Abstract
Objective
To undertake a meta-analysis of the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX (FOL [folinic acid], F [fluorouracil], IRIN [irinotecan], OX [oxaliplatin]) after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
Methods
This meta-analysis searched electronic databases, including Embase®, Medline, PubMed® and the Cochrane library, for eligible studies that reported the use of FOLFIRINOX and other drug regimens as second-line chemotherapy after failure of gemcitabine-based chemotherapy. Pooled analyses for progression-free survival (PFS), overall survival (OS), objective response rate (ORR), disease control rate (DCR) and grade 3/4 treatment-emergent adverse events (TRAEs) were undertaken.
Results
The analysis included six studies with a total of 858 patients. Compared with the three other second-line regimens, FOLFIRINOX had a significantly longer PFS (hazard ratio [HR] 0.68, 95% confidence interval [CI] 0.52, 0.89) and OS (HR 0.71, 95% CI 0.59, 0.86); and a significantly better ORR (HR 0.43, 95% CI 0.23, 0.80) and DCR (HR 0.71, 95% CI 0.58, 0.88). However, grade 3/4 adverse events were more frequently reported in patients administered FOLFIRINOX compared with the other three regimens.
Conclusion
FOLFIRINOX is recommended as a second-line chemotherapy regimen for patients with pancreatic cancer that have failed on gemcitabine-based first-line therapy.
Research Registry number: reviewregistry1300
Introduction
Pancreatic cancer is one of the deadliest malignant diseases that has an extremely poor prognosis. The estimated number of new cases in 2021 in the US was 60430, with 48220 expected to die of it. 1 Radical resection, which remains the only curative choice for pancreatic cancer, can only be performed on less than 15% of patients with localized lesions. 2 For patients with advanced pancreatic cancer, chemotherapy or chemoradiotherapy is usually the only choice for prolonging their life expectancy. Gemcitabine has been recommended as the first-line drug for adjuvant chemotherapy and palliative chemotherapy for decades. 3 New drug combinations, including FOLFIRINOX (FOL – folinic acid, F – fluorouracil [5-FU], IRIN – irinotecan, OX – oxaliplatin), have been demonstrated to be more effective than gemcitabine in pancreatic cancer patients. 4 Due to the toxicity and high rates of adverse reactions associated with FOLFIRINOX , even at a reduced dosage, the use of this regimen is usually restricted to patients with a good performance status (Eastern Cooperative Oncology Group [ECOG] 0–1). Compared with gemcitabine alone, gemcitabine plus albumin-bound paclitaxel (AG) showed a better survival benefit in patients with metastatic pancreatic cancer. 3 AG can also be used in patients with a poorer status (ECOG 0–2) compared with patients in the FOLFIRINOX group. 4 FOLFIRINOX and AG are the most commonly recommended first-line regimens for advanced pancreatic cancer.5,6 For patients with an even poorer performance status, gemcitabine is still be recommended as first-line chemotherapy. 5 Gemcitabine alone and regimens containing gemcitabine account for a large proportion of the first-line chemotherapy for patients with advanced pancreatic cancer.7,8
For patients that accept gemcitabine-based first-line chemotherapy, the subsequent recommended regimens are those combinations that contain 5-FU or capecitabine, including FOLFIRINOX, FOLFOX (FOL – folinic acid, F – fluorouracil [5-FU], OX – oxaliplatin), FOLFIRI (FOL – folinic acid, F – fluorouracil [5-FU], IRI – irinotecan), CAPOX (CAP – capecitabine, OX – oxaliplatin) or a single use of 5-FU and capecitabine infusions.9 –11 Single use of tegafur (S-1) and combination regimens containing S-1 are also widely used in Asian countries for patients with pancreatic cancer as subsequent therapy after gemcitabine-based chemotherapy. 12 The choice to use these regimens is usually based on the status of the patient and experience of the physicians. As second-line therapies, the differences in the effects of these regimens remains unclear.
This current systematic review and meta-analysis analysed previously published data regarding the treatment effects of different second-line chemotherapy regimens after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
Materials and methods
Search strategy
This meta-analysis followed the PRISMA 2020 guidelines. 13 Electronic databases, including Embase®, Medline, PubMed® and the Cochrane library, were searched from inception to October 2021. The search terms included “pancreatic cancer”, “folfirinox”, “gemcitabine” and “second-line chemotherapy”. Two authors (W.L. & K.T.) performed the electronic search independently in October 2021. Abstracts from the published articles were reviewed to determine their suitability for inclusion in the pooled analysis. Any discordance regarding study inclusion between the two authors were settled in discussion with a third independent author (L.W.). The quality of evidence provided by each study was evaluated using the Oxford Centre for Evidence-based Medicine scoring system. 14 In error, this meta-analysis was not registered so it was retrospectively registered at Research Registry: reviewregistry1300.
Eligibility criteria
Publications were included in this meta-analysis if they meet the following criteria: (i) patients with locally advanced and metastatic pancreatic cancer that failed on gemcitabine-based chemotherapy; (ii) second-line chemotherapy included FOLFIRINOX and other 5-FU-based chemotherapy or S-1; (iii) reported data included progression-free survival (PFS), overall survival (OS), objective response rate (ORR), disease control rate (DCR) and grade 3/4 treatment-related adverse events. Publications were excluded if they met any of the following criteria: (i) studies published in a language other than English; (ii) case reports or cohort studies including less than seven patients; (iii) patients that received FOLFIRINOX as the first-line chemotherapy; (iv) survival outcome data were unavailable. In the situation in which authors from the same institution had published a primary paper and then an updated analysis with a larger patient cohort, the most recent publication was included in the analysis.
Outcome measures for meta-analysis of comparative studies
The primary outcome measure evaluated was the hazard ratio (HR) for PFS, OS, ORR and DCR. Other information extracted from each study included author names, country, publication year, number of patients and severe adverse events. This analysis was mainly focused on the most commonly reported grade 3/4 treatment-related adverse events, including neutropenia, anaemia, thrombocytopenia, diarrhoea and nausea. Any discrepancies in study eligibility or data extraction were reconciled by a joint revaluation of the original article.
Statistical analyses
Two independent reviewers (W.L. & L.W.) extracted data from the selected articles by using a predefined data extraction form. To estimate HR and its variance, this was extracted from the study directly or required additional calculation depending on the method of data being presented: annual mortality rates, survival curves, number of deaths or percentage freedom from death. 15 For each study, the odds ratio (OR) was estimated by a method dependent upon the data provided. The simplest method consisted of the direct collection of ORs with 95% confidence interval (CI) described in the original study.
Meta-analysis of the data was conducted using a random-effects model by Stata 12.0 (StataCorp, College Station, TX, USA). Inter-study heterogeneity was assessed using the χ2 statistic and the I2 value to measure the degree of variation not attributable to chance alone. This was graded as low (I2 < 25%), moderate (I2 25% to 75%) or high (I2 > 75%). Publication bias was assessed using Egger’s and Begg’s tests. The significance level was set at P < 0.05.
Results
A flow chart showing the study selection process is presented in Figure 1. A total of 102 articles were identified by the literature search. After reviewing the titles and abstracts, 90 articles were not eligible as they were review articles, editorials, nonhuman studies or non-English articles, those not focusing on the research topic or others that did not meet the inclusion criteria. A total of 12 articles were identified as potentially eligible for this meta-analysis. However, two of these articles were case reports and four studies did not have comparative data for FOLFIRINOX. A total of six studies were eligible for inclusion. These included four retrospective trials, one prospective trial and one randomized controlled trial.16 –21

Flow diagram of eligible studies showing the number of citations identified, retrieved and included in the final meta-analysis of the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
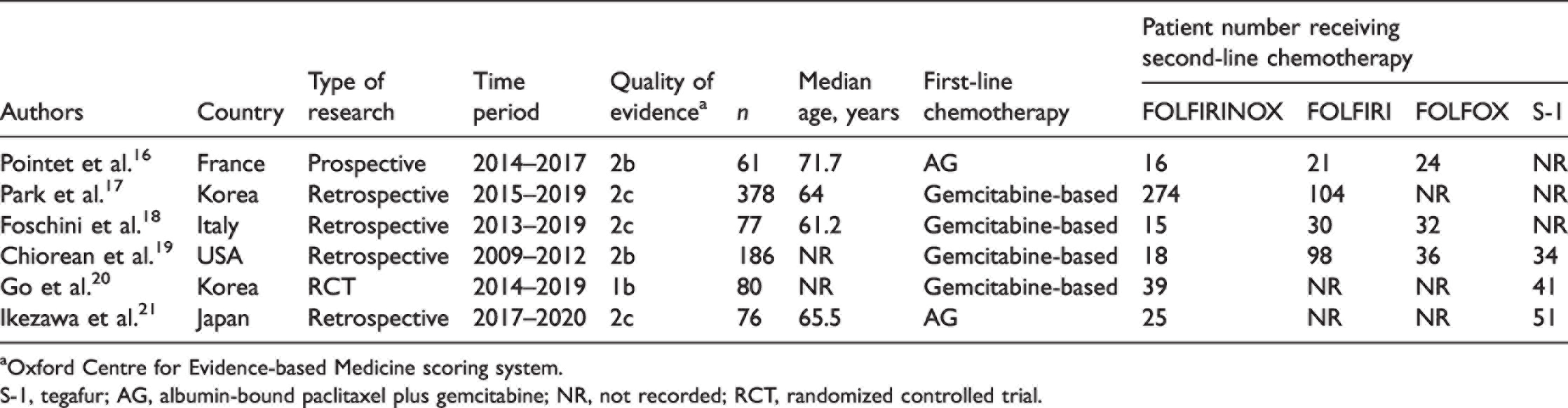
This meta-analysis included six studies that evaluated the value of FOLFIRINOX as a second-line chemotherapy for pancreatic cancer patients that failed gemcitabine-based chemotherapy.16 –21 The main characteristics of these six studies are presented in Table 1. The total number of patients considered in this meta-analysis was 858 with a median age of approximately 66 years. All of the studies measured the survival benefit of treatment with FOLFIRINOX as second-line chemotherapy compared with other second-line regimens. Two of the studies were from Korea and the other four were from France, Italy, USA and Japan. None of the studies included was from the same institution. The PFS, OS, ORR and DCR data for FOLFIRINOX and the other second-line regimens in the six studies are presented in Tables 2 and 3.
aOxford Centre for Evidence-based Medicine scoring system.
S-1, tegafur; AG, albumin-bound paclitaxel plus gemcitabine; NR, not recorded; RCT, randomized controlled trial.
Progression-free survival (PFS) and overall survival (OS) data from the six studies included in a meta-analysis to evaluate the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.16 –21

Data presented as hazard ratio 95% confidence interval).
S-1, tegafur; NR, not recorded.
Objective response rate (ORR) and disease control rate (DCR) data from the six studies included in a meta-analysis to evaluate the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.16 –21

Data presented as hazard ratio.
S-1, tegafur; NR, not recorded.
Table 4 presents the grade 3/4 treatment-emergent adverse events (TRAEs) from the second-line chemotherapy regimens used in the six studies included in this meta-analysis. These included neutropenia, anaemia, thrombocytopenia, diarrhoea and nausea. The most commonly reported grade 3/4 TRAEs for FOLFIRINOX were neutropenia (42.3%), anaemia (12.5%), nausea (8.1%), thrombocytopenia (4.3%) and diarrhoea (1.4%). A comparison between FOLFIRINOX and the other second-line regimens was undertaken. FOLFIRINOX was demonstrated to have a higher rate of grade 3/4 TRAEs compared with FOLFIRI, FOLFOX and S-1.
Treatment-emergent adverse events of ≥grade 3 from the six studies included in a meta-analysis to evaluate the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
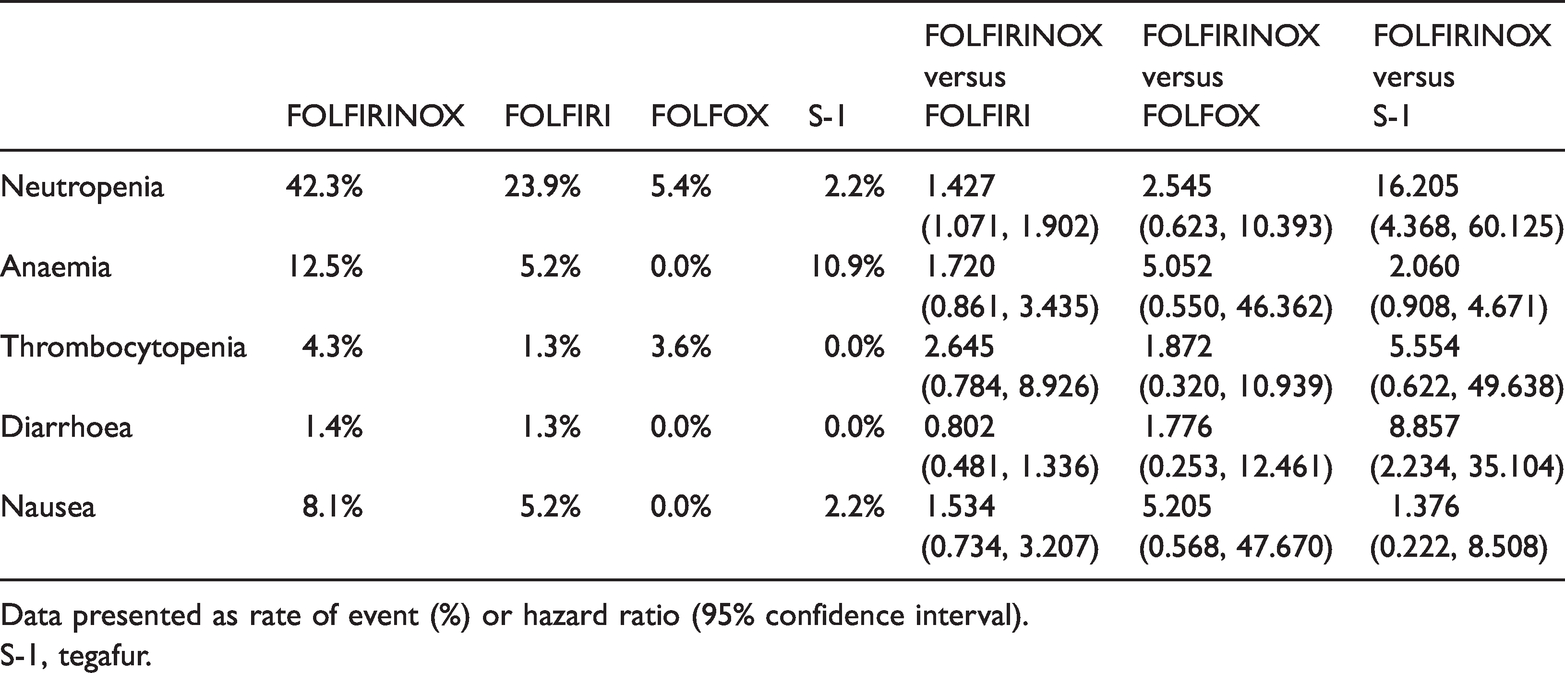
Data presented as rate of event (%) or hazard ratio (95% confidence interval).
S-1, tegafur.
The PFS in this analysis was defined as the time from the start date of gemcitabine-based chemotherapy to the date of first progression during second-line treatment or death for any reason. Compared with other second-line regimens, FOLFIRINOX had a significantly longer PFS (HR 0.68, 95% CI 0.52, 0.89) (Figure 2). A subgroup analysis demonstrated the following: (i) compared with FOLFIRI, FOLFIRINOX had a longer PFS, but the result was not significant (HR 0.78, 95% CI 0.40, 1.43); (ii) compared with FOLFOX, FOLFIRINOX had a longer PFS, but result was not significant (HR 0.73, 95% CI 0.48, 1.12); (iii) compared with S-1, FOLFIRINOX had a significantly longer PFS (HR 0.60, 95% CI 0.36, 0.98).

Forest plot of a meta-analysis (random-effects model) to evaluate progression-free survival from the six studies that evaluated the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
The OS in this analysis was defined as the time form the start date of the second-line regimen to the date of death for any reason. Patients alive were censured at the last follow-up date. Compared with the other second-line regimens, FOLFIRINOX had a significantly longer OS (HR 0.71, 95% CI 0.59, 0.86) (Figure 3). A subgroup analysis demonstrated the following: (i) compared with FOLFIRI, FOLFIRINOX had a longer OS, but result was not significant (HR 0.78, 95% CI 0.57, 1.07); (ii) compared with FOLFOX, FOLFIRINOX had a longer OS, but the result was not significant (HR 0.76, 95% CI 0.49, 1.18); (iii) compared with S-1, FOLFIRINOX had a significantly longer OS (HR 0.55, 95% CI 0.38, 0.81).

Forest plot of a meta-analysis (random-effects model) to evaluate overall survival from the six studies that evaluated the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
The ORR was calculated as the sum of all complete and partial responses. Compared with the other second-line regimens, FOLFIRINOX had a significantly higher ORR (HR 0.43, 95% CI 0.23, 0.80) (Figure 4). A subgroup analysis demonstrated the following: (i) compared with FOLFIRI, FOLFIRINOX had a higher ORR, but result was not significant (HR 0.61, 95% CI 0.27, 1.38); (ii) compared with FOLFOX, FOLFIRINOX had a significantly higher ORR (HR 0.38, 95% CI 0.16, 0.92); (iii) compared with S-1, FOLFIRINOX had a significantly higher ORR (HR 0.10, 95% CI 0.02, 0.55).

Forest plot of a meta-analysis (random-effects model) to evaluate objective response rate from the six studies that evaluated the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
The DCR was defined as the sum of all complete and partial responses and stable disease. Compared with other the second-line regimens, FOLFIRINOX had a significantly higher DCR (HR 0.71, 95% CI 0.58, 0.88) (Figure 5). A subgroup analysis demonstrated the following: (i) compared with FOLFIRI, FOLFIRINOX had a higher DCR, but result was not significant (HR 0.71, 95% CI 0.42, 1.22); (ii) compared with FOLFOX, FOLFIRINOX had a higher DCR, but result was not significant (HR 0.75, 95% CI: 0.52, 1.09); (iii) compared with S-1, FOLFIRINOX had a significantly higher DCR (HR 0.67, 95% CI 0.47, 0.97).

Forest plot of a meta-analysis (random-effects model) to evaluate disease control rate from the six studies that evaluated the treatment effects of different second-line chemotherapy regimens compared with FOLFIRINOX after failure of gemcitabine-based first-line therapy in patients with pancreatic cancer.
The Begg’s funnel plot (Figure 6) indicated the absence of publication bias.

Begg’s funnel plot to determine the publication bias of the studies included in this meta-analysis.
Discussion
Chemotherapy is the most commonly used treatment for advanced pancreatic cancer that cannot be radically resected. Gemcitabine used to be the most frequently used drug for adjuvant and palliative chemotherapy for pancreatic cancer. 22 New regimens such as FOLFIRINOX and AG have shown stronger effects in pancreatic cancer patients. 23 Yet the single use of gemcitabine and combined regimens containing gemcitabine are still applied in quite a high proportion of advanced pancreatic cancer patients.24,25 Patients that accept gemcitabine-based chemotherapy as first-line therapy usually receive single drug or combined regimens containing 5-FU, capecitabine or S-1 after disease progression. FOLFIRINOX is the most popular regimen if the patient’s status is good enough, but the differences in the effects and safety of these various regimens remains controversial.9,10
This current meta-analysis analysed six studies that enrolled 858 patients to evaluate the effects and safety of FOLFIRINOX compared with FOLFIRI, FOLFOX and S-1 as second-line regimens after failure of gemcitabine-based chemotherapy in advanced pancreatic cancer. Compared with patients treated with the other three regimens, patients treated with FOLFIRINOX had a significantly higher PFS, OS, ORR and DCR. For each subgroup analysis, FOLFIRINOX had a higher PFS, OS, ORR and DCR than the FOLFIRI and FOLFOX groups, but only the difference in the ORR between the FOLFIRINOX and FOLFOX groups was significant. Compared with S-1, FOLFIRINOX provided a significantly higher PFS, OS, ORR and DCR. This result showed that FOLFIRINOX was more effective as a second-line regimen after failure of gemcitabine-based first-line chemotherapy compared with FOLFIRI, FOLFOX and S-1. For those patients that failed gemcitabine-based therapy, these current findings suggest that FOLFIRINOX should be the first consideration as a second-line regimen.
This current meta-analysis also demonstrated that FOLFIRINOX treatment was associated with more severe TRAEs. In particular, more patients had severe neutropenia and anaemia after being administered this highly toxic regimen. Therefore, FOFIRINOX can only be used in patients after careful evaluation of their status. These TRAEs should be closely monitored and treated during the whole therapeutic process.
In conclusion, FOLFIRINOX is a good choice as a second-line chemotherapy regimen for patients with pancreatic cancer that have failed in gemcitabine-based chemotherapy as it demonstrated a longer PFS and OS and a better ORR and DCR. Although more severe TRAEs were associated with second-line FOLFIRINOX treatment, patients with careful evaluation and monitoring can get benefits from this therapy.
Footnotes
Author contributions
Wenjie Lu undertook the clinical investigation, data curation and wrote the manuscript. Lantian Wang undertook the formal analyses and wrote the manuscript. Xiawei Li undertook the data curation and wrote, reviewed and edited the manuscript. Kezhong Tang provided study conceptualization, funding acquisition and methodological guidance.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Zhejiang Province (no. LQ19H160021).