Abstract
Tumor imaging by ultrasound is greatly enhanced by the use of ultrasound contrast agents (UCAs), stabilized, gas-filled bodies. They are generally less than 7 μm to pass freely through the capillary bed. Development of a nano-sized agent would enable them to extravasate through the leaky pores of angiogenic vessels. We describe the development of an echogenic, nano-sized polylactic acid UCA by adaptation of a salting-out method. The viscosity of the initial colloidal suspension (concentration and molecular weight of protective colloid [polyvinyl alcohol (PVA)] and concentration of polymer) was key in determining particle size and polydispersity (increasing viscosity increased both). Addition of the porogens ammonium carbonate and camphor, required to produce hollow echogenic capsules, also increased the size (eg, 5 wt% PVA, mean solid nanocapsule size 386 ± 25 nm, polydispersity index 0.367 ± 0.14, and mean UCA size 640 ± 18 nm, polydispersity index 0.308 ± 0.027). Viscosity had the opposite effect on echogenicity of the resultant nano-UCA, which ranged from 21.69 ± 0.78 dB for 2% PVA to 12.1 ± 0.8 dB for 10% PVA. The UCA prepared with 10% PVA, however, had a longer half-life in the ultrasound beam (t½ > 15 minutes vs t½ < 10 minutes), suggesting a thicker shell. Optimization will require compromise among size, echogenicity, and stability.
Ultrasound Contrast Agents
Tumor imaging by ultrasound is greatly enhanced by the use of ultrasound contrast agents (UCAs), gas-filled bodies stabilized by surfactants, lipids, or polymers. 4 Ferrara and colleagues showed that the frequencies and pressures used in medical ultrasonography can generate a radiation force that displaces UCAs away from the source, pushing them toward the vessel wall and, hence, toward the pores in these vessels, through which a nanocapsule might be forced.5,6 In addition, abnormal angiogenic tumor vessels have uneven, slow, and multidirectional blood flow; hence, the residence time of the agent passing through the tumor is increased, adding to the potential for extravasation.2,3 These phenomena could be used to force nanocapsules through pores in the vasculature, resulting in a concentration of contrast in the tumor, reducing signal to noise, aiding visualization by uniquely identifying the tumor, and transporting any drug payload to the tumor interstitia.
Tumor Angiogenesis and Nanoparticles
Tumor angiogenesis is characterized by branching vessels with irregular diameters ranging from 10 to 200 μm7 and a lack of defining structures such as arterioles, capillaries, or venules. A crucial property of angiogenic vessels is that their rapid growth from established vessels renders them “leaky.” 8 The pore cutoff size has been reported from as low as 380 nm up to 780 nm.7,9 Pore size is an indicator of the stage and type of tumor being analyzed, with leakiness increasing concurrently with the histologic grade and malignant potential of the tumor. Inflammation has also been shown to increase the pore size of vessels, with nanoparticles up to 700 nm having been observed passing through inflamed capillary walls. 10 In most cases, the lymphatic drainage is also severely compromised, resulting in an accumulation of the nanoparticles at the tumor site. This phenomenon is generally referred to as the Enhanced Permeability and Retention (EPR) effect, and researchers are investigating methods of exploiting this for drug delivery.
Although the EPR effect can achieve high local concentrations of polymeric and nanoencapsulated drugs at a tumor site (10- to 50-fold higher than in normal tissue), accumulation is not always rapid and can take up to 2 days to reach a maximum. 11 As well as this “passive” targeting, nanoparticles can be conjugated with a ligand that will specifically interact with a cell surface receptor, that is, active targeting. There are various advantages to actively targeted entities; for example, multiple ligands can be attached (multivalent particles) and hence provide strong binding, and the attached capsules can be more readily taken into cells by virtue of receptor-mediated endocytosis. The group of Couvreur reported on both phenomena, employing folate grafted to PEGylated cyanoacrylate nanoparticles.12,13 Conjugated nanoparticles had a 10-fold higher apparent affinity for the folate-binding protein than free folate and were selectively taken up by the folate receptor-bearing KB3-1 cells. Posttargeting analysis of the cells found that the capsules were localized in the cell cytoplasm. However, an agent bearing targeting ligands must first extravasate to contact the cell that bears the receptors.
We propose here a nano-sized contrast agent that will be both visualized and forced toward the target cell by ultrasound. Other studies in our laboratory are investigating the potential of combining the nanocapsules with active targeting of these agents, following preliminary studies with a similar micrometer-sized agent. 14
Nanoscale Contrast Agents
Development of nanoscale UCAs for tumor imaging through angiogenesis (pore) targeting, in conjunction with new and improved imaging techniques, is an attractive concept. Acting against this idea is the fact that backscatter cross section
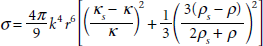
where 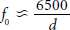
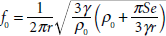
Patel and colleagues showed that when insonated at 2.25 MHz, hard-shelled “submicrometer” agents ranging in size between 500 nm and 1.3 μm expanded up to 18 times and an agent with a mean initial diameter of 800 nm expanded up to 30 times for a peak negative pressure near 1.5 MPa.
19
These experimental observations were in agreement with simulations based on the Rayleigh-Plesset (R-P) model. Improved signal to noise can be achieved when ultrasound harmonic components such as subharmonics and ultraharmonics (3
To date, the most successful nano-UCAs that have been described consist of liquid perfluorocarbon emulsions and only become echogenic when accumulated at the site of interest. 24
Contrast Agent Fabrication
Using our experience in producing hollow micrometer-sized UCAs coated with α-hydroxy poly esters,25,26 we investigated adaptation of a salting-out method to produce a nano-sized poly(D,L-lactide) (PLA) agent based on the same principle. The concept behind our micrometer-sized agents is that of encapsulating a sublimeable substance (porogen) that could subsequently be removed by lyophilization, leaving behind a void that is then charged with a gas. 25 The salting-out method relies on phase inversion from a water in oil to an oil in water emulsion brought about by adding a high-salt, aqueous solution of a protective colloid to a solution of polymer in a water-miscible organic solvent such as acetone. The salt causes salting out of the organic solvent, which is subsequently removed by the addition of a large volume of water into which the organic solvent diffuses. 27 The great advantage of this method for pharmaceutical purposes is that it does not involve toxic solvents. This article describes our investigations into the potential for salting out as a method for generating echogenic, polymer-shelled, nano-sized UCAs.
Materials and Methods
PLA 100 DL Low IV (Lakeshore Biomaterials, through SurModics Pharmaceuticals, Birmingham, AL, lot W2297-587) was purchased from Alkermes (Cincinnati, OH). Pharmaceutical grade ammonium carbonate was purchased from J.T. Baker (Phillipsburg, NJ), and (1R)-(+)-camphor was purchased from Sigma Chemical Co. (St. Louis, MO). Samples of polyvinyl alcohol (PVA), 88% mole hydrolyzed with molecular weights (MW) of 6,000 Da and 25,000 Da were purchased from Polysciences Inc (Warrington, PA), and PVA (Mowiol 4-88 and 8-88) with 88% mole hydrolyzed with a MW of 31,000 Da and 67,000 Da, respectively, and magnesium chloride hexahydrate were purchased from Sigma Aldrich (Fluka, St. Louis, MO). Acetone, hexane, methylene chloride, tetrahydrofuran, sodium chloride, potassium chloride, and isopropyl alcohol were all reagent grade and purchased from Fisher Scientific (Springfield, NJ).
Nanocapsule Preparation
The salting-out procedure described by Allémann and colleagues
27
was modified to produce echogenic capsules, incorporating camphor and ammonium carbonate as removable cores.
25
The organic phase consisted of camphor (10% w/w) and PLA 100 DL (2-17 wt%) dissolved in acetone (stirred with a magnetic stirrer), whereas the outer aqueous phase consisted of 60% w/w magnesium chloride hexahydrate, PVA, and deionized (DI) water. The aqueous to organic weight ratio was held constant at 2.5:1. To generate the first emulsion, 1 mL of 1 M ammonium carbonate (first aqueous phase) was added to the polymer-camphor solution and pulse sonicated at 110 W for 30 seconds (rate of 3 seconds on/1 second off). The second aqueous phase (20 g) was then added dropwise to the emulsion under mechanical stirring (BD6015 bench top, Caframo, Wiarton, ON) at 2,000 rpm with a three-blade propeller for ≈ 10 minutes. DI water (50 mL) was then added under stirring to cause acetone diffusion and create nascent nanoparticle suspension. The nanocapsules were collected and washed by centrifugation for 20 minutes (3 times) at 15,000 rpm (≈ 30,000
Acoustic Characterization
To determine the ability of the bubbles to generate backscattered ultrasound signals, in vitro tests were conducted at 37°C in a 100 mL custom-made vessel equipped with an acoustic window, set in an acoustic testing apparatus as shown in Figure 1, described previously. 23 The freeze-dried microcapsules were weighed and suspended in phosphate-buffered saline (PBS), and increasing doses of the suspension were added by pipette into 50 mL of PBS in the sample chamber at 37°C. The suspension was stirred with a magnetic stirrer and interrogated at 5 MHz with a spherically focused transducer (Panametrics, Waltham, MA) with a 12.7 mm diameter, focal length of 50.8 mm, −6 dB bandwidth of 91%, and a pulse length of 1.2 mm. 23 The frequency was chosen based on the medical imaging range and for comparison with previous results obtained in our laboratory. 28
A pulsar/receiver was used to pulse the transducers at a pulse repetition frequency of 100 Hz, resulting in a peak positive pressure amplitude of 0.69 MPa at the focus as measured by a 0.5 mm polyvinylidene fluoride (PVDF) needle hydrophone. The received signals were amplified and signal enhancement was analyzed using a

In vitro acoustic testing setup to test ultrasound enhancement and stability under simulated in vivo conditions (phosphate-buffered saline, pH 7.4, 37°C).
Size Distribution and Polydispersity Index
A Brookhaven Instruments Dynamic Light Scattering 90Plus Particle Size Analyzer (Holtsville, NY) was used to determine the relative size of the solid capsules and the polydispersity index (PDI). A concentration of 1 μg/mL was prepared of capsules in filtered (0.2 μm) PBS. The suspension was placed on a vortex mixer for about 2 minutes or until fully dispersed. The samples were run at 25°C, 678 nm wavelength (90°). For each sample, the analysis was performed a minimum of three times until the sample stabilized. The average of the maximum peak is reported.
A Malvern Zeta Sizer (nanoseries) particle-size analyzer (Malvern, UK) was used to determine the size, PDI, and zeta potential of hollow capsules. A concentration of 1 μg/mL of capsules in 0.2 μm filtered DI water was prepared and vortexed for 2 minutes to fully disperse the capsules. The samples were run at a temperature of 25°C using a polystyrene latex refractive index of 1.590. All values are quoted ± standard deviation (SD).
Morphologic Examination: Environmental Scanning Electron Microscopy
A Phillips (Eindhoven, The Netherlands) XL-30 scanning electron microscope (SEM) was used to image surface morphology. Freeze-dried samples were mounted on a metal stub using double-sided tape and platinum sputter coated prior to viewing.
Statistical Analysis
Statistical testing was performed using GraphPad
Results
Concentration of Protective Colloid: Influence on Particle Size for Solid Nanocapsules
An important step in preparing an emulsion is the creation of the appropriate droplet size, which, in turn, influences the final mean size of the particles. Parameters of the organic and aqueous phases that alter viscosity, such as PVA and PLA concentration and MW, were studied in the preparation of solid capsules to determine a basis to explore the formulation of echogenic particles.
Particle size decreased with increasing PVA concentrations, as demonstrated in detail in Table 1, together with PDI. The mean Z average diameters ranged from a high of 590.6 ± 56.8 with 2 wt% PVA to as low as 213.7 ± 11.3 using 15% PVA.
As the particle size dropped with increasing PVA concentration (viscosity), so did the PDI, approaching that of a monodispersed nanocolloid at 15%. At a PDI < .05, a dispersion can be considered to have narrow size distribution; for PDI > .05, the system is polydispersed. 29
Influence of Polyvinyl Alcohol (25 kDa), Concentration on the Particle Size, and Polydispersity Index
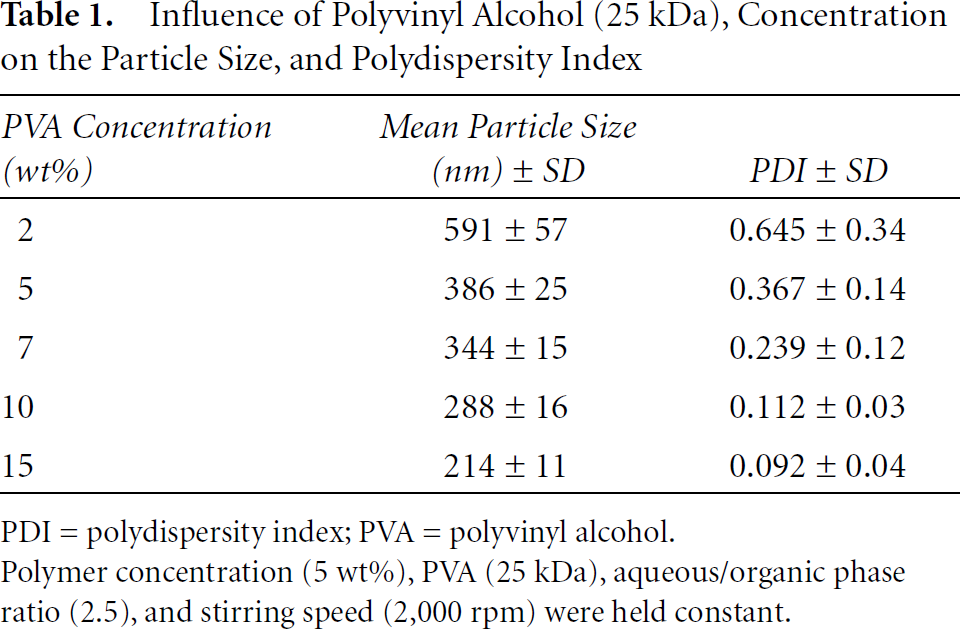
PDI = polydispersity index; PVA = polyvinyl alcohol.
Polymer concentration (5 wt%), PVA (25 kDa), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
Influence of the MW of the Protective Colloid on Particle Size
Given that the previous results confirm that viscosity impacts particle size, the effect of PVA MW was investigated next, choosing the PVA concentration of 10 wt%, which gave the smallest particle size, and a concentration of PVA, at which the solution was still manageable. A PLA polymer concentration of 5% was used and a constant hydrolysis of 88% on the PVA was maintained. As pointed out by Nakamae and colleagues, PVA is a popular stabilizer owing to its newtonian fluidity, superior primary wet tackiness, high strength, and creep-resistant film properties, and partially hydrolyzed PVA has high surface activity owing to a block distribution of residual acetyl groups.
30
Within the MW range tested (6, 25, 31, and 64 kDa), particle size varied linearly up to a PVA MW of 31 kDa ranging from 258 ± 25 nm, to 297 ± 24 nm, to 310 ± 22 nm. The value leveled off toward 64 kDa at 323 ± 29 nm, with the expected positive trend as MW increased. However, the results were shown not to be statistically significant in terms of the difference between the four groups by a one-way fixed ANOVA (
Influence of the Concentration of Polymer on Particle Size
As with PVA, PLA concentration has the ability to affect the viscosity of the emulsion and hence to influence the final particle size. PLA percentage varied from 2 wt% through 17 wt%.

Influence of polyvinyl alcohol (PVA) molecular weight on particle size. Polymer concentration (5 wt%), PVA (10 wt%), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.

Influence of poly(D,L-lactide) (PLA) concentration on particle size. Polyvinyl alcohol (10 wt%), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
A small increase in particle size (259 ± 18 nm, 272 ± 18 nm, 310 ± 26 nm, and 362 ± 21 nm) was seen when PLA concentrations in the organic phase increased from 2% through 17% (Figure 3). Given that sample yield was only about 50% of the initial PLA weight used, the 2 wt% samples were sparse. The results were shown to be statistically significant in terms of the difference between the four groups by one-way fixed ANOVA. Furthermore, post hoc comparison (Newman-Keuls) showed that samples prepared with 2, 5, and 10 wt% PLA were not significantly different from each other (
Morphology of Solid Nanoparticles
The optimized particles determined from the previously described studies were imaged using the SEM. The image, presented in Figure 4, shows that although the predominant size is submicrometer, a proportion of the capsules approaches 1 μm in diameter.
Nano-Sized UCA
To create hollow nanocapsules, sublimeable porogens were added to both the organic and the aqueous phases during preparation and later removed through lyophilization, leaving a void. PVA with a MW of 25 kDa was chosen as surfactant, based on the size results reported above. It was shown that PVA with this MW has the ability to stabilize the emulsion and still produce solid nanoparticles at a mean size of about 298 nm. The higher MW leads to a more stable particle, which is directly related to the surfactant strength.
The concentrations of PVA and PLA, shown above to have a role in determining the final capsule size in the salting-out procedure, were examined when porogens were added to the aqueous and organic phases. The mixing speed of 2,000 rpm and aqueous/organic phase ratio of 2.5 were held constant, as in the previous studies. Concentrations of 1 M ammonium carbonate in the aqueous phase and camphor (10 wt% of the weight of PLA) in the organic phase were used as the porogens, based on previous UCA studies in our laboratory. 26
Influence of Added Porogen on Particle Size at Different PVA Concentrations
In Table 2, the influence of added porogen on mean particle size for various PVA concentrations is presented and compared to solid capsules. Whereas size decreased as PVA concentration, and hence viscosity, increased, as before, the most interesting observation was that the addition of the two porogens increased the mean particle size in all cases, compared to the size for solid capsules. For 2%, 5%, 10% and 15% PVA, the hollow capsules increased in size over the solid capsules by 38%, 66%, 69%, and 22%, respectively. There was no discernible trend in PDI.

SEM of solid poly(D,L-lactide) (PLA) nanocapsules (×8,000 original magnification). Polyvinyl alcohol molecular weight (25 kDa) and concentration (10 wt%), PLA (5 wt%), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
Influence of Added Porogen on Mean Particle Size for Increasing Polyvinyl Alcohol Concentration
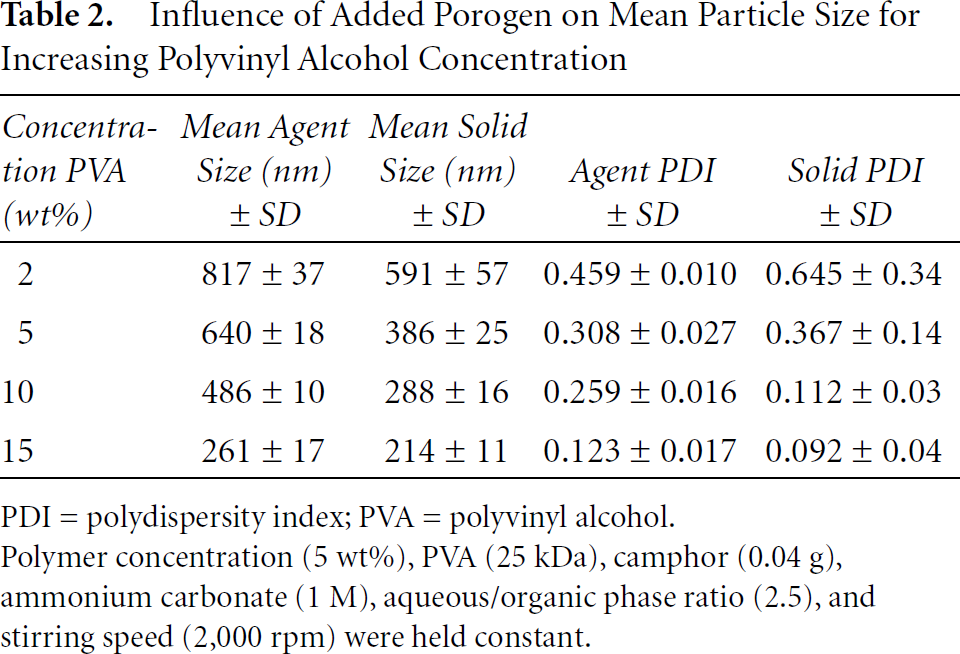
PDI = polydispersity index; PVA = polyvinyl alcohol.
Polymer concentration (5 wt%), PVA (25 kDa), camphor (0.04 g), ammonium carbonate (1 M), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
Influence of Added Porogen on Particle Size Using Different PLA Concentrations
The PLA concentration in the organic phase can also contribute to the viscosity and thus the particle size. PVA (25 kDa) was held constant at 5 wt%, and 0.04 g camphor, 1 M ammonium carbonate, 2.5 aqueous/organic phase ratio, and stirring speed (2,000 rpm) were used.
Table 3 shows the influence of added porogens. As with preparation of solid particles, no significant trend (
Influence of Added Porogens on Mean Particle Size at Different Poly(D,L-Lactide) Concentrations

PLA = poly(D,L-lactide).
Polyvinyl alcohol (25 kDa) concentration (5 wt%), camphor (0.04), ammonium carbonate (1 M), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
Morphology of Nanoparticles Prepared with Added Porogen
The optimized particles using a PVA concentration of 5 wt% and a PLA concentration of 5 wt% were imaged by SEM and are shown in Figure 5. The particles are well formed and spherical in shape. Although all particles are less than around 2.5 μm in diameter (measured against the 5 μm size bar), the population is not unimodal. Many submicrometer spheres are evident, together with a population of larger capsules up to the aforementioned 2.5 μm population.

SEM of poly(D,L-lactide) (PLA) salting out ultrasound contrast agent (×6,000 original magnification; bar size = 5 μm). Polyvinyl alcohol molecular weight (25 kDa) and concentration (5 wt%), PLA (5 wt%), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
Effect of Process Parameters on Echogenicity of the Nanocapsules
Capsules described in Table 2 (constant PLA 5%, increasing amounts of PVA [2 wt%, 5 wt%, 10 wt%, 15 wt%] in the aqueous phase) were examined to see if they displayed any echogenicity when insonated at 5 MHz. As seen in Figure 6, increasing the concentration of PVA resulted in a decrease in decibel enhancement, although the decrease was not directly proportional to the concentration.

Effect of polyvinyl alcohol (PVA) concentrations on the dose-response curve of poly(D,L-lactide) (PLA) nanocapsule. PLA (5 wt%), PVA (25 kDa), 0.04 g camphor, 1 M ammonium carbonate, aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant. Conducted at 37°C with a 5 MHz transducer. -♦- = 15 wt%; -■- = 10 wt%; -▲- = 5 wt%; -●- = 2 wt% PVA.

Effect of poly(D,L-lactide) (PLA) concentrations on the dose-response curve of nanocapsule. Polyvinyl alcohol (2 wt%), aqueous/organic phase ratio (2.5), stirring speed (2,000 rpm), camphor (0.04 g), and ammonium carbonate (1 M) were held constant. Conducted at 37°C with a 5 MHz transducer. -●- = 2%; -▲- = 5%; -■- = 10% PLA.
As the viscosity of the emulsion increased, the ability of the resulting nanocapsules to reflect ultrasound decreased. Maximum enhancement was seen with the lowest concentration (2 wt% PVA) of around 22 dB at a dose of 0.4 mg/mL, whereas 15 wt% PVA exhibited minimal enhancement (≈ 5 dB). It is highly encouraging that the nanocapsules are echogenic; however, it should be noted that the dose of particles required to achieve maximum echogenicity increased significantly (0.4 mg/mL vs 0.04 mg/mL) in comparison to the dose required to achieve the same enhancement using microbubbles (average diameter 1.2 μm) prepared from PLA of the same molecular weight in our laboratory by a water/oil/water emulsion method. 26 Replotting the data as enhancement at a dose of 0.4 mg/mL as a function of particle size (Figure 7) indicates a direct relationship between the mean size and echogenicity.
Effect of PLA Concentration
The concentration of polymer was not found to have a substantial influence on the dose-response curves of echogenic nanocapsules at low PVA concentrations, mirroring the observation of its lack of effect on the size of solid nanocapsules. Figure 8 shows the dose-response curves for a series using 2 wt% PVA, a protective colloid concentration that had been found to produce the most echogenic capsules. However, as the PVA concentration was increased, not only was the overall echogenicity decreased at any given PLA concentration, as seen in Figure 6, but at mutually high PLA and PVA concentrations, the echogenicity was also diminished; for example, at PVA of 10 wt%, a 2 wt% PLA concentration yields an echogenicity of 14.7 ± 0.7 dB, but this drops to 2.0 ± 0.17 dB at a PLA concentration of 10%. Table 4 tabulates the result.
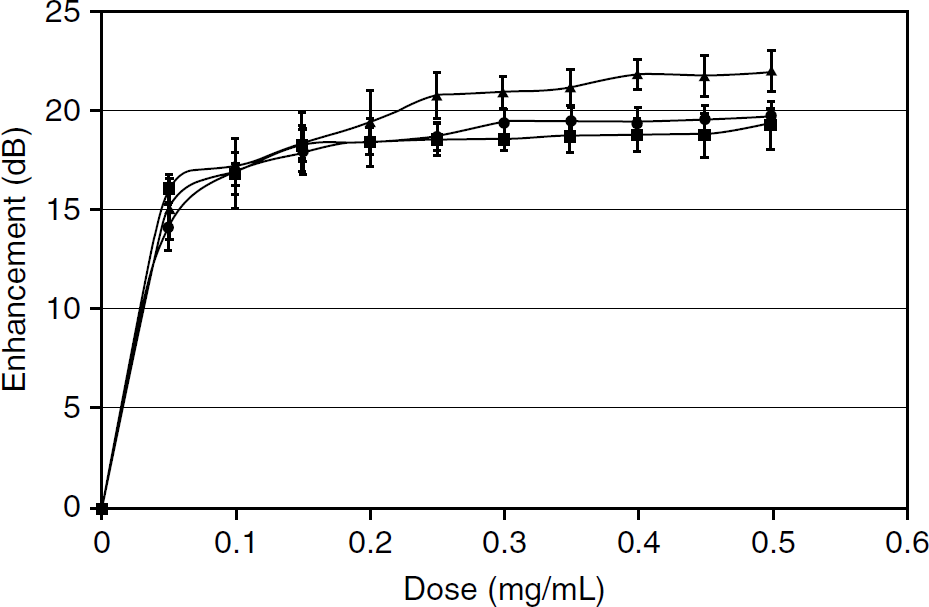
Relationship between enhancement and mean particle size of contrast agent in the salting-out procedure. Poly(D,L-lactide) (5 wt%), polyvinyl alcohol (25 kDa), camphor (0.04 g), 1 M ammonium carbonate, aqueous/organic phrase ratio (2.5), and stirring speed (2,000 rpm) were held constant. Conducted at 37°C with a 5 MHz transducer.
Stability of Echogenic Nanoparticles in the Ultrasound Field
Figure 9 shows the normalized signal decay over a period of 15 minutes for nanocapsule preparations prepared with different concentrations of protective colloid PVA. Increasing the concentration of PVA, which plays a role in shell strength, decreases the signal loss. Samples prepared with 10 wt% PVA were the most stable in the ultrasound field, losing about 40% of signal in comparison to 60% loss of signal exhibited by the 2 wt% PVA samples, which had a half-life of 9.4 minutes. One-way fixed ANOVA indicated a significant difference among the groups. Further statistical analysis (Newman-Keuls) showed that samples prepared with 2 wt% and 5 wt% PVA were not significantly different (α > .05), but there was a significant difference among the group of samples.
Influence of Poly(D,L-Lactide) and Polyvinyl Alcohol Concentration on Particle Echogenicity

PLA = poly(D,L-lactide); PVA = polyvinyl alcohol.
PVA (25 kDa), camphor (0.04 g), ammonium carbonate (1 M), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant.
When investigating the effect of PLA concentration on robustness under 5 MHz insonation, samples prepared with 10 wt% PLA showed the smallest loss, losing about 35% of signal in 15 minutes compared to 50% loss of signal exhibited by the 2 wt% PLA samples (Figure 10). As with the samples prepared at different PVA concentrations, one-way fixed ANOVA did show a significant difference among the three groups, and statistical analysis (Newman-Keuls) again showed that samples prepared with 2 wt% and 5 wt% PLA were not significantly different (α > .05), whereas a significant difference among the group of samples prepared with 2 wt% and 5 wt% in comparison to the 10 wt% PLA was observed (α < .05). The increased PLA concentration in the organic phase therefore enhances the stability of the capsule but also contributes to a decrease in the signal enhancement shown in the dose-response curves (see Figure 6).

Effect of polyvinyl alcohol (PVA) concentration on the normalized time response of poly(D,L-lactide) (PLA) nanocapsules. PLA (5 wt%), PVA (25 kDa), camphor (0.04), ammonium carbonate (1 M), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant. Conducted at 37°C with a 5 MHz transducer. -●- = 2%; -▲- = 5%; -■- = 10% PVA wt% prepared with 2 wt% and 5 wt% in comparison to the 10 wt% PVA (α < .05).

Effect of poly(D,L-lactide) (PLA) concentration on the normalized response of PLA. Polyvinyl alcohol (PVA) (5 wt%, 25 kDa), camphor (0.04 g), ammonium carbonate (1 M), aqueous/organic phase ratio (2.5), and stirring speed (2,000 rpm) were held constant. Conducted at 37°C with a 5 MHz transducer. -●- = 2%; -▲- = 5%; -■- = 10% PVA wt%.
Discussion
Contrast Agent in the Nano Size Range
In studying the salting-out method for development of a nano-sized UCA, we made particular note of the current literature, which indicated that nanocapsule properties were variable owing to the fact that the viscosity of the emulsion can change significantly depending on the concentration of protective colloid (PVA) in the aqueous phase. The results reported in the literature, however, appeared to be operator dependent. Murakami and colleagues showed an increase in diameter size, relating it to an increase in the viscosity of the emulsion. 31 In contrast, Allémann and colleagues reported a decrease in size with increasing viscosity, relating it to steric stabilization. 32 Nakamae and colleagues and Quintanar-Guerrero and colleagues showed similar decreased size with increasing PVA concentrations while preparing PLA particles via the emulsion diffusion method.30,33 Zweers and colleagues reported intermediate results and showed a decrease in size with PVA concentrations from 0 to 2 wt% but an increase in size from 5 to 10 wt%. 34 They attributed this effect to a decreased efficiency of mixing owing to the increased viscosity of the emulsion. Scholes and colleagues reported a biphasic effect with the particle size decreasing from 1 to 8 wt% but then increasing with increasing PVA concentrations up to 15 wt%. 35 The particle size is dependent on the raw droplet size formed during the emulsion stage. It is possible that the decrease in size can be attributed to the polymer chains of the PVA interacting at the emulsion droplet surface. Galindo-Rodriquez and colleagues investigated the physicochemical properties of PVA and reported a decrease in size with increasing PVA concentration. 36 This was believed to be due to a decrease in surface tension with an increase in steric stabilization. The increasing PVA percentage was proposed to present more PVA molecules available for stabilization, therefore producing an increased steric effect of absorbed PVA chains at the interface. At lower concentrations, there are not enough polymer chains to cover the emulsion droplet, decreasing uniformity and stabilization. As PVA increases to the point where there is a sufficient amount, the particle size becomes steady, resulting in uniform size particles. 36 We saw similar results both with solid (see Figure 2 and Table 1) and hollow (echogenic) particles (see Table 2); the viscosity of the emulsion exercised a critical influence on both size and PDI, both of which were much broader at lower PVA concentrations. The amount of PVA determines the size but also the stability and viscosity of the emulsion. As the PVA concentrations in the aqueous phase decreased, the particle size increased, as well as the ability to control the size distribution. When PVA concentrations increased, the emulsion also became significantly viscous, causing a negative effect on producing echogenic particles. The PLA polymer concentration was less influential (see Figure 3), indicating that the effect was primarily on stabilization of the emulsion droplets by the protective colloid. However, the results suggest two separate groups, defined by a limiting viscosity. A similar phenomenon in an emulsification-diffusion system was explained by Kwon and colleagues as an increase in the viscosity of the emulsion producing higher shear forces in mixing. 37 It was proposed that a limit is reached at which the viscosity of the emulsion is affected enough to have an influence on particle size. At lower PLA concentrations, the viscosity of the emulsion is not modified enough to affect the size of the droplets. However, increases in PLA concentration can lead to higher resistance to shear forces and less efficient stirring.
In all cases, the addition of porogens to covert solid particles to an echogenic agent resulted in an increase in size (see Table 3). This is similar to results obtained by Allémann and colleagues on encapsulation of the neuroleptic drug savoxepine methanesulfonate. 32 In the salting-out method, high concentrations of a salt and a protective colloid are first dissolved in water to form a viscous gel, which is then added dropwise to a solution of the polymer and the porogens to be encapsulated in a water-miscible organic solvent (acetone), under vigorous stirring. An oil-in-water emulsion is formed, and on addition of a large volume of water, the acetone diffuses from the nanodroplets into the aqueous phase. At this point, the polymer precipitates and encapsulates the other compound present in that phase. Salting out is associated with high encapsulation efficiencies in encapsulation of lipophilic compounds. 32 It is probable that in our case, only the hydrophobic porogen (the camphor) is encapsulated, and the ammonium carbonate added with sonication to the initial organic phase to mimic the successful methods using an water/oil/water emulsion for micrometer-sized agents is removed with the other salts.
The morphologic examination of the UCA (see Figure 5) gave results similar to those found for solid nanoparticles (see Figure 4). Although the predominant size is submicrometer, a proportion of the capsules are 1 μm in diameter and above, and this may have had a significant influence on the echogenicity seen in the samples.
Influences on the Echogenicity of the Nanocapsules
The primary task of this investigation was to produce not only nano-sized capsules but also a diagnostically viable UCA. Given that the resonance frequency is dependent on UCA diameter (
The time-response curves (see Figure 9 and Figure 10) suggest that the percentage of PVA used to produce the capsules affects the stability of the bubble. The results strongly suggest that there is a point where the increased concentration of PVA acts to increase the strength of the capsule's shell, decreasing signal decay over time. This observed effect of PVA on the agent's ability to withstand insonation may be due to several conditions. In any encapsulation process, the surfactant, such as PVA, contributes to the stabilization of the shell. The salting-out procedure also makes use of PVA but more in the role of stabilizing colloid. Additionally, in comparison to the double emulsion method we used to produce a micrometer-sized UCA, for example, isopropyl alcohol and hexane are employed to harden the shell of the microcapsule, a step that is absent in the salting-out procedure because salt hydration causes polymer precipitation. Therefore, it is hypothesized that the shell thickness is smaller when using salting out and, hence, the UCA is more susceptible to ultrasound energy.
The increased PLA concentration in the organic phase enhances the stability of the capsule but also contributes to a decrease in the signal enhancement shown in the dose-response curves (see Figure 9). Because it was noted that PLA concentration had little effect on the capsule size (see Figure 3), and assuming that the higher polymer concentration did not go toward making a larger number of capsules, the size results combined with the acoustic results suggest that the increased polymer concentration went into forming thicker nanocapsule walls. This points to the critical role of the surfactant PVA in determining emulsion size.
Conclusions
A novel approach was undertaken to develop a nano-sized UCA. By adjusting the protocol of the salting-out process and evaluating the effects of the process parameters, we were able to show the ability to produce echogenic capsules within the nano size range and control the characteristics of the capsules. It was determined that the viscosity of the emulsion has a considerable effect on particle size and echogenicity and PLA concentration affected stability and echogenicity. Increasing PVA concentration in the aqueous phase resulted in smaller particles but decreased in vitro ultrasound enhancement. There is, then, a tradeoff between viscosity and the ability to effectively produce an UCA via the salting-out method. Producing an UCA via the salting-out method has advantages. Various parameters can be altered, such as organic solvents or salting-out agents to tune capsules to the desired purpose. It is possible, then, to establish and modify specific experimental parameters to obtain the desired size and/or acoustic characteristics.
Future investigation is needed to establish the specific influences that parameters have to obtain particles of a desired size and echogenicity and to separate the two. The possibility that echogenicity is derived from a small population of micrometer-sized capsules nested among nonechogenic nanocapsules has to be considered.
Footnotes
Acknowledgments
We would like to thank Kelleny Oum for assistance with the SEM, Shashank Siri and Seunglee Kwon for invaluable discussions regarding preparation of nanocapsules, and John Eisenbrey for advice in preparing the manuscript.
Financial disclosure of authors: This work was funded in part by National Institutes of Health grant HL52901.
Financial disclosure of reviewers: None reported.
