Abstract
Introduction:
Most patients with pancreatic cancer experience systemic recurrence within 1 to 2 years after radical pancreatectomy. Phellinus linteus (PL) has demonstrated anti-inflammatory, antioxidant, and anti-cancer properties, suggesting potential as an adjunct to cancer therapy. This study aimed to evaluate the long-term oncological impact of perioperative PL in resected pancreatic cancer.
Method:
This retrospective cohort study included 407 patients who underwent curative resection and adjuvant chemotherapy for pancreatic cancer at Severance Hospital (2012-2022). Among them, 103 patients who began PL postoperatively and continued throughout treatment were assigned to the PL group; 304 patients without PL intake comprised the control group.
Results:
The mean overall survival (OS) was significantly longer in the PL group (47.0 months; 95% CI: 42.8-51.1) than in the control group (35.0 months; 95% CI: 30.3-39.7; P < .001). Recurrence-free survival (RFS) showed a borderline improvement (P = .053). PL use was marginally associated with improved OS in multivariate analysis (HR: 0.614; 95% CI: 0.376-1.002; P = .051). Subgroup analysis showed no significant OS or RFS benefit with PL in patients receiving FOLFIRINOX. However, among patients treated with non-FOLFIRINOX regimens, PL use led to significantly better OS (43.9 months vs 35.0 months; P = .021), though RFS remained similar. Notably, the OS of the non-FOLFIRINOX + PL group was comparable to that of the FOLFIRINOX group (P = .332) and superior to the non-FOLFIRINOX control group (P = .021).
Conclusion:
PL may enhance survival in resected pancreatic cancer, particularly in patients receiving non-FOLFIRINOX chemotherapy, supporting its role as a potential adjunct when FOLFIRINOX is not feasible.
Introduction
Pancreatic cancer is one of the most aggressive and lethal forms of cancer, with a dismal 5-year survival rate of less than 10%, because of late diagnosis and limited effective treatment options. 1
Margin-negative pancreatectomy is essential for the cure of pancreatic cancer; however, most patients have a recurrence within 1 to 2 years after surgery (especially in the liver, lungs, or peritoneum), and thus, postoperative adjuvant chemotherapy should be mandatory. 2 The remarkable development of anti-cancer drugs, as evidenced by the therapeutic effect of FOLFIRINOX as postoperative adjuvant chemotherapy, has provided hope that pancreatic cancer can be cured in the near future. 3 However, conventional chemotherapeutic approaches often fail to provide significant improvements in survival rates, highlighting the urgent need for alternative therapeutic strategies.
In recent years, there has been growing interest in the use of nutraceuticals—natural products with medicinal properties—as potential adjuncts in cancer therapy. 4 Among these, Phellinus linteus (Pl), known as Sanghuang mushroom in Korea, has garnered significant attention. This medicinal mushroom has been used for centuries in traditional Asian medicine and is known for its diverse bioactive compounds that exhibit a wide range of pharmacological effects, including anti-inflammatory, antioxidant, and anti-cancer effects.5,6
PL is rich in polysaccharides, polyphenols, and triterpenoids, which are believed to contribute to its therapeutic effects.5,6 Research has shown that these compounds can modulate various molecular pathways involved in cancer progression, including apoptosis, angiogenesis, and metastasis. 7 The potential of PL as a nutraceutical in cancer treatment is supported by a growing body of preclinical studies demonstrating its ability to inhibit tumor growth and enhance the efficacy of conventional chemotherapeutic agents. 8
In the context of pancreatic cancer, PL has shown promise in several in vitro and in vivo studies.9-11 The findings of these studies suggest that the bioactive components of the mushroom can induce apoptosis in pancreatic cancer cells, suppress tumor growth, and improve overall survival rates in animal models. 12 Furthermore, its anti-inflammatory and immune-modulating properties may help mitigate the adverse effects of chemotherapy, thereby improving the quality of life of patients undergoing treatment.
We previously performed a pivotal study highlighting the potential mechanisms through which PL exerts its anti-cancer effects. 13 In that study, we reviewed the potential role of the polysaccharides extracted from PL, which significantly inhibited the proliferation of pancreatic cancer cells by inducing cell cycle arrest and promoting apoptosis. 13 These findings are consistent with those of other studies, suggesting that PL can activate key apoptotic pathways, thereby enhancing the susceptibility of cancer cells to chemotherapeutic agents.9-11 We also investigated the synergistic effects of the PL extract and conventional postoperative adjuvant chemotherapy for patients with resected pancreatic cancer. 8 It was found that the combination of PL with gemcitabine, a standard chemotherapeutic agent for pancreatic cancer, resulted in a marked increase in cancer cell death compared to gemcitabine alone. This synergistic effect is attributed to the ability of PL to modulate the tumor microenvironment, reduce inflammation, and enhance immune responses, thereby improving the overall therapeutic outcomes. 8
In the present study, based on our accumulated clinical experience, we investigated the potential long-term oncological role of the perioperative application of PL as a nutraceutical in patients with resected pancreatic cancer. In addition, we examined the adverse events that occurred during adjuvant chemotherapy in patients who received PL.
Methods
Study Population and Design
This retrospective cohort study was conducted at a single institution. The study population consisted of patients diagnosed with pancreatic cancer at Severance Hospital from January 2012 to June 2022 who underwent curative resection followed by adjuvant chemotherapy. The study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14
Mesima® (Phellinus linteus mycelium extract) is produced by Han Kook Shin Yak Pharmaceutical Co., Ltd. (Choongnam, Korea) under GMP-certified conditions using standardized manufacturing processes to ensure product quality and consistency. The product has been approved by the Korean Food and Drug Administration. The supplement was administered orally at a dose of 1100 mg, 3 times a day, starting after surgery and continuing during adjuvant chemotherapy.
Some patients who underwent curative resection for pancreatic cancer were informed about PL and a previous pivotal study, and were prescribed the supplement before initiating adjuvant chemotherapy based on patient preference. Clinical data of patients who received PL and those who did not were retrospectively collected from electronic medical records (EMRs).
As shown in Figure 1, a total of 439 patients who were diagnosed with pancreatic ductal adenocarcinoma (PDAC) and underwent surgery at Severance Hospital between January 2012 and June 2022 were identified. After excluding 11 patients who underwent palliative procedures such as bypass surgery, a total of 428 patients were included in the final analysis. Among them, 124 consented to receive PL and began supplementation before undergoing adjuvant chemotherapy. Of these 124 patients, 103 who started taking PL before chemotherapy were designated as the PL group, whereas 21 who began taking PL during chemotherapy were excluded. The remaining 304 patients who did not undergo PL were assigned to the control group.

Flow chart of patients who received adjuvant chemotherapy after pancreatic resection, showing the inclusion of patients in the study.
Data Collection
The clinicopathological factors, perioperative management, short-term outcomes, and pathological results of the patients were collected from electronic medical records, including pathology reports, progress notes, and surgical records. Patient baseline comorbidity and general condition were classified using the American Society of Anesthesiologists grade. 15 Operative type was divided into distal pancreatectomy (DP), central pancreatectomy (CP), pancreaticoduodenectomy (PD), pylorus preserving pancreaticoduodenectomy (PPPD), and total pancreatectomy (TP). Surgical approach was classified as open pancreatectomy, laparoscopic pancreatectomy, or robot-assisted pancreatectomy. Postoperative complications were graded according to the Clavien–Dindo classification system, with severe complications defined as those of grade IIIa or higher. 16 Tumor stage was determined according to the American Joint Committee on Cancer (AJCC) eighth edition TNM staging system. 17 The variable “symptoms” was defined based on whether the patient presented with clinical symptoms that led to further evaluation and the eventual diagnosis of pancreatic cancer. Relevant symptoms included jaundice, weight loss, abdominal pain, dyspepsia, cholangitis, general weakness, anorexia, and nausea/vomiting. Vascular resection was defined as intraoperative resection of the superior mesenteric vein, splenic vein, or portal vein. The adjuvant chemotherapy regimen was categorized as either FOLFIRINOX-based or non-FOLFIRINOX-based. The non-FOLFIRINOX group included gemcitabine-based and other chemotherapy regimens. The duration of PL administration was recorded from outpatient clinic records. To evaluate the potential adverse effects of PL, liver function tests and serum creatinine levels were collected during the preoperative period and at each hospital visit for adjuvant chemotherapy administration. Chemotherapy-related adverse events were evaluated and graded according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. 18 Only grade 3 or higher toxicities were considered clinically significant and included in the analysis. The analysis focused on hepatotoxicity and nephrotoxicity. Information about the adjuvant chemotherapy regimen, recurrence, and cancer-related deaths was obtained from outpatient records and data from the Korean National Health Insurance Service.
Statistics
We conducted a comparative analysis between patients in the PL and control groups. Categorical variables were analyzed using the chi-square test or Fisher’s exact test, while continuous variables were assessed, after checking for normality, using either the Student’s t-test or Mann-Whitney U test. Recurrence-free survival (RFS) and overall survival (OS) were compared using the Kaplan-Meier method and log-rank tests. Median survival times were reported when estimable. In cases where fewer than 50% of patients experienced the event of interest, median survival could not be calculated, and mean survival times were reported instead. To minimize potential bias resulting from differing treatment periods between the 2 groups, the follow-up duration was limited to 60 months from the initiation of treatment.
The Cox proportional hazards model was used to analyze the prognostic factors associated with cancer-related deaths. For multivariate analysis, stepwise regression was performed with a threshold P-value less than .2, using backward elimination, and the therapeutic effect of PL was analyzed after adjusting for other factors that may influence OS. Variables included in the multivariable Cox regression analysis were selected based on either statistically significant differences between the 2 groups or prior evidence of prognostic relevance in pancreatic cancer. For instance, variables previously reported to be associated with survival—such as perioperative transfusion, postoperative complications, TNM stage, resection margin status, vascular resection, ASA grade, and adjuvant chemotherapy regimen (eg, FOLFIRINOX vs gemcitabine)—were included in the model.19,20 Conversely, variables that differed between groups but have not been shown to affect prognosis in previous studies (eg, minimally invasive surgery vs open surgery) were excluded from the multivariable analysis.21-25 To minimize the confounding effects of chemotherapy regimens, which may directly affect survival, we conducted a subgroup analysis by stratifying patients according to their treatment regimen. Cases with missing values in key variables were excluded from the analysis. Statistical significance was considered at a P-value less than .05, and all statistical analyses were performed using SPSS Version 24 (IBM, Chicago, IL, USA) and R 3.6.3.
Results
Patient Demographics
A comparison of the clinicopathological results between the 103 patients in the PL group and 304 patients in the control group is presented in Table 1. There were no significant differences between the 2 groups in terms of preoperative age, weight, or comorbidities. In contrast, a significant difference was observed in the presence of symptoms between the groups (P = .012), with details provided in Supplemental Table 1.
Baseline Demographic and Clinical Characteristics of the Subjects.

Continuous variables are presented as the mean ± standard deviation or median with interquartile range based on the normality test results.
Abbreviations: BMI, body mass index; ASA, American Society of Anesthesiologists; PD, pancreaticoduodenectomy; PPPD, pylorus-preserving pancreaticoduodenectomy; DP, distal pancreatectomy; TP, total pancreatectomy; CP, central pancreatectomy; MIS, minimally invasive surgery, PL, Phellinus linteus.
Regarding the timing of surgery, most patients in the PL group underwent surgery after 2018, as shown in Supplemental Figure 1a. Temporal variations were also noted in both the surgical approach and the selection of adjuvant chemotherapy regimens (FOLFIRINOX vs non-FOLFIRINOX), as illustrated in Supplemental Figures 1b and 1c.
A significantly higher proportion of patients in the PL group underwent minimally invasive surgery (MIS), including both robot-assisted and laparoscopic approaches, compared to the control group (P < .001). Furthermore, a notable difference in adjuvant chemotherapy regimens was observed: only 6.9% of patients in the control group received FOLFIRINOX, whereas 45.6% of those in the PL group received this regimen (P < .001).
Intraoperative transfusion rates, estimated blood loss, and tumor size also differed significantly between the 2 groups. In contrast, no significant differences were observed in the type of surgery performed, presence of vascular resection, TNM stage, resection margin status, or the incidence of postoperative complications.
Survival Analysis
The total follow-up duration was 13,825 person-months (1152.1 person-years). The mean follow-up time was 34.0 months, and the median follow-up was 33.0 months (IQR: 16.5-46.0). Survival analysis was conducted to evaluate the long-term oncologic outcomes of patients who underwent PL treatment. The mean OS time was 47.0 months for the PL group (95% confidence interval [CI]: 42.8-51.1) and the median OS time for the control group was 35.0 months (95% CI: 30.3-39.7), showing a statistically significant difference, as illustrated in Figure 2A (P < .001). Analysis of RFS showed that the PL group exhibited a trend toward improved recurrence rates, although this was borderline significant (P = .053; Figure 2B). In terms of FOLFIRINOX regimen and non-FOLFIRINOX regimen, the median RFS of patients with FOLFIRINOX regimen was 24.0 months (95% CI: 10.3-37.7), whereas the median RFS of patients with non-FOLRINOX regimen was 13.0 months (95% CI: 11.4-14.6; log-rank test, P = .009). The mean OS of patients with FOLFIRINOX regimen was 49.2 months (95% CI: 44.0-54.5), whereas the median OS of patients with non-FOLFIRINOX regimen was 37.3 months (95% CI: 35.2 vs 39.5; log-rank test, P = .002).

Kaplan-Meier curves for (A) overall survival and (B) recurrence-free survival, comparing the Phellinus linteus (PL) group and the control (C) group in the entire patient cohort; (C) overall survival and (D) recurrence-free survival among patients who received FOLFIRINOX as adjuvant chemotherapy, comparing the PL group and the C group; (E) overall survival and (F) recurrence-free survival among patients who received other regimens as adjuvant chemotherapy, comparing the PL group and the C group; and (G) overall survival among patients receiving FOLFIRINOX, non-FOLFIRINOX + PL, and non-FOLFIRINOX + no PL.
Prognostic Factors Affecting Survival in Patients with Resected Pancreatic Cancer
A Cox proportional hazards model was used to identify the prognostic factors influencing survival in patients who underwent pancreatic cancer resection. The results are presented in Table 2. In the univariate analysis, significant prognostic factors for overall survival included PL medication (HR: 0.439, 95% CI: 0.279-0.691, P < .001), perioperative transfusion (HR: 1.804, 95% CI: 1.270-2.562, P = .001), vascular resection (HR: 1.757, 95% CI: 1.233–2.504, P = .002), N stage (N1: HR: 1.729, 95% CI: 1.284-2.328, P = .003; N2: HR: 2.531, 95% CI: 1.739-3.682, P < .001), resection margin status (R2: HR: 2.446, 95% CI: 1.203-4.975, P = .014), and use of FOLFIRINOX as an adjuvant chemotherapy regimen (HR: 0.426, 95% CI: 0.243-0.749, P = .003).
Univariate and Multivariate Analyses of Prognostic Factors Associated with Overall Survival in Patients Undergoing Adjuvant Chemotherapy After Pancreatic Resection for Pancreatic Cancer.
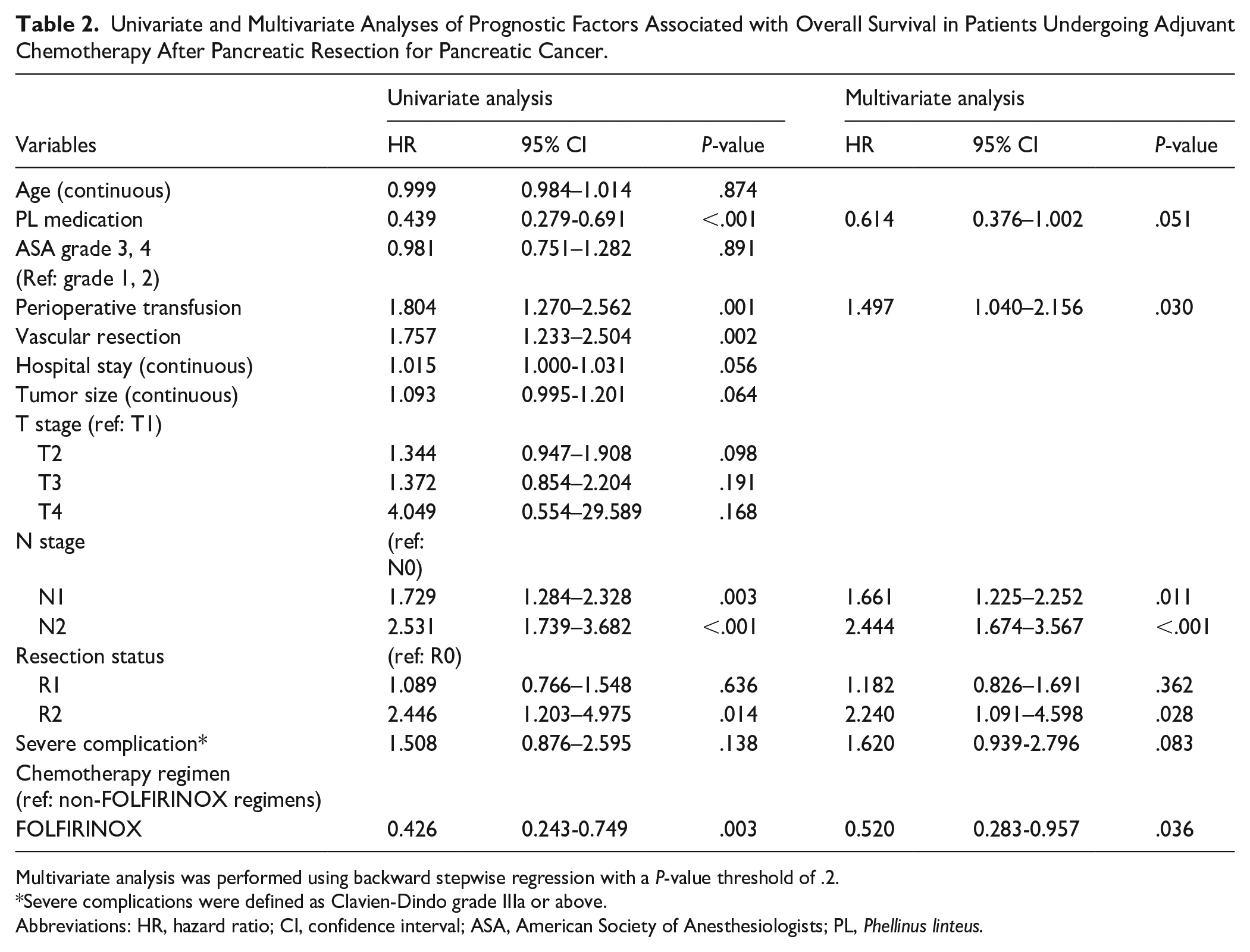
Multivariate analysis was performed using backward stepwise regression with a P-value threshold of .2.
Severe complications were defined as Clavien-Dindo grade IIIa or above.
Abbreviations: HR, hazard ratio; CI, confidence interval; ASA, American Society of Anesthesiologists; PL, Phellinus linteus.
In the multivariate Cox regression analysis, PL medication showed a marginal association with improved survival (HR: 0.614, 95% CI: 0.376-1.002, P = .051). Statistically significant independent prognostic factors included perioperative transfusion (HR: 1.497, 95% CI: 1.040-2.156, P = .030), N stage (N1: HR: 1.661, 95% CI: 1.225-2.252, P = .011; N2: HR: 2.444, 95% CI: 1.674-3.567, P < .001), resection margin status (R2: HR: 2.240, 95% CI: 1.091-4.598, P = .028), and FOLFIRINOX use (HR: 0.520, 95% CI: 0.283-0.957, P = .036).
PL Improves Survival in Resected Pancreatic Cancer with non-FOLFIRINOX Chemotherapy
Subgroup analysis was conducted to demonstrate the effect of PL on improving survival by stratifying patients according to whether they received FOLFIRINOX or non-FOLFIRINOX regimen as adjuvant chemotherapy. Among patients who received FOLFIRINOX as adjuvant chemotherapy, there was no statistically significant difference in overall survival (OS) between the 2 groups (FOLFIRINOX + PL group: mean OS, 44.9 months; 95% CI: 41.0-48.7 vs FOLFIRINOX control group: mean OS, 43.1 months; 95% CI: 33.3-53.0; P = .107).
Similarly, there was no significant difference in recurrence-free survival (RFS) between the groups (FOLFIRINOX + PL group: median RFS, 24.0 months; 95% CI: 14.5-33.5 vs FOLFIRINOX control group: median RFS, 14.0 months; 95% CI: 8.2-19.8; P = .292; Figure 2C and D). In the subgroup of patients who received non-FOLFIRINOX chemotherapy, long-term outcomes were compared between those who received PL and those who did not. In terms of overall survival (OS), the PL group showed a statistically significant improvement compared to the control group (non-FOLFIRINOX + PL: mean OS, 43.9 months; 95% CI: 38.0-49.8 vs non-FOLFIRINOX control group: mean OS, 35.0 months; 95% CI: 30.0-40.0; P = .021; Figure 2E).
However, there was no significant difference in recurrence-free survival (RFS) between the 2 groups (non-FOLFIRINOX + PL: median RFS, 24.0 months; 95% CI: 14.5-33.5 vs non-FOLFIRINOX control group: median RFS, 14.0 months; 95% CI: 8.2-19.8; P = .292; Figure 2F).
Finally, we analyzed long-term outcomes among 3 subgroups: (1) patients who received FOLFIRINOX, (2) patients who received non-FOLFIRINOX chemotherapy with PL, and (3) patients who received non-FOLFIRINOX chemotherapy without PL.
The mean OS in the FOLFIRINOX group was 49.2 months (95% CI: 44.0-54.5), while the non-FOLFIRINOX + PL group had a mean OS of 43.9 months (95% CI: 38.7-49.8), with no statistically significant difference between the 2 groups (log-rank test, P = .332).
In contrast, the non-FOLFIRINOX control group (without PL) had a median OS of 35.0 months (95% CI: 30.0-40.0), which was significantly lower than both the FOLFIRINOX group (log-rank test, P = .001) and the non-FOLFIRINOX + PL group (log-rank test, P = .021), as shown in Figure 2G.
Duration of PL Intake and Adverse Events
Among the 103 patients who elected to receive PL following curative resection for pancreatic cancer, the median duration of PL intake was 6.0 months (95% CI: 3.0-11.0). The distribution of PL intake duration is illustrated using a violin plot (Figure 3). The time interval from surgery to the initiation of adjuvant chemotherapy was significantly shorter in the PL group compared to the control group (median, 46.0 days [IQR, 37.0-57.0] vs 51.0 days [IQR, 45.0-65.5]; P < .001). Chemotherapy-related hepatotoxicity and nephrotoxicity were analyzed according to CTCAE version 5.0. Table 3 summarizes the incidence of grade 3 or higher toxicity across treatment regimens in both the PL and control groups.

Violin plot of duration of PL prescription in patients who received adjuvant chemotherapy after pancreatic resection.
Comparison of Grade ≥ 3 Hepatotoxicity and Nephrotoxicity between PL and Control Groups According to Chemotherapy Regimen.

There were no cases of grade 4 or higher hepatotoxicity or nephrotoxicity observed in the study population. One patient who received a gemcitabine-based regimen in the control group was excluded from the toxicity analysis due to missing follow-up data, as the patient received adjuvant chemotherapy at an outside institution.
Abbreviations: CTCAE, Common Terminology Criteria for Adverse Events; PL, Phellinus linteus.
Grade 3 or higher hepatotoxicity was observed in 15 patients (4.9%) in the control group and 12 patients (11.7%) in the PL group. When stratified by chemotherapy regimen (FOLFIRINOX, gemcitabine-based, and other agent-based regimens), no significant difference in hepatotoxicity was noted between the 2 groups for the FOLFIRINOX or other regimens. However, among patients receiving gemcitabine-based regimens, the incidence of grade ≥ 3 hepatotoxicity was significantly higher in the PL group compared to the control group (4.2% vs 15.1%; P = .008). Notably, all hepatotoxicity cases were grade 3, and no grade 4 or higher events were observed.
Discussion
Pancreatic cancer is well known to be one of the most lethal malignant diseases of the gastrointestinal system. 1 The difficulty in treating pancreatic cancer mainly stems from the difficulty of early detection and its biological characteristics. Pancreatic cancer often presents with minimal symptoms, and the initial symptoms can be very vague. Therefore, pancreatic cancer is usually found to be at an advanced stage at the time of diagnosis. Margin-negative pancreatectomy is thought to be the most effective monotherapy to cure the disease; however, resectable pancreatic cancer accounts for less than 20% of cases. 2 In addition, most patients with resected pancreatic cancer show a very high recurrence rate (especially in the liver, lung, and peritoneum), and the 5-year survival rate after surgery is still low. 26 Recurrence is mainly caused by micrometastases or residual cancer cells, which highlights the need for additional adjuvant therapy. 26
In the treatment of pancreatic cancer, nutraceutical studies have shown great potential as adjuvant therapies.13,27 Nutraceuticals are bioactive substances based on dietary ingredients, and various health improvement effects have been reported, including anti-cancer effects. 28 In particular, natural compounds, such as curcumin, resveratrol, and epigallocatechin gallate, have anti-inflammatory, antioxidant, and antiproliferative properties, which may contribute to the inhibition of the growth and metastasis of pancreatic cancer cells.13,27 In addition, these nutraceuticals may work synergistically with traditional chemotherapy and radiotherapy treatments and may help overcome treatment resistance. 8 However, further research and clinical trials are required to clinically demonstrate these benefits.
In a previous study, we investigated the potential oncological role of PL as a nutraceutical agent for treating pancreatic cancer.8,13 PL is a medicinal mushroom that is attracting attention for its anti-cancer effects, and its efficacy has been demonstrated in various studies.5,12,27,29 Its main active ingredients include polysaccharides, beta-glucan, and various polyphenols, which strengthen the immune system and exert anti-inflammatory and antioxidant effects.5,12 PL extract has been reported to inhibit the proliferation of cancer cells and promote apoptosis, especially in pancreatic cancer cell lines. 11 In addition, it has the potential to increase the therapeutic effect by showing a synergistic effect when combined with existing anti-cancer treatments. 8 In this context, several prior studies have demonstrated the potential of PL as an adjunct to conventional anti-cancer therapy. At our institution, two retrospective studies have reported on the efficacy of PL in resected pancreatic cancer.8,13 The first, involving 53 patients, showed improved disease-free survival (11.0 months vs 2.8 months, P = .017) without a significant OS difference. 8 The second, a propensity score-matched study of 217 patients, demonstrated improved DFS and OS, along with higher adherence to adjuvant chemotherapy. 13 Another external study involving gastric cancer patients showed that PL contributed to improved outcomes in patients receiving postoperative adjuvant therapy, with no serious adverse effects reported.8,13,30 The current study builds upon these findings and serves as a follow-up investigation to evaluate the clinical impact of PL in patients with resected pancreatic cancer.
To assess the internal and external validity of our findings, we compared the long-term survival outcomes of patients in our study with those reported in the PRODIGE 24/CCTG PA.6 trial, a landmark phase III study on adjuvant chemotherapy in pancreatic cancer.31,32 That trial included 493 patients and demonstrated a significant survival benefit with FOLFIRINOX over gemcitabine-based therapy: the median overall survival was 53.5 months (95% CI: 43.5-58.4) versus 35.5 months (95% CI: 30.1-40.3; HR, 0.68; 95% CI: 0.54-0.85; P = .001), and the median disease-free survival was 21.4 months (95% CI: 17.5-26.7) versus 12.8 months (95% CI: 11.6-15.2; HR, 0.66; 95% CI: 0.54-0.82; P < .001). In our study, the long-term survival outcomes for each adjuvant chemotherapy regimen were generally comparable to those reported in the PRODIGE 24/CCTG PA.6 study.31,32
Differences in baseline clinical characteristics between the PL and control groups must be considered when interpreting the validity of this study. Notably, the timing of surgery differed substantially between the 2 groups, leading to differences in follow-up duration. To mitigate potential bias, we limited the follow-up period to 60 months. Nonetheless, substantial differences remained in both the chemotherapy regimens and surgical approaches used. FOLFIRINOX became the standard of care for adjuvant chemotherapy following the publication of the PRODIGE 24/CCTG PA.6 trial in 2018.31,32 Prior to this, most patients received gemcitabine-based regimens.33,34 Despite the survival advantage associated with FOLFIRINOX, some patients continued to receive alternative regimens due to intolerance or toxicity concerns. 35
To account for these confounding effects, subgroup analyses and multivariable Cox regression models were used to evaluate the independent impact of PL on survival. Furthermore, with the growing adoption of minimally invasive surgery (MIS), particularly in more recent years, a higher proportion of patients undergoing surgery during the later study period received laparoscopic or robot-assisted procedures. Given that recent literature, including studies from our institution, has shown that MIS achieves long-term oncologic outcomes comparable to open surgery in pancreatic cancer, we opted not to include surgical approach as a covariate in the multivariable analysis, as it was not considered a decisive prognostic factor for adjuvant chemotherapy effectiveness.21-25
Consistent with previous studies, established prognostic factors such as resection margin status, nodal stage, and use of FOLFIRINOX were independently associated with long-term survival in our analysis.21-25 While the effect of PL use did not reach the conventional levels of statistical significance, it demonstrated a marginal association with improved survival. These findings suggest a potential benefit that warrants further investigation through prospective or randomized controlled trials.
In the present study, a higher proportion of patients in the PL group than the control group received FOLFIRINOX as adjuvant chemotherapy, which might explain the higher survival rates observed in the PL group. However, even after adjusting for chemotherapy regimens and other factors affecting survival in the multivariate Cox analysis, PL supplementation was marginally associated with improved survival.
Among patients treated with FOLFIRINOX, the addition of PL did not appear to yield additional survival benefits. Given the small sample size in this subgroup, caution is warranted in interpreting these findings. In contrast, patients who received non-FOLFIRINOX regimens, such as fluorouracil- or gemcitabine-based therapies, appeared to benefit from the addition of PL, suggesting a potential role in less intensive chemotherapy settings.
The enhanced survival observed in the PL group likely reflects the beneficial effects of PL in patients treated with regimens other than FOLFIRINOX. This suggests that PL is a potential strategy to enhance the effectiveness of chemotherapy, particularly for patients who need to receive alternative regimens because of the side effects of FOLFIRINOX. In addition, despite no statistically significant differences, a minor tendency toward superior long-term oncological outcomes was observed in patients in the PL group treated with FOLFIRINOX. Based on this observation, the potential oncological role of PL should be re-evaluated even in patients undergoing FOLFIRINOX treatment in the near future, because a very small proportion of the patients in control group (6.9%) were treated with FOLFIRINOX.
The median duration of PL intake was approximately 6 months. Although the median interval between surgery and the initiation of adjuvant chemotherapy differed by several days between the 2 groups and reached statistical significance, this difference is unlikely to be clinically meaningful. Thus, PL administration does not appear to have substantially delayed the initiation of adjuvant chemotherapy. During the study period, a statistically significant difference in the incidence of grade ≥ 3 hepatotoxicity was observed among patients receiving gemcitabine-based chemotherapy, with fewer events in the PL group compared to the control group. However, no grade 4 or higher hepatotoxicity was reported in either group, and no grade 3 or higher nephrotoxicity was observed in the PL group. Based on previous clinical studies, PL has demonstrated a favorable safety profile, with most adverse events limited to mild gastrointestinal symptoms or skin reactions.30,36
When considering the fact that most patients with resected pancreatic cancer experience systemic recurrence within 1 or 2 years after surgery, potent and effective postoperative adjuvant chemotherapy should be mandatory. 26 However, pancreatic surgery is associated with high complication rates and delays in functional recovery of the patients. 37 Therefore, most patients can receive postoperative adjuvant chemotherapy in 1.5 to 3 months after radical pancreatectomy. 37 It has been hypothesized that residual cancer cells contribute to the systemic recurrence of pancreatic cancer after surgery. 38 Moreover, it has been noted that the interval from immediately after radical pancreatectomy to the initiation of postoperative chemotherapy is the specific period when the remaining cancer cells can be activated due to the influence of pancreatectomy-related cytokines, but no conventional systemic chemotherapy can be used during the recovery period after radical pancreatectomy.39-41 This particular period is regarded as the “window period” of pancreatic cancer treatment when no anti-cancer treatment is available.42,43 To enhance the effectiveness of postoperative chemotherapy, it has also been hypothesized that the attenuation of potential residual cancer cells during this period would be helpful. Systemic anti-cancer drugs that can be used during this window period (1) should help remove the remaining cancer cells, (2) should not adversely affect the patient’s recovery after surgery, (3) should not reduce the patient’s immune function, and (4) should not adversely affect standard postoperative chemotherapy. With this in mind, it is highly likely that herbal medicines with proven anti-cancer effects have potential to be used in the window period. For example, PL used in this study is a possible option for application during the window period after pancreatic cancer surgery. We confirmed an improvement in long-term survival rates in patients who received PL after surgery.
This study has several limitations inherent to its retrospective design, including potential selection bias related to treatment regimens, surgical approach, and postoperative management. In particular, the difference in surgical timing between the PL and control groups may have introduced confounding, as patients in the PL group were more likely to receive FOLFIRINOX and undergo minimally invasive surgery. To mitigate these biases, we limited the follow-up period to 60 months and performed stratified and multivariable analyses. In addition, the prescription and adherence to PL were not standardized, and patients who survived longer naturally had extended durations of PL intake, making it difficult to assess the direct relationship between treatment duration and survival. Finally, subgroup analysis within the FOLFIRINOX cohort was limited by a small sample size, and further studies are warranted to clarify potential interactions between PL and specific chemotherapy regimens. Further research is also needed to elucidate the biological mechanisms underlying the potential survival and anti-recurrence benefits of PL. Nevertheless, through methodical statistical adjustment, this study was able to demonstrate the potential adjunctive benefit of PL in the context of established pancreatic cancer treatment protocols.
Conclusions
The prognosis of pancreatic cancer has improved in recent years due to the development of more effective chemotherapeutic agents such as FOLFIRINOX and gemcitabine/nab-paclitaxel (GEM-ABX). However, patients must endure significant adverse effects, and the use of intensive anti-cancer agents is often restricted during the perioperative period, adversely affecting quality of life. Although this study was retrospective in nature, the observed improvement in 5-year survival among patients who received PL was notable and warrants further investigation. Moreover, no grade 4 or higher hepatotoxicity or nephrotoxicity was observed, indicating that PL has a favorable safety profile. Based on these findings, a prospective randomized clinical trial is needed to further validate the concept of the postoperative “window period” and to explore the potential oncologic role of nutraceuticals, such as PL, in the adjuvant treatment of pancreatic cancer.
Supplemental Material
sj-doc-2-ict-10.1177_15347354251353499 – Supplemental material for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study
Supplemental material, sj-doc-2-ict-10.1177_15347354251353499 for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study by Juwan Kim, Seung Soo Hong, Sung Hyun Kim, Ho Kyoung Hwang and Chang Moo Kang in Integrative Cancer Therapies
Supplemental Material
sj-docx-1-ict-10.1177_15347354251353499 – Supplemental material for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study
Supplemental material, sj-docx-1-ict-10.1177_15347354251353499 for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study by Juwan Kim, Seung Soo Hong, Sung Hyun Kim, Ho Kyoung Hwang and Chang Moo Kang in Integrative Cancer Therapies
Supplemental Material
sj-jpg-4-ict-10.1177_15347354251353499 – Supplemental material for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study
Supplemental material, sj-jpg-4-ict-10.1177_15347354251353499 for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study by Juwan Kim, Seung Soo Hong, Sung Hyun Kim, Ho Kyoung Hwang and Chang Moo Kang in Integrative Cancer Therapies
Supplemental Material
sj-xlsx-3-ict-10.1177_15347354251353499 – Supplemental material for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study
Supplemental material, sj-xlsx-3-ict-10.1177_15347354251353499 for Perioperative Clinical Usage of Phellinus Linteus as a Nutraceutical for Non-FOLFIRINOX-Based Postoperative Adjuvant Chemotherapy for Resected Pancreatic Cancer: A Retrospective Cohort Study by Juwan Kim, Seung Soo Hong, Sung Hyun Kim, Ho Kyoung Hwang and Chang Moo Kang in Integrative Cancer Therapies
Footnotes
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of our institution (approval number: 2022-1556-001).
Consent to Participate
The study was approved by the Institutional Review Board of our institution (approval number: 2022-1556-001. All participants provided written informed consent prior to participating. Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
Consent for Publication
Not applicable.
Author Contributions
JK analyzed the data and wrote the manuscript. HKH, SHK, and SSH critically revised the manuscript for important intellectual content. CMK supervised and designed the study. All authors have approved the final version of the manuscript for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Han Kook Sin Yak Pharmaceutical Co., Ltd., Choongnam, Korea [grant number 2022-31-1119]. The funder had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The research data have been included as Supplemental Material.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
