Abstract
Background:
This trial evaluated the efficacy of melatonin versus placebo in alleviating radiotherapy-induced fatigue, anxiety, and depression in breast cancer patients.
Methods and materials:
This randomized, triple-blind, parallel-group, single-center clinical trial was conducted in the radiation oncology department of Namazi Hospital in Shiraz, Iran. Eligible breast cancer patients scheduled for adjuvant radiotherapy were randomly assigned to receive either 20 mg oral melatonin daily or a placebo. All participants underwent conventional radiotherapy (50 Gy total, delivered as 2 Gy per day). Patients in the melatonin group received the intervention starting on the first day of radiotherapy and continuing until treatment completion. Assessments were performed at baseline and 1 week after the intervention using the Multidimensional Fatigue Inventory and Beck’s Depression and Anxiety checklists. Data were analyzed using SPSS version 22 with a significance level set at P < .05.
Results:
Both groups (n = 50 each) had comparable baseline characteristics. Baseline MFI fatigue (P = .06), Beck Anxiety Inventory (P = .5), and Beck Depression Inventory (P = .9) scores showed no significant differences between groups. Post-intervention, the melatonin group had significantly lower scores than the placebo group (Mann–Whitney U test, P < .001 for all): MFI fatigue (median: 42.5 vs 55), Beck Anxiety Inventory (median: 28.5 vs 38), and Beck Depression Inventory (median: 29 vs 38). In the placebo group, changes were minimal: MFI fatigue scores were nearly unchanged (median: 56 vs 55), with no significant differences in Beck Anxiety Inventory (median: 38 vs 38, P = .08) or Beck Depression Inventory scores (median: 38 vs 38, P = .3). Within the melatonin group, the Friedman test showed significant reductions from baseline to post-intervention in MFI fatigue (median: 61-42.5), Beck Anxiety Inventory (median: 38-28.5), and Beck Depression Inventory scores (median: 38.5-29; P < .001 for all).
Conclusion:
Melatonin administration during radiotherapy significantly alleviated fatigue, anxiety, and depression in breast cancer patients.
Introduction
Breast cancer is a remarkable public health challenge worldwide, and it is noteworthy that its incidence has been rising rapidly, with recent estimates aligning with the latest reported data from Iran. 1 It has been shown that breast cancer is not a single unit of disease but appears considerably heterogeneous in both histopathological and clinical dimensions. Current classifications of breast tumors by the World Health Organization report more than 20 diagnostic subtypes associated with this disease. 2 Most breast tumors arise from a common origin, specifically from epithelial tissue. 3 Invasive carcinoma predominantly manifests as invasive ductal carcinoma, not otherwise specified (NOS). This disease comprises a heterogeneous group of tumors that cannot be easily categorized into a specific subtype based on their morphological and histological characteristics.
Various factors influence the prognosis of breast cancer, among which the primary tumor size and extend of lymph node involvement play a crucial role. 4 These 2 parameters demonstrate strong prognostic effects on the management and treatment of patients. Other prognostic factors, such as the presence of lymphovascular invasion, the degree of response to neoadjuvant therapies, and immunohistochemical findings, are also essential to evaluate in this context. 5 Currently, standard treatments for non-metastatic breast cancer involve a combination of surgery, radiotherapy, chemotherapy, and hormonal therapy.6 -9
A breast cancer diagnosis profoundly affects the physical, psychological, and social well-being of women. 10 Throughout the disease trajectory, patients may experience physical changes, particularly during treatment as a result of adverse effects. These changes can negatively influence self-perception, confidence, self-worth, and personal acceptance. Additionally, pain and suffering caused by the disease, anxiety about family members’ future, fear of death, treatment-related side effects, reduced functional capacity, body image disturbances, and sexual difficulties collectively contribute to a decline in the mental health of breast cancer patients. 11 Moreover, the psychological consequences of mastectomy or lumpectomy, combined with reduced social functioning from various treatments, disrupt the physical, psychological, social, and spiritual well-being of patients, ultimately diminishing their quality of life. Accordingly, addressing, assessing, and enhancing mental health and quality of life represent critical components of comprehensive breast cancer care. 12
Among the various effects of breast cancer and its treatments, cancer-related fatigue stands out as one of the most profound.13,14 The pathophysiology of cancer-related fatigue has not been fully elucidated but some of the likely causes may include the presence of malignant cells and the effects of adjuvant therapies, compounded by underlying psychological conditions.15,16 Inflammatory conditions resulting from malignancy and adjuvant treatments,17,18 neuroendocrine changes, 19 and inadequate responses from the autonomic nervous system are all among the primary mechanisms proposed and various interventions have been introduced to overcome this challenge.15,20,21 In this study, we specifically focused on fatigue induced by adjuvant radiotherapy, a common adverse effect that is distinct from general cancer-related fatigue or other contributing factors.
Melatonin, an endogenously secreted hormone from the pineal gland, primarily exerts effects related to chronobiotic activity and the regulation of sleep-wake cycles through seasonal and circadian patterns.22,23 Given the wide-ranging effects of melatonin (antioxidant, oncostatic, antiproliferative, and immunomodulatory characteristics),24 -27 its role in alleviating fatigue associated with malignancies and their treatments has been investigated. 28
Melatonin exerts its effects through multiple mechanisms, including its potent antioxidant properties, which reduce oxidative stress—a known contributor to cancer progression and treatment-related side effects. It also exhibits oncostatic effects by inhibiting cancer cell proliferation and inducing apoptosis, particularly in hormone-dependent cancers like breast cancer, via modulation of estrogen receptor signaling and suppression of aromatase activity. Additionally, melatonin’s regulation of circadian rhythms may mitigate fatigue and psychological distress by stabilizing sleep-wake cycles disrupted by cancer and its therapies. Recent evidence also suggests that breast cancer patients exhibit altered melatonin profiles, with lower serum levels correlating with increased depressive symptoms and sleep disturbances. 29 Overall, studies have indicated that melatonin levels are lower in cancer patients compared to individuals without malignancies, and the use of melatonin supplements has been associated with improvements in sleep quality as well as the quality of life of these patients.30-32
While previous studies have explored melatonin’s effects on cancer-related fatigue, few have specifically targeted radiotherapy-induced symptoms in a triple-blind, randomized controlled trial (RCT) design, making this study a novel contribution to the field. Moreover, consensus regarding the definitive efficacy of melatonin in reducing fatigue levels and enhancing the quality of life in cancer patients is still lacking. 33 Therefore, this clinical trial aims to compare the efficacy of melatonin versus placebo administration in alleviating radiation-induced fatigue among breast cancer patients on adjuvant radiotherapy.
Methods and Materials
This parallel-group, triple-blind, single-center and randomized clinical trial was conducted in the radiation oncology department of Namazi Hospital affiliated to Shiraz University of Medical Sciences in Shiraz, Iran. The study took place from March 2023 to September 2024. Prior to the initial stage of the study, the study protocol was submitted toEthics Committee of Shiraz University of Medical Sciences (Ethics Code: IR.SUMS.MED.REC.1400.604). Following this, the study protocol was registered with the Iranian Registry of Clinical Trials (Registration Code: IRCT20220907055912N1). Additionally, prior to enrollment in the study, a written informed consent form was obtained from all participants.
In this study, patients with breast cancer who were candidates for adjuvant radiotherapy were included. The inclusion criteria consisted of an age range of 18 to 65 years, a performance status of KPS ≥ 70 (or ECOG score of 0-1), a recent diagnosis of breast cancer with definitive histopathological confirmation, an indication for adjuvant chemotherapy, an indication for adjuvant radiotherapy, and an average interval of 3 to 4 weeks between chemotherapy and radiotherapy, utilizing a conventional radiotherapy regimen (2 Gy daily) for a total dose of 50 Gy to the whole breast/chest area. The exclusion criteria were the participants’ refusal to cooperate, pregnancy, a history of prior breast or chest irradiation, concurrent chemotherapy, a history of connective tissue diseases such as scleroderma or systemic lupus erythematosus, systemic infection, non-healed scars at the radiotherapy site, a history of uncontrolled underlying conditions such as diabetes and hypertension, the presence of metastasis at the time of presentation, and the use of tobacco or narcotics. Also, the withdrawal criteria during the study included receiving a total radiotherapy dose of less than 50 Gy to the whole breast, interruption of radiotherapy for more than 2 weeks, improper and irregular use of the medication or placebo, and retraction of signed informed consents.
Following the acquisition of informed consent, patients were randomly assigned to 2 groups: (1) to receive either melatonin tablet (20 mg daily) or to receive placebo, during their adjuvant radiotherapy treatment. Patient enrollment was consecutive based on the order of presentation to initiate radiotherapy. Assignment to study groups was accomplished using randomly blocked allocation with blocks of 2. For this method, the size of each block was determined (block size of 2), followed by the creation of a list of blocks with numbers assigned (1. AA 2. BB 3. AB 4. BA). Random numbers between 1 and 4 were selected, and the treatment assignment list was generated based on these random numbers. For allocation concealment, sealed envelopes were used. The medication and placebo (which were similar in appearance, consistency, and odor) were placed in identical containers and coded only as A and B. The patients, the treating physician, and the evaluator of treatment side effects were all blinded to group assignments; only an individual outside the research team (a pharmacist preparing the medications) was aware of which group received either the active medication or the placebo.
For patient treatment design, two tangential fields were employed to encompass the entire breast/chest tissue. If regional lymph node was indicated, level I axillary lymph nodes were treated with 2 tangential fields, while level II, III, and supraclavicular lymph nodes were addressed in 2 anterior-posterior fields. The total dose of radiotherapy and the dosage per session were uniformly set at 50 Gy, with 2 Gy per day for all patients.
The commercially available melatonin tablets in the Iranian pharmaceutical market are 10 mg, and are produced by Jalinus Pharmaceutical Company (Tehran, Iran). Based on previous study by Sedighi Pashaki et al, 28 patients were instructed to take 2 melatonin/placebo tablets before bedtime from the beginning day to the end of radiotherapy. The 20 mg dose was administered orally at 10 PM each night to align with the natural circadian peak of melatonin secretion.
Prior to the intervention and 1 week after the intervention, patients were assessed using the Multidimensional Fatigue Inventory (MFI) questionnaire. This 20-item instrument comprises 5 domains: general fatigue (4 items), physical fatigue (4 items), mental fatigue (4 items), reduced activity (4 items), and reduced motivation (4 items). The response range for each question is based on a five-point Likert scale from “strongly agree” to “strongly disagree,” with a scoring system from 1 to 5 for each item; for some items, reverse scoring is also applied. Therefore, the total score for each domain ranges from 4 to 20, and the cumulative fatigue score, determined by summing the scores from each domain, can range from 20 to 100. A higher score indicates greater fatigue levels. This questionnaire has been translated into Persian, with confirmed reliability and validity. 34 Anxiety and depression were assessed using the Beck Anxiety Inventory (BAI) and Beck Depression Inventory (BDI), which were validated, previously.35,36
The BAI and BDI, each comprising 21 items scored from 0 to 3, were administered at baseline (pre-radiotherapy) and 1-week post-intervention to assess anxiety and depression, respectively, consistent with the MFI timeline.
In order to determine the sample size, the results of the study by Sedighi Pashaki et al 28 regarding the impact of melatonin on fatigue levels in patients with breast cancer were utilized. According to the findings of this study, the incidence of severe fatigue after intervention was reported to be 42.1% in the intervention group and 83.3% in the control group. Therefore, using the sample size determination formula below:
and considering a power of 90% and a confidence level of 99%, the sample size was estimated to be at least 34 patients in each group. Given the potential for dropouts during the study, a minimum of 50 patients were included in each group.
The collected data were categorized and subsequently entered into SPSS 22 software. Descriptive statistics, including measures of central tendency, measures of dispersion, and frequency distribution, were utilized to describe the collected data. To compare categorical variables between the studied groups, the Chi-square test (or Fisher’s exact test) was used. For comparing quantitative variables between 2 groups, the independent t-test was employed if the data distribution was normal; otherwise, the Mann-Whitney U test was applied. Prior to this phase, the Kolmogorov-Smirnov test was used to assess the normality of the quantitative variables. For comparing changes in quantitative variables across independent groups over time, repeated measures ANOVA (or its non-parametric equivalent) was utilized. The correlation between quantitative variables was examined using Pearson’s correlation coefficient. In all calculations, a P-value of less than .05 was considered statistically significant.
Results
A total of 100 breast cancer patients eligible for adjuvant radiotherapy were randomly assigned to 2 groups: the intervention group (n = 50) and the placebo group (n = 50; Figure 1).
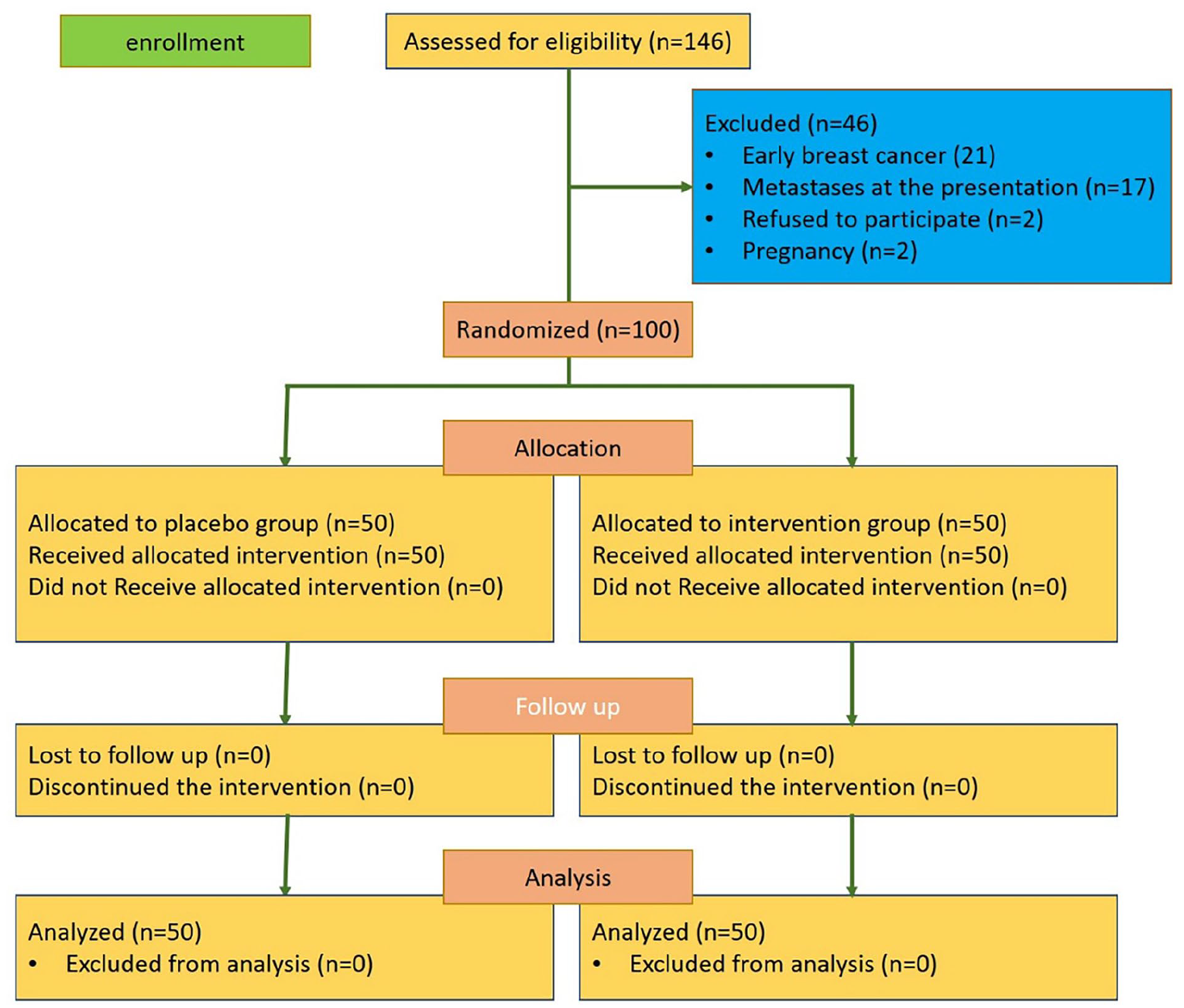
Flow diagram of patient recruitment in the present clinical trial.
The patients in both groups were similar in terms of age (P = .07), underlying conditions (P = .3), site of involvement (P = .1), disease stage according to AJCC TNM staging (P = .6), and molecular subgroup (P = .09; Table 1).
Patients’ Background Information in the 2 Groups Before Treatment.

Abbreviations: DM, diabetes mellitus; ER, estrogen receptor; Her2, human epidermal growth factor receptor 2; HTN, hypertension; IHD, ischemic heart disease; IQR, interquartile range; LIQ, lower inner quadrant; LOQ, lower outer quadrant; PR, progesterone receptor; UIQ, upper inner quadrant; UOQ, upper outer quadrant.
The Mann Whitney U test.
Chi-squared test.
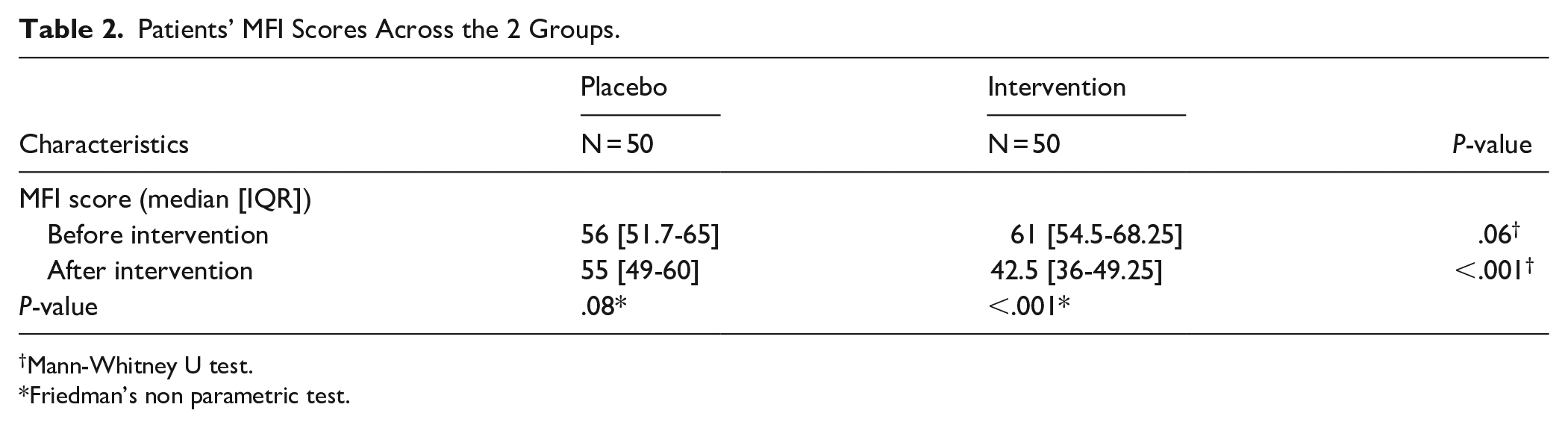
Mann-Whitney U test results showed that the MFI fatigue score (P = .06), Beck’s Anxiety score (P = .5), and Beck’s Depression score (P = .9) prior to the intervention were comparable between the 2 groups. However, in the intervention group, post-intervention scores for MFI fatigue (median 42.5 vs 55), Beck’s Anxiety (median 28.5 vs 38), and Beck’s Depression (median 29 vs 38) were significantly lower compared to the placebo group (Mann-Whitney U test; P < .001). An exploratory analysis of melatonin response by cancer stage (I, II, III) showed no significant differences in MFI fatigue score reductions (P = .7, Kruskal-Wallis test), suggesting melatonin’s efficacy is consistent across stages in this sample.
On the other hand, the Friedman test indicated that for the placebo group, the changes in MFI fatigue score (median pre-intervention 56 vs post-intervention 55; P = .08), Beck’s Anxiety score (median pre-intervention 38 vs post-intervention 38; P = .3), and Beck’s Depression score (median pre-intervention 38 vs post-intervention 38; P = .2) were not significant over the study period. Conversely, in the intervention group, the Friedman test revealed a significant reduction in MFI fatigue score (median pre-intervention 61 vs post-intervention 42.5), Beck’s Anxiety score (median pre-intervention 38 vs post-intervention 28.5), and Beck’s Depression score (median pre-intervention 38.5 vs post-intervention 29) after the intervention compared to scores before (P < .001 for all analyses). Tables 2 to 4 illustrate the comparison of MFI fatigue scores, Beck’s Anxiety scores, and Beck’s Depression scores across the 2 groups, as well as respective changes in these scores during the study for each group.
Patients’ MFI Scores Across the 2 Groups.
Mann-Whitney U test.
Friedman’s non parametric test.
Patients’ Beck’s Anxiety Scores Across the 2 Groups.

Mann-Whitney U test.
Friedman’s non parametric test.
Patients’ Beck’s Depression Scores Across the 2 Groups.

Mann-Whitney U test.
Friedman’s non-parametric test.
Discussion
The present study aimed to investigate the efficacy of melatonin administration during adjuvant radiotherapy for breast cancer patients. By focusing on radiotherapy-induced fatigue, this study isolates a key treatment-related symptom, though broader cancer-related factors may also play a role. Overall, the results indicated that the administration of this drug was associated with a significant reduction in levels of fatigue, anxiety, and depression among the patients. These findings suggest a practical clinical benefit, as reducing fatigue, anxiety, and depression could enhance patients’ ability to complete radiotherapy regimens and maintain daily functioning.
Previous research has explored the effects of melatonin on fatigue across various clinical settings, generally indicating its efficacy. For example, Sedighi Pashaki et al investigated melatonin’s impact on fatigue in 78 breast cancer patients undergoing adjuvant treatments (chemotherapy and radiotherapy). Patients were randomly assigned to receive either 20 mg of melatonin daily or a placebo, starting 1 week before treatment and continuing for 1 month after completion. The study found that the melatonin group experienced significantly lower fatigue scores on the Brief Fatigue Inventory (BFI) and reduced fatigue severity compared to the placebo group, which showed no significant changes. 28 Sedighi Pashaki et al conducted a 2-year intervention on the same group of patients as part of their clinical trial; the researchers reported the long-term effects of melatonin administration in breast cancer patients, showing that during post-intervention period, not only was the fatigue score significantly lower in the melatonin group, but also there was a notable reduction in fatigue scores in the intervention group. Moreover, prolonged use of melatonin, even after the completion of adjuvant treatment in women with breast cancer, was associated with a significant decrease in fatigue levels related to malignancies and their treatments. 37
In another study, Semiglazova et al 38 investigated the effects of melatonin and metformin administration during neoadjuvant therapy in patients with breast cancer, and found that the use of melatonin caused a significant reduction in patient fatigue in the intervention group. The findings from the studies conducted by Sedighi Pashaki et al and Semiglazova et al. regarding the significant effects of melatonin in reducing fatigue levels in breast cancer patients are in line with the results of the present study. Additionally, the findings of this study demonstrated that the administration of melatonin significantly improved the participants’ anxiety and depression levels. Other studies have indicated the effect of melatonin administration on alleviating patients’ anxiety levels, 39 depression,29,39 sleep quality,29,39,40 quality of life,32,41,42 and tolerance of oncology treatments. 43 Melatonin plays a role in synchronizing the circadian rhythm and consequently regulates fatigue and the sleep-wake cycle. It appears that through this physiological function, melatonin exerts its effects on levels of fatigue, anxiety, and depression. 44
The significant reductions in fatigue, anxiety, and depression observed here may stem from melatonin’s multifaceted actions, including its antioxidant effects that counteract radiotherapy-induced oxidative stress, and its circadian regulation that stabilizes disrupted sleep-wake cycles. These mechanisms likely synergize to improve patients’ psychological and physical well-being, a hypothesis supported by studies linking melatonin to enhanced quality of life in cancer patients. 32
However, some published studies do not support such a conclusion. For example, the study by Mukhopadhyay et al 45 evaluated the effects of melatonin in preventing fatigue and other symptoms in breast cancer patients undergoing radiotherapy; in their phase III double-blind, placebo-controlled clinical trial, the administration of melatonin not did not improve levels of fatigue and other symptoms in the early stages of radiotherapy. In another clinical trial by Lund Rasmussen et al, 33 the effect of 20 mg melatonin daily administration in metastatic patients was investigated, and no benefits were reported in fatigue levels in patients with malignancies who were candidates for palliative treatment; such differences regarding this type of inefficiency of melatonin on the fatigue levels in these patients may be attributed to the small sample size, different assessment tools, and a population with varied histories of receiving different lines of oncological treatments.
The present study had some limitations since conducting a phase II clinical trial may pose risks to the generalizability of its results. Also, because participants of the present study were evaluated solely during radiotherapy, the outcomes of long-term melatonin administration remain to be explored in other studies. Furthermore, since the patients in this study were treated with a conventional radiotherapy regimen, the efficacy and safety of melatonin administration in other dose-per-fraction approaches, such as hypofractionated regimens, still remain unclear. Particularly, the daily visits of patients in the conventional and hypofractionated approaches differ fundamentally, which are likely to influence the fatigue levels experienced by the patients. Moreover, the lack of assessment of serum melatonin levels restricted the researchers’ ability to evaluate adherence to therapy. The reliance on self-reported scales rather than clinical diagnoses for anxiety and depression may limit diagnostic precision, though these tools are standard in research settings. Chronotype was not assessed in this study, which could influence individual responses to melatonin timing; future research should explore this factor. Sleep, a critical factor interrelated with fatigue and influenced by melatonin, was not evaluated as an outcome due to the study’s focus on fatigue, anxiety, and depression, and limited resources for polysomnography or sleep diaries. This omission is notable, as melatonin’s sleep-enhancing effects could mediate its impact on fatigue, warranting investigation in future trials.
While this study focused on radiotherapy-induced fatigue, other factors such as cancer itself, psychological stress, or comorbidities could contribute to fatigue. The randomized, triple-blind design minimizes these confounding effects by evenly distributing such variables across groups.
Conclusion
The results of the present study showed that melatonin administration during radiotherapy significantly reduced fatigue, anxiety, and depression in breast cancer patients, potentially improving their quality of life and treatment tolerance.
Footnotes
Acknowledgements
We would like to thank all the participants who made the completion of this research possible. We extend our thanks to the Clinical Research Development Unit of Vasei Hospital, affiliated with Sabzevar University of Medical Sciences, for their kind support.
Ethical Considerations
The protocol of present study was approved by Ethics Committee of Shiraz University of Medical Sciences (approval code IR.SUMS.MED.REC.1400.604).
Consent to Participate
Prior to enrollment in the study, the participants were provided with clear explanations regarding both treatment protocols and random allocation; also, written informed consents were obtained from all participants.
Consent for Publication
Not applicable.
Author Contributions
S.S.Y. and A.M. contributed to the conception, design, and final drafting of the manuscript. S.S.Y. and S.A.J. contributed to data collection. S.S.Y. and S.A.J. developed the statistical design and analysis. J.S.W. contributed to the primary drafting of the manuscript. A.M. supervised the study. All authors approved the final version for submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present article is extracted from the residency thesis of the first author, which was supported by the Research Vice-Chancellor of the Faculty of Medicine at Shiraz University of Medical Sciences, Shiraz, Iran (Thesis No. 23481, approved on Feb 15, 2022).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data sets used and/or analyzed during the current study are available from the corresponding authors per reasonable request.
Code Availability
The custom code was used.
Clinical Registry Approval
The study protocol was registered with the Iranian Registry of Clinical Trials (Registration Code: IRCT20220907055912N1).

