Abstract
Finding the rate constants (rates of transition) of radiopharmaceuticals between organs or different regions of interest (ROIs) in nuclear medicine quickly and accurately provides rich physiological data that is essential for determining drug dosages and performing precise radiation dosimetry. This article describes a novel exact analytical approach to achieving this goal. This can be done in a very short time period at the beginning of treatment with application of tracer levels of radiopharma, and using activities in the ROIs construct the rate constants. Here, using randomly chosen rate constants, time activity curves (TACs) for a four-compartment model are created, and using different sampling regimens of these TACs, the rate constants are reconstructed in a large number of simulations. The least square error (LSE) and relative LSE in the reconstructed parameters are shown to be better than
Keywords
Introduction and Motivation
With the continued refinement of radiological science, the reliability of quantitative data has improved markedly. However, the results of nuclear imaging studies are principally considered qualitatively, with quantitative assessment typically limited to measurements of standard uptake value within regions of interest (ROIs) which correspond to organs and neoplasms. This has not been due to lack of effort by scientists, engineers, and clinicians; underlying mathematical and statistical complexities have limited progress, particularly for modalities that require time-dependent data acquisition in the clinical setting, relating the time activity curves (TACs) for different organs or ROIs in real time.
The physiologically based pharmacokinetic (PBPK) models with which we are concerned are a mainstay of the literature in physiology and nuclear medicine.1,2 In the language of differential equations, such models provide the equations of motion
Here, we present a novel approach to solving this inverse problem by applying a new mathematical result that produces highly accurate results in a very short time period and completely analytically. The mathematical theory and algorithm presented in Ellingson and Jafari
4
side-steps the need to perform an elaborate optimization and almost instantaneously solves the problem for the rate constants, based on sampling of the TAC from multiple ROIs. Furthermore, the proposed
Theory
In nuclear medicine and radiopharmaceutical therapy (RPT), the injected radiopharmaceutical (RP) distributes over time throughout tissues, commonly modeled as flow from one compartment to another throughout the body. A multicompartment model is often used to mathematically describe these transitions (see Steiner et al., 3 Hobbs, 5 Siegel et al., 6 Nestorov, 9 Gospavic et al., 10 and the references therein) in terms of a system of differential equations.
The solution of this system represents the activity in each compartment as a linear combination of exponential functions of the form:
for an 

An Example Compartmental Model.
where 
This problem, which is also known as the de Prony problem (Gaspard de Prony, 1755–1839), has received significant attention over the past 50 years by well-known numerical and computational analysts (see, e.g. Golub and Pereyra, 11 Pereyra, 12 Sarkar and Pereira, 13 Paluszny et al., 14 Hussen and He, 15 and the many references therein). Although not fully incorporated into practice, currently these parameters can be determined using numerical optimization methods that fit the data acquired on gamma cameras, single photon emission tomography (SPECT), or positron emission tomography (PET) machines. The limiting factors seem to be that data acquisition is required to be over long periods of time, frequently over equally spaced time points, and can be quite time consuming. A good fit may be achieved using gradient descent and similar methods 3 ; however, this involves long post-processing steps and the numerical methods are complex. Exponential curve fitting routines are also used and are based on least square error (LSE) minimization (regression)7,16; however, the latter methods suffer from isolation of each compartment and do not view the system as a whole.
Here, we present a complete analytical solution to this problem and demonstrate how these analytical methods may be implemented on nuclear medicine platforms to provide nearly instantaneous results. In particular, this solution eliminates the uniform sampling requirements of the de Prony/Sarkar
13
methods (matrix pencil method (MPm)) and can be achieved over short time periods up to statistical concerns. Having attained the
Existence and Uniqueness of the Solution
For nondegeneracy and as is the case in fully dissipative systems, as in RPT, we assume that the eigenvalues
With 
where
Let
What is occurring here is a change of basis in the expression of the original equation from a basis consisting of
If
For the detailed proofs of these results and several motivating examples, the reader is referred to Ellingson and Jafari.
4
Please note that the proofs presented in Ellingson and Jafari
4
extend far beyond applications of this method to RPT, and the numerical considerations of that article delve deeper into the algorithmic nuances of this method. Relating the
Determining the Rate Constants
Returning to the multicompartmental models presented in the introduction for PBPK models, and generalizing them appropriately to the case of
Since we are approaching this through the lens of nuclear medicine, we will no longer consider the concentrations of a pharmaceutical and instead consider the activity of an RP. These have a proportional relationship, so such a change is merely a rescaling of the system.
Let 
with the initial condition
where 
where 
where
For simplicity of presentation, we first consider the case in which 
where
Given the activity measurement, 

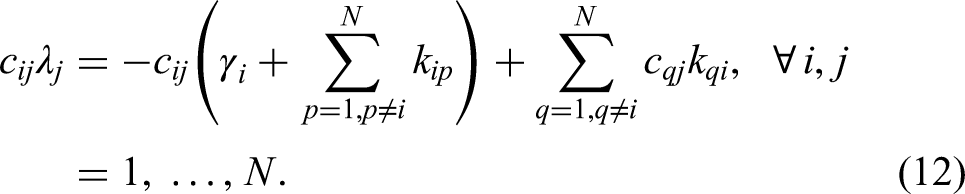
Nonzero Forcing Functions
We consider here forcing functions that are relevant to this application within nuclear medicine. Forcing functions that are being considered share the property that, at some point in time, uptake stops. That is to say, there exists some
Only using measurements taken at 
where
If we wish to also consider forcing functions which include an exponential decay factor in lieu of being zero-valued after some amount of time, this is also acceptable so long as the decay occurs on a shorter time scale than the dynamics of the system to allow for measurements to be taken without significant influence by
Results and Validation
To demonstrate the validity of this technique, we propose an example instance of the forward problem and, from the forward problem solution alone, reconstruct the system, that is, solve the inverse problem.
Suppose that the following randomly chosen 
with initial condition
Using these values in (8), we have
Solving the forward problem (see, e.g. Steiner et al. 3 ), this yields the model TACs in Figure 2.
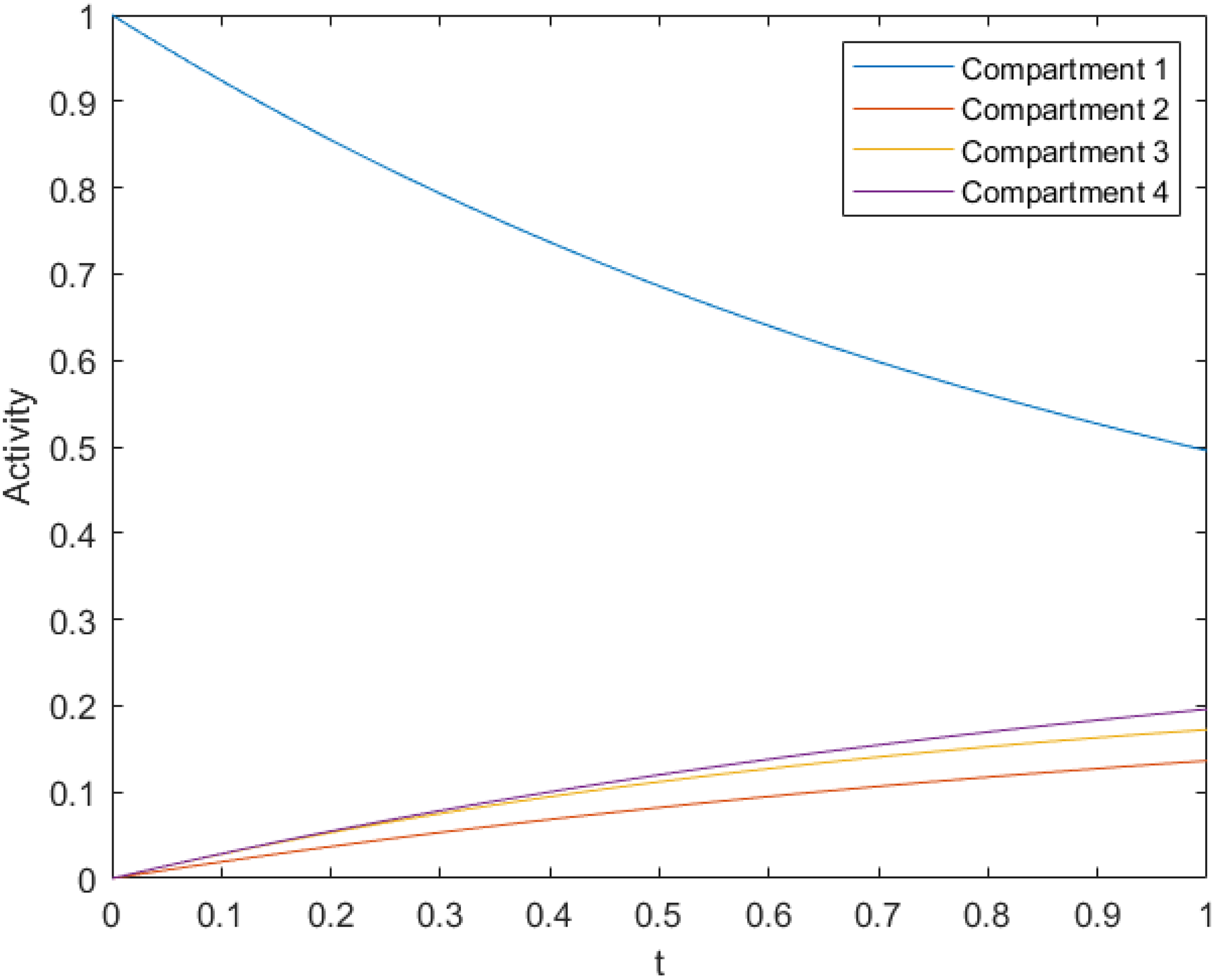
Time Activity Curves for Four-Compartment Model.
Sampling these curves on the interval 
The reader should compare the values in the matrices (14) and (15) entry-wise. The resulting relative error from 
This agreement is encouraging and repeatable with randomized
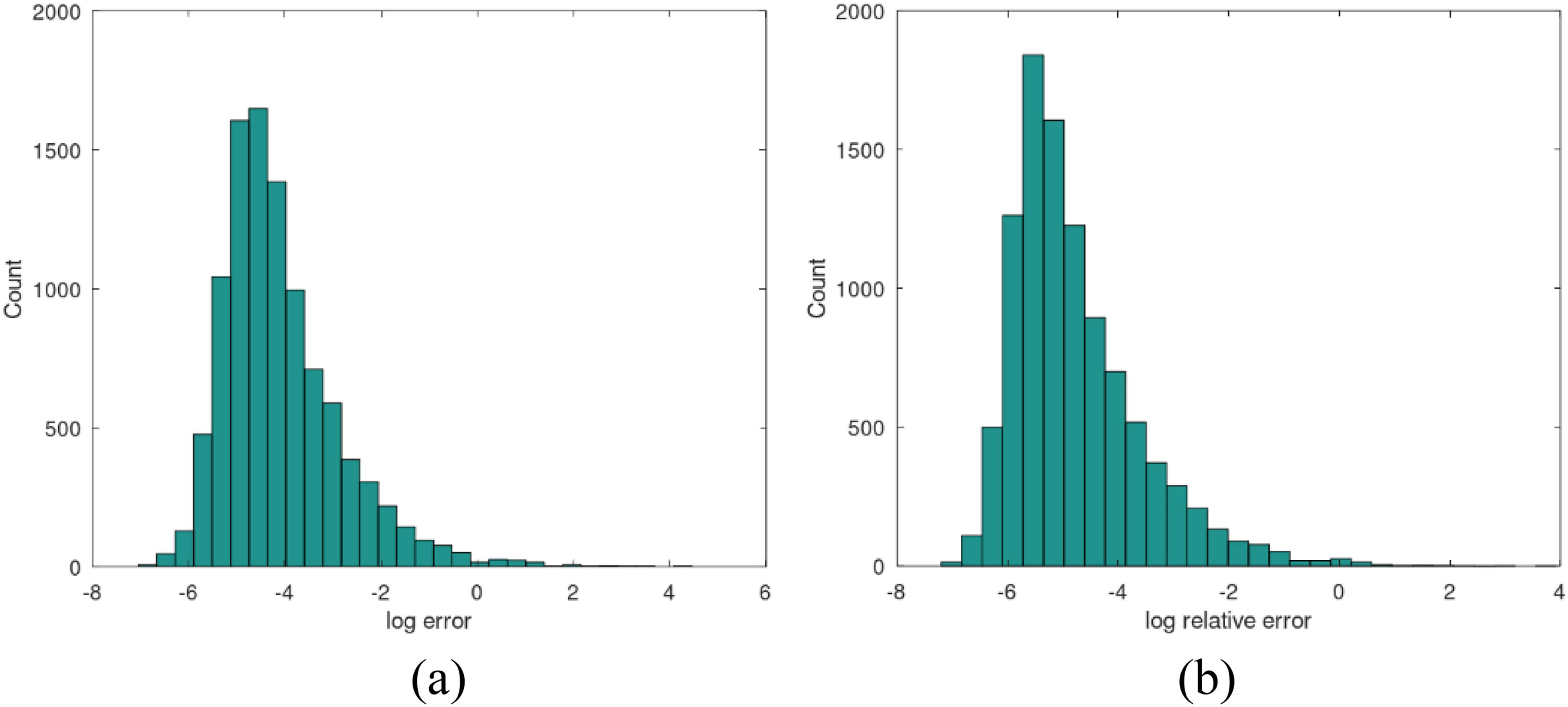
Comparison of
The choice of sampling interval also affects the agreement between the model parameters in an evocative manner. Returning to the model parameters we first considered in (14), we once again sample on the interval
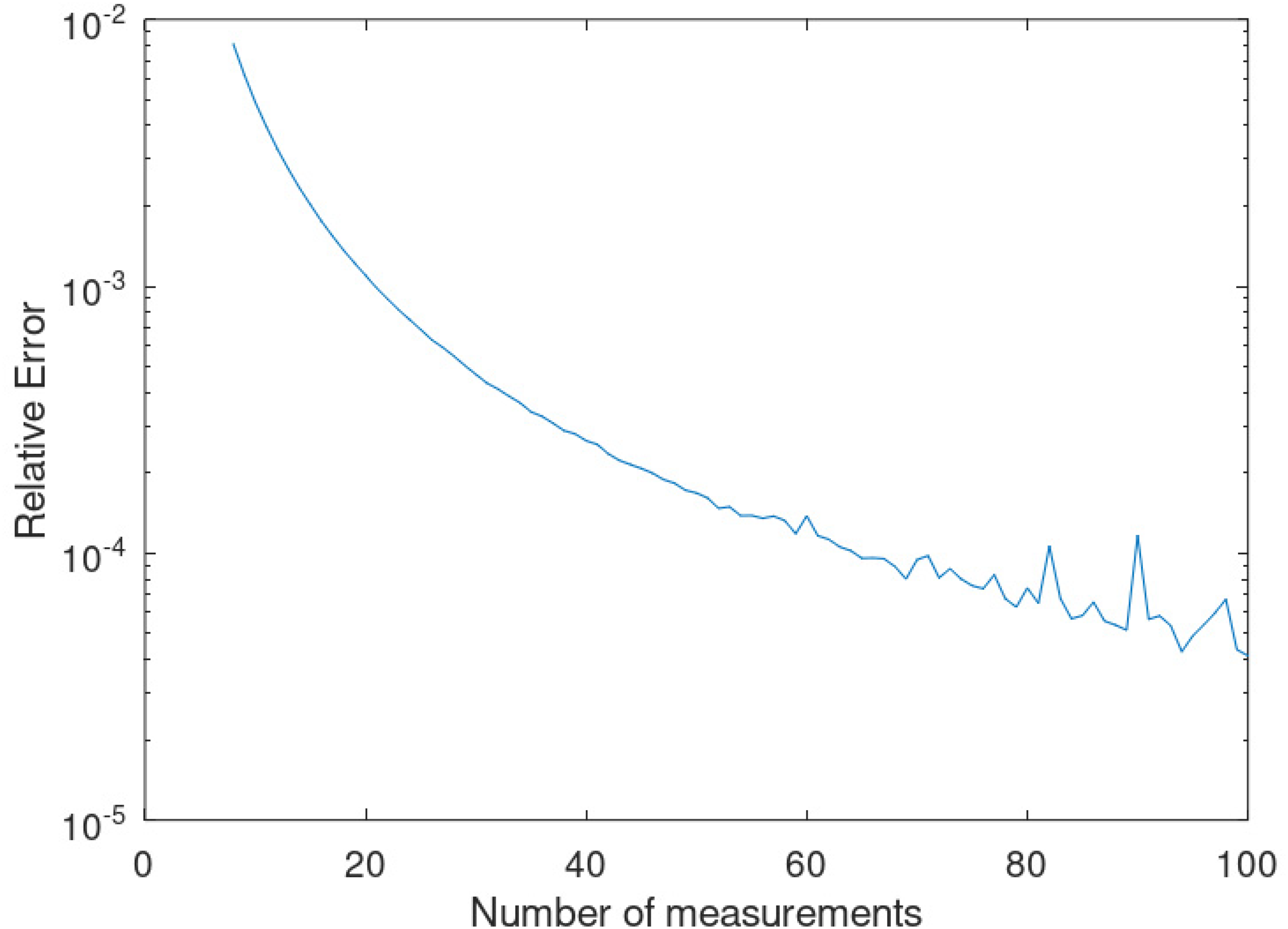
Convergence of


An important point arises here that needs clarification. In the nuclear medicine setting, acquiring the activity in an ROI at different times often requires the patient to return to the lab and have the activity re-measured. Hence, at best, one may expect to have three to five data points along the TACs. Table 1 gives the relative errors in
Discussion
Comparison of the parameters in (14) and their estimation in (15) shows that the described algorithm faithfully determines the rate constants from activity measurements. The relative LSEs in the 16 parameters are
Data acquisition remains a limiting factor for diagnostic accuracy and, therefore, clinical utility. This is illustrated in Table 1. These limitations arise primarily from estimations of the integrals

Relative Error in Parameter Estimates Using Linear Interpolation (Trapezoidal Rule) (a) and Using Cubic Spline Interpolation (b).
The novel mathematical approach presented here has numerous immediate and potential applications in imaging modalities. The radiologist may feel that the computational methods currently utilized are sufficient for clinical purposes, particularly with relatively simple interpolations or standard reconstructions that provide both a semi-quantitative and overall qualitative characterization of a particular pathology when interpreted within a clinical picture. However, this result is of immediate benefit in the acquisition of nuclear medicine images. Until now, the acquisition was dependent on photon counts reaching a sufficient threshold as a function of time, balancing radiation dose and signal-to-noise ratios with a reasonable (physiological and comfort) duration under the camera. Simple linear or spline interpolation is utilized to assist in data processing. The method presented here allows for a reduction in RP dose by tailoring the dosage to the individual patient in a quantitatively precise manner.
Nuclear medicine provides an additional dimension of patient physiology by utilizing receptor- or function-specific ligands or molecules bound with radioactive agents for imaging, treatment, or both. PET and SPECT technology also provide higher clinical certainty with localization that will benefit from the aforementioned methods. In addition, whole-body imaging in PET and SPECT acquisitions can show the early flow phases as the radiotracer is distributed, reaches the target and critical organs, and decays or is excreted physiologically. Each patient’s physiology may be unique and the method can profile their specific physiologic distribution to optimize the study protocol to answer the clinical question.
The next logical step in this effort toward personalized medicine is to create a model with parameters unique to the patient that determines the RP kinetics of a small dose delivered to the patient to optimize the dose of a theranostic agent to the index tumor. Further administrations and overall protocols can be determined based on radiologic response, with further research required to evaluate treatment protocols and long-term outcomes. Furthermore, the parameters from this patient-specific model can constitute a new set of quantitative imaging biomarkers that might allow for improved characterization of both disease and natural variation. This high-dimensional data can be studied using machine-learning methods and incorporated into the features of neural networks.
PKs within other modalities, including photon-counting CT, MRI, and contrast-enhanced mammography, can further characterize a pathology by relating to and confirming with histological studies. Specific ROIs, such as index tumors, can be targeted for this purpose under each modality to which the above method can be applied to provide subcharacterizations with opportunity for improved prognostic accuracy.
Applying such a model to tracer kinetics may require some additional discussion. The problem of radioactive decay is a nonissue; nuclear medicine systems already make corrections for the physical half-life of the tracer. One might reasonably point out how the transfer coefficients may vary according to physiological conditions. However, within small time intervals, it is reasonable to assume that the transfer coefficients are relatively stable. We make the critical observation of an application where the relative stability gives us the opportunity to describe how these transfer coefficients vary according to physiological conditions. This could yield a phenomenological account of how transfer coefficients vary under differing physiological conditions. Examples of such phenomena that might alter these transfer coefficients include tissue saturation and altered metabolism due to disease. A further generalization that will be considered in future research is relaxing the homogeneity assumption by accounting for spatial distribution within compartments.
A concern some readers might have regarding the clinical viability of the technique described is the number of data points required. For the analytical method to determine a unique solution,
The limitations of this method may be reduced to uncertainties in the parameter estimations due to system noise. Additional mathematical work is being performed to characterize the outcome of this method in noisy systems and the sensitivity of the estimations to noise.
Advantages Over Existing Methods
Optimization methods for parameter fitting must contend with the nonconvexity and high-dimensionality of the problem posed. Convergence to local minima would yield high agreement between the observed and simulated TAC but disagreement between the ground truth parameter values and those found by the optimization procedure. In this event, the physiological significance of these parameters would be lost. Additionally, optimization methods are computationally intensive.
The MPm of Sarkar et al. 13 requires uniform sampling in the time domain. This makes the method impractical for certain long-term dynamics in this setting. In addition, MPm involves singular value decomposition of the Hankel matrix that is constructed from the dataset, which can be computationally intensive for large datasets. In addition, in the case of short data length, MPm can suffer from spectral leakage that would affect the accuracy of the parameter estimations. It is well-known that this method is also sensitive to noise and is poorly conditioned in certain settings; however, we will postpone a comparison of the two methods in the noisy setting to an upcoming article.
The analytical method described overcomes many of these challenges by directly solving for the parameters. This allows for the study of multicompartmental models with many more compartments than were able to be previously considered.
Variable Transfer and Excretion Coefficients
We alluded to the notion of variable transfer and excretion coefficients earlier. Under Michaelis–Menten kinetics, the flux rate of activity between containers 
where
To the radiologist, the problem solved is one that initially might seem clinically unimportant since PET data are sufficiently abundant such that linear or spline interpolation provides all the qualitative information they need. However, the accuracy and precision of the PK estimates described allow for a confident approach to patient care and creation of individualized protocols.
Conclusion
We conclude with some immediate observations. The methods discussed here emphasize that the choice of sampling the TAC is completely arbitrary in determining the rate constants associated with compartmental models. The algorithm and the data presented show that the accuracy and precision of recovering the rate constants are excellent, whether the data are sampled randomly or uniformly, and how it relates to the number of data points sampled. The appearance of the iterated integrals or the moments of
We have already performed simulations by adding statistical noise to the TAC readings and reconstructed the rate constants from these noisy data. Interestingly, where the sampling is relatively dense, the results are consistent with the results presented here and fit the randomly chosen rate constants quite well; not unexpectedly, for coarser sampling regimens, the errors become somewhat larger. The analysis of the statistical uncertainties associated with the noisy activity data is proving to be quite extensive. Once the results on the statistics of the problem are completed, applications of these models to patient data will be center stage to bring these results into clinical practice. Without these models, applications to patient data are unverifiable.
Finally, a natural extension of this technique is dealing with the situation where we do not know the exact number of exponential basis functions in the sum. The over-determined problem has already been considered in Proposition 3 in Ellingson and Jafari,
4
while the under-determined problem will require optimization over the set of parameters
Several topics for future work have already been discussed above and in Section “Dicussion.” It can be useful to collect these topics here. Of course, this is by no means an exhaustive list.
The homogeneity assumption within each ROI will directly impact the calculation of RP dosage and the organ radiation dose. Introducing diffusion and other partial differential equation-based models that account for RP distribution in each ROI would improve the dose estimates. This is an interesting topic and one that is worthy of pursuing. Introducing the patient-specific PK model parameters as a physiologically meaningful quantitative imaging biomarker into machine-learning methods for diagnostics, therapeutics, and prognostics. Using Michaelis–Menten kinetics, the rate constants are dependent on the concentration of activity in each compartment. These models lead to saturation of activity/concentrations and to nonlinear systems of differential equations, whose solutions may not be sums of exponentials. Fractional calculus models have been used by various authors to model biological phenomena. The reader is referred to Khan et al.,19,20 and the references therein for inquiries in this direction. Finally, the study of sensitivity and stability of the estimates of the rate constants to noise and other nonstochastic parameters (e.g. patient motion) is interesting to achieve full clinical birth of these models.
Abbreviations
Least square error
Pharmacokinetic
Positron emission tomography
Single photon emission computed tomography
Time activity curve
Time-integrated activity
Physiologically based pharmacokinetic
Standard uptake value
Radiopharmaceutical
RP kinetics
RP therapy
Region(s) of interest
Matrix Pencil method
Footnotes
Acknowledgments
We gratefully thank the editor and expert (anonymous) referees of this article for their careful review of this manuscript and comments.
Research Ethics and Patient Consent
Not applicable; no human or animal subjects were used in this research, nor was any patient data used or referenced.
Author Contributions
All authors contributed to the development of the ideas of this article, reviewed and discussed the results, and assisted with the writing of this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
No patient studies were carried out in creating this manuscript. The generated data are simulated. No AI tools were used to generate or alter the data demonstrated in this article.
