Abstract
In the current study, we investigated the chemopreventive activity of arabinoxylan rice bran, MGN-3/Biobran, against chemical induction of glandular stomach carcinogenesis in rats. Gastric cancer was induced by carcinogen methylnitronitrosoguanidine (MNNG), and rats received MNNG alone or MNNG plus Biobran (40 mg/kg body weight) for a total of 8 months. Averaged results from 2 separate readings showed that exposure to MNNG plus Biobran caused gastric dysplasia and cancer (adenocarcinoma) in 4.5/12 rats (9/24 readings, 37.5%), with 3.5/12 rats (7/24 readings, 29.2%) showing dysplasia and 1/12 rats (8.3%) developing adenocarcinoma. In contrast, in rats treated with MNNG alone, 8/10 (80%) developed dysplasia and adenocarcinoma, with 6/10 rats (60%) showing dysplasia and 2/10 rats (20%) developing adenocarcinoma. The effect of combining both agents was also associated with significant suppression of the expression of the tumor marker Ki-67 and remarkable induction in the apoptotic gastric cancer cells via mitochondrial-dependent pathway as indicated by the upregulation in p53 expression, Bax expression, downregulation in Bcl-2 expression, an increase in Bax/Bcl-2 ratio, and an activation of caspase-3. In addition, Biobran treatment induced cell-cycle arrest in the subG1 phase, where the hypodiploid cell population was markedly increased. Moreover, Biobran treatment protected rats against MNNG-induced significant decrease in lymphocyte levels. We conclude that Biobran provides protection against chemical induction of glandular stomach carcinogenesis in rats and may be useful for the treatment of human patients with gastric cancer.
Introduction
In 2015, in the United States alone, about 24 500 cases of stomach cancer were diagnosed and more than 10 000 people died from this type of cancer. 1 Gastric cancer was placed as the second leading cause of cancer-related deaths throughout the world. 2 In 2012, there were approximately 723 000 deaths from gastric cancer and 951 500 new cases worldwide. With a mortality rate close to 75%, stomach cancer is the fourth most common malignancy in the world. 3 Despite progress in options for treatment, gastric cancer is most frequently discovered in advanced stages, 4 and there is a low survival rate over 5 years at 27%. 2 Gastric cancer is thought to arise from chronic inflammation caused by Helicobacter pylori. 5 For several decades, the development of gastric cancer has been theorized to go through several stages, as follows: normal mucosa → chronic active gastritis → atrophy → intestinal metaplasia (I/II/III) → dysplasia → gastric cancer. 6
N-methyl-N′-nitro-N-nitrosoguanidine (MNNG) is a biochemical tool used as a carcinogen and mutagen. MNNG has been identified as a possible human carcinogen listed as an IARC Group 2A carcinogen. 7 It induces chromosomal aberrations, sister chromatid exchanges, and DNA strand breaks. Since its introduction by Mandell and Greenberg, 8 and earlier studies by Sugimura and Fujimura, 9 MNNG has been an excellent model for the induction of colorectal cancers. The organ specificity of MNNG correlates well with the level of DNA methylation after oral administration in rats. Earlier studies suggest that the host and environmental factors significantly influence gastric carcinogenesis induced by MNNG. 10 Further studies have examined the hormonal aspect of MNNG in relation to its carcinogenic potency for the gastric epithelium. The action of MNNG has been shown to be affiliated with glucocorticoid and androgen receptors, as well as the turnover of hydrocortisone and dihydrotestosterone in the glandular stomachs of mice. 11 In the current study, we used MNNG to induce adenocarcinoma in male Wistar rats and examined the effects of MGN-3/Biobran, an arabinoxylan rice bran, on its growth.
With the limitations of conventional therapies, such as surgery, chemotherapy, and anti-inflammatory drugs,12,13 scientists have been searching for other methods to combat adenocarcinoma, and they have found regulated dietary patterns, as well as the relationship between gastric cancer and diet, to be important factors in the declining incidence of adenocarcinoma. 14 Since gastric cancer is most frequently discovered in its advanced stage, it is of particular interest to investigate the natural dietary fiber MGN-3/Biobran for its ability to protect against the development of chemically induced gastric cancer in rats.
MGN-3/Biobran is an arabinoxylan extracted from rice bran that is treated with hydrolyzing enzymes from Shiitake mushrooms. 15 MGN-3/Biobran has been examined for its various biological activities. It has been characterized as a radioprotector against whole-body γ-irradiation 16 and as an antiviral agent with the ability to significantly reduce the viral load in patients with chronic hepatitis C virus infection. 17 In addition, Biobran has shown its potential as an anticancer agent. It exhibits antitumor activity in mice bearing a solid Ehrlich carcinoma (SEC) tumor 18 and synergizes with conventional therapies for the treatment of hepatocellular carcinoma patients. 19 The anticancer activity was attributed to the ability of Biobran to act as a potent biological response modifier that activates dendritic cells20-22 and enhances the activity of natural killer (NK) cells,18,23-27 increases human T and B cell mitogen response, 15 and augments phagocytosis by macrophages. 28 Further studies showed that Biobran has the ability to sensitize human leukemic cell surface CD95 receptors that are involved in the triggering of apoptosis. 29 Also, it sensitizes human and murine breast cancer cells to chemotherapeutic agents, such as daunorubicin and paclitaxel.30,31
The current study was undertaken to investigate the chemopreventive activity of Biobran against chemical induction of glandular stomach carcinogenesis in rats and elucidate the possible mechanisms behind its action. Results indicate that Biobran exhibited a protective effect against chemically induced adenocarcinoma in rats via induction of apoptosis in cancer cells.
Materials and Methods
Methylnitronitrosoguanidine
A total of 200 mg/kg body weight of MNNG (Sigma-Chemical, St Louis, MO) was orally administered to the rats daily for 2 weeks.
MGN-3/Biobran
Biobran is an arabinoxylan extracted from rice bran that is treated with hydrolyzing enzymes from Shiitake mushrooms. The main chemical structure of Biobran is an arabinoxylan with a xylose in its main chain and an arabinose polymer in its side chain. 15 Biobran was freshly prepared by dissolving in 0.9% saline solution and given intraperitoneally (IP; 40 mg/kg body weight every other day) for a total of 8 months. Biobran was kindly provided by Daiwa Pharmaceuticals Co Ltd, Tokyo, Japan.
Animals
Male Wistar rats aged 4 months (~120 g body weight) were used in this study. The animals were obtained from the Research Institute of Ophthalmology (Giza, Egypt) and acclimatized for 1 week prior to the commencement of the study. Rats were caged individually with light and temperature control (20 ± 2°C) and were fed standard laboratory cube pellets.
Experimental Design
Thirty-eight rats were divided into 4 groups: (1) control untreated (7 rats), (2) Biobran treated (9 rats), (3) MNNG treated (10 rats), and (4) MNNG plus Biobran treated (12 rats). Using the standard protocol for induction of gastric cancer, rats were given carcinogen MNNG at dose 200 mg/kg body weight once daily by oral gavage for 2 weeks, followed by oral administration of NaCl (1 mL/rat) once every 3 days for 4 weeks. Concomitantly with chemical induction, the rats were given Biobran alone (group 2) at a dose of 40 mg/kg body weight every other day or MNNG plus Biobran (group 4) for a total of 8 months. By the end of the treatment (8 months), animals were sacrificed and examined for different parameters in the gastric tissues.
Sample Collection and Gastric Tissues Preparation
After the treatment period (8 months), blood samples were collected from rats by puncturing the orbital venous plexus using heparinized capillary glass tubes into containers. For tissue preparation, animals were sacrificed by cervical dislocation, and the stomach was opened along the greater curvature, excised, and carefully examined. Then the stomach was divided into 2 parts symmetrically along the greater and lesser curves. One part was used to prepare cells in suspension for the flow cytometry studies; the other part was fixed in 10% formaldehyde for histopathology examination.
Flow Cytometric Analysis
Cell Preparation
Tumor tissues were excised from gastric cancer bearing rats under different treatment conditions, cut into pieces, and gently rubbed through fine nylon gauze (40-50 mesh count/cm, HD 140 Zuricher Buteltuch fabrik AG). Samples were then washed through the gauze with Tris/EDTA (ethylenediaminetetraacetic acid) buffer of pH 7.5 (3.029 g of 0.1 M Tris-(hydroxymethyl) aminomethane + 1.022 g of 0.07 M HCl + 0.47 g of 0.005 M EDTA). Cells were suspended in phosphate-buffered saline (PBS), centrifuged for 5 minutes at 200 to 300 g, resuspended in sterile PBS (cell density approximately 1 × 106 cells/mL), and then fixed in 70% ice-cold ethanol in PBS and stored at −20°C until used.
Cell-Cycle Analysis by Propidium Iodide (PI)
Cell cycle is a series of events that takes place in cell duplication, and monitoring cell cycles can provide much information about disease diagnosis and therapy prognosis. Tumor cell suspensions were centrifuged and the cell pellets were resuspended in 1 mL PI solution for 30 minutes in the dark and subsequently analyzed by flow cytometry. Data analysis was conducted using the DNA analysis program MODFIT (Verity Software House, Inc, Topsham, ME). Computer software calculated the coefficient of variation around the G0/G1 peak as well as the percentage of cells in each phase (G0/G1, S, and G2/M) of the DNA cell cycle for each sample. An aneuploid cell population was considered present if a distinct peak, in addition to the G1 diploid peak, deviated more than 10% from the diploid internal standard, or if the G1 itself deviated more than 10% from a corresponding G2/M peak. The apoptosis index (AI)/ proliferation index (PrI) ratio was calculated.
Annexin-V/PI
Annexin-V is a protein that binds to phosphatidylserine (ps) residues that are exposed on cell surfaces of apoptotic, but not normal, cells. During apoptosis, the ps groups are exposed to the exterior of the cell membrane. This binding of ps with Annexin-V is an established biochemical marker of apoptosis. Induction of apoptosis caused by Biobran treatment in gastric cancer cells was quantitatively determined through flow cytometry using the Annexin-V-conjugated Alexafluor 488 Apoptosis Detection Kit following the manufacturer’s instructions (BD Biosciences, San Jose, CA). The early apoptotic cells stained with Alexa488 give green fluorescence and present in the lower right quadrant of the fluorescence-activated cell-sorting histogram, and the late apoptotic cells stained with both Alexa488 and propidium iodide give red-green fluorescence and present in the upper right quadrant of the fluorescence-activated cell-sorting histogram.
Proliferation Marker Ki-67
Ki-67 is a cell-proliferation marker for tumor progression commonly used as an early predictor in cancer cells. Gastric cells suspended in PBS at a concentration of 1 × 106 cell/mL were prepared, and flow cytometric analysis was performed using mouse Ki-67 antibody protein (sc-7846 Per CP; Santa Cruz Biotechnology, Santa Cruz, CA) according to the manufacturer’s instructions.
Analysis of Apoptotic Regulators p53, Bax, and Bcl-2 Expression
For the determination of the expression of pro-apoptotic proteins p53 and Bax or anti-apoptotic protein Bcl-2, stomach cells (1 × 106) from rats treated or not with Biobran were incubated with mouse anti-p53, anti-Bax, or anti-Bcl-2 for 1 hour at room temperature, and then with FITC-conjugated goat anti-rabbit antibody. Cells were washed thoroughly and analyzed on a flow cytometer (Becton Dickinson, San Jose, CA). A total of 20 000 cells were acquired for analysis using CellQuest software, and histogram plot of FITC-fluorescence (x-axis) versus counts (y-axis) has been shown in logarithmic fluorescence intensity.
Quantification of Caspase-3 Activity
ab65613Caspase 3 (active) FITC Staining Kit (Abcam) was employed to determine the activity of caspase-3. The assay utilizes the caspase-3 inhibitor, DEVD-FMK, conjugated to FITC (FITC-DEVD-FMK) as a marker. FITC-DEVD-FMK is cell permeable, nontoxic, and irreversibly binds to activated caspase-3 in apoptotic cells. The FITC label allows detection of activated caspase-3 in apoptotic cells directly by flow cytometry. Cells were mixed with FITC-DEVD-FMK and incubated for 0.5 to 1 hour at 37°C incubator with 5% CO2. Cells were centrifuged at 3000 rpm for 5 minutes and the supernatant was removed. Cells were resuspended in 300 µL of Wash buffer. Samples were kept on ice and analyzed by flow cytometry using the FL-1 channel.
Histopathological Examination of Tumor Tissue
The stomach tissues from each group were examined for histopathological changes at 8 months after exposure to MNNG. Tissues were fixed in 10% formalin solution and fixed overnight in cassettes. The paraffin-embedded tissues were sectioned on a microtome to a thickness of 4 µm, stained with hematoxylin and eosin (H&E), and observed under light microscopy for signs of hyperplasia, dysplasia, and cancerous lesions. In addition, the cancer incidence and cancerous lesions were calculated as percentage by dividing the number of rats with cancer or lesions by total number of rats of the same group. Gastric tissues from rats treated with MNNG alone (10 rats) and with MNNG plus Biobran (12 rats) were examined initially for the presence of dysplasia, and then reexamined for the presence of cancer. Tumor tissues were examined twice, and the results averaged, for incidences of both dysplasia and adenocarcinoma.
Lymphocyte Level
At 8 months after exposure to MNNG, blood samples from rats under different treatment conditions were drawn by heart puncture using heparinized plastic syringes. Blood was quickly transferred into anticoagulation test tubes for measuring lymphocyte levels.
Statistical Analysis
Values were reported as mean ± SE, and significance of the differences between mean values was determined by one-way analysis of variance (ANOVA), coupled with the Newman-Keuls multiple comparison test or by Student’s t test where appropriate. Different pathological lesions were evaluated by Fisher’s exact test or χ2 test wherever appropriate. P < .05 was considered statistically significant.
Results
Rats under different treatment conditions were examined for the following parameters: histopathology of gastric cancer, Ki-67 expression, cell-cycle analysis, apoptosis, apoptotic regulators, and lymphocyte levels.
Histopathology
Percentages of Dysplasia and Cancer
The percentages of dysplasia and cancer were examined in histopathological tissues of H&E stained gastric mucosa at 8 months. Gastric tissues from 10 rats treated with MNNG alone and from 12 rats treated with MNNG plus Biobran were examined for the presence of dysplasia, and then examined again for the presence of cancer. The reported results are the average of 2 separate readings. Figure 1 shows that treatment with MNNG alone caused gastric dysplasia and adenocarcinoma in 8/10 rats (80%): 6/10 rats (60%) showed dysplasia, and 2/10 rats (20%) developed adenocarcinoma. In contrast, rats treated with MNNG in the presence of Biobran showed significantly lower incidence of dysplasia and adenocarcinoma, with a total of 4.5/12 rats (9/24 readings, 37.5%; P < .01) showing either condition: 3.5/12 rats (7/24 readings, 29.2%; P < .01) had dysplasia, and only 1/12 rats (8.3%) had adenocarcinoma.

Percentage of animals showing dysplasia or adenocarcinoma after treatment with both MNNG and Biobran. Animals were treated under different conditions: control untreated, Biobran alone, MNNG alone, and MNNG plus Biobran. The percentages of dysplasia and adenocarcinoma were examined at 8 months posttreatment. No dysplasia or adenocarcinoma were detected in the control untreated or Biobran alone treated rats. Each group contains 7 to 12 rats. *P < .01 compared to MNNG plus Biobran.
Histopathology Examination for Gastric Tissues
Histopathological changes of H&E-stained tissues of the gastric mucosa from rats at 7 weeks after carcinogen treatment showed gastric tissues had chronic superficial gastritis with regenerative atypia. At 16 weeks, gastric tissues had mild to moderate chronic inflammatory cell infiltration in the mucosa and submucosa, and focal intramucosal lymphoid aggregate. Neither dysplasia nor malignancy was detected at 16 weeks.
At 8 months, gastric mucosa from rats under different treatment conditions (control, carcinogen MNNG, and MNNG plus Biobran) were examined. The gastric mucosa from 7 control untreated rats were examined and showed the body and the antrum to be within normal limits. We did not detect hyperplasia, dysplasia, or carcinoma in the control tissues (Figure 2A-D). The gastric mucosa from MNNG-treated rats showed hyperplastic mucinous glands and mild- and high-grade gastric glandular dysplasia (Figure 2E-H). In addition, invasive well-differentiated keratinizing cell carcinoma was also detected (Figure 2I). In contrast, rats treated with MNNG and Biobran showed patchy and small areas of mild dysplasia in only 3.5/12 tissues (7/24 readings, 29.2%), while patchy and small areas of high-grade dysplasia/carcinoma in situ were seen in only 1/12 rats (8.3%). This suggests that Biobran treatment decreased the extent of gastric dysplasia and adenocarcinoma.

Histopathological examination of H&E-stained gastric tissues at 8 months. A-D shows gastric tissues from control untreated rats are within normal limits. (A) Normal body of stomach (4×). (B) Mucosa from the body of stomach (10×). (C) Normal antrum of stomach (4×). (D) Mucosa from the antrum of stomach (10×). (E) Section showing gastric glandular dysplasia (2×). (F) Section of stomach antrum showing mild dysplasia of glands and hyperplastic mucinous glands (10×). (G) Section of stomach showing high-grade glandular dysplasia (image is limited to a small focus; (40×). (H) Well-differentiated adenocarcinoma of the stomach (4X); (I) Gastroesophageal mucosa within normal limits (2X).
Ki-67 Expression
Effect of MNNG and Biobran treatments on the expression of tumor proliferation marker Ki-67 was examined. Rats treated with MNNG alone showed 50.8% Ki-67 expression in gastric tumor, while rats with MNNG plus Biobran showed 39.8%, representing a 21.6% decrease (Figure 3).

Effect of MNNG and Biobran treatments on Ki-67 expression of stomach tumor cells of rats as determined by flow cytometry. Each value represents the mean ± SE of 6 rats/group. *Significantly different from MNNG group at .01 level (% change of MNNG group).
Cell-Cycle Analysis
We examined the effects of MNNG and Biobran treatments on the different phases of cell cycle in stomach tumor cells. Data in Figure 4A show that Biobran treatment in the presence of MNNG caused cell-cycle arrest in the subG1 phase, where the hypodiploid cell population was markedly increased by 115.8% (P < .01), as compared to the group treated with MNNG alone. In addition, AI/PrI ratio showed a marked increase by 66.65% posttreatment with MNNG plus Biobran, as compared with MNNG alone, which represents an increase of 1.67-fold (Figure 4B).

Quantification of Apoptosis by Annexin-V/PI Staining
Annexin V/PI was used to analyze the level of apoptosis (early, late, and total). Results in Table 1 show that Biobran is a potent apoptotic agent as demonstrated by its ability to induce high levels of apoptotic cancer cells by 63.7%. In addition, Biobran was effective in inducing early apoptosis and late apoptosis by 230.1% and 20.2%, respectively, as compared to carcinogen-treated rats.
Effect of Biobran Treatment on Apoptosis in Tumor Tissues as Determined by Annexin-V/PI Double Staining a .
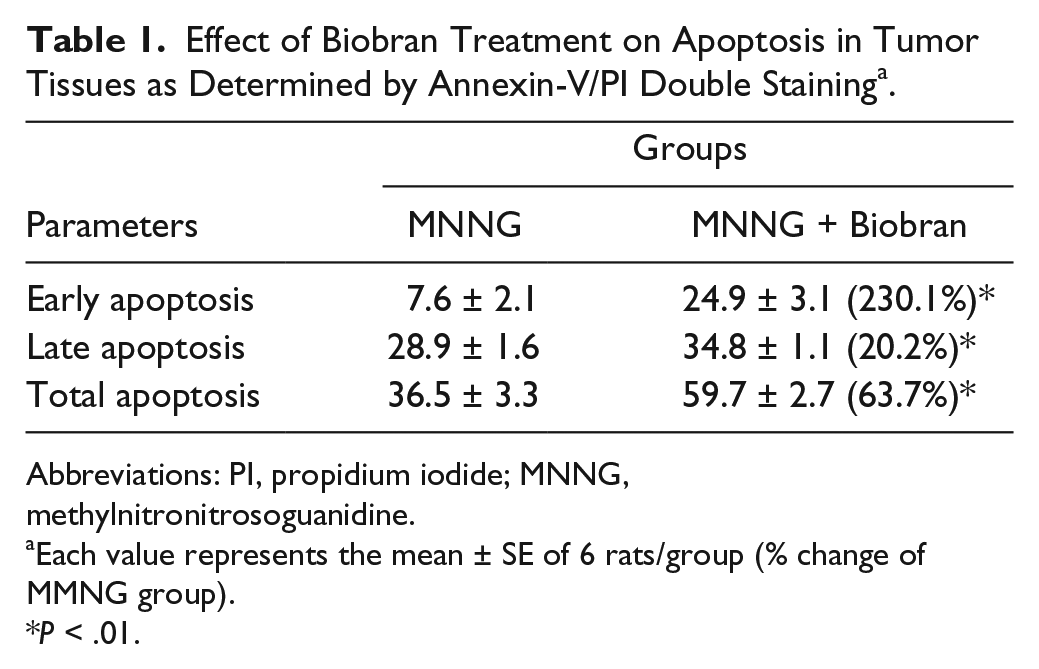
Abbreviations: PI, propidium iodide; MNNG, methylnitronitrosoguanidine.
Each value represents the mean ± SE of 6 rats/group (% change of MMNG group).
P < .01.
Percentage of Apoptotic Regulators
Effects of MNNG and Biobran on the percentage of apoptotic regulators in gastric tumor cells were examined by flow cytometry. Results in Table 2 show that Biobran induced apoptosis in gastric cancer cells in vivo via the mitochondria-dependent pathways as indicated by 37.3% increase in P53 expression, 49.3% increase in Bax expression, 15.1% decrease in Bcl-2 expression, 75.7% increase in Bax/Bcl-2 ratio, and 34.8% increase in caspase-3 activity, as compared with MNNG-treated rats.
Effect of Biobran on the Percentage of Apoptotic Regulators in Gastric Tumor Cells as Determined by Flow Cytometry a .

Abbreviation: MNNG, methylnitronitrosoguanidine.
Data represent the mean ± SE of 6 rats/group and % change of MNNG group.
P < .05, **P < .01; as compared with untreated MNNG group (% of change from MNNG group).
Lymphocytes Levels
The effects of MNNG and Biobran on the percentages of lymphocytes were examined. Figure 5 shows rats treated with MNNG alone showed significant decrease in the percentage of lymphocytes (−23.3% of control untreated rats; P < .01). On the other hand, Biobran supplementation caused recovery of lymphocytes, which remained within normal levels (P < .05).
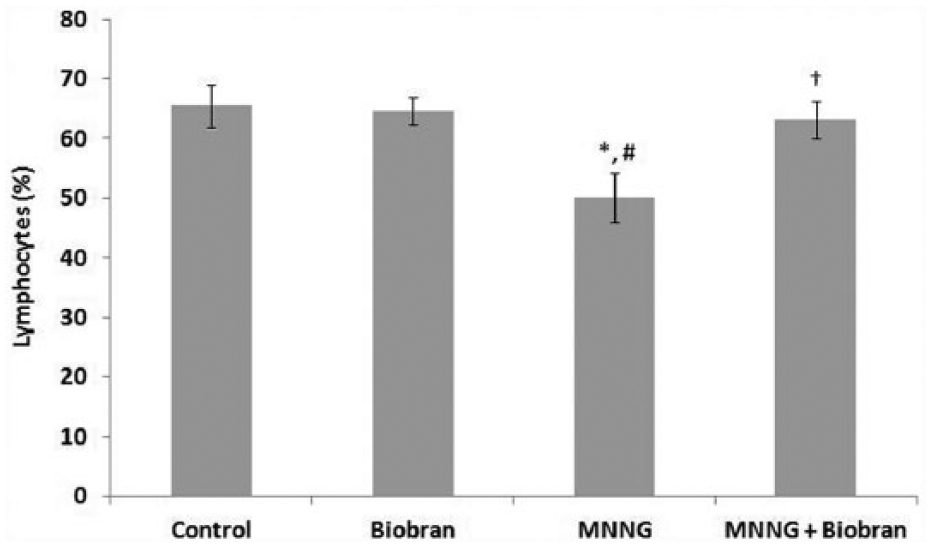
Percent lymphocytes. Each value represents the mean ± SE of 6 rats/group. *Significantly different from control group at .01 level. #Significantly different from Biobran group at .01 level. †Significantly different from MNNG group at .05 level.
Discussion
MGN-3/Biobran is a natural product extracted from rice bran whose effectiveness in the treatment of cancer has been widely examined in both animal models and humans with different types of malignancies. Results of the current study indicated that treatment with Biobran caused a marked suppression of the chemical induction of glandular stomach carcinogenesis in rats, as indicated by significant reduction in the incidence of animals bearing gastric dysplasia and adenocarcinoma. Exposure to MNNG plus Biobran caused gastric dysplasia and adenocarcinoma in 37.5% rats, as compared to 80% rats treated with carcinogen MNNG alone. The histopathological examination was further confirmed by a significant decrease in the expression of the tumor marker Ki-67. The anti-gastric-tumor effect of Biobran here is in accordance with earlier studies by us and others, which demonstrated the antitumor effect of Biobran in different animal models. This includes SEC tumor,18,32 neuroblastoma, 33 and liver cancer. 34 In addition, the synergistic effect of Biobran with conventional therapies (CT) for the treatment of hepatocellular carcinoma patients has been shown to result in greater reduction in tumor size, less recurrence of cancer, and higher survival rate, as compared with patients treated with CT alone. 19 Markus et al also described a successful treatment with full recovery of patient with metastatic hemangiopericytoma posttreatment with Biobran. 35
The apoptotic properties of Biobran may represent a mechanism by which this agent induces gastric tumor regression. Treatment with Biobran resulted in significant increase in the level of apoptotic gastric cancer cells as compared with MNNG-treated rats. The high apoptotic effect by Biobran was also noticed in SEC cells in vivo as determined by flow cytometry and the histopathological examination. 18 The Biobran-induced apoptosis in gastric cancer cells occurs via cell-cycle arrest of gastric cancer cells in the sub-G1 phase where the hypodiploid cell population was markedly increased, also via mitochondrial-dependent pathway as indicated by upregulation of P53, Bax expression, downregulation of Bcl-2 expression, and an increase in Bax/Bcl-2 ratio. Changes in these apoptotic molecules can cause disruption of the outer mitochondrial membrane and the release of cytochrome C that ultimately activates caspase-3. Biobran treatment should therefore have also caused an increase in caspase-3 activity, and we indeed measured an increase by 34.8% as compared with MNNG-treated rats. We have noted that treatment with Biobran induced apoptosis in human breast cancer cells via activation of capase-8, -9, and -3. 36 Moreover, we observed an increase in the levels of early and late apoptosis in gastric cancer cells posttreatment with Biobran, which further confirms the ability of Biobran to act by initiating mitochondrial-mediated apoptosis. Several medicinal herbs have been found to induce apoptosis in different cancer cells through the mitochondria-dependent pathway, including human osteosarcoma cells by aspidin PB, an extract from Dryopteris fragrans (L.) Schott 37 ; human breast cancer ZR-75-1 cells by Esculetin, a natural coumarin compound 38 ; and colorectal cancer by oleanolic acid, a natural pentacyclic triterpenoic acid. 39
Gastric tumor regression by Biobran was observed in the histopathology of the gastric tissue as well as the downregulation of Ki-67 expression and the maximization of the apoptosis/proliferation ratio. Such results are in accordance with studies of SEC tumor regression posttreatment with Biobran 40 and of tumor volume inhibition by the medicinal herb Melissa officinalis. 41 Induction of apoptosis in gastric cancer cells posttreatment with Biobran was associated with an increase in both the early and late apoptotic gastric cancer cells together with significant inhibition in the viable cell count. These data agree with our recent studies of Biobran-induced early and late apoptosis in EAC cancer cells. 40 The exact mechanism by which Biobran exerts its apoptotic effect is not fully understood but could be related to the ability of Biobran to sensitize the surface CD95 receptors that are involved in the triggering of apoptosis. 29
The immunomodulatory effects of Biobran may represent another mechanism by which this agent suppresses the growth of adenocarcinoma. Chemical carcinogens are known to be potent immune suppressive agents. Our earlier studies and those of others show that exposure to carcinogens causes dysfunction in the activity of immune cells.42-44 In the current study, we examined the effects of MNNG and Biobran on lymphocyte levels. Lymphocytes are a type of white blood cell that comprise the body’s immunologically competent cells and their precursors. Results from rats treated with MNNG alone showed significant decrease in the percentage of lymphocytes; however, treatment with Biobran resulted in a protection against MNNG-induced lymphocytopenia. Other studies have shown that Biobran is a potent biological response modifier that activates immune cells that possess anticancer activity, including dendritic cells,20-22 NK cells,23-27 CD8+T cells, 22 and interferon production.21,22,24 The ability of Biobran to suppress the neuroblastoma growth in vivo was strongly associated with stimulation of NK cell cytotoxic activity and upregulation of NK cell activation markers. 33
The current results also show that exposure to MNNG causes a decrease in red blood cell (RBC) indices (lower RBC count and Hb and Hct levels) and an increase in platelet levels. However, rats that received MNNG plus Biobran showed steady levels of RBC indices and platelet levels for 8 months (data not shown). Taken together, these findings show that Biobran protects the white blood cell count and overall stability of hematopoietic tissues against carcinogens. Additionally, we have observed that Biobran protected against an MNNG-induced reduction in spleen and liver weights (data not shown).
Data of the current study showed that Biobran specifically induced cancer cell apoptosis. Biobran has previously been shown to be a safe, nontoxic agent. Its biosafety is evidenced by the following: the LD50 is greater than 36 g/kg; the Ames test for mutagenicity is negative; and the subchronic toxicity study in rats, antigenicity study, and genotoxic testing all demonstrate that Biobran is nontoxic.45,46 Biobran has also been shown to be safe when investigated in humans using blood chemistry analysis including liver enzymes (SGOT and SGPT). 23 In addition, HCC patients that were treated with CT plus Biobran, as compared with CT alone, showed lower α-fetoprotein and alanine transaminase levels. 19 In the current study, rats treated with Biobran for 8 months showed no observable changes in their behavior, body weight (not shown), or survival.
We conclude that Biobran provides protection against chemical induction of glandular stomach carcinogenesis in rats. The present study highlights Biobran’s role as a potent apoptotic agent in malignant cells that may be used as an effective adjuvant for the prevention and treatment of gastric cancer.
Footnotes
Authors’ Note
Data from this study were partially presented at American Association for Cancer Research (AACR) annual meeting; April 5-9, 2014; San Diego, CA. The sponsors had no role in the study design; in the collection, analysis, or interpretation of data; in the writing of the article; or in the decision to submit the article for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Diawa Pharmaceutical Co Ltd, Tokyo, Japan; Grant #T0099108 (M. Ghoneum); and by NIH-NIMHD Grants U54MD007598 (formerly U54RR026138) and S21 MD000103 (D. Pan).
