Abstract
Introduction:
Although breast cancer is common worldwide, if diagnosed early and treated on time, the probability of recovery is high and patients often experience a long life. Reducing the quality of life is a common side effect in patients. Melatonin may have an important role in fatigue, sleep disorders and, as a result, the health-related quality of life (HRQoL) in people. About 184 patients with breast cancer were enrolled in 2 groups: intervention with daily melatonin intake of 18 mg for 3 years (93 patients) and the control group with placebo intake (91 patients). Health-related quality of life and the effect of melatonin on increasing that were evaluated with the EORTC QLQ-C30 questionnaire, third edition at the beginning, 2 months later and 3 years after the beginning of the study.
Results:
The general score of the HRQoL was significantly different both in the passage of time and in the comparative study of the 2 groups, and it was better in the melatonin group (P < .05).
Conclusion:
Long-term use of 18 mg of melatonin for 3 years in patients with non-metastatic breast cancer can lead to an increase in the patients’ quality of life.
Introduction
Breast cancer is the major cause of cancer-related deaths in women worldwide.1,2 Despite many years of research, cancer is still a main cause of death. Breast cancer is common worldwide, but in cases of early diagnosis and treatment, the probability of recovery is high and patients often have a long life. Reducing the QoL in patients is a common side effect of breast cancer.3-5 In a study, it was shown that breast cancer patients who have sleep problems, sadness and fatigue before chemotherapy, experience a lower quality of life during treatment than other patients who do not have these symptoms. 6 HRQoL is a multidimensional conception that includes the social, physical and mental aspects of a person’s health and wellbeing. According to the European Organization for Research and Treatment of Cancer (EORTC), the HRQoL is the ability to do daily physical or mental activities and enjoy social well-being, along with the individual’s satisfaction with the levels of disease control and performance. 7 For example, cancer patients may experience physical, emotional, social, and practical challenges. Fatigue, pain, lymphedema, nausea, shortness of breath, constipation, decreased sleep, loss of appetite, diarrhea, impaired mobility, menopausal symptoms, loss of sexual desire, anxiety, depression, fear of recurrence, mastectomy or hair loss, financial and social problems are some of the common factors of reducing the quality of life in cancer patients.5,8,9 Research results show that HRQoL assessments can improve the quality of care that patients receive and also improve their health status. Therefore, it has become as important as patient’s survival in treatment decisions in modern oncology. 10
The day/night rhythm-regulated hormone, melatonin (N-acetyl-5-methoxytryptamine) has an important relationship with fatigue, sleep disorders and, as a result, with the health-related QoL in cancer patients. According to literature, alteration in melatonin levels causes depressive disorders.11,12 In addition, melatonin has antioxidative, oncostatic, antiproliferative, and immunomodulatory effects.13-15 The expression level of genes effective in apoptosis and immune response is increased by melatonin, but it reduces the expression of cell metastasis genes, for example angiogenesis, mitosis and telomerase activity. 16 According to research, the level of melatonin in cancer patients is lower than healthy people and melatonin can improve QoL and sleep disturbance in them in addition to anti-tumoral effects,17-20 In this study, the effect of melatonin on the QoL of cancer patients was assessed during chemotherapy and radiation therapy.
Methods and Materials
Cases of Study and Questionnaire
The study was a double-blinded clinical trial in which volunteers were randomly placed in placebo or control group. Female with non-metastatic breast carcinoma stages I-III, at least 18 years of age were enrolled in the study from Mahdieh Diagnostic and Treatment Center and Be’sat Hospital of Hamedan, Iran. Patients were candidates for receiving radiotherapy and chemotherapy. Also, patients with Eastern Cooperative Oncology Group performance status (ECOG PS) 2 or more were included. The intervention group with daily use of melatonin (101 patients) and the control group with consumption of placebo (102 patients) were included in the study. Out of 101 patients in the melatonin group, 93 patients remained in the study until the end; 6 patients were excluded due to the occurrence of metastasis or disease recurrence, and 3 patients did not continue the study. Out of 102 patients in the placebo group, 91 patients remained in the study; 8 patients were excluded due to the occurrence of metastasis and 3 patients did not continue the study. The data collection method was using European Organization for the Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30), third edition. It has 30 questions that measures the quality of life of cancer patients. This questionnaire contains 5 functional scales (social, physical, cognitive, role and emotional), 3 symptom scales (nausea, fatigue, pain and vomiting) and a global health and quality of life scale. The remaining items evaluate the other symptoms mainly reported by cancer patients (shortness of breath, sleep disturbance, loss of appetite, diarrhea and constipation) also the financial effect of the treatment of the disease. Patients who are pregnant or in the lactation period, patients with uncontrolled hypercalcemia, or use of methylphenidate, warfarin and sleeping pills by patients resulted in exclusion from the study. Systolic blood pressure less than 100 mm Hg and TSH less than 0.5 or more than 5.5 were also excluded. Death, lack of consent to continue the study, the occurrence of metastasis or depression and treatment discontinuation, were the other exclusion criteria. Simple random sampling method was used for selecting patients. The Research Ethics Committee of the Hamadan University of Medical Sciences was approved the study (IR.UMSHA.REC.1401.903). In addition, the study received the Iranian Registry of Clinical Trials code (IRCT20221205056712N1). Informed consent was signed by patients before the study.
Study Design and Primary Endpoints
Randomization was prepared by https://www.randomization.com. All of the hospital staffs and patients were not informed of the study groups. Any side effect of chemotherapy resulted in exclusion of the study, based on the Common Terminology Criteria for Adverse Events. The subscale of Physical functioning (PF2) consists of questions 1 to 5 of the questionnaire, which examine physical problems in daily activities such as walking, or eating. Role functioning subscale (RF2) includes questions 6 and 7 of the questionnaire. In this subscale, the amount of limitation in performing daily activities and hobbies that are effective in fulfilling their role in life is investigated. The subscale of Emotional functioning (EF) includes questions 21 to 24 of the questionnaire which evaluates the feeling of tension, nervousness, depression and anxiety of the patients. The Cognitive functioning (CF) subscale includes questions 20 and 25 of the questionnaire. This subscale investigates memory disorders and general cognitive disorders of patients. The Social Functioning (SF) subscale includes questions 26 and 27 of the questionnaire which evaluates the disorders and problems in the social activities and family relationships of the patients. In all the above subscales, the number 1 means little problem and the number 4 means more severe problems of the patients. Functional Scale (FS) subscale includes RF2, PF2, EF, CF, and SF subscales. Global health status (QoL) scale includes 30 and 29 questions of the questionnaire. This subscale examines the general state of the QoL and health level, in which the number 1 means a very poor level and the number 7 means a very good level of health and QoL of the patients. The Fatigue (FA) subscale includes questions 10, 12, and 18 of the questionnaire. This subscale evaluates the need for rest, tiredness and weakness of the patients. Nausea and vomiting (NV) subscale includes questions 14 and 15 of the questionnaire. This subscale investigates the presence of nausea and vomiting in patients. The Pain subscale (PA) consists of questions 19 and 9 of the questionnaires. This subscale examines the presence of pain in patients. The Dyspnea subscale (DY) consists of question 8 of the questionnaire. This subscale examines shortness of breath in patients. The Insomnia (SL) subscale consists of question 11 of the questionnaire. This subscale examines insomnia in patients. The Appetite loss (AP) subscale includes question 13 of the questionnaire. This subscale evaluates the loss of appetite in patients. The Constipation (CO) subscale consists of question 16 in the questionnaire. It investigates constipation in patients. The Diarrhea subscale (DI) consists of question 17 of the questionnaire. It investigates diarrhea in patients. The Financial difficulties (FI) subscale includes question 28 of the questionnaire. It evaluates economic problems in patients. The symptom scales (SS) which includes a set of sub-scales Fatigue/ Dyspnoea/Nausea and vomiting/ Insomnia/Pain/Appetite loss/Diarrhoea/Financial difficulties and Constipation examine the clinical symptoms that patients declare. In all the mentioned cases, the number 1 means little problem and the number 4 means more severe problems of the patients.
Treatment Schedule
Randomly, patients were treated with melatonin or placebo (control group). Oral melatonin 18 mg by RAZAK Laboratory, Tehran, Iran was taken every night from 3to 7 days before the start of adjuvant treatment, placebo tablets were made at Hamadan University of Medical Sciences, 4 weeks after the last adjuvant treatment session, the patients were evaluated by the EORTC QLQ-C30 by a person unaware of the treatment methods. Finally, information related to each patient, including age, quality of life and health, was recorded in a checklist. At the beginning, 2 months later and 3 years after the beginning of the study, evaluation was done according to the EORTC QLQ-C30 questionnaire.
All patients received doxorubicin, cyclophosphamide, and paclitaxel every 14 days with granulocyte colony stimulating factor for 8 cycles. Her2/neu positive cases, were received Trastuzumab, within and after the prescription of paclitaxel. The conventional radiotherapy of breast/chest wall with local node irradiation (if needed) was done in both groups.
Registration of Data, Follow-Up and Quality Assurance
Patients were visited in mentioned oncology clinics, every week during the radiotherapy and before each chemotherapy session. One month after completion of chemotherapy and radiotherapy, the patients were visited again. Patients were inquired about the consumption of placebo or melatonin. The present study is a follow-up of the previous study. 21 Information of each patient, including age, sex, quality of life, type of adjuvant treatment was recorded in a checklist and assessment was done by an investigator who were uninformed to the groups.
Statistical Methods
As mentioned, this study is a follow-up of the previous study, SPSS V16 was used for data analysis. Descriptive analysis was performed for all data; First, the K-S test was performed and with the availability of parametric conditions and normal distribution, the independent sample test was used, to evaluate the average comparison among different subgroups, the ANOVA test was used, and to compare the nominal and rank qualitative variables, Pearson’s correlation coefficient test was used. P < .05 were determined to be statistically significant.
Results
From the melatonin group, 93 patients remained in the study until the end. And from the placebo group, 91 patients remained in the study (Figure 1). The average age in the intervention group was 50.32 years and in the control group was 47.02 years. There was no significant relationship between the age and the study results in 2 groups (P = .223). Specifications of the patients at the beginning of the study are shown in Table 1.

Screening, randomization, follow up and analysis.
Specifications of the Patients at Baseline.
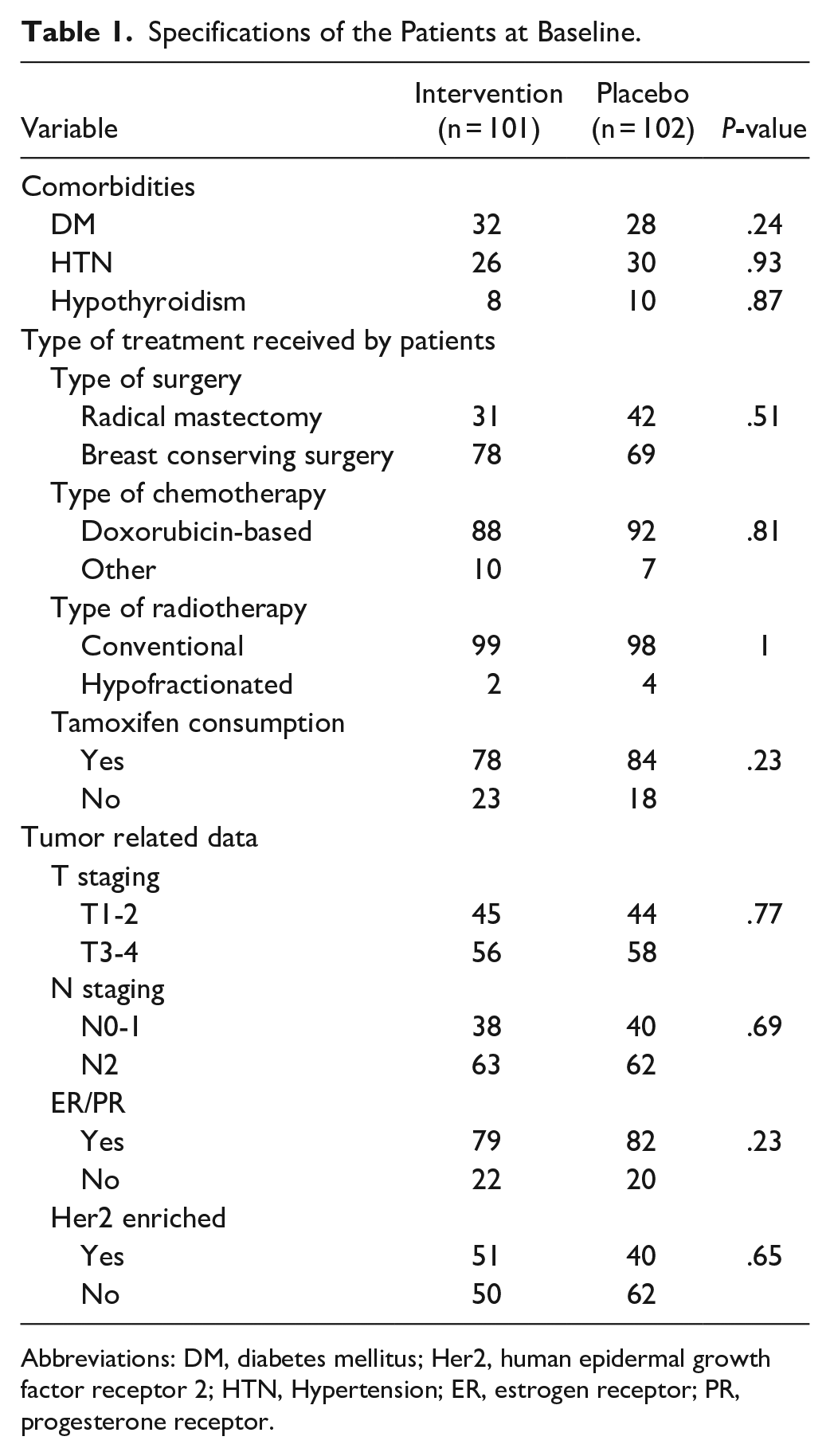
Abbreviations: DM, diabetes mellitus; Her2, human epidermal growth factor receptor 2; HTN, Hypertension; ER, estrogen receptor; PR, progesterone receptor.
Table 2 shows the mean and standard deviation of subscales of the HRQoL scores over time across groups. The results of the study were presented according to the subscales of the questionnaire in Table 2.
Comparison of the Mean HRQoL Scores Over Time Across Groups for Various Subscales.

Abbreviations: PF2, physical functioning; RF2, role functioning; EF, emotional functioning; CF, cognitive functioning; SF, social functioning; FS, Functional Scale; QoL, global health status; FA, fatigue; NV, nausea and vomiting; PA, pain; DY, dyspnea; SL, insomnia; AP, appetite loss; CO, constipation; DI, diarrhea; FI, financial difficulties; SS, symptom scales.
Obtained by the repeated measurements Analysis of Variance during the time.
Table 3 shows the results of repeated measurement ANOVA to assess the effect of time, group and interaction of time and group.
The Results of Repeated Measurement ANOVA to Assess the Effect of Time, Group and Interaction of Time and Group.

Discussion
The current study was a clinical trial with the aim of determining the effect of melatonin on increasing HRQoL in female patients with non-metastatic breast cancer in a 3-year follow-up. Breast cancer patients were enrolled in 2 intervention groups with daily consumption of 18 mg of melatonin for 3 years and control group with placebo consumption. In this study, the Persian form of the EORTC QLQ-C30 questionnaire (third version) was used to measure HRQoL. The analysis of the data related to the performance scale showed that the condition of the subscales of physical performance, role playing, and cognitive performance in the group of melatonin-user patients was significantly better than the placebo group and also over time; However, in the 2 subscales of emotional (or excitement) and social performance, the 2 groups did not differ significantly from each other despite the significant difference over time. In general, the results of examining patients’ function scales showed a significant amelioration in the melatonin group in comparison to placebo. In examining the symptom scales of the patients, in the melatonin group, fatigue, nausea and vomiting, shortness of breath, decreased sleep, and constipation were significantly reduced compared to the placebo group; However, in relation to pain, loss of appetite, diarrhea and economic issues, this difference was not significant despite the better condition of the melatonin group. The score of the QoL was different both in the passage of time and in the comparative study of the 2 groups, and it was significantly better in the melatonin group than in the placebo group. Melatonin regulates the sleep–wake cycle and alterationin its level causes depressive disorders,11,22,23 moreover it has oncostatic, antioxidative, antiproliferative, and immunomodulatory effects.13-15 On the other hand, cancer patients produce lower amounts of melatonin compared to the healthy people. Based on the findings, melatonin supplements help patients to overcome sleep disturbances and improve quality of their life, besides potential anti-tumoral effects in higher doses, such as the inhibition of tumor cell proliferation and metastasis and anti-angiogenic effects, particularly in hormone-positive breast cancer which is the most common type. Melatonin can modify estrogen pathways efficiently. Studies showed a relationship between low melatonin levels and sleep disturbances and consequently mood disorders in patients with breast cancer.17-20,24 In Innominato et al study, the results indicated that taking 5 mg of melatonin before going to bed for 2 months in 32 patients with metastatic breast cancer, treated with hormone or trastuzumab was associated with improvement in fatigue intensity, sleep status, global QoL and cognitive and social function scales. But in the field of emotional performance, role playing and reduction of pain, constipation, loss of appetite, shortness of breath, diarrhea, nausea and vomiting did not have a significant effect. 18 The results of the mentioned study confirmed the present study. Palmer et al study, showed that consumption of 20 mg of melatonin for 10 days before and during adjuvant chemotherapy caused neuroprotective effects and reduced side effects of adjuvant chemotherapy and improved mental function, depression and sleep of breast cancer patients. 19 Contrary to previous findings, there was not any evidence of therapeutic effect of melatonin in the Hansen et al study, in this study, women aged 30 to75 years with breast cancer randomly received 6 mg of melatonin or placebo for 3 months. The results showed that use of 6 mg melatonin had no significant positive effects on the cognitive performance of breast cancer patients. 25
Conclusion
The long-term use of 18 mg melatonin for 3 years, in persons with non-metastatic breast cancer can result in to an increase in the overall QoL and improvement in physical function, individual role-playing, social and cognitive function. It reduces negative symptoms such as fatigue, nausea, vomiting, shortness of breath, loss of sleep and constipation.
Footnotes
Acknowledgements
We appreciate all staffs of Besat Medical Education Center of Hamadan University of Medical Sciences and Mahdieh Diagnostic/treatment center and Charity Center of Hamedan. We also grateful for Cooperation of all the patients in this study.
Correction (May 2024):
Article updated to correct second author's name from “Fatemeh Sheida” to “Fateme Sheida”.
Author’s Note
Fatemeh Sheida is also affiliated to Gastrointestinal and liver Diseases Research Center, Guilan University of Medical Sciences, Rasht, Iran.
Author Contributions
Study concept and design: Z.K.A and R.A; acquisition of data: F.Sh and SR.H and H.M; statistical analysis and interpretation of data: L.T; drafting of the manuscript: N.B; critical revision of the manuscript: Z.K.A and N.B.
Data Availability Statement
All of the study data and their analysis are accessible through communication with the corresponding author and consent of the research team members.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Hamadan University of Medical Sciences, Deputy of research and technology (grant number: 37649).
Ethical Approval Statement
Research Ethics Committee of the Hamadan University of Medical Sciences was approved the study (IR.UMSHA.REC.1401.903).
Clinical Trial Registration Number
The study received the Iranian Registry of Clinical Trials code (I IRCT20221205056712N1).
