Abstract
Purpose:
Radiotherapy-induced oral mucositis is the most common side effect in nasopharyngeal carcinoma (NPC) patients. We aimed to evaluate the efficacy and safety of Rabdosia rubescens drop pills in NPC patients with radiation-induced oral mucositis (RTOM).
Methods:
The study involved 40 NPC patients who were given Rabdosia rubescens drop pills thrice daily from the start of radiation therapy. The study monitored the incidence and severity of oral mucositis and oral pain. The main outcomes measured were the occurrence rate of oral mucositis, grade 3 oral mucositis, oral pain assessment, and changes in immunological function, body weight, BMI, NRS2002, and albumin levels.
Results:
In the study, 38 patients completed the treatment. The incidence rates of Grade 0 to 3 oral mucositis were 5.26%, 21.05%, 47.37%, and 26.32% respectively. Pain levels were mild (42.11%), moderate (13.16%), and severe (13.16%). The onset of Grade 1, 2, and 3 oral mucositis occurred at 18, 24, and 30 days respectively. Grade 3 oral mucositis was associated with body weight, BMI, NRS2002 score, and albumin levels. Post-treatment, there was a decrease in CD4+/CD8+, CD3+, and CD4+ immune cells, but an increase in CD8+ cells. Mild to moderate gastrointestinal adverse events were observed in 13.2% of patients.
Conclusion:
Rabdosia rubescens drop pills administration can reduce the incidence and severity of radiotherapy induced oral mucositis. Our finding suggested a positive impact of Rabdosia rubescens drops pills upon administration to NPC patients.
Introduction
Nasopharyngeal carcinoma (NPC), also known as lymphoepithelioma, develops from the nasopharyngeal epithelium and is the most frequent malignant tumor of the nasopharynx.1,2 It is a relatively rare malignancy worldwide, but its geographical distribution is dramatically variable.3,4 Approximately two-thirds of worldwide cases are diagnosed in the east and south-east Asia, and the remaining one-third are diagnosed in other parts of Asia and some parts of Africa. NPC is largely endemic to China, particularly Southern China, with incidence rates ranging from 15 to 50 per 100 000.5,6 A complex etiology consists of genetic and environmental factors, and Epstein-Barr virus (EBV) infection is responsible for the disease. 7 The common therapeutic options for NPC are chemotherapy and radiotherapy.8-10 Oral mucositis is among the most common complication in patients with NPC. Oral mucositis not only impairs the quality of life of the patient but also interrupts radiotherapy or chemoradiotherapy in approximately 20% of the patients.11,12 Acute mucositis caused by radiation is a common complication of radiotherapy for NPC and manifested in the form of mucosal hyperemia, erosions, and ulcers.13,14
Severe radiation-induced mucositis is often accompanied by significant pain, discomfort, and impaired eating and swallowing ability. Additional nutrition support and analgesic treatment are required to relieve the symptoms. Severe radiation-induced mucositis may even lead to reduced or unplanned interruption of radiotherapy.15,16 Therefore, finding safe, efficient, and cost-effective medications to prevent and treat radiation-induced mucositis is of major therapeutic importance. Rabdosia rubescens drop pills are prepared from Rabdosia rubescens using patented technology. These drop pills are clinically used to treat sore throat, hoarseness, and other symptoms caused by heat-toxin congestion and excessiveness due to the anti-tumor, antibacterial, anti-inflammatory, and immunoregulatory effects of the pills.17,18 In this study, we aim to assess the efficacy and safety of Rabdosia rubescens drop pills for the prevention and treatment of acute mucositis caused by radioation for NPC patients on a scientific basis. The patients were administered Rabdosia rubescens drop pills and assessed for reduction of mucositis grade, adverse events, and effects on indices, such as nutrition and immunity were monitored to evaluate the quality of life and physical condition from multiple perspectives.
Materials and Methods
Study Subjects
This was a prospective, single-centered and single-armed design, phase II clinical trial performed on NPC patients in the Sichuan Cancer Hospital in Chengdu, China between January and November 2018. A total of 40 NPC patients were enrolled in this trial. All the patients were informed about the study procedures and purpose and they provided signed informed consent before enrollment (Ethics Committee Approval Number: SCCHEC-02-2017-051 and Ethics Committee Approval Date 2017-12-28).
The criteria for inclusion in the study were as follows: (1) The participant must have a histopathologically confirmed diagnosis of squamous carcinoma with no evidence of distant metastasis; (2) The clinical stage of the participant must be between I and IVB, as per the UICC 8th system staging; (3) The participant’s age must be between 18 and 70 years; (4) The participant must have undergone radical chemoradiotherapy; (5) The participant must have a Karnofsky score of 70 or above; (6) The participant must have an expected survival time of at least 6 months.
Patients with any of the following conditions were excluded from the study: (1) active oral infectious diseases, such as oropharyngeal candidiasis, herpes facialis; (2) elderly patients with xerostomatitis; (3) individuals with recurrent or residual NPC; (4) patients who used 5-fluorouracil as neoadjuvant or concurrent chemotherapy; (5) patients who received concurrent molecular targeted therapy; (6) patients with any known or suspected allergy to the drug; (7) patients with severe blood, kidney, nervous system, heart or liver disease or mental illness; (8) patients who took part in any previous clinical trials involving other drugs within 3 months of this investigation; (9) other patients whom researchers deemed unsuitable to participate in the study.
Study Design
Rabdosia rubescens drop pills were produced by Henan Bainian Kangxin Pharmaceutical Co., Ltd. (Approval No.: Z20150003, 270 pills/bottle, lot number: YBZ00202015; shelf-life: 24 months). Study participants were instructed to rinse their mouth with water before each dose and then slowly swallow a total of 24 Rabdosia rubescens drop pills. The administration schedule was three times per day following meals. The course of treatment was started from the first day of radiotherapy and continued until either the radiotherapy course was completed or the patient was diagnosed with oral mucositis of grade 3. Administration of antibiotics and hormones to patients diagnosed with oral mucositis of grades 0 to 2 was prohibited.
Chemotherapy and Radiotherapy
In this study, all patients were treated with concurrent chemoradiotherapy using an intensity-modulated radiation therapy technique (IMRT). IMRT is among the most important recent developments in oncology which precisely conform radiations to the target site sparing normal tissues from unnecessary radiation exposure. The radiation is provided by a standard radiotherapy instrument called LINAC (linear accelerator) equipped with a freely movable multi-leaf collimator made of lead (Pb) thin leaves. The presence of lead leaves assures the targeting of diseased tissue and spares normal healthy areas. The radiation dose is adjusted according to the tumor size and organ involved. 19 The total radiotherapy dose used in this study was 66 to 74 Gy (2.1-2.3 Gy per section) for gross tumor volume (GTV), 60 to 70 Gy (2.0-2.2 Gy per section) for GTV in lymph node (GTVln), 60 to 66 Gy (1.8-2.0 Gy per session) for clinical target volume-1 (CTV-1), 54 to 60 Gy (1.8-2.0 Gy per session) for clinical target volume-2 (CTV-2), and 50 Gy (1.8-2.0 Gy per session) for neck prevention. The limited dose for organ at risk (ORA) was set according to the requirements of RTOG0225. The specific radiotherapy technique and prescribed dose were adopted from the relevant studies of this center. The chemotherapy regimen was cisplatin (100 mg/m2) and the patients received three cycles of concurrent chemotherapy. Treatment of patients with 5-fluorouracil was prohibited in this study.
Study outcomes assessments
The key outcome measures of this study were the incidence rate of oral mucositis, oral pain assessment, change in immunological function, body weight, BMI, NRS2002, and albumin concentration. Throughout the treatment period, patients were assessed for inflammation of the oral mucosa, including the retropharyngeal mucosa, and any oral pain in these patients was scored daily from 8:00 AM to 10:00 AM according to the Common Terminology Criteria for Adverse Events (CTCAE) v3.0 criteria and Numerical Rating Scale (NRS) standard. The grades of oral mucositis and oral pain were recorded.
Other observations included: (1) Nutritional status assessment within 24h of admission and once a week during chemoradiotherapy. Body mass index and nutrition risk screening 2002 (NRS2002) were recorded. (2) Immune function tests, including peripheral blood T cell subsets (CD3+, CD4+, CD8+, CD4+/CD8+), NK cells and C-reactive protein (CRP) were performed every 2 weeks during chemoradiotherapy. The range of CD3+, CD4+, CD8+, and CD4+⁄CD8+ ratios in patients were assessed through flow cytometry (Epics XL-MCL; Beckman Coulter). Briefly, whole blood (50 μL) was mixed with monoclonal antibody (each T lymphocytes reagent tube containing 5 μL) and incubated in a dark place for 15 minutes at room temperature. The erythrocytes were lysed using a fluorescence-activated cell lysing solution containing 15% formaldehyde and 50% diethylene glycol (Becton Dickinson). The tubes were vortexed and further incubated for 15 minutes in the dark at ambient temperature. Cells were injected into a “flow cell” analyzed, and counted using a fluorescence-activated cell sorting counter (Becton Dickinson cytometer). The change in cells’ counts was plotted for each patient against time (in weeks). The primary endpoints for the efficacy analysis included the incidence of oral mucositis and change in grade over the course of treatment. Secondary endpoints included changes in body weight and oral pain grade during radiotherapy.
Statistical Analysis
Statistical analyses were performed using the R language package (version 3.5.0) and demographic characteristics were analyzed for the full analysis set (FAS), and efficacy index and adverse events were analyzed for the Per Protocol Set (PPS). Generalized estimating equations (GEEs) were used to detect changes in immune biomarkers and indices over time. Paired t-test was used to compare differences in Weight, BMI, NRS2002, ALB, CRP, CD3+ T cells, CD4+ T cells, CD8+ T cells, NK cells, and CD4+/CD8+ T cells of the RTOM Grade 0 to 2 group and Grade 3 group patients. For all statistical analyses, the statistically significant level was set at .05 (P < .05).
Results
Basic Characteristics
In total, 40 patients were enrolled in our study, and two patients withdrew from the study (one patient took 5-fluorouracil during treatment, and the other discontinued treatment due to an epileptic seizure). The data of all 40 patients were used for the Full Analysis Set (FAS) and the Per Protocol Set (PPS) analysis was applied to the 38 subjects who completed the study. The demographic data of all the patients are listed in Table 1. The mean weight of patients was 63.06 ± 13.39kg before radiotherapy and 59.1 ± 12.40kg after treatment, with a mean weight loss of about 3.96 kg. According to the NRS2002 score, 17.5% of patients were malnourished before radiotherapy and 37.5% after radiotherapy with an increase of 20% (P < .0001).
Basic Characteristics of Patients.

Abbreviation: KPS, Karnofsky performance status.
Patients experienced the most serious malnutrition in the 6th week after radiotherapy, with the NRS2002 score of 4.7 ± 1.87. During concurrent chemoradiotherapy, BMI showed a significant decrease from 23.26 ± 3.94 before radiotherapy to 20.71 ± 3.68 after radiotherapy (P < .0001); however, there was no significant difference in albumin concentration during radiotherapy (P = .19) (Table 2). Dynamic weight changes, BMI, NRS 2002, and Albumin (ALB) during the trial are shown in Figure 1.
Dynamic Changes in the Nutritional Index During the Study.

NRS2002: nutrition risk screening 2002.
Abbreviations: BMI, body mass index; ALB, albumin.
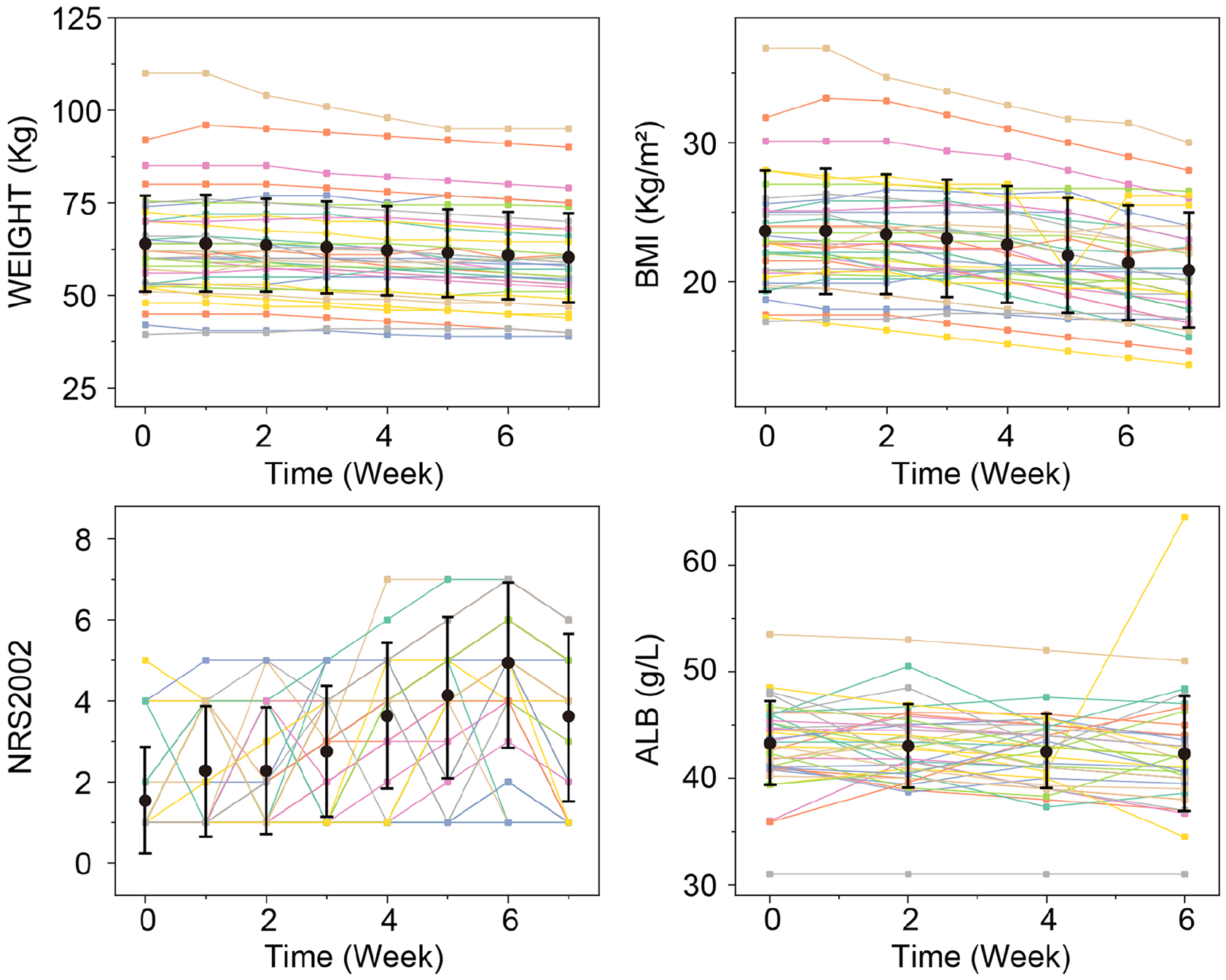
Dynamic changes of weight, BMI, NRS 2002 and Albumin (ALB) during the study.
The immune function of NPC patients treated with concurrent chemoradiotherapy at different times (in weeks) was dynamically observed, in which CD3+ and CD4+ immune cells decreased, while CD8+ immune cells increased. Thus, the ratio of CD4+/CD8+ decreased significantly (P < .0001). The function of non-specific immune NK cells was significantly enhanced, with a mean value of 21.22 ± 10.54 before radiotherapy and 26.39 ± 13.34 after radiotherapy (P = .0064) (Table 3). Dynamic changes in immune functions during the study are shown in Figure 2.
Dynamic Changes in Immune Functions During the Study.

Abbreviation: CRP, C-reactive protein.

Dynamic changes in immune functions during the study.
Efficacy
The time to the development of grade 1, 2, or 3 oral mucositis was 18.06 ± 7.67, 24.22 ± 8.48, and 30.3 ± 9.04 days, respectively counted from the start of chemoradiotherapy. At the end of the study, the incidence of grade 0, 1, 2, or 3 oral mucositis was 5.26% (2/38), 21.05% (8/38), 47.37% (18/38), and 26.32% (10/38), respectively. It is reported in the literature that the incidence of Grade 3 RTOM in NPC patients under routine care is 53.70% at the completion of radiation. Rabdosia rubescens drop pills effectively reduced the incidence of grade 3 radiation-induced oral mucositis (Table 4). At the completion of the study, a total of 68.42% of patients had oral pain, including 42.11% mild, 13.16% moderate, and 13.16% severe pain patients (Table 5). Previous results from our hospital indicated that gargling with compound borax (10 mL) resulted in a 75% incidence of severe oral pain.
Incidence of Oral Mucositis During the Study.

Incidence of Oral Pain During the Study.

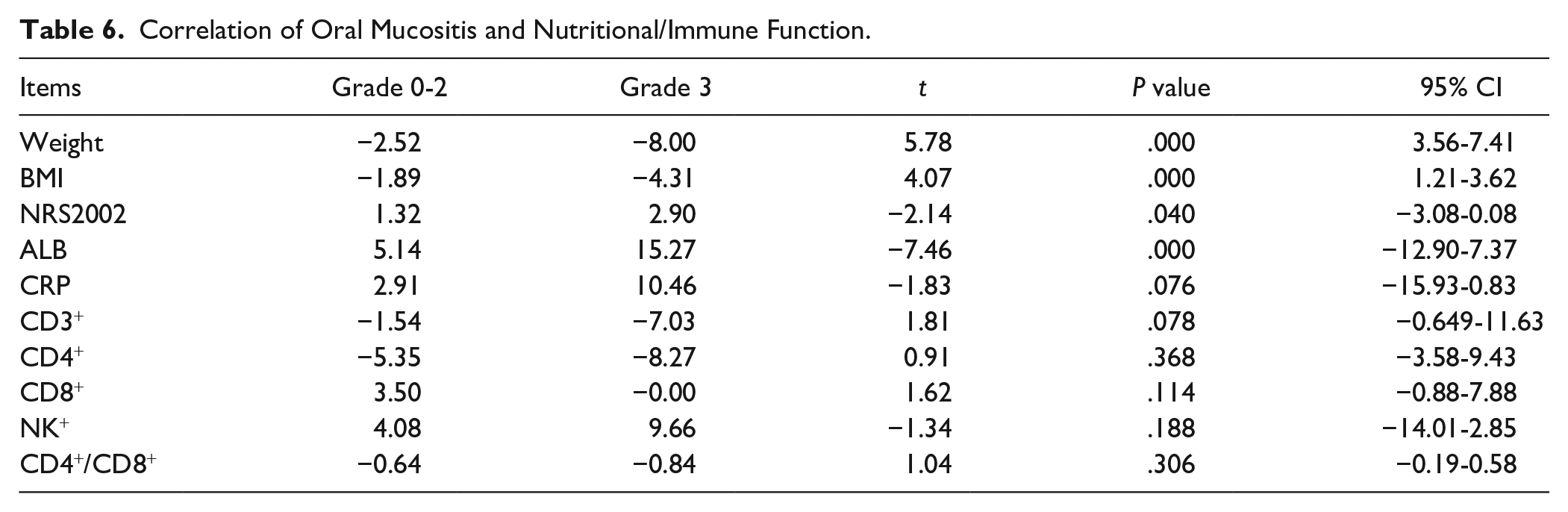
To assess a potential relationship between RTOM and nutritional status, patients were separated into subgroups according to the severity of RTOM grade 0 to 2 group and grade 3 group. There were 28 (73.7%) patients in the grade 0 to 2 group, and 10 (26.3%) patients in the grade 3 group. Table 6 shows that Grade 3 oral mucositis was strongly associated with body weight, BMI, NRS2002 score, and albumin levels (P < .05). CRP and NK cells were increased, while CD3+ T cells, CD4+ T cells, and CD4+/CD8+ T cells levels were decreased in the two subgroups. There were no significant differences in CRP, NK cells, or CD3+ T cells, CD4+ T cells, CD8+ T cells and CD4+/CD8+ T cells between the two subgroups (P > .05) (Table 6).
Correlation of Oral Mucositis and Nutritional/Immune Function.
Safety evaluation
The enrolled patients experienced grade 1 to 2 adverse reactions, including nausea and vomiting (73.68% [28/38]), leucopenia (60.53% [23/38]), abdominal distention (10.53% [4/38]), hyperlipidemia (10.53% [4/38]), venous thrombosis (7.89% [3/38]), constipation (5.26% [2/38]), increased alanine aminotransferase concentration (5.26% [2/38]), fever (5.26% [2/38]), and anemia (5.26% [2/38]). Grade 3 adverse reactions included leukopenia (21.05% [8/38]) and nausea and vomiting (2.63% [1/38]). No Grade 4 adverse reactions were observed in this study. There were five adverse events related to the study drug, including abdominal distention (10.5% [4/38]) and diarrhea (2.6% [1/38]), and other adverse reactions were all related to chemotherapy drugs (Table 7).
Incidence of Adverse Events in 38 Patients.

Abbreviation: ALT, alanine aminotransferase.
Discussion
Radiation-induced oral mucositis (RTOM) refers to inflammatory or ulcerative lesions of the oral mucosa caused by ionizing radiation and can be divided into acute and chronic RTOM. Acute RTOM usually occurs after the radiotherapy dose reaches 20 Gy. The incidence of oral mucositis caused by chemoradiotherapy in patients with head and neck tumors is more than 85%, and more than 40% of the patients experience Grade 3 to 4 RTOM.20,21 Severe oral mucositis could lead to dose reduction and/or unplanned interruption of the radiotherapy, patient weight loss, and poor quality of life. As a result, additional analgesics and parental nutrition support therapies are required to ameliorate the patients’ symptoms. Furthermore, it also leads to an increased risk of serious infections and prolonged hospital stay of the patients, thus adversely affecting the long-term treatment efficacy.22-24 The cytotoxic effects of radiation directly damage mucosal epithelial cells. Reactive oxygen species produced by radiation trigger an oxidative stress response in the body, destroying cells, tissues, and blood vessels, and activating NF-κB to promote the release of inflammatory factors, thereby exacerbating tissue damage and causing ulcers and bacterial and fungal infections.25,26 While there are several agents such as amifostine, benzydamine, allopurinol, or erythropoietin mouthwash mentioned in the literature that have been explored for their potential in preventing radiation-induced oral mucositis, it is important to note that no method or specific drug has demonstrated high efficacy in preventing this condition in patients. Current clinical treatment primarily focuses on alleviating symptoms, promoting healing, and preventing/treating concurrent infections. Despite ongoing research and the identification of various agents, the search for a highly effective preventive medication for radiation-induced mucositis remains a challenge.27,28
Oridonin is the most abundant active constituent in Rabdosia rubescens and has strong biological activity.29,30 The antibacterial activity of oridonin is associated with changes in cell wall permeability, DNA metabolism, and overall morphological integrity in bacteria. 31 Oridonin is also able to induce apoptosis in HEp-2 cells at least in part through mechanisms related to altered autophagic activity and ROS production as a result of caspase-9 targeting. 32 Clinical studies have shown that Rabdosia rubescens preparation can effectively reduce oral ulcer area, pain level, congestion, and other symptoms in patients. 33 At present, most of the Rabdosia rubescens-related experimental studies are basic research. The clinical effectiveness and safety of Rabdosia rubescens drop pills for the therapy of radiation-induced mucositis have not been previously reported. In this study, we assessed its efficacy and safety in patients undergoing radiochemotherapy for nasopharyngeal carcinoma treatment. At the completion of radiation, the incidence of grade 3 RTOM in patients taking Rabdosia rubescens drop pills was 26.32%. A total of 68.42% of patients had oral pain, including 55.27% mild-to-moderate pain, and the mucosal injury occurred at 18.06 ± 7.67 days. It is reported in the literature that the incidence of Grade 3 RTOM in NPC patients under routine care is 53.70% at the completion of radiation. The initial onset of mucosal injury was observed at 14.5 ± 6.5 days, and the incidence of moderate or severe oral pain in patients with routine care is 74.0%. 34 Oropharyngeal and esophageal mucosal injury and oral pain may lead to the inability to eat normally and even increase the psychological burden in patients, while Rabdosia rubescens drop pills can effectively control the severity of the disease, delay the occurrence of radiation-induced mucositis, and reduce oral pain.
The incidence of malnutrition among head and neck cancer patients who receive radiotherapy ranges from 44% to 88%, according to the most recent study on the nutritional condition of such patients. 35 According to published reports, 56% of NPC patients experience around 5% weight loss following radiotherapy. 36 In a study, patients with head and neck cancer suffered considerable weight loss 4 weeks following the initiation of radiotherapy. 37 The major causes of weight loss and malnutrition in NPC patients are oral mucositis, oligoptyalism, difficulty in opening the mouth, and gastrointestinal reactions.38,39 This study showed that 37.5% of patients were malnourished at the end of radiation, with a mean weight loss of about 3.96 kg. The patients experienced the most severe malnutrition in the sixth week of radiotherapy compared to the previously published 4 weeks. Weight loss is inevitable in NPC patients during chemoradiotherapy; however, the severity and timing of weight loss in patients have been improved compared with previous data, which may be closely related to the control of oral pain and RTOM upon administration of Rabdosia rubescens drop pills with chemoradiotherapy. At the same time, it is of positive significance to take necessary nutritional interventions to keep patients in good physical condition for successful completion of treatment.
B lymphocytes, T lymphocytes, and NK cells are human immune cells that contribute to anti-tumor immune responses. 40 CD4+ and CD8+ mature T cells exist in the body, of which CD4+ cells mainly mediate cellular immune responses and promote the secretion of various inflammatory cytokines, thus playing a role in killing tumor cells, 41 while CD8+ cells are mainly involved in humoral immune response removing tumor cells through the biological activity of immunoglobulins. 42 This study found that the levels of CD8+ cells increased considerably during chemoradiotherapy coupled with Rabdosia rubescens drop pills administration. On the other hand, CD3+, CD4+, and CD4+/CD8+ cells gradually decreased, suggesting the involvement of immunosuppressive factors that can inhibit CD4+ cell maturation and differentiation in the tumor cell microenvironment. In addition, the proportion of CD8+ cells increased while the proportion of CD4+/CD8+ cells decreased, allowing the human body to enter an immunosuppressive state, thus causing immune dysfunction. Although T lymphocytes were inhibited, the proportion of NK cells was increased, indicating the immune response of NK cells was enhanced, and abnormal cellular immune function in tumor patients could be related to the activation of the proliferative ability of NK cells. 43 This initial assessment of the Rabdosia rubescens drops pills for the prevention of oral mucositis in NPC patients undergoing radiotherapy suggests positive results.
Limitations
This study has several important limitations that should be considered when interpreting the results. Firstly, this was a single-centered and single-armed study design. One of the primary limitations of a single-arm study is the absence of randomization, which may introduce selection bias. In the absence of randomization, patient characteristics and outcomes could be influenced by factors other than the intervention itself. Additionally, findings from a single-arm study may have limited generalizability to broader populations or settings. The lack of a control group makes it challenging to assess the true effectiveness of the intervention across diverse patient groups. For more reliable and generalizable results, future studies should employ parallel-group designs with placebo-controlled comparisons. The relatively small sample size of the current study is another potential limitation. A sufficiently large sample would be needed to verify the robustness of the results. Larger, multi-center trials with adequate statistical power would help confirm the findings and provide more definitive conclusions.
Conclusions
According to the findings of this prospective single-arm phase II clinical trial, the use of Rabdosia rubescens drop pills can decrease the occurrence and severity of oral mucositis in NPC patients undergoing radiotherapy. Furthermore, it has the potential to reduce the incidence of malnutrition by maintaining oral mucositis caused by radiotherapy at a grade lower than 3. To further investigate the efficacy of Rabdosia rubescens drop pills, longer follow-up times and higher sample sizes are required in future randomized controlled trials.
Footnotes
Acknowledgements
We are grateful to all the participants in the study, and the healthcare professionals from Sichuan Cancer Hospital in Chengdu.
Authors’ Contributions
Lu Li was responsible for the project administration. Yecai Huang contributed to the writing of the original draft and the methodology. Peng Xu was in charge of data curation. Mei Lan contributed to the methodology, investigation, and data curation. Churong Li was involved in the methodology. Yunxiang Qi contributed to the writing of the original draft and the methodology. Ke Xu was involved in the methodology and data curation. Bosen Li contributed to the methodology, investigation, and data curation. Yukun Luo was involved in the methodology and investigation. Qinghua Jiang contributed to the writing of the original draft. Shanshan Peng and Jinyi Lang were responsible for data analysis. Mei Feng supervised the writing, analyses, and revision of the manuscript. All authors provided feedback on previous versions of the manuscript and read and approved the final manuscript.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Sichuan Province Science and Technology Program (No. 2021JDRC0146); Sichuan Province Science and Technology Program (No. 2022YFS0047); Oncology Medicine and Engineering Innovation Fund (No. ZYGX2021YGCX006); Sichuan Province Science and Technology Program (No. 2020YFS0424); and Sichuan Medical Association Project (No. 20210221020001).
Ethics Approval and Consent to Participate
Subjects (or their guardians) have given their written informed consent. Ethical approval for the study was obtained from the ethics committee of Sichuan Cancer Hospital in Chengdu (Ethics Committee Approval Number: SCCHEC-02-2017-051 and Ethics Committee Approval Date 2017-12-28). All methods were performed in accordance with the relevant guidelines and regulations.
