Abstract
Emerging trends for cancer chemotherapy show promising developments with the better understanding of molecules delivering more potent and powerful capabilities. But these are severely limited because of increased side effects and higher probability of tumor recurrence. In this scenario, putative exploration of the indigenous and untapped resources modulating immune system to deliver adequate but potent chemopreventive effects appeals considerable interest. However, these require rigorous scientific validation with regard to potency compared with the existing drugs. Aegle marmelos (Linnaeus) Correa (family Rutaceae), a plant component of polyherbal formulation, Indukantha Ghritha, is known for its widespread medicinal values. But the chemopreventive potential has not been explored in comparison to existing anticancer agents. Our attempt contributes the scientific evidence for beneficial immunoprophylactic and antitumor functions in mice challenged with ascites tumors, Dalton’s lymphoma ascites, and Ehrlich’s ascites carcinoma either alone or in combination with cyclophosphamide and 5-fluorouracil. Specifically, the petroleum ether extracts of this plant (AMPE) prophylactically activated a cascade of host defense mechanisms by stimulating or restoring total white blood cell count, macrophage phagocytosis, hematopoiesis, lymphocyte proliferation and functions (CD4+ and CD8+) either naturally or under conditions of impaired immunity like in ascites tumor or during standard agent chemotherapy. Overall, AMPE also elicited strong antitumor effects by increasing median survival time and life span, while reducing murine ascites tumor volume and viable tumor counts on par with cyclophosphamide and 5-fluorouracil especially when administered prophylactically. This study also identified 2 putative components, xanthorrhizol and marmelosin, which could be imparting the immunoprophylactic and antitumor effects in transplantable tumor models. Thus, our attempts provide sufficient proof to warrant further to test this drug in higher animal models or in patients with high risk for tumor recurrence and/or immunocompromised diseases.
Keywords
Introduction
Although the insight and appreciation that nature combined with chemical modifications provide robust candidates with great chemical diversity in drug discovery solutions, ethnomedicine has its own value imparting only the beneficial effects with no side effects or toxicity. Biological immune response modifiers act in part by augmenting or modulating the host defense mechanisms that are normal or already activated, thereby strengthening the response against the diseases. 1 Evidence from the literature points out that more than half of the tested plants are not scientifically validated for their therapeutic use in the modern medicine. However, some of the plants that have been reported to modify or stimulate immune system components are Tinospora cordifolia, 2 Withania somnifera, 3 Piper longum, 4 and Enicostema axillare. 5
Earlier studies have shown that Indukantha Ghritha (IG), an ayurvedic polyherbal drug possesses immunomodulatory function, which makes it a potential adjuvant to chemotherapy in tumor-induced mice. 6 Because of this being a polyherbal formulation, the total effect of the IG could very well be the synergistic effect of the different plants. But, as a targeted therapeutic approach, it was important to study separate biologically active components of IG imparting this multitude of effects. One such component of IG, Aegle marmelos (L) Correa commonly known as Bengal quince or Stone apple, native to the Indian peninsula, Sri Lanka, Burma, Thailand, and Indo-China has been proved to possess various beneficial activities, including anticancer,7-9 immunomodulatory, 10 antimicrobial, 11 anti-inflammatory, 12 antioxidant,9,13 and antihyperglycemic 14 properties. Roots of A marmelos form an essential ingredient of Dhasmoola, IG, etc. which are herbal medicines commonly prescribed by Ayurvedic practitioners. However, chemopreventive or immunoprophylactic potential and its possible implication in eliciting protective antitumor immunological responses have been widely lacking.
The current study evaluated a comprehensive immunoprophylactic and ascites tumor regression competencies of the petroleum ether extract of A marmelos (AMPE) in comparison with the standard chemotherapeutic drugs, cyclophosphamide (CTX) and 5-fluorouracil (5-FU). This study highlights the importance of AMPE as a chemopreventive drug restoring a variety of host immune system functions as a prerequisite to retard recurring tumor growth, thereby providing strong antitumor efficacy, especially when administered prophylactically in murine transplantable tumors.
Materials and Methods
Preparation of Plant Extracts
Fresh plant roots of Aegle marmelos (L) Correa were identified and procured from reliable sources and identified by a qualified botanist. Voucher specimen (010405/AM) was also deposited for future identification. Briefly, roots were shade-dried, powdered (250 g), and extracted with 2 L petroleum ether solvent in a Soxhlet apparatus for 18 to 24 hours. This afforded a solid residue (yield = 4%) after solvent elimination under reduced pressure. The crude extract obtained (AMPE) was filtered through a 0.45-µm Millipore after dissolving in 0.1% dimethyl sulfoxide (DMSO, Sigma-Aldrich, St Louis, MO). This was confirmed to be endotoxin free and stored at −20°C until use.
Gas Chromatography–Mass Spectrometry Analysis
Parent petroleum ether extracts were chemically analyzed in a gas chromatography–mass spectrometry system for qualitative analysis of compounds present in the extracts. The analyses were carried out on Agilent 6890 gas chromatography system equipped with 5973 N mass selective detector, 7683 autosampler (Agilent Technologies, Santa Clara, CA) and CP SIL 8CB capillary column of length 30 m, 0.25 mm internal diameter, and 0.25 µm film thickness. The column oven was programmed from 50°C hold for 2 minutes, and raised to a final temperature 300°C in steps of 10°C/min. The column was held at the final temperature for 10 minutes. Helium at the flow rate of 1.0 mL/min was used as the carrier gas. The injection port and the interface temperatures were 250°C and 280°C, respectively. The analysis was conducted in the electron ionization mode at 70 eV. The ion source and quadrupole temperatures were 230°C and 150°C, respectively. One microliter samples were injected with the autosampler in split injection mode with 10:1 split ratio. The components of the AMPE were identified by comparison of their mass spectra with those of a computer library (Wiley database). As routine quality control, all the batches of AMPE were ensured to contain xanthorrhizol (17.72 retention time) by comparison of the mass spectra along with other identifiable compounds such as 9,12-octadecadienoic acid, piperine, and 1,3-benzodioxole (Figure 1).

Gas chromatography–mass spectrometry analysis of Aegle marmelos. (A) Total ion chromatogram (TIC) of petroleum ether fraction yielding 4 major identifiable peaks. (B) Mass spectra of particular peaks in the TIC—peak 1 (xanthorrhizol); peak 2 (9,12-octadecadienoic acid); peak 3 (piperine); peak 4 (1,3-benzodioxole).
High-Performance Thin Layer Chromatography Fingerprinting
Ten-microliter aliquots of the A marmelos petroleum ether extract, ethyl acetate extract (external control), and the standard, marmelosin were applied on silica gel 60 F254 precoated high-performance thin layer chromatography plates, 10 × 10 cm (Merck, Darmstadt, Germany) using Camag Linomat-V applicator and eluted in solvent system, ethyl acetate:toluene:glacial acetic acid (55:45:1). Samples were applied using Camag Linomat-V automated thin layer chromatography applicator at a constant application rate of 15 nL/s using nitrogen aspirator. Plates were developed in a twin-trough chamber (10 cm × 10 cm) presaturated with the above mobile phase for 20 minutes at room temperature. After heating the plate at 100°C for 5 minutes, derivatization of the chromatogram was performed by Camag glass reagent sprayer by spraying the hot plate with anisaldehyde–sulfuric acid. The plate was observed after 30 minutes under ultraviolet (366 nm) light in Camag UV cabinet and the fluorescent image was documented. The corresponding digital scanning profiling was carried out with a Camag TLC scanner III. Documentation of chromatograms was carried out with digital camera. Marmelosin was detected in the petroleum ether extracts of A marmelos whereas a comparative ethyl acetate extract did not show the standard (Figure 2).

Chromatographic fingerprinting of Aegle marmelos extracts. (A) High-performance thin layer chromatography plates exposed to ultraviolet, fluorescence, or visible light showing tracks: S1, marmelosin; T1, ethyl acetate fraction; T2, petroleum ether fraction. (B) High-performance thin layer chromatogram of above extracts and reference standard.
Maintenance of Experimental Animals
Inbred colonies of male and female BALB/c mice weighing 25 ± 4 g housed under hygienic conditions were acclimatized for 1 week prior to all experiments. Optimum temperature of 25°C ± 2°C and humidity of 50% ± 5% was maintained with illumination cycle set to 12-hour light and 12-hour dark. Dalton’s lymphoma ascites (DLA) and Ehrlich’s ascites carcinoma (EAC) were originally procured from reliable sources, but were freshly aspirated at the time of experiments from 15- to 17-day old DLA or EAC tumor-bearing mice. All the experiments were reviewed and approved by Institutional Animal Ethics Committee and Committee for the Purpose of Control and Supervision of Experiments on Animals, India.
Study Design for Immunological, Hematological, and Antitumor Functions
BALB/c mice (n = 8 per group) of either sex were used for the hematological and immunological study. To determine whether the period of initiation of the drug plays a role in the immunological or hematological assessments, AMPE was administered either 48 hours before (AMPEB) or 48 hours after (AMPEA) the EAC or DLA tumor induction (1 × 106 cells per mouse), daily for 14 days. CTX and 5-FU purchased from Dabur Pharmaceuticals Ltd (Sahidabad, India) were used as the gold standards. Experimental groups were assigned as group 1, vehicle (0.1% DMSO); group 2, AMPE (300 mg/kg body weight started on day 0); group 3, EAC; group 4, EAC + AMPEB; group 5, EAC + AMPEA; group 6, EAC + 5-FU (30 mg/kg body weight × 6 alternating days); group 7, EAC + 5-FU + AMPEB; and group 8, EAC + 5-FU + AMPEA. Similarly for DLA, group 9, DLA; group 10, DLA + AMPEB; group 11, DLA + AMPEA; group 12, DLA + CTX (50 mg/kg body weight × 4 alternating days); group 13, DLA + CTX + AMPEB; and group 14, DLA + CTX + AMPEA. Additionally, 6 groups (n = 12 per group) for the antitumor studies were assigned as group 1, EAC; group 2, EAC + 5-FU; group 3, EAC + AMPEB; group 4, DLA; group 5, DLA + CTX; and group 6, DLA + CTX + AMPEB. The antitumor studies were extended up to 20 days or till the mouse died, whichever was earlier. Half the animals from this set of experiments were used to determine the median survival time. Experiments were terminated 24 hours after the last doses by cervical dislocation and the blood was drawn by cardiac puncture to assess hematological, immunological, and antitumor responses.
Hematological Parameters
Blood hemoglobin, RBC, total and differential white blood cell (WBC) counts were estimated in the mice as described earlier. 6 The differential count of WBC was performed on a minimum of 200 cells to quantify the percentages of lymphocytes, neutrophils, and eosinophils in the blood smear.
Bone Marrow Cellularity
Mice were sacrificed after the respective drug/tumor/reference agent treatment time points. Briefly, epiphyseal plates on both the ends of femur bone were cut to flush out bone marrow cells with 2 mL of chilled Hank’s balanced salt solution. Cells were dissociated by passing through a 22-gauge needle and the viability was checked using trypan blue. Results were calculated as million cells per femur. Both femora were processed to get the average counts.
Lymphocyte Proliferation Assay
Lymphocytes were isolated from the above experimental mice blood using ficoll-paque (Pharmacia Fine Chemicals, Uppsala, Sweden) mediated gradient centrifugation. These were then cultured in RPMI-1640 (Sigma-Aldrich, St Louis, MO) containing 10% fetal bovine serum in the presence or absence of mitogen phytohemagglutinin (Gibco BRL, Gaithersburg, MD) in 96-well microtiter plates at 37°C in a 5% CO2 atmosphere for 72 hours. The stimulation index was measured by determining the ratio of the stimulated lymphocyte optical density to an unstimulated control optical density by the MTS (Promega, Madison, WI) assay using standard manufacturer’s protocol. The experiments were done in triplicates and the absorbance was read at 490 nm.
Immunophenotyping of Lymphocytes
To characterize the effect of AMPE on the immune status in mice, whole blood from the experimental animals were used for the enumeration of lymphocyte subsets by flow cytometry as mentioned earlier. 6 Briefly, after preparing the cells, surface antigen markers were determined using FITC-labeled antibodies, anti-CD3, anti-CD4, anti-CD8, and anti-CD56 (BD Biosciences, San Jose, CA), and then analyzed immediately on a FACS Calibur (Becton Dickinson, San Jose, CA). On flow cytometric analysis, lymphocytes were gated according to their forward versus side scatter properties and displayed as dot plots, which were converted to percentages. For each sample, 10 000 gated cells were analyzed using BD CellQuest Software. The percentages of positive cells were calculated by subtracting the respective number of isotype labeled negative cells. 15
Macrophage Phagocytosis Assay
To investigate macrophage phagocytosis by AMPE, peritoneal exudate cells from either vehicle (0.1% DMSO) or AMPE-treated mice were collected by injecting 5 mL chilled RPMI-1640 into the peritoneum. The glass-adherent macrophages were separated by adhering peritoneal exudates cells over glass Petri dish in 5% CO2 at 37°C for 3 hours. The nonadherent cells were washed and the rest were incubated with zymosan yeast suspension (Saccharomyces cerevisiae, Sigma-Aldrich, St Louis, MO) in the ratio 1:5 (macrophage:yeast) for 2 hours at 37°C and stained. Viability was determined and macrophages ingesting 3 or more yeast cells were taken as positive and the percentage of phagocytosis was determined by counting at least 200 cells per slide.
Tumor Volume, Median Survival Time, and Percentage Increment in Life Span
DLA or EAC mice treated with AMPEB, CTX, or 5-FU were used to ascertain antitumor activity. To calculate volume of the ascites tumor, mouse abdomen was incised to collect the ascites fluid (V1) in a sterile Petri dish, which was then transferred to a graduated centrifuge tube. The peritoneal cavity was irrigated thoroughly with isotonic saline to prevent coagulation. The volume of saline added (V2) to the same graduated tube was noted. Volume of ascites tumor (V3) was calculated by subtracting the volume of saline added previously from the total volume of ascites fluid + saline, V3 = (V1 + V2) − V2 as described. 16 The median survival time (MST) was determined as explained elsewhere. 17 The increment in life span was calculated by the formula % ILS = (T − C)/C × 100, where ILS is the increment in life span, T represents MST of the treated group, and C represents the MST of the DLA or EAC tumor group.
Packed Cell Volume, Viable and Nonviable Tumor Cell Count
The ascites fluid aspirated from the peritoneal cavity of the DLA or EAC mice treated with AMPEB, CTX, or 5-FU were divided into 2 parts. One part was used to determine the packed cell volume as reported previously. 18 The cells in the other part were separated by centrifugation and stained with trypan blue (0.4% in normal saline). The viable and nonviable tumor cells counts were calculated using Neubauer hemocytometer using standard protocols.
Statistical Analysis
Results are expressed as the mean ± standard deviation. Statistical significance (P < .05) was evaluated by one-way analysis of variance followed by the Tukey–Kramer multiple comparisons or Mann–Whitney test using GraphPad (GraphPad Software Inc, San Diego, CA).
Results
Prophylactic AMPE Stabilizes Hematological and Immunological Functions in Tumor-Induced Mice
AMPE by itself showed significant increase in the total WBC counts (P < .01), percentage of neutrophils (P < .001), bone marrow cells (P < .01), macrophage activation (P < .001), and clonal proliferation of lymphocytes (P < .05) compared with the vehicle control (Table 1). Unexpectedly, lymphocyte counts were largely affected (P < .001). Animals treated with AMPE 48 hours prior to EAC inoculation displayed elevated hematological parameters (P < .001) compared with the EAC alone, while showing a normalized, but slightly enhanced total WBC and neutrophil counts than the control (Table 1). Bone marrow cellularity of the femur (P < .01) and stimulation index of lymphocytes (P < .001) were also increased with AMPE pretreatment compared with the EAC tumor-alone group. Mice treated with AMPE 48 hours after EAC tumor induction largely indicated a decrease (P < .01), but an overall basal level normalization of the blood picture and lymphocyte functions (Table 1) compared with tumor alone mice. Although later treatment of the drug in tumor-challenged mice was effective in increasing lymphocyte counts (P < .001), it was not up to the basal levels. Irrespective of the period of initiation of the drug (48 hours before or 48 hours after), EAC + 5-FU + AMPE–treated mice showed sharp increase in total WBC (P < .01) and bone marrow cells (P < .001) as shown in Table 1. Overall, AMPE positively gains control of the total hematological and immunological functions either by stimulation when administered by itself or by normalization when challenged with transplantable tumors.
Hematological and Immunological Functions in Control/EAC/EAC + 5-FU Mice on AMPE Treatment.

Abbreviations: AMPE, petroleum ether extract of Aegle marmelos; EAC, Ehrlich’s ascites carcinoma; 5-FU, 5-fluorouracil; Hb, hemoglobin; RBC, red blood corpuscles; TC, total count; BMC, bone marrow cellularity; LPA, lymphocyte proliferation assay; SI, stimulation index; MPA, macrophage phagocytosis assay.
AMPE was administered in control or EAC or EAC + 5-FU mice either 48 hours before (AMPEB) or 48 hours after (AMPEA) tumor induction. Data represent the mean ± standard deviation (n = 8) on day 15. Superscript letters a, b, and c represent statistical differences at P < .001, P < .01, and P < .05, respectively, versus EAC or EAC + 5-FU group using Tukey’s multiple comparisons test or Mann–Whitney test.
Mice treated with AMPE either 48 hours before or 48 hours after DLA tumor displayed 2-fold increase in lymphocyte counts (P < .001) and proliferation (P < .05), elevated levels of hemoglobin (P < 0.05) and bone marrow cells (P < .05) compared with the DLA mice (Table 2). Irrespective of whether AMPE was given before or after tumor induction, neutrophil and total counts (P < .001) were strongly reverted. CTX-treated DLA-challenged mice regardless of AMPE treatment before or after tumor instillation, showed strong surge in the total counts and bone marrow cells (P < .001), which was otherwise suffering myeloablation (Table 2). Of note, CTX + AMPE before DLA inoculation induced lymphocyte proliferation (P < .05), whereas after DLA inoculation, it enhanced hemoglobin levels (P < .01). Like in EAC, administration of AMPE 48 hours before DLA inoculation markedly showed the strongest correlation as observed by the recouped antitumor immune responses. Thus, the total immunoprophylactic effect is once again evident, especially when this drug is administered prior to establishing tumor, which in turn may be used to prevent tumor recurrence in the actual clinical scenario.
Hematological and Immunological Functions in DLA/DLA + CTX Mice on AMPE Treatment.

Abbreviations: AMPE, petroleum ether extract of Aegle marmelos; DLA, Dalton’s lymphoma ascites; CTX, cyclophosphamide; Hb, hemoglobin; TC, total count; BMC, bone marrow cellularity; LPA, lymphocyte proliferation assay; SI, stimulation index.
AMPE was administered in DLA or DLA + CTX mice either 48 hours before (AMPEB) or 48 hours after (AMPEA) tumor inoculation. Data represent the mean ± standard deviation (n = 8) on day 15. Superscript letters a, b, and c represent statistical differences at P < .001, P < .01, and P < .05, respectively, versus DLA or DLA + CTX group using Tukey’s multiple comparisons test.
AMPE Largely Restores Helper and Cytotoxic T Cells When Administered Prior to Establishing Tumors
AMPE treatment in control mice resulted in an overall increase in all the subsets evaluated (Figure 3A). In particular, CD3+ (P < .01), CD4+ (P < .001), CD8+ (P < .05), and CD56+ (P < .01) cells showed significant increases compared with the vehicle. More strikingly, AMPE by itself increased CD4/CD8 ratio to 1.78 from the control ratio of 1.54 (data not shown). AMPE treatment 48 hours prior to EAC tumor inoculation showed ~1.5-fold increase (P < .001) in all the subsets (Figure 3B). Of note, AMPE pretreatment significantly recuperated CD3+, CD4+, and CD8+ counts, in an attempt to plateau with normal levels, which was originally reduced in tumor mice. CD56+ cells also augmented this effect. This increase in the subsets (P < .05), except for CD4+ was evident on AMPE treatment even after EAC tumor establishment (Figure 3C). As for the CD4/CD8 counts, earlier treatment of AMPE had a better ratio of 1.63 compared with 1.52 by the later treatment. However, EAC-challenged mice had a reduced CD4/CD8 ratio (1.39). Lymphocyte subsets were hugely reduced when 5-FU was introduced to combat the tumor. Combination of AMPE + 5-FU prior to establishing EAC clearly indicated an advantage as evident by the increased (P < .05) CD3+, CD4+, and CD8+ cells (Figure 3C). However, once EAC was established, AMPE + 5-FU showed negligible effect (P < .05) on the CD8+ and CD56+ cells.

Distribution of cell surface antigen markers. (A) Mice were treated with either vehicle or petroleum extract of Aegle marmelos (AMPE; 300 mg/kg body weight) for 14 days and lymphocytes were isolated from whole blood. Immunophenotyping was done by flow cytometry and the results are expressed as percentage of CD3+, CD4+, CD8+, or CD56+ cells. (B) Effect of AMPE either before (AMPEB) or after (AMPEA) Ehrlich’s ascites carcinoma (EAC) tumor inoculation. (C) Comparison between the efficacy of 5-FU and AMPE in mice challenged with EAC tumor. (D) Effect of AMPE either before (AMPEB) or after (AMPEA) Dalton’s lymphoma ascites (DLA) tumor inoculation. (E) Comparison between the efficacy of cyclophosphamide (CTX) and AMPE in DLA mice. Data represent the mean ± standard deviation (n = 8). Statistical significant differences at a (P < .001), b (P < .01), and c (P < .05) versus control or tumor alone or tumor + reference drug, using Mann–Whitney or Tukey’s test.
Regardless of the initiation schedule, AMPE treatment in DLA mice resulted in the significant increase (P < .001) of CD3+ and CD4+ cells (Figure 3D). However, CD8+ cells were increased (P < .01) only in the pretreatment group. CD4/CD8 ratios were also higher in both the AMPE groups (1.57) compared with the DLA (1.42). Surprisingly, none of the subsets showed any significance either with CTX or in combination (Figure 3E). Altogether, AMPE showed an overall increase of the subsets by itself while also restoring CD3+, CD4+, and CD8+ counts in mice challenged with EAC and DLA tumors.
Prophylactic AMPE Exhibits Strong Antitumor Activity in Murine Ascites Tumors
Antitumor activity of AMPE was ascertained by evaluating tumor regression and survival in EAC or DLA mice. The mode of treatment, that is, before or prophylactic was chosen based on the positive immunopotentiating responses of AMPE in the tumor-bearing hosts. This efficacy was compared with standard anticancer agents such as 5-FU and CTX. Our results show that AMPE prophylactically decreases packed cell volume and volume of ascites while increasing nonviable tumor cell count and MST (P < .001) compared with the respective tumor-alone controls (Table 3). As expected, reference drugs also replicated this effect. Most important, AMPE contributed to increase in MST of EAC mice by 1.6-fold, whereas it was only 2.1 fold for 5-FU, being a standard drug. On the other hand, in the DLA mice, MST was increased by 1.4-fold by AMPE whereas it was only a fraction more for CTX. The percentage increment in life span by AMPE was 62.86 and 42.22 for EAC and DLA, respectively (Table 3). Significantly reduced but normal trends of body weights (P < .05) were observed for all the treatment groups compared with the mice challenged with tumor (data not shown). The increase in body weight seen in the tumor-alone group is perhaps because of the increased tumor burden. Spleen weights were reduced considerably (P < .001) in the standard anticancer drug treated groups whereas it was either improved or normalized in the presence of AMPE (data not shown). This decrease might indicate the nonspecific effect on the hematopoiesis by 5-FU and CTX at the time of tumor cell killing. Nonetheless, the present outcome reveals the significant impact of AMPE in eliciting beneficial immune responses with strong tumor impeding capabilities in both the transplantable tumor models, with a slight advantage for EAC.
Antitumor Activity of Prophylactic AMPE on EAC and DLA Ascitic Tumors.
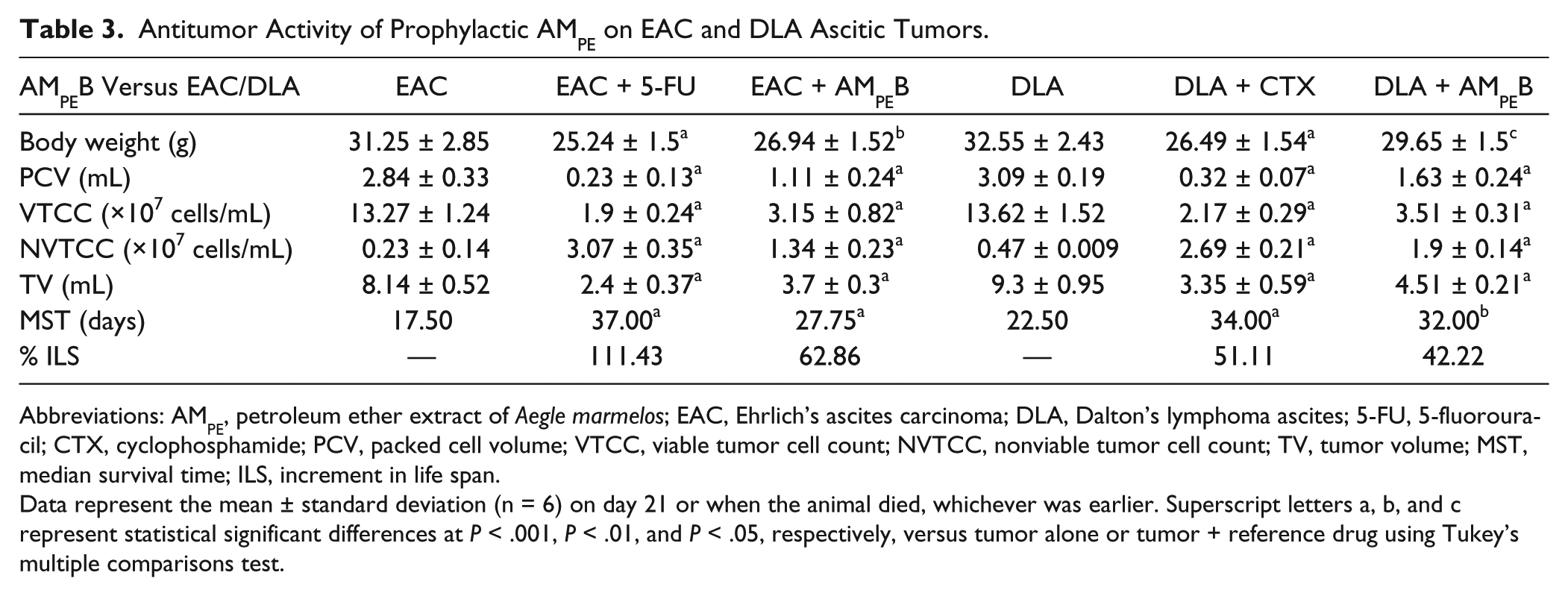
Abbreviations: AMPE, petroleum ether extract of Aegle marmelos; EAC, Ehrlich’s ascites carcinoma; DLA, Dalton’s lymphoma ascites; 5-FU, 5-fluorouracil; CTX, cyclophosphamide; PCV, packed cell volume; VTCC, viable tumor cell count; NVTCC, nonviable tumor cell count; TV, tumor volume; MST, median survival time; ILS, increment in life span.
Data represent the mean ± standard deviation (n = 6) on day 21 or when the animal died, whichever was earlier. Superscript letters a, b, and c represent statistical significant differences at P < .001, P < .01, and P < .05, respectively, versus tumor alone or tumor + reference drug using Tukey’s multiple comparisons test.
Discussion
Coupling the properties of modifying immune systems of the host while delivering powerful antitumor responses offers a lethal therapeutic synergy against cancer. Despite numerous pharmacological activities reported for A marmelos (L.) Correa, an extensive study with regard to its mechanism of chemopreventive immunity, and comparing its activity with the existing drugs such as CTX and 5-FU has not been attempted. The present study reports a comprehensive immunological and antitumor evaluation of A marmelos using 2 different murine ascites tumors. This study demonstrated that administration of AMPE prior to establishing DLA or EAC tumor; in a scenario where the initial tumor has been treated or resected and the chances of recurrence are high, showed robust stimulation of the immune responses with strong antitumor activity. Our data show the evidence of activated macrophages, stimulated lymphocyte clonal expansion, increased CD4/CD8 ratio, and enhanced helper and cytotoxic T cells, which was even better than the CTX and 5-FU treatment. This plant drug also increased the survival and life span of the tumor-bearing mice by greatly reducing viable ascites tumor cell counts.
An interesting finding in this study is that the AMPE was able to stimulate or restore the immune responses when challenged with ascites tumors. Importantly, blood picture, lymphocyte clonal expansion, and macrophages were significantly improved, which are central for the activation of the immunological responses. In particular, preponderance of neutrophils was apparent in controls treated with this drug. Neutrophils are important modules orchestrating dendritic cells, monocytes, macrophages, and lymphocytes to initiate and maintain immune responses. 19 Conversely, excess activation of the neutrophils can severely impair T-cell activation in advanced stages of cancer, 20 which is consistent with our observation where the lymphocytes were outnumbered by neutrophils in ascites tumors with a concomitant T-cell anergy. In the presence of the drug, lymphocyte counts, and their functional capacity to respond to mitogen was either increased or revamped when challenged with tumor, which indicate the stimulatory effect this drug can deliver against opportunistic pathogens. Macrophage phagocytosis is yet another determinant of the immune defense mechanism, which ingests pathogens, cancer cells, tissue debris and so on,21,22 which is a vital function stimulated by AMPE for the widespread tumor clearance. Bone marrow is the site of hematopoiesis and any stress, including tumor can cause severe imbalance. Our data show that treatment with reference agents such as CTX/5-FU cause myeloablation, which further weaken the immune system to combat cancer in mice. But, combination treatment of agents with AMPE restored functionality, indicating the ability of this drug to be also effective under immunocompromised conditions.
In the antitumor immunity, T cells are the predominant immune cells that combat either by direct or indirect cytotoxic mechanisms aided by the production of cytokines such as TNF-α and interferon-γ. These in turn activate natural killer (NK) cells, which specifically target tumor cells in the absence of antibody production. Indeed, immunophenotyping data showed increasing CD3+, CD4+, CD8+, and CD56+ cells, and especially helper and cytotoxic subsets when the drug was started prior to tumor induction. Lymphocyte ablation already induced by the tumor was worsened by the treatment with 5-FU. This was effectively counteracted by the prophylactic administration of AMPE. CD4 and CD8 T cells are involved in cell-mediated immunity, which coordinates immune responses against tumors, whereas the CD8 cells and NK cells directly kill cancer and other infected cells.23,24 Proper CD4/CD8 ratio is an indication of the functioning of the immune system and a ratio less than 1.5 is associated with impaired immunity. 25 This drug was able to constantly maintain a CD4/CD8 ratio >1.5 irrespective of the treatment regimen, which clearly strengthens the overall immunostimulatory role under drastic immunocompromised circumstances. Thus, the mechanism underlying cell-mediated antitumor immunity of AMPE involves a deadly combination of cytotoxic T cells and natural killer cells, which can virtually kill any altered nucleated host cell, along with the neutrophils and macrophages, which altogether remove the rest of the tumor debris.
To our knowledge, levamisole is the only immunomodulatory drug approved by the Food and Drug Administration in patients with colon cancer. But this has limited drug utility because of toxic side effects. 26 The present drug, AMPE, exhibits remarkable ability to balance immunity under stress by also delivering powerful antitumor responses without any side effects or toxicity. This unique ability to compensate the impaired immune defense mechanisms by also not causing auto immune disorders is definitely a virtue. The primary endpoint of a valued anticancer drug in a preclinical setting is its ability to reduce tumor size and increase life span. 27 Interestingly, the results of this study showed better tumor-specific responses on both the aspects.
Aegle marmelos is a diverse chemical factory containing many bioactive compounds. Our chromatographic analyses mainly identified xanthorrhizol and imperatorin (marmelosin) as major components. Both these compounds have been shown to inhibit cancer cell growth and proliferation28-34 and there are no reports on its immunoprophylactic or immunomodulatory potential. Although this drug is promising, it is yet to be determined whether this identified individual compounds can alone impart this chemopreventive immunity or whether a synergistic effect is desired to attain maximal activity.
Conclusion
In conclusion, considering AMPE is very effective prophylactically, it has the potential to be developed as a drug candidate, especially for the chemoprevention of the cancer where the current drugs have miserably failed. Although further studies are warranted, therapeutic testing of this natural drug in cancer patients with high risk for recurrence or in patients suffering from immunocompromised disease conditions, will improve its drug utility thereby reducing the morbidity and mortality rates.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was in part financially supported by the Ayurveda, Yoga and Naturopathy, Unani, Siddha and Homeopathy (AYUSH), New Delhi, India. The first author (SKG) was supported by the Senior Research Fellowship (45/20/2006/BMS/TRM) from Indian Council of Medical Research (ICMR), New Delhi, India for the study.
