Abstract
Background:
This retrospective study investigated the efficacy and safety of chemotherapy (CTX) integrated with Traditional Korean Medicine (TKM) in patients with metastatic pancreatic cancer, in a single Korean center.
Methods:
From January, 2014 to February, 2019, patients with metastatic pancreatic cancer who had received CTX were enrolled. Overall survival (OS), demographic characteristics, and adverse events were examined. Statistical analysis was utilized to evaluate the differences in characteristics and to compare the survival rates between the CTX group and CTX+TKM group. Kaplan-Meier curves were used to compare the differences in survival time. A Cox regression analysis was performed to determine the hazard ratio of the risk of mortality.
Results:
A total 37 participants were included and visited a TKM hospital 7.4 ± 8.3 months after being diagnosed with metastatic pancreatic cancer. The median age of the participants was 62 years; 26 patients (70.3%) had an Eastern Cooperative Oncology Group (ECOG) performance status between 0 and 2, and 23 patients (62.2 %) had first-line CTX failure. The median OS of all patients was 3.8 months (95% CI, 3.1-4.6). The CTX + TKM group showed longer survival (4.1 months; 95% CI, 2.4- .8) than the CTX group (2.4 months, 95% CI 0.2-4.6) but this was not statistically significant (P = .217). Chemotherapy with TKM treatment for more than 30 days (CTX + TKM ≥ 30) significantly prolonged median OS (9.1 months; 95% CI, 3.6-14.5; P = .025) compared to chemotherapy alone. Cox hazard ratio analysis revealed that CTX + TKM ≥ 30 and prior chemotherapy were significantly independent prognostic factors for OS. The main herbs in the TKM treatment were Rhus verniciflua Stokes and Astragalus. Severe adverse events with respect to TKM treatment were not reported.
Conclusions:
TKM treatment integrated with chemotherapy may prolong OS in patients with metastatic pancreatic cancer compared to chemotherapy treatment alone. More rigorous prospective clinical trials are needed to confirm this result.
Introduction
Pancreatic cancer remains a lethal malignancy and is the 5th leading cause of cancer-related death in Korea. 1 More than half of pancreatic cancer patients present either unresectable or metastatic cancer at diagnosis. The 5-year survival for metastatic pancreatic cancer is approximately 2%, despite recent advances in conventional medicine.2-5
Traditional Korean Medicine (TKM) has been used intensively as an integrative cancer therapy for relieving cancer-related symptoms, alleviating the adverse events of conventional treatments, sensitizing the anticancer effects of chemotherapy, and prolonging survival time in cancer patients. A range of anticancer molecular targets of TKM, including inflammation, invasion, metastasis, protein kinases, the cell cycle, apoptosis, growth factors and receptors have been identified and extensive scientific research regarding commonly used TKM herbs for cancer treatment has been undertaken. 6
A recent study reported that Rhus verniciflua Stokes (RVS)-based TKM has potential benefits for overall survival (OS) and clinical symptoms without significant adverse events in patients with advanced or metastatic pancreatic cancer. 7 In a preclinical study, RVS suppressed the invasion and migration of pancreatic cancer cells via downregulation of the JAK/STAT and Src/FAK signaling pathways. 8 A case study reported that a patient with metastatic pancreatic cancer treated with RVS and Astragalus-based TKM combined with chemotherapy showed 29 months of prolonged progression-free survival. 9 Astragalus has an immune-modulating effect and Astragalus-based herbal medicine improves cancer-associated anorexia, 10 increases OS, tumor response rate, performance status, and reduces chemotherapy-associated adverse events in non-small cell lung cancer and colorectal cancer.11-14
This retrospective study investigated whether chemotherapy integrated with TKM, including RVS and Astragalus as the main herbs, prolonged OS in patients with metastatic pancreatic cancer in a Korean medicine cancer center.
Patients and Methods
Eligibility Criteria
From January, 2014 to February, 2019, pancreatic cancer patients who had visited and been treated at the Korean Medicine Cancer Center in Kyung Hee University Hospital at Gangdong were reviewed retrospectively, based on electronic medical records. Inclusion criteria were as follows: (1) histological or cytological diagnosis of pancreatic cancer; (2) pathologically and radiologically confirmed metastatic cancer of the pancreas based on the American Joint Committee on Cancer (AJCC) Cancer Staging Manual; (3) underwent at least 1 prior chemotherapy cycle; (4) aged 18 years or older. Exclusion criteria were as follows: (1) prior history of using TKM treatment; (2) unavailable National Health Insurance Corporation survival data.
Demographic characteristics including sex, age, Eastern Cooperative Oncology Group (ECOG) performance status, chemotherapy regimen, number of prior chemotherapy lines, period of TKM administration, and interval between the metastatic diagnosis of pancreatic cancer and day of the first visit were obtained from medical records. This study was approved by the Institutional Research Board of the Kyung Hee University Hospital at Gangdong (Seoul, Republic of Korea) (IRB No. 2019-04-001).
Traditional Korean Medicine Treatment
Experienced TKM doctors at Kyung Hee University Hospital at Gangdong performed the TKM treatment. RVS extract and Astragalus-based herbal medicine were generally administered orally, 2 or 3 times a day, 30 minutes after a meal.
RVS extract was manufactured at Kyung Hee University Medical Center as follows. RVS, which was purchased from Kyung Hee Pharmacy, was roasted for 1 hour at 180°C and extracted with distilled water for 2 hours. The extract was filtered for to remove allergens, and concentrated under a vacuum. The concentrate was lyophilized to a powder and packed in capsules containing 350 mg per capsule (yield 3.4%). The quality of the RVS extract was tested and monitored according to the standards of our hospital (fisetin >0.6%, urushiol not detected).
The Astragalus-based herbal medicine consisted of Astragalus membranaceus 24 g, Atractylodes macrocephala 12 g, Poria cocos 12 g, Pinellia ternata 12 g, Citrus unshiu 12 g, Agastache rugosa 4 g, Alisma orientale 12 g, Plantago asiatica 8 g, Spatholobus suberectus 8 g, Zizyphus jujuba 12 g, Glycine max 8 g, Crataegus pinnatifida 6 g, Hordei fructus germinatus 6 g, Prunus mume 6 g, and Glycyrrhiza uralensis 6 g, per day, as described in our previous report. 10 The prescription was made by adding the herbal medicines in 4.6 L of water and boiling at 102°C to 103°C for 2.5 hours. Individual prescriptions were aliquoted into 120 cm3 pouches at the hospital pharmacy.
TKM treatment was administered concurrently or sequentially with chemotherapy. In addition, rescue therapies were administered intermittently to control patients’ symptoms and maintain the best supportive care.
Outcome Measures
The primary endpoint of this study was a comparison of OS of patients treated with chemotherapy only (CTX group) and patients treated with TKM-integrated chemotherapy (CTX + TKM group). OS was defined as the time from the first day of TKM treatment or visiting our medical center to the time of death from any cause or the end of the study (February 28, 2019), or time of loss to follow-up. All patients who received TKM started TKM treatment on their first visit. Based on the median period of TKM administration and data from previous studies,7,15 the effect of TKM was analyzed by dividing the period of TKM administration into 30 days more than this value (CTX + TKM ≥ 30 group) and 30 days less than this value (CTX + TKM < 30 group).
For patients treated with TKM or chemotherapy, toxicity was evaluated as a grade from 1 to 4 based on the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0.
Statistical Analysis
The Kruskal-Wallis test or One-Way Analysis of Variance (ANOVA) test was used depending on the normality of variables to compare the groups. Survival curves were estimated using the Kaplan-Meier method and statistically compared using the log-rank test. The Cox proportional hazard model analysis of OS, sex, age, ECOG status, number of chemotherapy lines, and period of TKM treatment was used as a multivariate approach for assessing potential predictors of OS. A P-value <.05 was considered statistically significant, and all analyses were performed using SPSS version 18.0 (Statistical Package for the Social Sciences, Inc, Chicago, IL).
Results
Demographic and Clinical Characteristics
A total of 37 patients with metastatic pancreatic cancer were included in this study (Figure 1). The median age was 62 years (range 36-83 years) and 26 (70.3%) patients had an ECOG performance status of 0 to 2. About 23 (62.2%) patients received surgery. At baseline, 2 (5.4%) patients had not received prior chemotherapy, 23 (62.2%) patients had received 1 prior chemotherapy regimen, and 12 (32.4%) patients had received 2 or more chemotherapy regimens. Twenty-three (62.2%) patients received 5-Flurouracil-based chemotherapy and 17 (45.9%) patients received gemcitabine-based chemotherapy. The mean interval between the initial diagnosis of metastatic pancreatic cancer and the first visit to our center was 7.4 ± 8.3 months (Table 1).

Flowchart of subjects.
Baseline Demographics and Clinical Characteristics of Patients (n = 37).
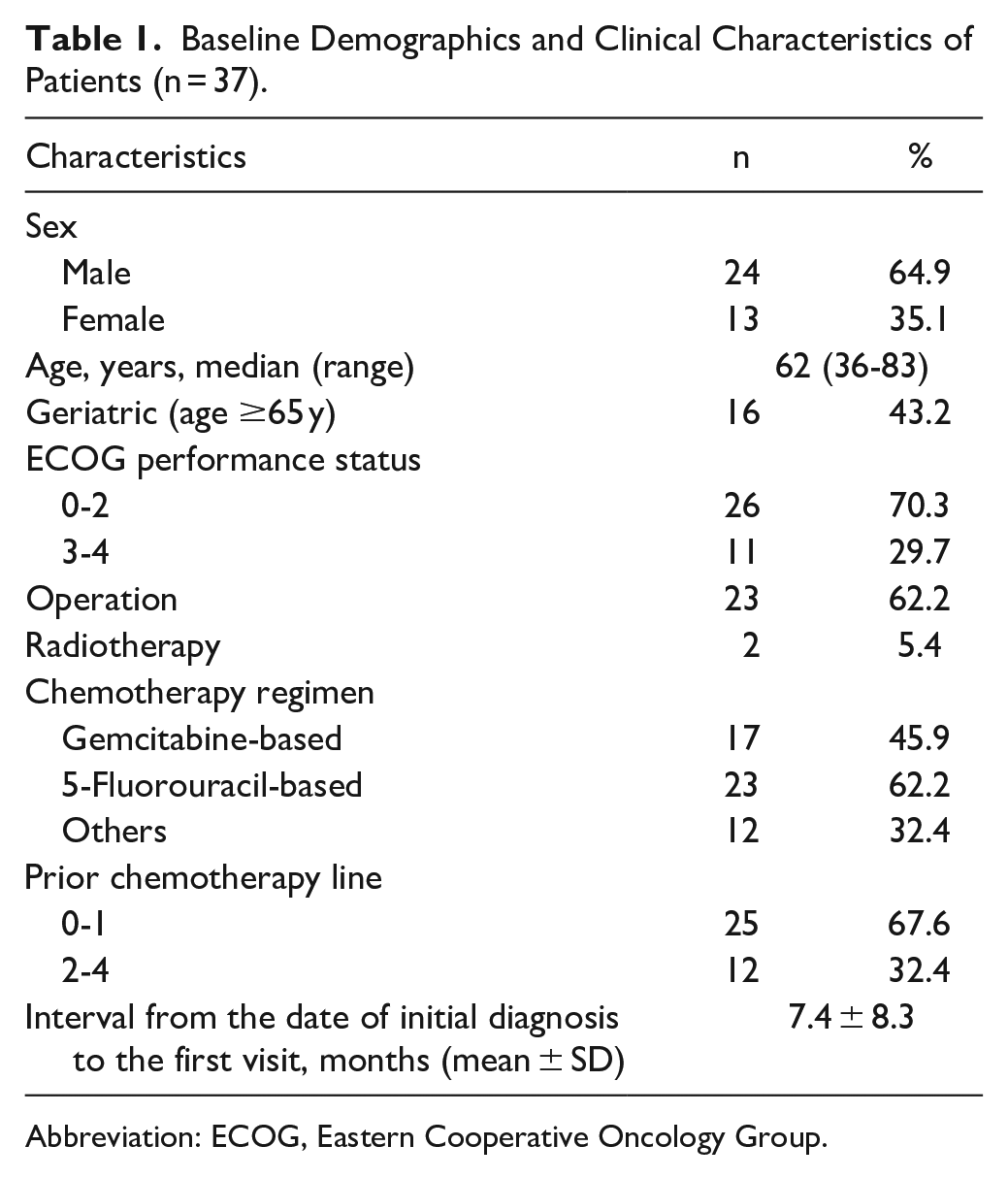
Abbreviation: ECOG, Eastern Cooperative Oncology Group.
Out of all the patients, 25 (67.6%) patients received TKM treatment with chemotherapy (CTX + TKM group) and 12 (32.4%) received chemotherapy only (CTX group). The median period of TKM treatment was 26 days (range 3-279 days). Among the CTX + TKM group, 10 (27.0%) patients underwent TKM treatment for more than 30 days (CTX + TKM ≥ 30 group) and 15 (40.5%) patients underwent TKM treatment less than 30 days (CTX+TKM<30 group). There were no significant differences in characteristics between the CTX group, CTX + TKM < 30 group, and the CTX + TKM ≥ 30 group (Table 2).
Baseline Characteristics of Patients According to Use of Traditional Korean Medicine.

Abbreviations: CTX, Chemotherapy; TKM, Traditional Korean Medicine; SD, Standard deviation; ECOG, Eastern Cooperative Oncology Group.
Survival Outcomes
From January, 2014 to February, 2019, 31 patients died and the remaining 6 patients were alive at the end of the study. The median OS for all patients was 3.8 months (95% confidence interval [CI], 3.1-4.6), and the estimated 1-year survival rate was 8.2% (Figure 2).
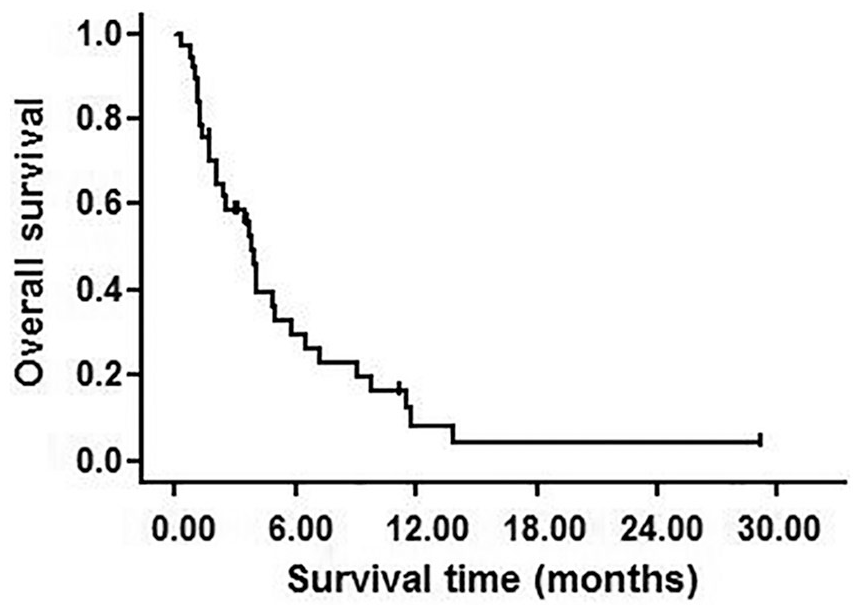
Kaplan-Meier survival plots for overall survival of patients. The median overall survival was 3.8 months (95% CI, 3.1-4.6), and the estimated 1-year survival rate was 8.2%.
The median OS of the CTX group and CTX + TKM group were 2.4 months (95% CI, 0.2-4.6) and 4.1 months (95% CI, 2.4-5.8), respectively, but this was not statistically significant (P = .217; Figure 3a). For the CTX + TKM group, the CTX + TKM ≥ 30 group showed more prolonged survival time than the CTX + TKM < 30 group (9.1 months [95% CI, 3.6-14.5] vs. 2.5 months [95% CI, 0.5-4.5], P = .028; Figure 3b). The median OS of the CTX + TKM ≥ 30 group was significantly longer than the median OS of the CTX group (9.1 months vs. 2.4 months, P = .025; Figure 3c). There were no significant differences in characteristics between the CTX group and the CTX + TKM ≥ 30 group.

Overall survival of patients with metastatic pancreatic cancer treated with chemotherapy integrated with or without Traditional Korean Medicine. (a) Overall survival of the CTX group and the CTX + TKM group. The median overall survival of the CTX group (n = 12) and the CTX + TKM group (n = 25) were 2.4 months (95% CI, 0.2-4.6) and 4.1 months (95% CI, 2.4-5.8), respectively (P = .217). (b) Overall survival of the CTX + TKM < 30 group and the CTX + TKM ≥ 30 group. The median overall survival of the CTX + TKM <30 group (n = 15) and the CTX + TKM ≥ 30 group (n = 10) were 2.5 months (95% CI, 0.5-4.5) and 9.1 months (95% CI, 3.6-14.5) respectively (P = .028). (c) Overall survival of the CTX group and the CTX + TKM ≥ 30 group. The median overall survival of the CTX group (n = 12) and the CTX + TKM ≥ 30 group (n = 10) were 2.4 months (95% CI, 0.2-4.6) and 9.1 months (95% CI, 3.6-14.5), respectively (P = .025).
According to the Cox hazard ratio model, inclusion in the CTX + TKM ≥ 30 group had a lower hazard ratio (HR) for mortality risk, independently (adjusted HR, 0.32 [95% CI, 0.13-0.79], P = .014). In addition, a prior chemotherapy line was also an independent prognostic factor for OS. The mortality risk in patients who received 2 or more chemotherapy lines was higher than that of 1 or less chemotherapy line (adjusted HR, 2.63 [95% CI, 1.22-5.68], P = .014; Table 3).
Univariate and Multivariate Analyses of Predicting Factors Associated with Overall Survival in Patients with Metastatic Pancreatic Cancer.
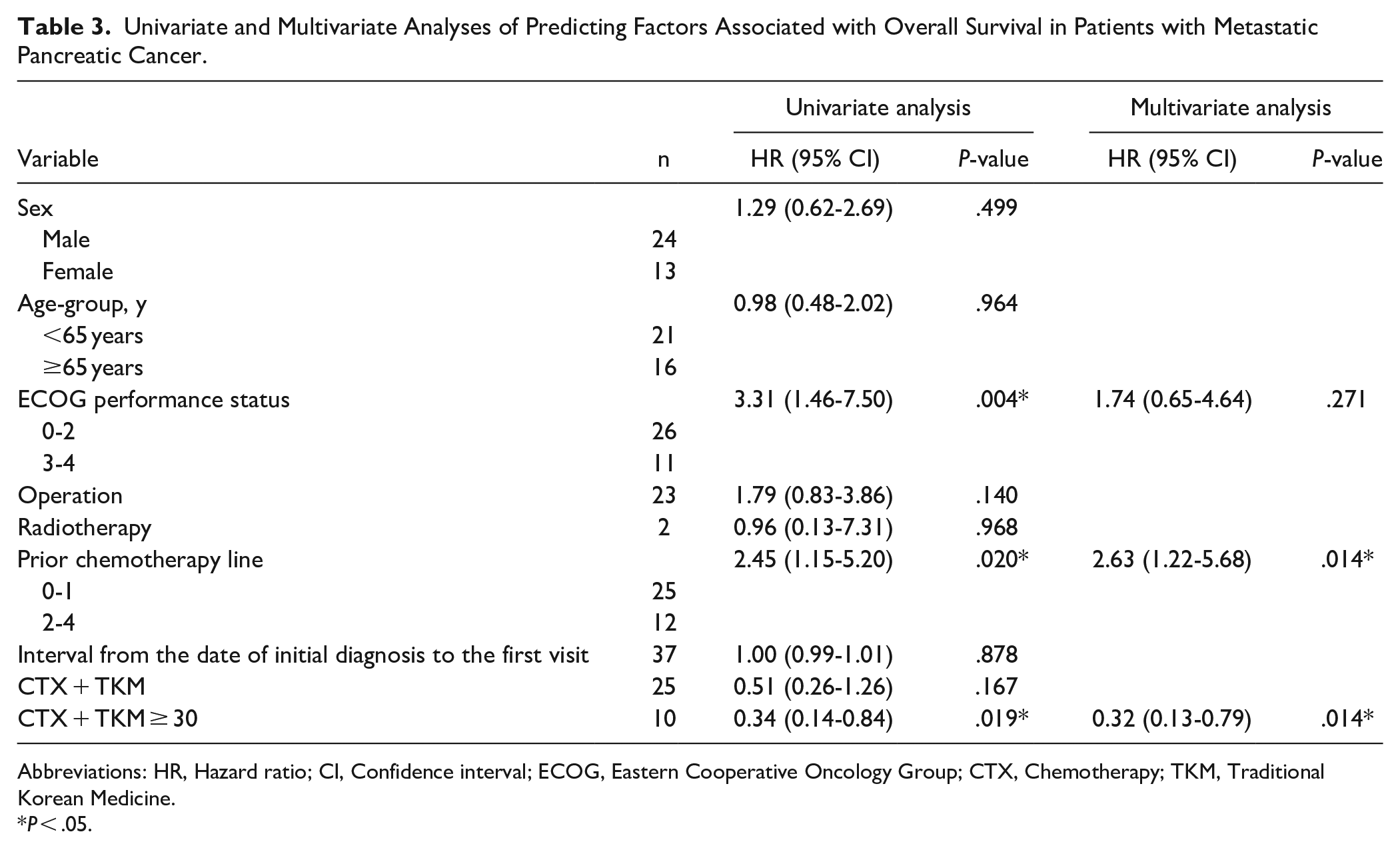
Abbreviations: HR, Hazard ratio; CI, Confidence interval; ECOG, Eastern Cooperative Oncology Group; CTX, Chemotherapy; TKM, Traditional Korean Medicine.
P < .05.
Adverse Events and Safety
Hematological toxicity and adverse events of grade 3 or higher of the CTCAE related to TKM treatment were not observed. Mild fatigue and dyspepsia were observed in 10 patients and 3 patients, respectively, with toxicity grade 2. These symptoms were reversible and diminished without reducing TKM treatment.
Discussion
We retrospectively analyzed patients with metastatic pancreatic cancer to investigate the benefits of TKM treatment integrated with chemotherapy. RVS extract and Astragalus-based herbal medicine were administered mainly for TKM treatment. Median OS, which was defined as the time from the first date of TKM treatment to the time of death, was 3.8 months in all participants. Considering that the duration between the initial diagnosis of metastatic pancreatic cancer and the first visit to our center was 7.4 ± 8.3 months, the survival time of all patients in this study was extended to approximately 11 to 12 months. These results are consistent with the median OS of 8 to 11 months in external control patients with metastatic pancreatic cancer who received palliative chemotherapy. 16 The median OS of 9.1 months in patients who underwent TKM treatment integrated with chemotherapy for more than 30 days (CTX + TKM ≥ 30 group) was significantly longer than the median OS in the patients who received CTX only (CTX group), and inclusion in the CTX + TKM ≥ 30 group was identified as an independent prognostic factor.
The standardized chemotherapeutic regimens for metastatic pancreatic cancer include 5-fluorouracil and gemcitabine as single agents or combination regimens such as FOLFIRINOX and gemcitabine/nanoparticle albumin-bound paclitaxel. 17 Although the survival outcome of metastatic pancreatic cancer has improved to 11.1 months owing to the development of conventional chemotherapy, severe adverse events to chemotherapy not only degrade quality of life but also require reduction or termination of chemotherapy. 18 Besides, as many patients with metastatic pancreatic cancer show poor treatment performance, finding effective conventional anti-cancer treatments still represents a major challenge to these vulnerable patients.19,20
TKM treatment including herbal medicines as integrative adjuvant cancer therapies has the reported benefits of prolonging survival outcome, reducing side effects associated with conventional treatment, preventing metastasis, and improving quality of life in cancer patients.21,22 According to a prior case series study reported by Wong et al, 23 pancreatic cancer patients with experience of chemotherapy using herbal medicines showed significantly prolonged overall survival of 15.2 months. In another previous retrospective study, Kuo et al 15 reported that pancreatic cancer patients who received traditional herbal medicine for more than 30 days showed lower mortality risk and higher OS than patients who did not receive traditional herbal medicine. In addition, Yang et al 24 also showed that pancreatic cancer patients who received traditional herbal medicine for more than 2 months showed longer median OS than patients who did not (19 months vs. 8 months, P < .001). The outcomes of these studies support the potential benefit of TKM treatment in patients with metastatic cancer who received chemotherapy, as in the present study.
RVS has been traditionally prescribed in Korea for detoxifying and resolving blood stasis and masses. 25 The active ingredients of RVS are fustin, fisetin, butein, sulfuretin, gallic acid, and kaempferol. These substances have been reported to exhibit anti-oxidant, apoptotic, anti-proliferative, anti-angiogenic, and anti-tumor effects in various cancer cells, including pancreatic cancer.8,25-27 In a previous clinical study, receiving RVS extracts more than 30 days prolonged the survival outcome in pancreatic cancer (9.8 months vs. 6.0 months, P < .036), and was an independent prognostic factor in survival (P = .001). 7 These results suggest that RVS could be beneficial in prolonging the survival of pancreatic cancer patients.
Astragalus is one of the most widely used medicinal plants for relieving fatigue, lack of appetite, and immune dysfunction in cancer patients. The major active compounds of Astragalus, such as glycosides, polysaccharides, and flavone are demonstrated to have the clinical benefits of enhancing chemosensitivity and reducing chemotherapeutic agent-associated adverse reactions.28,29 We previously reported that an Astragalus-based herbal medicine, which has the same composition as that used in this study, improved cancer-associated anorexia. 10 According to previous systematic reviews, Astragalus-based herbal medicine combined with chemotherapy for the treatment of non-small cell lung cancer and colorectal cancer may increase OS, efficacy of tumor response rate, and performance status, and reduce chemotherapy-derived toxicities including neutropenia, anemia, thrombocytopenia, fatigue, anorexia, nausea, and vomiting.11-14 These chemosensitizing and chemoprotective effects of Astragalus-based herbal medicines are expected to show similar results for pancreatic cancer.
This study has some limitations. First, this study was a small sample-sized, retrospective study performed at a single center, and immortal time bias can affect the survival outcomes. Second, there were long intervals between the diagnoses of metastatic pancreatic cancer and the first visits to the hospital. We also presume that only a small number of cancer patients had an open discussion about TKM treatment with their physicians or TKM doctors in Korea, at their initial diagnoses. 30
In conclusion, TKM treatment may improve the survival of patients with metastatic pancreatic cancer treated, or being treated, with chemotherapy. A well-designed large-scale prospective study is required to support this result.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (HF20C0038).
