Abstract
Keywords
Introduction
Cancer is a leading cause of death worldwide with an estimated 14.1 million new cases and 8.2 million cancer deaths occurring worldwide in 2012. 1 Conventional cancer treatment, including surgery, chemotherapy, and radiotherapy, which focuses on eliminating cancer cells, can be effective in the short term. 2 However, such approaches are insufficient to eradicate all malignant cells, especially in advanced cancer, resulting in recurrence. Repeated cycles of such intensive treatment can suppress the immune system, promote chemotherapy- and radiotherapy-resistant tumors, as well as local and systemic toxicity. 3 Immunotherapy, which uses nonchemical, biological substances called biological response modifiers (BRMs) or immunomodulators to induce, boost, or restore the body’s natural defense capability to fight cancer, 4 is now being acknowledged as an important cancer treatment strategy that can work in combination with conventional therapies.5,6
BioBran/MGN-3 Arabinoxylan (MGN-3) is a natural blend of hemicelluloses derived from partially hydrolyzed rice bran with shiitake mushroom enzymes (Lentinus edodes mycelia extract). It is developed and manufactured in Japan by Daiwa Pharmaceutical Co, Ltd, and marketed worldwide as a nontoxic food supplement under different brand names such as BioBran (Globally), Lentin Plus (Japan/Asia), Ribraxx (Australia/New Zealand), BRM4 (United States), and others. 7 The main chemical structure of MGN-3 is an arabinoxylan with a xylose in its main chain and an arabinose polymer in its side chain. The result of a methylation analysis suggests that a complex structure of heteropolysaccharide (arabinogalactan, arabinoxylan, arabinan, β-1,3:1,4-glucan) is behind the immunomodulatory mechanisms and antitumor activity of MGN-3. 8 MGN-3 has been promoted as a plant-based BRM for enhancing the depleted immune system during and after conventional cancer treatment, claiming support from research findings and good clinical results. 9
Objectives
To perform a comprehensive literature review of the available evidence on the effects and efficacies of MGN-3 as a complementary therapy to support conventional cancer treatment.
Methods
Systematic searches were conducted using research databases including PubMed, ProQuest, MEDLINE, EBSCOhost (All), Cochrane CENTRAL, Embase, and EBM Review (All) without any restriction in year of publications. Keywords used include “MGN-3,” “BioBran,” “arabinoxylan,” “rice bran,” and “plant immunomodulators” in combination with “cancer” or “chemotherapy.” Additionally, the collected papers on MGN-3 published by BioBran Research Foundation, 10 the official website of MGN-3 (https://biobran.org), the English language website of Daiwa Pharmaceutical (http://www.daiwa-pharm.com/english), and the references of included articles were searched manually. The criteria for inclusion were (1) primary research reports published in English inclusive of in vitro, in vivo, clinical study, and case reports and (2) research studies focused on the effects of MGN-3 on any form of cancer alone or as a combination therapy, or the combined effects of MGN-3 with one or more chemotherapeutic agents, or the effects of MGN-3 on cancer patients.
Results
Search Results
Systematic searches were conducted by 2 authors (DM and SLO) independently between March to May 2017. The searches yielded 166 unique records after duplications were removed. After initial screening, 48 articles were assessed for eligibility. Sixteen articles were excluded with reasons as follows: duplication (5),11-15 not cancer focused (7),16-22 not primary study (3),23-25 and 1 case report article published in Japanese was also excluded. 26 Thirty full-text articles and 2 conference abstracts were included in this review, consisting of 7 in vitro studies;27-33 1 article reporting results from both in vitro and in vivo studies; 34 7 in vivo studies;35-41 6 clinical case reports;42-47 and 11 clinical studies,48-58 with one of them also including case reports of selected follow-up patients. 48 The literature search flow is summarized in Figure 1.

Literature search flow diagram (based on PRISMA [Preferred Reporting Items for Systematic Reviews and Meta-Analyses] 2009).
In Vitro Studies
The characteristics of the in vitro studies are listed in Table 1. In these studies, both the immunomodulating and synergistic anticancer effects of MGN-3 were demonstrated.
Characteristics of the Included In Vitro Studies.

Immunomodulating Anticancer Effect
The growth of squamous cell carcinoma (SCC13) was arrested after incubating in MGN-3 for up to 72 hours. 27 Tumor cell growth was arrested with the increase in apoptosis through the CD95 death receptor pathway. An increase of more than 200% in the rate of apoptosis of MGN-3 pretreated tumor cells (human T cell leukemic HUT 78) to the agonistic anti-CD95 antibody was observed. The effect was shown to be dose dependent. 28 Compared with resting NK cells, MGN-3 stimulated NK cells induced a higher expression of the activation associated receptors CD25 and CD69. Statistically significant increases in cytotoxicity of MGN-3 stimulated NK cells against several cancer cells were consistently observed, but cytotoxicity was absent against normal cells. 34
Synergistic Anticancer Effect
MGN-3 works synergistically with natural anticancer substances such as Saccharomyces cerevisiae (Baker’s yeast) and curcumin, as well as chemotherapy drugs (daunorubicin and paclitaxel) in a dose-dependent manner.
Ghoneum and Gollapudi29,30 confirmed the ability of MGN-3 to accelerate the phagocytosis of S cerevisiae in breast cancer and enhance the yeast-induced apoptosis of breast cancer cells (BCCs).29,30 After incubating different BCCs (MCF-7, ZR-75, HCC70) with heat-killed S cerevisiae and MGN-3, a 2-fold increase was observed in the attachment and uptake of yeast by the treated MCF-7 cells, compared with untreated cells, in a time-dependent manner. 30 Treatment with MGN-3 also resulted in a 1.8- to 2.5-fold increase in the percentage of apoptosis in different BCC cell lines. 29 The yeast-phagocytosis and apoptosis effect was not present in nontumorgenic breast cells. 30
Culturing human multiple myeloma (MM) cell line U266 with MGN-3 (50 or 100 μg/mL) and curcumin (2.5-10 μM) for 3 days, Ghoneum and Gollapudi 32 also showed that the proliferation of U266 cells was inhibited by MGN-3 alone or curcumin alone. 32 However, an optimal synergistic effect was observed with a combination of 100 μg/mL MGN-3 plus 10 μM curcumin, characterized by an 87% decrease in U266 cell numbers and a 2.6-fold increase in the percentage of apoptotic U266 cells. 32
The survival rates of BCCs (MCF-7 and HCC70 cells) cultured for 3 days with different concentrations of daunorubicin (1 × 10−9 to 1 × 10−6 M) with or without MGN-3 (100-1000 μg/mL) were measured. 31 MGN-3 increased the accumulation of daunorubicin in the cancer cells and significantly decreased the cell survival of MCF-7 (by 5.5-fold) and HCC70 cells (by 2.5-fold) as compared to BCCs treated with daunorubicin alone. 31 Through a similar experimental design in a later study, MGN-3 was demonstrated to sensitize non-metastatic human BCC MCF-7 and metastatic murine BCC 4TI to paclitaxel and to increase their susceptibility to the chemotherapeutic agent by over 100-fold. The synergistic effects include causing DNA damage, enhancing apoptosis, and inhibiting cell proliferation of BCCs. 33
In Vivo Studies
The characteristics of the in vivo studies are listed in Table 2. Through various murine models, these studies showed that MGN-3 could potentially prevent cancer, inhibit its growth, and work synergistically with other chemotherapeutic agents.
Characteristics of the Included In Vivo Studies.

Preventive Effect Against Carcinogenic Agents
Significantly lower incidences of dysplasia and gastric cancer were observed in male Wistar rats fed with carcinogen methylnitronitrosoguanidine (MNNG) plus MGN-3, compared with those administered MNNG alone. 39 The preventive effects of MGN-3 observed include suppression of Ki-67 tumor marker, upregulation of apoptotic gastric cancer cells via the mitochondrial-dependent pathway, and protection against decrease in lymphocyte levels. 39
Male albino rats pretreated with MGN-3 before receiving carcinogenic N-nitrosodiethylamine (NDEA) and carbon tetrachloride (CCl4) showed a significant reduction in liver tumor incidence, marked decrease in the percentage of preneoplastic foci in hepatic parenchyma, and inhibition in the development of hepatocellular carcinoma, compared with controls. MGN-3 treated rats were also able to maintain close to normal levels of hepatic diagnostic markers. 41
Inhibition of Cancer Growth
Female Swiss albino mice bearing solid Ehrlich carcinoma (SEC) tumors were treated with intraperitoneal injections of MGN-3.37,38 Compared with controls, MGN-3-treated mice exhibited a significant delay in tumor growth measured by tumor volume (63.27%) and tumor weight (45.2%), without any observed adverse side effects due to the treatment. 37 The anticancer mechanisms of MGN-3 were shown to be immunomodulating through upregulation of tumor necrosis factor-α (TNF-α) and interferon-γ (IFN-γ), and downregulation of interleukin-10 (IL-10), as well as inducing apoptosis in the SEC cells. 37 Elevated levels of antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, catalase, and glutathione-S-transferase, in blood and liver of MGN-3-treated animals comparable to the normal levels were observed. 38 The effects of MGN-3 treatment were more pronounced in mice that were treated earlier rather than later.
NOD-scidIL-2Rgnull mice inoculated with neuroblastoma cells were treated with intravenous NK cellular therapy using either fresh NK cells or NK cells activated with MGN-3 overnight. The mice treated with MGN-3 activated NK cells had upregulated NK cell activation markers, significant neuroblastoma growth inhibition, and higher survival rate compared to control groups. 34
Synergistic Effects With Chemotherapeutic Agents
Female Swiss albino mice inoculated with Ehrlich ascites carcinoma cells were treated with paclitaxel only, MGN-3 only, or MGN-3 plus paclitaxel. Tumor volumes were significantly suppressed in MGN-3 plus paclitaxel group (88% smaller relative to controls), compared with paclitaxel only group (77%), and MGN-3 only group (59%). 40
MGN-3 may also potentially protect against the adverse effects of chemotherapeutic agents. Sprague-Dawley derived albino rats fed with MGN-3 were healthier, gained weight, and had a lower incidence of diarrhea and gross intestinal pathology, after being administered a dose of either cisplatin or doxorubicin, compared with control. 35 Similarly, MGN-3 given either orally or intraperitoneally to BALB/c female mice was shown to protect them from severe weight loss associated with the injection of cisplatin. 36
Case Reports
The included case reports42-48 narrated a total of 14 successful MGN-3-treated clinical cases. The patient characteristics and outcomes are summarized in Table 3.
Patient Characteristics and Outcomes of the Included Clinical Case Reports.

Most patients were older than 50 years with only 2 patients of younger age, and 2 patients with age unreported. Types of primary cancer in these cases include leukemia (2), prostate (1), breast (1), colorectal (2), pancreas (1), liver (3), lung (2), skin (1), and ovary (1). Most patients started taking MGN-3 either in conjunction with conventional cancer treatment, or after completion of conventional treatment. Dosages of MGN-3 reported in these cases were 3 g/d, 6 g/d, and 45 mg/kg per body weight (BW), administered orally. In some cases, MGN-3 was combined with other forms of complementary therapies, including ACM π water, 43 Bio-reproducing Protein (BRP), 44 and mistletoe lectin.46,47
In nonterminal cancer cases, reported results were: improvements in tumor markers, immunocompetence profile, and initial symptoms 44 ; patient conditions were stable with no sign of cancer recurrence at follow-up 48 ; and patients showed improved quality of life (QoL) reported as subjective improvements in sleep, appetite, digestion, physical activity and decrease in anxiety and pain; reduced adverse effects during chemotherapy, and cancer remission.46,47 In one self-treated case, the patient refused further conventional treatment for metastatic lung tumor after the initial removal of his skin lesion. The metastatic lung tumor became undetectable after 34 months of self-treatment with MGN-3. 45 In 3 cases of terminal cancer with poor prognosis, the patients were reported to be able to survive much longer beyond the initial estimated lifespans with improved QoL, and even normal working lives.42-44
Clinical Studies
Eleven clinical studies were included in this review. Among the 11 clinical studies, 5 are nonrandomized, pre-post intervention studies,48,50,51,57,58 and 6 are randomized controlled trials (RCTs).49,52-56 The characteristics of these studies are summarized in Table 4 (nonrandomized studies) and Table 5 (RCTs).
Characteristics of the Included Nonrandomized Clinical Studies.

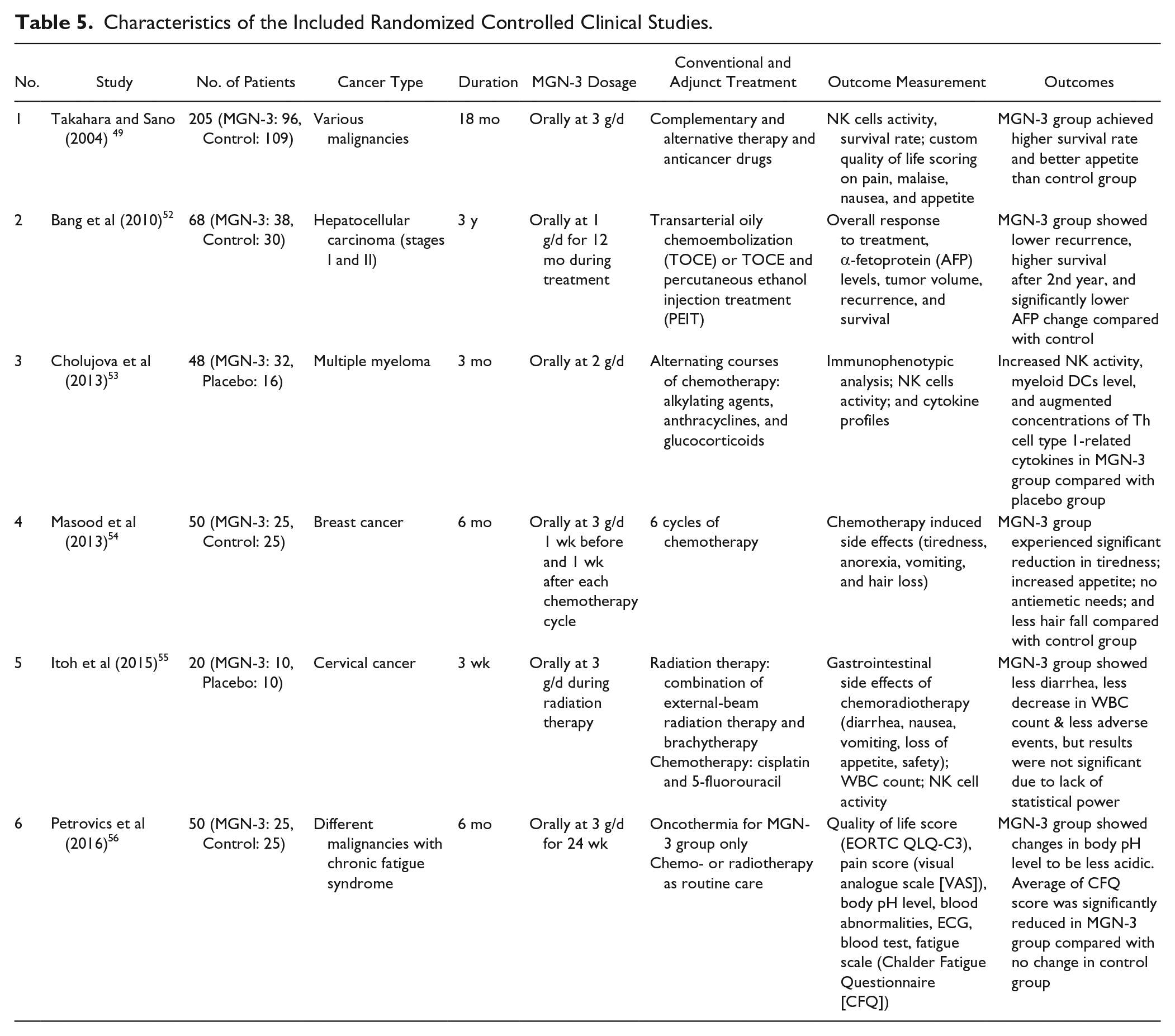
Characteristics of the Included Randomized Controlled Clinical Studies.
Nonrandomized, Pre-post Intervention Studies
The immunomodulating effects of MGN-3 were examined by Ghoneum and Brown. 48 Thirty-two cancer patients (various malignancies) with depressed NK cell activity post conventional cancer treatment were treated with MGN-3 for 2 weeks. A significant increase in NK cell activity up to tenfold was observed. Increase in NK cell granularity and binding capacity, improvement in T and B cell proliferation in vivo, and improvement in tumor-associated antigens were observed in a selected number of patients. 48 Similarly, Tsunekawa 50 reported normalization of NK cell activity and improvement in white blood cell (WBC) count in a small study of 16 cancer patients. These were patients with various malignancies who went through conventional cancer treatment before starting the MGN-3 therapy for 6 months. No subjective or objective adverse effects were recorded.
In another study, however, no substantial change in NK cell counts was observed in 22 cancer patients after taking MGN-3 for 2 months. 51 Instead, an increase in the T helper (Th) lymphocytes to T regulatory (Treg) lymphocytes (CD4+ CD25+) ratio (Th:Treg) was recorded, demonstrating the effect of anticancer immunity restoration of the MGN-3 therapy. Nonetheless, due to the small number of patients, the results were not statistically significant. 51 More recently, Golombick et al 57 reported that the combination therapy of MGN-3 and curcumin helped 20 patients with early B-cell lymphoid malignancies to delay disease progression by increasing neutrophil count and reducing the raised erythrocyte sedimentation rate (ESR). No significant change in NK cell counts was detected. Again, results from this study lack statistical power.
The QoL improvement effects of MGN-3 were studied by Hajto et al. 58 A total of 35 patients with various malignancies were administered a combination of mistletoe lectin and MGN-3 as a complementary therapy for a minimum of 6 months during or after conventional cancer treatment. Patients responded to a custom binary-response questionnaire with 8 questions regarding various aspects of their QoL after treatment, including pain, anxiety, physical activity, appetite, and sleep, reporting a subjective improvement of physical activity and decrease in side effects as the most important benefits of the complementary therapy. 58
Randomized Controlled Studies
The QoL improvement effects of MGN-3 were previously studied in a large RCT with 205 progressive cancer patients in late stages (III-IV) of various malignancies. 49 Checking patient QoL by observation and enquiry during the study using a custom grading scale, Takahara and Sano 49 reported that, patients who received MGN-3 plus standard complementary and alternative therapy (CAT) achieved a higher survival rate (54.2% vs 35.8%) and a better appetite than the control group who received only standard CAT. There was no clear difference between groups in terms of percentage changes in increasing and decreasing NK activity, but the MGN-3 + CAT group had a higher percentage of patients with unchanged NK activity compared with the CAT only group. 49 It was unclear whether this study was sufficiently blinded.
The RCT by Bang et al 52 can be regarded as the first clinical evidence for the antitumor efficacy of MGN-3. The participants of this 3-year RCT consisted of 68 patients with liver cancers of stage I or II. A higher survival rate after the second year (35% vs 6.7%) and lower recurrence of the disease (31.6% vs 46.7%) for liver cancer patients taking MGN-3, compared with the controls, were the results of MGN-3 applied in conjunction with conventional cancer treatment in 38 patients. 52 Thirty patients receiving only conventional therapies were assigned as controls. Significant decreases in the tumor marker level (α-fetoprotein [AFP]), and tumor volumes, as compared with baseline, were also observed in the MGN-3 group whereas the control group showed no significant changes in either AFP or tumor volume. 52
MGN-3 was shown to reduce chemotherapy-induced side effects among breast cancer patients who were undergoing 6 cycles of chemotherapy in another RCT with 50 patients. 54 MGN-3 was given to 25 patients 1 week before and 1 week after each cycle of chemotherapy, with the control group receiving only chemotherapy. Side effects were assessed by a custom questionnaire given to patients at the start of each cycle. Significant reductions in tiredness, increased appetite, no anti-emetic needs, and reduced hair loss compared with the control group were reported. 54
In cervical cancer patients undergoing chemoradiotherapy, MGN-3 was shown to be better than placebo in reducing the diarrheal side effect in a clinical trial reported by Itoh et al., using a custom symptom scoring system. 55 Adverse events were graded by the National Cancer Institute scale (Common Terminology Criteria for Adverse Events [CTCAE] v3.0). Only 2 patients in the MGN-3 group developed grade 2 (moderate) events during chemotherapy, with the control group tending to develop adverse events of higher grades than did the MGN-3 group. 55 Patients taking MGN-3 also showed less decrease in WBC count. However, the results from this pilot trial with only 20 patients were not significant due to the lack of statistical power. 55
For patients with chronic fatigue syndrome (CFS) due to cancer or cancer treatment, MGN-3 plus Oncothermia was shown to be able to reduce the CFS symptoms in an RCT with 50 CFS patients diagnosed with various malignancies. 56 The 25 patients who received 3 g/d of MGN-3 and Oncothermia once a week had significantly lower mean scores of Chalder Fatigue Questionnaire (CFQ) compared with baseline after 6 months of treatment. No significant change in mean CFQ scores was recorded in the control group who received only conventional chemo or radiotherapy treatment. 56
The effects of MGN-3 on innate immunity were studied by Cholujova et al 53 in an RCT with 48 multiple myeloma patients (MGN-3 group, 32; placebo, 16). Detailed blood analyses were performed before and during the 3 months of treatment. Significant increase in NK activity, myeloid dendritic cell level, and augmented concentrations of Th cell type 1-related cytokines were observed in the MGN-3 group. No significant change in the placebo group was observed. Hence, MGN-3 clearly improved the innate immunity profile of the patients compared with placebo. 53
Risk of Bias Assessment
We have assessed the risk of bias of the included RCTs using the Cochrane Risk of Bias tool. 59 The results of the assessment are summarized in Figures 2 and 3. Because of insufficient information provided on randomization and blinding, most included RCTs have unclear risk in at least one or more items in selection bias, performance bias, and detection bias. Only the study by Itoh et al. 55 is considered low risk in these items. Notwithstanding, this study has a high risk of attrition bias due to a high percentage of participants being excluded from assessment (30%). 55 The risks of attrition bias and reporting bias are low in most other studies except the study by Masood el al, 54 which has unclear risk of bias across all items of assessment due to insufficient detail in published information. We assess the study by Takahara and Sano 49 to have a high risk in other bias. It was conducted by a commercial hospital specialized in CAT for cancer patients, thus the risk of bias cannot be excluded due to conflict of interest. With industry influence being a potential source of other bias, only the study by Petrovics el al 56 provided disclosure on funding and clarified the roles of industry partners in the study, which the other studies did not.

Risk of bias summary: Review authors’ judgements about each risk of bias item for each included study.

Risk of bias graph: Review authors’ judgements about each risk of bias item presented as percentages across all included studies.
Discussion
Current evidence supports MGN-3 to be a BRM. MGN-3 is an evidence based and standardized arabinoxylan concentrate from plant origin which, similar to bacterial pathogen associated molecular pattern molecules, can stimulate the body’s natural immune system to protect against cancer growth. 46 The effect of MGN-3 in upregulating the cytotoxic activities of NK cells, by multiple-fold, has been most prominently demonstrated.34,48-50,53 The anticancer effect of NK cells is the subject of intense current research and NK cell immunotherapy is being touted as the future of cancer immunotherapy. 4 Effectiveness of NK activity has been associated with good prognosis in patients with metastatic cancers.60,61 This may explain the life-prolongation effect of MGN-3 as reported in a number of clinical studies49,52 and case reports.42-44 However, MGN-3 treatment does not increase the absolute NK cell counts as noted in clinical studies,51,57 which is a limitation. Cancer patients with low NK cell counts (<0.15 × 109/L) tend to have lower survival rates,62,63 indicating that lower basic level of natural antitumor activity can reduce the efficacy of a BRM. While they may benefit from the upregulated NK activity with MGN-3 treatment to a certain extent, 49 combining NK cellular therapy 64 with MGN-3 may further improve prognosis in these patients; an approach that warrant future clinical investigation. 34
Other immunomodulatory effects of MGN-3 include the following: increase susceptibility of cancer cells to undergo apoptosis mediated by CD95 death receptors 28 ; influence plasma cytokine production (upregulated TNF-α and IL-12, while downregulating IL-10) 37 ; enhance the activity of antioxidant enzymes 38 ; improve T and B cell proliferation 48 ; improve Th cell concentration,51,53 downregulate Treg cells 51 ; and activate dendritic cells.13,22,53 These effects have been presented in depth by Ghoneum 65 in a recent review, supporting the use of MGN-3 as an effective BRM in cancer therapy.
MGN-3 has been tested in clinical trials as an adjunct therapy during conventional chemo- and radiotherapy with effects that include (1) improvement of immunoprofile,53,55 (2) reduction of side effects (diarrhea, nausea, pain, tiredness, anorexia, and vomiting),52,54-56,58 and (3) improvement in treatment outcomes. 52 Outcome improvement and reduction of side effects have been mostly attributed to an improved balance of the innate immune system. However, MGN-3 has also been shown to work synergistically with daunorubin 31 and paclitaxel33,40 in preclinical studies, as well as transarterial oily chemoembolization (TOCE) and percutaneous ethanol injection treatment (PEIT) for liver cancer treatment in a clinical trial, 52 by sensitizing the cancer cells to these agents. More research is needed to explore such synergistic effects with other chemotherapeutic agents and radiation therapy to fully explore the potential of MGN-3 as a combination therapy in conventional cancer treatment.
As a follow-up therapy after conventional cancer treatment, MGN-3 has demonstrated effects in restoring weakened immunoprofile,48-51 improving QoL and survival rate of late-stage cancer patients, 49 as well as reducing recurrence. 52 Such results are consistent with the clinical experiences reported in the published case reports.42-47 As such, MGN-3 may also be considered as part of the follow-up care after conventional cancer treatment.
No adverse reaction was reported in studies that explicitly monitored potential MGN-3 induced adverse effects in vivo 37 and in a clinical study. 50 No adverse event due to MGN-3 was reported in any of the included clinical trials or clinical case reports. Furthermore, the safety MGN-3 has been studied in a series of animal studies. The median lethal dose (LD50) of MGN-3 is more than 36 g/kg, and the “no observed adverse effect level (NOAEL)" is 200 mg/kg/d or higher. 66 Therefore, typical MGN-3 dosages of 3 to 6 g/d or 45 mg/kg BW/d are extremely safe.
At present, there is no study on the pharmacokinetics of MGN-3 and the achievable level of MGN-3 in serum is unknown (personal email communication with Dr Mamdooh Ghoneum at Charles Drew University of Medicine and Science on August 4, 2017). Since concentrations of MGN-3 used in some of in vitro studies were very high (500-1000 μg/mL),28,29,31,33 it is unclear whether these concentrations can be of clinical relevance. Although the MGN-3 dosages (5-50 mg/kg/BW) used for in vivo studies34,35,37-41 were more closely matched to the typical dosage used in human studies, without understanding of the pharmacokinetics of MGN-3, questions remain whether the higher bioavailability via intraperitoneal injection in some animal studies36-41 can be applicable to humans.
The clinical research on MGN-3 is still at its early stage with only a small number of RCTs available in the literature. Most of them are small pilot trials with limited participants (N < 100) and short duration (<=6 months). At the moment only one RCT is available to support clinical evidence. 52 The other RCT that has adequate size (N = 205) and duration (18 months) 49 suffers from methodological limitations including inadequate blinding, no placebo control, and potential risk of bias due to conflict of interest. Hence, large, well-designed, long-term placebo-controlled RCTs are needed to further evaluate the effects of MGN-3 as a complementary therapy to support conventional cancer treatment.
Conclusion
Current evidence from preclinical studies, clinical case reports, and small clinical trials suggests that MGN-3 can be an effective BRM to complement the conventional cancer treatment through upregulating the patient’s immune system, especially in boosting the NK cell activity. MGN-3 appears safe in its application with no adverse event reported to date. It may be used as a complementary immune therapy to reduce side effects, improve treatment outcomes, and enhance long-term survival rate. The combination of MGN-3 with new biological targeting treatments may open new perspectives in the tumor therapy. Nevertheless, we call for additional study into the pharmacokinetics of MGN-3, as well as more well-designed RCTs to confirm its efficacies and strengthen the evidence to support its clinical application.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
