37] leading to a remarkably reproducible inter- and intraexperimental transfection efficiency [38]. The exact location of the FeO particles within the cells, however, remains unclear. Microscopic cell analyses revealed the occurrence of such particles, both intracellular and extracellular, that are attached to the cell membrane (Figure 8A). The same pattern was found for FeO/TA-labeled cells. Although the presence of TA molecules at the surface of the FeO particles is supposed to enhance complex formation, thereby facilitating the penetration of cellular membranes, it appears that some particles remain sticking to the outer leaflet of the plasma membrane. Concerning iron that has been internalized into cells, confocal microscopy experiments using house-synthesized chimerical USPIO-Cy5.5 particles showed that iron within T cells is located in the cytoplasm (Figure 8B).
X-ray spectroscopy experiments and NMR

Microscopy of labeled T cells. (A) Different examples of primary cultured T cells labeled with SPIO + PLL after cytospin centrifugation. The overview image yields an estimate on the percentage of labeled cells. The three examples below show a cell containing FeO particles in the cytoplasm (left), a cell with FeO particles sticking to the cell membrane (middle), and a dead cell full of iron (at the bottom of the right image). Prussian blue staining was used to visualize the iron content. (B) Confocal image and 3-D reconstruction (Imaris®
It has been established that intracellular tagging of T cells with 0.02 pg iron SPIO per cell can be achieved [22]. By using TAs, the ratio between FeO particles and TA should be defined in order to achieve a proper formation of complexes small enough for efficient endocytosis. In our studies the formation of aggregates between T cells and FeO/TA complexes has been observed over a considerable range of TA concentrations (i.e., 0.5 and 1 μg/mL) due to the adherent behavior of activated T cells (e.g., with PEI in Figure 8C). Although PEI does not form iron clustering (Figure 1A), it had the strongest effect on the IL-2 secretion of T cells (Figure 4). Not surprisingly, PEI displayed the most severe toxic effect on T cell viability of all the TAs evaluated (Figure 3B) at low concentrations. On the other hand, labeling efficiency of the SPIO–PEI combination was highest, resulting in approximately 60% of T cells containing/presenting FeO. This indicates that the increased immune activation state of T cells tends to increase the labeling efficiency. This higher number of selected lymphocytes not only includes T cells with internalized iron particles but also a large number of T cells with FeO/TA complexes sticking to their surface. Figure 8C shows clear aggregations of activated T cells after the SPIO–PEI labeling process. Staining of these cells with annexin-V-based fluorescent dye (Vybrant™ Apoptosis Assay, Molecular Probes) revealed high fluorescence intensity in such clusters. As annexin V targets externalized phosphatidylserine, which migrate to the outer leaflet of the plasma membrane during apoptosis, the high signal intensity is indicative of programmed cell death. We speculate that the formation of aggregates of activated T cells with FeO/TA complexes might in fact lead to premature cell death.
Another undesired consequence of this kind of surface labeling is the ability of iron particles to interfere with the basic function of cell surface signaling. FeO particles sticking to the outer membrane of the T lymphocytes could inhibit the normal immune response by steric hindrance of the antigen access to the cell surface receptors.
In summary, our results showed that labeling of primary cultured rat T cells after a fresh extraction from the spleen and thymus is feasible. Mixing of TA with dextran-coated FeO nanoparticles led to the formation of complexes, which enhances the labeling efficiency, compared to spontaneous uptake in the absence of TAs. Determination of physicochemical properties of the FeO/TA particles demonstrated that large polymers of PLL led to the formation of the highest positively charged particles suited for efficient uptake. Moreover, qualitative and quantitative assessment of iron content within cells showed that the number of labeled T cells was highest when using large PLLs as TAs. Hence, from a practical point of view and balancing toxicity versus labeling efficiency, the combination of SPIO (22.5 μg/mL) and PLL (1.5 μg/mL) yielded the best results for labeling primary cultured T cells. Although PLL showed no toxicity at concentrations used routinely (i.e., about 1 μg/mL), our findings revealed that the use of polyamine TA provoked a significant T cell activation. As a consequence, labeling occurs not only intracellularly but also on the surface of cells. This labeling is not negligible and may result in premature phagocytosis of labeled T cells by macrophages, which could lead, for instance, to unspecific labeling of macrophages. By providing a detailed characterization of the biophysical properties of FeO particles coupled with specific TAs and their capacity to label primary cultured T cells, the results from this study should aid in the determination of efficient procedures to tag T cells with FeO/TA complexes that would allow in vivo MR visualization after adoptive transfer of cells.
Footnotes
Acknowledgments
The authors thank Catherine Cannet, Marinette Erard, and Brigitte Greiner (Novartis Institutes for Biomedical Research) for technical support on histology and microscopy; Dr. Rainer Kneurer for helpful discussions on the biophysical properties of dextran-coated iron particles; and Dr. Claire Corot (Laboratoire Guerbet, Paris) for providing Sinerem (AMI-227).
