Abstract
Background:
Hepatic metastases are a leading cause of mortality in gastrointestinal cancers, often exacerbated by the limited efficacy of conventional chemotherapeutics. Biobran/MGN-3, a rice bran-derived arabinoxylan, has shown promise in sensitizing cancer cells to paclitaxel.
Objective:
This study evaluates the hepatoprotective potential of Biobran/MGN-3 in a murine model of Ehrlich ascites carcinoma (EAC).
Methods:
Female Swiss mice inoculated with EAC were treated with paclitaxel (10 mg/kg) and Biobran (40 mg/kg) concomitantly for 30 days. Tumor volume along with liver function enzymes were quantified, and hepatic tissue was examined using Masson’s trichrome staining, histopathology, and transmission electron microscopy.
Results:
Biobran administration significantly reduced tumor volume and liver enzyme levels, indicating improved hepatic function. Collagen deposition around hepatic capillaries was markedly diminished. Histological analysis revealed restoration of normal lobular architecture, while ultrastructural examination showed enhanced nuclear integrity, bile duct morphology, and cytoplasmic organelle preservation. Notably, Biobran activated Kupffer cells to target neoplastic cells infiltrating hepatic parenchyma.
Conclusion:
Biobran/MGN-3, in combination with paclitaxel, confers substantial hepatoprotective effects in EAC-bearing mice, supporting its potential as a nutraceutical adjunct in cancer therapy.

Introduction
Numerous cancer-related fatalities are caused by hepatic malignancies. Hepatic metastases are the main factor contributing to gastrointestinal cancers mortality, which are the second highest cause of cancer-related deaths in the USA. 1 In 2020, there were 830 200 liver cancer deaths and 905 700 new cases worldwide. This count of new cases and fatalities might rise up by 55% by 2040 according to the “Global Burden of Primary Liver Cancer in 2020 and Estimates through 2040.” 2 The usual first-line treatment of hepatic malignancies is chemotherapy, including paclitaxel, doxorubicin, 5-fluorouracil, and cisplatin. However, these chemotherapeutic agents failed to curtail the disease. Paclitaxel is an effective antitumor drug against a wide diversity of malignancies. It is a member of the microtubules-focusing family called taxanes, which augments formation and stability of microtubules preventing polymerization. 3 Paclitaxel and its vehicle Cremophore EL have many serious side effects including congestive heart failure, anaphylactoid hypersensitivity, neuropathy, diarrhea, mutagenic and carcinogenic effect.4 -7
Dietary biological response modifiers (BRMs) may be used to combat the toxicity linked to high doses of chemotherapy, via different mechanisms such as increasing the effectiveness of treatment, promoting cancer cell death by apoptosis rather than necrosis, lowering drug resistance, or boosting drug accumulation within cancer cells.8,9 In addition, our earlier studies investigated the development of an efficient, natural treatment agent known as MGN-3/Biobran, an arabinoxylan rice bran treated with carbohydrate-hydrolyzing enzymes from Shiitake mushrooms. It has xylose in its main chain and an arabinose polymer in its side chain. 10 Our earlier studies and that of others have demonstrated the free radical scavenging ability of Biobran/MGN-3 and related compounds,11,12 and the enhancement of anticancer activity by human natural killer cells (NK) in healthy subjects13,14 and in cancer patients. 15 Biobran/MGN-3 is also known as a potent chemo-sensitizing agent that possesses great potential for adjuvant therapy in the treatment of cancer. It sensitized human leukemic cells HUT78 to CD-95 antibody 16 and breast cancer cells to daunorubicin. 17 In addition, Biobran revealed a potent tumor inhibitory effect via induction of apoptosis in animals bearing tumors. 18 Furthermore, the chemoprotective effect of Biobran/MGN-3 was reported against cisplatin in mice 19 and cisplatin and daunorubicin in rats. 20
Our earlier studies demonstrated the ability of Biobran intake with liver cancer drugs to reduce cancer recurrence and increase the survival of hepatic cancer patients. 21 Furthermore, we have documented the advantageous outcomes of combining Biobran with paclitaxel on the proliferation of metastatic (4T1) and nonmetastatic (MCF-7) breast cancer cell lines in vitro. 22 In vivo studies also showed the enhancement of the apoptotic ability of a low-dose of paclitaxel on tumor cells in mice by Biobran, which was associated with significant inhibition of tumor growth. 23 Accordingly, this study aimed to evaluate the hepatoprotective effect of Biobran in mice inoculated with Ehrlich ascites carcinoma (EAC), assess the potential synergistic or additive impact of its combination with paclitaxel on liver integrity, measure alterations in liver function enzymes as biochemical indicators of hepatic damage or protection, examine histochemical changes in liver tissue architecture, and visualize ultrastructural modifications in hepatocytes using electron microscopy to determine subcellular effects of the interventions.
Materials and Methods
Reagents
Paclitaxel (Taxol)
100 mg/16.7 ml starting concentration of paclitaxel (Taxol) was purchased from Bristol-Myers Squibb Inc. in Princeton, New Jersey, 08543. Six milligrams of paclitaxel, 527 mg of pure Cremophor® EL (polyoxyethylated castor oil), and 49.7% (v/v) dehydrated alcohol, USP are all included in each milliliter of the sterile nonpyrogenic solution.
Biobran/MGN-3
This is a modified hemicellulose developed by combining hemicellulose from rice bran with several Shiitake mushroom hydrolyzing enzymes. The primary chemical structure of Biobran/MGN-3 is an arabinoxylan with xylose in the main chain and an arabinose polymer in the side chain. 10 The Biobran was graciously donated by Daiwa Pharmaceutical Co. Ltd., Tokyo, Japan.
Preparation of Ehrlich Ascites Carcinoma (EAC) Cells
EAC is a hyperdiploid undifferentiated breast cancer with extensive invasive capabilities, rapid proliferation rate, short lifespan, and 100% malignancy. The murine EAC cells for this investigation were originally acquired from the National Cancer Institute at Cairo University, Egypt. They were maintained in vivo by injecting 2.5 × 106 cells into female Swiss albino mice intraperitoneally on a weekly basis. Solid tumors were induced in mice via subcutaneously implanting of 0.2 ml of ascites fluid including 2.5 × 106 live EAC cells,24,25 into their lower back. Viability was assessed using the Trypan blue dye exclusion technique and was observed to be 95% or above.
Animals
Thirty female Swiss albino mice, each weighing between 19 and 25 g, were obtained from the National Cancer Institute at Cairo University in Egypt. Mice were kept in our animal research facilities with 12-hour light and dark cycles. Animals were given regular cube pellets and water ad libitum. The pellets purchased from Misr Oil & Soap Company in Cairo, Egypt, and were composed of casein (12.5%), lipids (1.0%), wheat flour (80%), bran (3.3%), olive oil (2.3%), DL-methionine (0.5%), a blend of vitamins and salt (0.2%), and water (0.2%). The proportion of total calories consists of around 18% protein, 73% carbohydrate, and 9% fat. The study was approved by the Ethics Committee of the Experimental Animal, Faculty of Science, Mansoura University, Egypt, with approval number is Sci-Z-P-113.
Experimental Design
The animals were randomly allocated into 6 groups (5 animals per group), allowed a week for acclimation according to ARRIVE guidelines, and subsequently inoculated with EAC. Various treatment sets commenced intraperitoneally (i.p.) starting on day 9 post-inoculation and continued until day 30 post-inoculation (Figure 1) as follows:
Group 1. (Control): mice served as untreated controls and received no treatment.
Group 2. (Biobran): mice were given 40 mg/kg18,23 of Biobran alone 3 times a week (i.p.) for 30 days.
Group 3. (EAC): mice inoculated with 2.5 × 106 viable Ehrlich ascites cells into their lower back and left untreated for 30 days.
Group 4. (EAC + paclitaxel): mice bearing EAC and treated (i.p.) with 10 mg/kg 26 paclitaxel alone every other day for 30 days.
Group 5. (EAC + Biobran): mice bearing EAC and treated (i.p.) with Biobran alone (40 mg/kg) alone 3 times a week for 30 days.
Group 6. (EAC + Biobran + paclitaxel): mice bearing EAC and treated (i.p.) with Biobran (40 mg/kg, 3 times a week) in combination with paclitaxel (10 mg/kg, every other day) concomitantly for 30 days.

Experimental timeline.
Body Weight Changes (BW) and Tumor Volume (TV)
The initial body weight was recorded at the beginning of tumor inoculation. BW was monitored weekly, the last BW and tumor weight were measured to calculate the net final body weight, BW gain and % of change of the different groups at the end of the experiment. Tumor volume was measured 5 times during the experiment on day 9, 14, 20, 25 and 30 successively, and estimated via this equation: TV (mm3) = 0.5 A * B2; where A is the major axis and B is the minor axis. 27 The significance of the differences in BW and TV due to the different treatments paclitaxel and Biobran alone and in combination was compared to normal untreated mice (Control) and to the untreated tumor bearing mice (EAC).
Liver Function Analysis
We examined the liver function enzymes that include alanine aminotransferase (ALT), aspartate aminotransferase (AST), and gamma-glutamyl transpeptidase (γGT). Serum levels of ALT and AST were detected as previously described, 28 and serum γGT 29 using ELI Tech Clinical Systems kit (France).
Histopathological Examination
Liver samples were collected for histological processing, cut into tiny pieces, promptly fixed in 10% buffered formalin for 24 hours, dehydrated in escalating series of ethyl alcohol, cleaned in xylene for 20 minutes, then embedded in paraffin wax. Using a microtome, sections 4 to 5 m thick were cut, mounted on a glass slide, and stained with hematoxylin and eosin (H&E), before being inspected under a light microscope. Histopathological scoring in between the treatment sets was conducted by semi-quantitatively by a single, non-blinded observer as (−) is nil, (+) is low observed, (++) is moderately observed, (+++) is highly observed for: neoplastic metastasis; leucocytic infiltration; dilated or destructed veins; fatty infiltration; fibrosis.
Hepatic Collagenous Fibers Intensity
To assess hepatic fibrosis, paraffin-embedded liver tissue was stained with Masson’s trichrome. Sections (4 -5 mm) were seen under a light microscope and captured on camera (DFC Leica camera). The blue stain is used to detect collagen fibers and shows the severity of hepatic fibrosis. 30 ImageJ software, version 1.48d (created by Wayne Rasband, National Institutes of Health, Bethesda, Maryland, USA), was used to perform image analysis techniques.
Ultrastructural Studies
After fixing the liver tissues of the different groups with 4F1G fixative in PBS (pH 7.2), they were postfixed in 3 changes of 1% cool osmium tetroxide in PBS (pH 7.2) for 1 hour. After dehydrating the samples in ethanol of varying concentrations, they were encapsulated in Epson-Araldite resin. Following uranyl acetate and lead citrate staining, 31 ultrathin sections were examined under a 60 kV JEOL electron microscope (Tokyo, Japan).
Statistical Analysis
Statistical analyses were done by GraphPad Prism® software version 7. The standard error of the mean, or mean ± SEM, was used to report the data. One-way analysis of variance (ANOVA) in conjunction with the Tukey multiple comparison test was used to assess the significance of the variations in mean values. P-values less than .05 were regarded as significant. The statistical significance is shown by the symbols *, **, ***, and **** at P < .05, P < .01, P < .001, and P < .0001, respectively.
Results
Biobran Attenuated BW, TW and TV Changes
Figure 2A illustrates that the net final body weight of the control mice was around 23.77 ± 0.21 g, representing a 15.96% increase from their initial body weight. Similarly, Biobran-treated mice demonstrated an increase in net final body weight (24.19 ± 0.2 g, 17.66%) relative to their initial body weight. In contrast, the untreated EAC group exhibited a reduced net end body weight gain of 22.31 ± 0.43 g, indicating a 10.88% rise. Treatment with paclitaxel in the inoculated animals led to a little increase in net final body weight (0.42%) compared to the EAC group. The administration of Biobran to EAC-bearing mice yielded a net final body weight increase of 4.68%. The enhancing effect was maximized in mice after exposure to Biobran alongside paclitaxel, leading to a net final body weight increase of 18.1% (P < .05) versus to the EAC alone group.
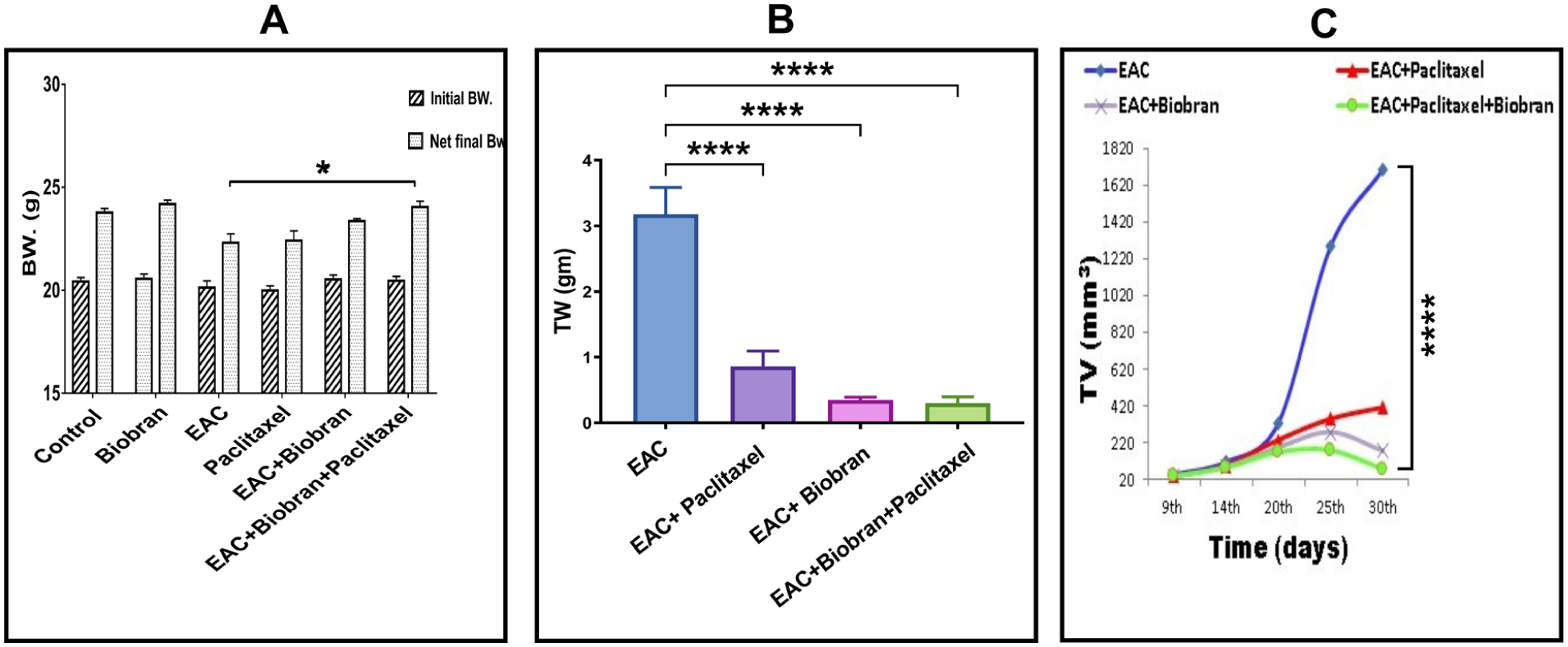
Effect of Biobran and/or paclitaxel treatment on (A) body weight (BW, g). (B) Tumor weight (g). (C) Tumor volume (mm3). Data are expressed as mean ± S.E. (n = 5). *Significant change at P < .05, ****Significant change at P < .0001.
Figure 2B and C illustrate that administration of paclitaxel alone resulted in a substantial decrease in both TW and TV by 72.9% and 76.1%, respectively, with P < .0001. Likewise, administration of Biobran alone resulted in a substantial reduction in TW and TV by 89.1% and 89.7%, respectively, with P < .0001 in comparison to the untreated EAC control group. The concurrent administration of paclitaxel and Biobran led to a significant decrease in both TW and TV, surpassing the reductions obtained from each therapy individually by 90.6% and 95.7%, P < .0001, respectively, compared to the EAC group.
Liver Function Enzymes
Figure 3 depicts the serum levels of liver function enzymes ALT, AST, and γGT in mice under different experimental conditions at day 30 post-tumor inoculation. EAC group showed a significant elevation in AST, ALT, and γGT levels (450.4%, 266.6%, and 92.7% respectively vs the control group, P < .0001). Treatment with paclitaxel into EAC bearing mice caused marked decrease in the levels of liver function enzymes as follow (34.3%, P < .001) for ALT; (42.2%, P < .0001) for AST and (16.6%, P < .05) for γGT as compared with EAC group. Furthermore, Biobran therapy in EAC-bearing mice demonstrated reductions in the aforementioned parameters as (53.3%, 48.2%, and 42.2%, P < .0001 respectively) compared to the EAC group alone. Interestingly, co-administration of Biobran and paclitaxel markedly improved liver enzyme elevations, with reductions of 68.9%, 64.1%, and 40% in AST, ALT, and γGT levels, respectively (P < .0001), in the EAC + Biobran + paclitaxel group versus the EAC group.

Effect of Biobran and/or Paclitaxel on serum level of liver function enzymes. (A) ALT level. (B) AST level. (C) γGT level in the different experimental groups. Data are expressed as mean ± S.E. (n = 5). *Significant change at P < .05, **Significant change at P < .01, ***Significant change at P < .001, ****Significant change at P < .0001.
Histopathological Observations of Liver Tissue
Liver tissues obtained from Control and Biobran groups showed normal appearance of hepatic lobular architecture with normal multigonal, mononucleated hepatocytes. Blood sinusoids were lined with a single fenestrated endothelial layer and Kupffer cells (Figure 4A and B). Meanwhile EAC-inoculated mice revealed fibrosis: neoplastic cells with hyperchromatic nuclei were observed in the centrilobular regions and inside the dilated and/or destructed portal veins (Figure 4C and D). Loss of architecture was represented in hepatocellular necrosis, degeneration and vacuolization with frequent mitotic activity was also seen.

Histopathological investigation of liver tissue. (A & B) Represent the Control and Biobran group respectively showing normal hepatic cords (hc), blood sinusoids (bs), Kupffer cells (kp) and central vein (cv) lined with intact endothelial layer (E). (C & D) EAC group showing degenerated central vein (dcv) with eroded endothelial cells (EE), and different mitotic figures of neoplasm (red arrow) invasion in-between the dilated portal vein (green arrow) and the degenerated hepatocytes with vacuolated cytoplasm, few binucleated hepatocytes (bn) and karyorrhexes nuclei (kr). (E) EAC + Paclitaxel group showing infiltrated of few neoplastic cells (T) between eosinophilic hepatocytes (black arrow). (F) EAC + Biobran group showing active Kupffer cells (kp) and few apoptotic (A), vacuolated and binucleated (bn) hepatocytes. (G) EAC + Biobran + Paclitaxel group demonstrating improved hepatocytic architecture. (H&E, ×400, inset ×1000).
On the other hand, EAC + Paclitaxel group revealed a remarkable improvement in the portal veins with infrequently encountered neoplastic cells in the centrilobular regions. However, hepatic parenchyma showed dilatation in blood sinusoids (Figure 4E). EAC + Biobran group showed a partial improvement in liver parenchyma, including lesser necrotic and degenerative changes, vacuolation of hepatocytes and scanty mitosis. In addition, dilated blood sinusoids with numerous Kupffer cells were also observed (Figure 4F). Surprisingly, the cotreatment of Biobran with paclitaxel highlighted evidence of robust enhancement in most of the hepatic tissue, together with prominent Kupffer cells. However, several hepatocytes continue to exhibit mild vacuolization (Figure 4G).
The histopathological scoring in between the treatment sets could be summarized in this table as (−) is nil, (+) is low observed, (++) is moderately observed, (+++) is highly observed (Table 1):
The Histopathological Scoring in Between the Treatment Sets.

Hepatic Collagenous Fibers Levels
Examination of liver tissues obtained from normal Control and Biobran group displayed a negligible amount of collagenous fibers around the central vein and blood sinusoids (Figure 5A and B). However, liver tissue excised from the EAC-bearing mice showed a remarkable increase in the collagenous fibers (P < .0001) around central veins, blood sinusoids and scattering fibers around the neoplastic cells in the destructed hepatic parenchyma comparing to control normal mice (Figure 5C). Similarly, liver tissues excised from EAC + Paclitaxel and EAC + Biobran groups also showed a marked increase in collagenous fibers but to a lesser extent. All showed a considerable amount of collagenous fibers around the neoplastic cells near the veins and in the blood sinusoids (Figure 5D and E). In contrast, it was of great interest to show that liver samples obtained from animals cotreated with both Biobran and paclitaxel revealed an obvious decrease in the collagenous fibers (P < .0001) that was higher than either treatment alone in comparison with EAC-group alone. (Figure 5F and G).

(A-G) Liver tissue stained with Masson’s trichrome stain for collagen fibers. (A & B) Liver sections of control and Biobran groups showing a negligible amount of collagen fibers. (C) EAC-untreated animals showing a remarkable amount of collagen fibers around the central and portal vein. (D) EAC + Paclitaxel displaying a large amount of collagen around the central vein. (E) EAC + Biobran: showing a remarkable amount of collagen fibers around the central vein and the bile duct. (F) EAC + Biobran + Paclitaxel displaying a significant improvement in the liver architecture and a negligible amount of collagen fibers around the central vein and centrilobular blood sinusoids. (G) Quantitative analysis of the fibrotic area in hepatic tissue of different treatment groups. ****Significant change at P < .0001. (Masson’s trichrome stain ×400).
Ultrastructural Observations of Liver Tissues
The ultrastructural investigation of both normal Control and Biobran groups showed hepatocytes with typical structure (Figure 6A-F). However, liver samples obtained from mice inoculated with EAC appeared highly affected. The nuclei appeared blebbed, partially chromatinolysed or pyknotic with remarkable cytoplasmic accumulation of fat droplets (Figure 7A and B). In another focus, a metastasized neoplastic cell could be seen nearby collapsed blood sinusoids with degenerated endothelial cells (Figure 7B). A large myelin-figure was observed nearby a destructed bile cuniculus (Figure 7C). Additionally, hepatic sinusoids collapsed with a dense collagen fiber in the perisinusoidal space enclosing a pyknotic Kupffer cell (Figure 7D). Hepatocytes from paclitaxel alone showed some recovery from their pathological changes. Nuclei were rounded with abundant fibrilo-granular euchromatin (Figure 8A). The bile canaliculi restored their normal shape and microvilli (Figure 8B). Interestingly, in the blood sinusoids of this group, a morphologically intact Kupffer cell with long processes was observed in a state indicating the involvement with a metastatic neoplastic cell. Within the same focus, a relatively large Kupffer cell characterized by numerous microvilli was noted, exhibiting features morphologically consistent with a reactive phenotype (Figure 8C).

Illustrating ultrastructure examination of hepatic tissue of Control (A-C) and Biobran (A-F) groups showing normal nucleus (N) with 2 nucleoli (nu), numerous mitochondria (m), glycogen granules (g), bile caniculus (bc) with numerous microvilli (mv) and guarded by zona occulodenta (zo) and zona adherens (za), blood sinusoids (bs) with normal endothelial cells (E) and Kupffer cell (kp) in the space of Disse (Di). Smooth and rough endoplasmic reticulum (sER & rER).
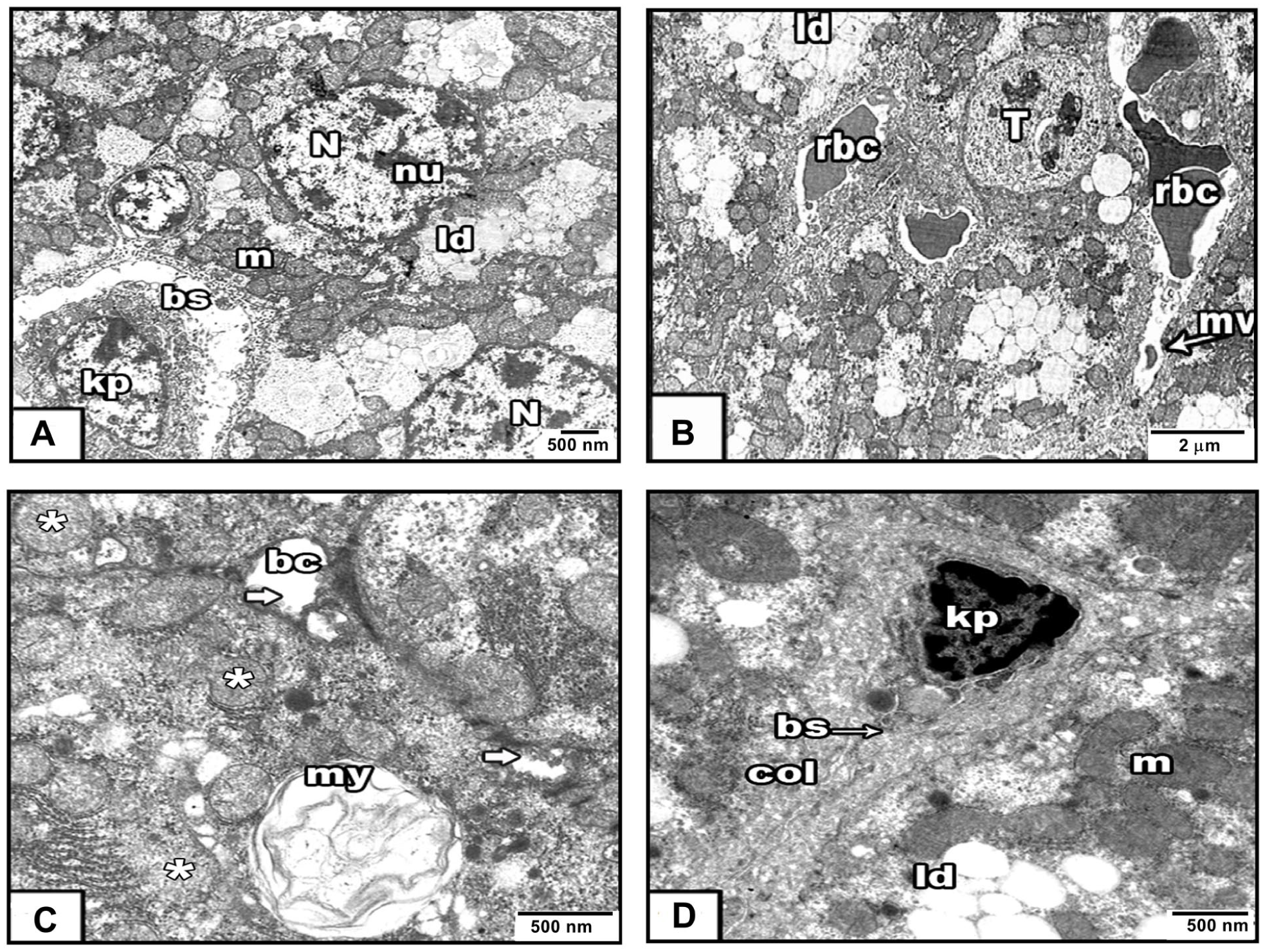
(A-D) Illustrating ultrastructure examination of hepatic tissue of EAC group. (A & B) Showing abnormal nucleus (N) with more than 2 nucleoli (nu), grouped mitochondria (m), wide blood sinusoid (bs) with Kupffer cell (kp). Invasive lipid droplets (ld), and a metastasized tumor cell (T) nearby collapsed blood sinusoids with degenerated microvilli (mv) that filled with red blood corpuscles (rbc). (C) Showing degenerated bile canaliculi (bc) without microvilli (white arrow) and myelin figure (my) nearby them. (D) Showing a dense collagenous fibers surrounding blood sinusoid (bs) and Kupffer cell (kp).

(A-E) Illustrating ultrastructure examination of hepatic tissue of the different treatment sets. (A-C EAC + Paclitaxel group): (A) Shows a rounded nucleus (N) with 2 nucleoli (nu), mitochondria (m), lipid droplets (ld), and rough endoplasmic reticulum (rER). (B) A bile canaliculus (bc) with few microvilli (mv) is sealed by zonula adherens (za) dilated rough (rER) and smooth (sER) endoplasmic reticulum, and a peripheral Golgi apparatus (Ga). Blood sinusoid (bs) containing electron-dense components (black arrow) and Kupffer cells (kp) targeting metastatic cells in the Disse space are shown in (C). (D & E EAC + Biobran group): (D) displaying normal rounded nucleus (N) with distinct nucleoli (nu), blood sinusoids (bs) with red blood cell (rbc) and active Kupffer cell (kp), Dilated rough endoplasmic reticulum (rER). Figure (E) shows elongated mitochondria (m) and bile canaliculus (bc) with numerous microvilli (mv) and guarded by zonula occludent (zo) and zonula adherens (za). (F-H) EAC + Biobran + Paclitaxel group): (F) A typical hepatocyte with rounded nucleus (N), multiple electron-dense mitochondria (m), blood sinusoids (bs) loaded with Kupffer cells (kp), and numerous red blood cells (rbc) surrounded by endothelial cells E. (G) The bile canaliculus (bc) has typical microvilli (mv), a strong zonula occludes (zo) and zonula adheres (za), mitochondria (m), glycogen particles (g), and lysosomes (ly). Degenerated electron-lucent mitochondria(black arrow). See Figure (H) for blood sinusoids (bs) with active Kupffer cells (kp) attacking and killing a metastatic tumor cell (T).
Hepatocytes from EAC + Biobran group showed moderate improvement with rounded nuclei, identifiable nucleoli, and little heterochromatin. Dilated rER and electron-dense mitochondria were seen in the cytoplasm. Additionally, a well-developed Kupffer cell with electron dense rER was noticed in the space of Disse (Figure 8D). Bile canaliculi showed a little improvement (Figure 8E).
The combined treatment of Biobran + Paclitaxel revealed a marked improvement in the liver architecture. The hepatocyte possessed typical nuclei with a large amount of electron-lucent euchromatin (Figure 8F). Bile canaliculi and their microvilli appear normal (Figure 8G), cytoplasm showed abundant glycogen particles and electron-dense rounded and elongated mitochondria. However, a few fat droplets, secondary lysosomes and electron-lucent vacuoles were observed; in addition, intact blood sinusoids with well-formed Kupffer cells (Figure 8F and G). In another focus, a few metastasized tumor cells were observed in a state morphologically consistent with degradation, suggestive of enhanced engagement by Kupffer cells within a blood sinusoid filled with electron-lucent materials (Figure 8H).
The Proposed Mechanism of Biobran/Paclitaxel Combination on EAC-Liver Metastasis
Our recent findings revealed a method by which Biobran enhances the efficacy of Paclitaxel in liver protection against metastatic EAC, which might be illustrated in Figure 9.

Hypothetical preclinical mechanism of Biobran & paclitaxel in EAC-metastasis.
Discussion
Various hepatic and immune cell types, including liver sinusoidal endothelial cells, Kupffer cells (KCs), hepatic stellate cells, hepatocytes, dendritic cells, resident NK cells, and infiltrating monocytes, macrophages, and neutrophils, are recognized for their roles in the progression and sustenance of liver metastasis. 32 Liver tumors typically exhibit a poor response to systemic chemotherapy, which commonly diminishes only a small fraction of tumor mass and is linked to significant adverse effects. 33 Prior research has indicated that Biobran/MGN-3 (arabinoxylan rice bran) demonstrates anti-cancer properties in multiple experimental models, including solid Ehrlich ascites carcinoma (EAC), 18 glandular stomach carcinoma, 34 and neuroblastoma. 35 Biobran has demonstrated the ability to amplify the tumor-regressing effects of radiation 36 and to improve the apoptotic response to low-dose paclitaxel in mice with EAC. 23 The observed effects were linked to reduced proliferation, elevated DNA fragmentation, and an increased apoptosis-to-proliferation ratio. Moreover, Biobran has exhibited hepatoprotective effects against N-nitrosodiethylamine (NDEA)-induced liver carcinogenesis in rats, where it decreased liver enlargement, lowered liver enzyme increases (AST, ALT, ALP, and GGT), 37 and diminished fibrotic and inflammatory markers IkappaB-alpha (IκB-α) mRNA expression and nuclear factor kappa-B (NF-κB/p65) expression. 38
Our histological and ultrastructural findings demonstrate that Biobran, paclitaxel, and their combination effectively mitigate EAC-induced liver metastases. Although the lack of a clinical benchmark, like silymarin, prevents comparison with recognized hepatoprotective medications, the study’s internal validation demonstrates a considerable reduction in harm compared to the untreated tumor. Meanwhile, the EAC model offered a consistent framework for monitoring short-term metastatic responses over 30 days; however, the long-term clinical ramifications of these combined medicines require additional exploration.
The present study demonstrates the superior hepatoprotective of the co-treatments against EAC-induced liver metastasis compared to monotherapy. While the EAC model typically induces severe cachexia, the preservation of body weight in the co-treated group suggests a systemic mitigation of tumor burden and toxicity, extending beyond simple local hepatic effects. These findings are parallel, but do not replicate previously reported hepatoprotective outcomes.37,38 Histological analysis and single non-blinded semi-quantitative scoring demonstrated that the combined treatment attenuated preneoplastic alterations and reduced collagen deposition, indicating a significant improvement in tissue organization. These findings establish a clear quantitative trend that provides a strong foundation for future investigations. To further build upon this data, subsequent studies should incorporate automated stereological counting and multi-observer validation to enhance the high-resolution precision of these results. Because molecular assessments were not conducted, the specific pathways underlying these changes remain speculative.
It is well established that collagen deposition patterns are well-recognized indicators of fibrotic and cirrhotic processes which can cause the creation of portosystemic shunts within the liver and perhaps the formation of a connective tissue diffusion barrier in the space of Disse between hepatocytes and their vascular supply. 43 In this study, we observed increased collagen deposition in the livers of EAC-bearing mice, and only the combined Biobran-paclitaxel therapy substantially reduced collagen accumulation to near-control levels.
Ultrastructurally, immunomodulation activity and resident KCs in the untreated group appeared overwhelmed by tumor infiltration, seemingly incapable of checking metastatic colonization. In contrast, the co-treated group displayed KCs with distinct morphological signs of activation, including preserved cellular processes. Since KCs are the liver’s primary line of defense against metastatic seeding exerting cytotoxicity via cytokine release and phagocytosis,44 -53 their improved structural integrity in the co-treated group implies a restoration of immune surveillance. This aligns with Biobran’s established role as a biological response modifier.13 -16,39 -42 While molecular markers were not assessed, the morphological data supports the hypothesis that combination therapy synergistically enhances the hepatic immune response, limiting metastatic seeding and subsequent fibrosis.
Conclusion
The present findings suggest the hepatoprotective potential of Biobran/MGN-3 when co-administered with paclitaxel in EAC-bearing mice. The treatment was observed to attenuate hepatic alterations and fibrosis, while maintaining features morphologically consistent with improved structural integrity of the liver tissue. Observations suggestive of enhanced Kupffer cell engagement and the preservation of lobular architecture support the further preclinical evaluation of Biobran as a potential chemo-adjunct in experimental metastatic models. It is important to note that as no standard benchmark compound (eg, silymarin) was included, these results are interpreted based on within-group comparisons rather than relative potency against established clinical standards. These exploratory results provide foundational proof-of-concept, warranting further investigation using human-derived xenograft or syngeneic murine models to define the precise mechanisms of action and the broader scope of this intervention within a preclinical framework.
Footnotes
Acknowledgements
The authors express their gratitude to Professor Nariman Badr Eldin for her valuable guidance in the preparation of this work.
Ethical Considerations
The study was approved by the Committee of the Ethics of Animal Experiments of the University of Mansoura, Egypt, approval number is Sci-Z-P-2022-113.
Author Contributions
M. A. and D. A. participated in the conceptualization, design, data collecting, and compilation, and provided final approval of the manuscript. M. G. and M. A. collaborated on data analysis, interpretation, manuscript preparation, and approved the final article. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.*
