Abstract
Introduction
Mucositis is characterized by the damage to the epithelium of the oropharyngeal cavity and gastrointestinal tract. Chemotherapy- and radiotherapy-induced oral mucositis represents a therapeutic challenge frequently encountered in cancer patients. 1 It occurs in almost all patients with radiotherapy for head and neck cancers. 2 This debilitating side effect causes significant morbidity and may delay the cancer treatment plan as well as increase therapeutic expenses. 3 Although there are many traditional forms of treatment such as mouthwashes and local anesthetics, most are ineffective. Antimicrobials have not demonstrated consistent efficacy in the prevention or treatment of oral mucositis. 4 Many agents such as topical sucralfate, 5 topical granulocyte macrophage colony stimulating factors, 6 prostaglandin E analogue misoprostol,7,8 topical corticosteroids, 9 and parenteral radioprotector amifostine 10 have been tried with varying response rates. Botanical products are currently being investigated in the management of functional disorders of the gastrointestinal tract. Iberogast, an extract of multiple herbal drugs, partially improves the histopathological features of 5-fluorocil (5-FU)-induced mucositis in rats.11,12 Grape seed extract improves parameters of small intestinal mucositis in rats and may enhance chemotherapy effectiveness.13,14 The nutritional approach may be the means to raise cancer therapy to a new level as supplementing or supporting the body with natural phytochemicals, which can not only reduce adverse side effects but also improve the effectiveness of chemotherapeutics. 15 To date, no single efficient intervention or agent for the prophylaxis or management of mucositis has been identified. The current approach is to seek an agent that combines multiple strategies for the prevention and treatment of oral mucositis. Date palm pollen (DPP) was selected as an ideal mucosal protector in this pilot study. It is easier to identify and characterize as a botanical treatment, and its chemical constituents have been well documented. Moreover, it can be formulated in different delivery systems.
Date palm pollen is the male reproductive cell of the date palm (
The DPP used in this study was identified using morphological information obtained from light and scanning electron microscopy. Each individual cell has unique size, shape, and surface characteristics (Figure 1). DPP contains a variety of bioactive components that are important for the vitality and function of the cell, such as estrone, estradiol, and estriol. 18 Estrogens are known to protect and preserve vascular structures as well as enhance cell regeneration. 18 Reported studies suggest that estrogens improve the supply of nutrients and oxygen to mucosa. 19 DPP also contains rutin and carotinoids, compounds that have powerful antioxidant effects. Antioxidants are known to prevent DNA damage. 20 DPP contains natural lipids, cholesterol, and fatty acids such as palmitic acid, linoleic acid, and myristic acid. These lipids mimic the lipids found naturally in epithelial layers of the oral mucosa. 21 In addition, DPP contains triterpenoids, 22 compounds that are known to possess anti-inflammatory and antibacterial actions. 23

SEM image of DPP.
The objective of this pilot study was to evaluate whether DPP would be effective in the prevention and treatment of oral mucositis induced by radiation and chemotherapy in patients with head and neck cancers.
Materials and Methods
The study was conducted in accordance with International Conference on Harmonization (ICH) Guidelines for Good Clinical Practice (GCP), the Declaration of Helsinki. Prior to initiation of the study, approval was obtained from the facility medical research committee, institutional review board, and by the department of oncology. This study enrolled 20 subjects recently diagnosed with varying head and neck cancers, prior to exposure to first-line treatment. Subject ECOG (Eastern Cooperative Oncology Group) performance status 24 level ranged from 0 to 1. The first 10 subjects enrolled were treated with DPP administered orally daily at bedtime, as a swish and swallow suspension. The next 10 subjects in the control group received the facility’s standard of care for the treatment of oral mucositis, which consists of an oral antifungal (20 mg/g miconazole oral gel), rebamipide (an amino acid derivative of 2-(1H)-quinolinone) for mucosal restoration, and local and oral analgesics as needed. Objective oral assessments were made at baseline and every 2 weeks during the 30-day observation period. The treatment with DPP was given for 6 weeks during the required radiation and/or chemotherapy regimen. The sample size of the study was not determined by statistical power considerations, but is considered adequate for proof of concept. Table 1 summarizes the subject demographics, disease characteristics, and disease treatment plan within the treatment group.
Subject Characteristics Within DPP-Treated Group and Control Group.

Abbreviation: DPP, date palm pollen.
Chemotherapy treatment of cisplatin 100 mg/m2 every 3 weeks for 3 cycles. Radiation treatment of 3D conformal radiotherapy 50 Gy to 72 Gy in 200 cGy conventional fractionation.
Pure DPP for oral administration was supplied in powder form to be mixed in water by the subject prior to administration in following proportions: 2 g of the powder formulation added to 125 mL of water and taken as a swish and swallow. Treatment was self-administered, nightly for 42 days starting 1 day prior to initiation of chemotherapy or radiation treatment. ClinAvenir LLC, Los Altos, California, supplied the DPP clinical samples. No other treatments for oral mucositis were allowed during the treatment in the course of the study for those subjects in the DPP treatment group. Subjects in the control arm were allowed standard of care treatment for oral mucositis. A complete oral examination using the Oral Mucositis Assessment Scale (OMAS), 25 including digital photographs of the oral cavity, were completed at days 0, 15, and 29 of the study. In addition, the clinical consequences of treatment on mouth pain and impact on swallowing were monitored by subjective visual analog scale (VAS) completed by the subjects. The OMAS is an easily used and reproducible tool specifically designed for use in clinical trials that measures objective and subjective indicators of oral mucositis including ulceration/pseudomembrane formation (0-3 in each area), and erythema (0-2 in each area) in 9 areas of the oral cavity and subjective outcomes of mouth pain, ability to swallow, and function. 25 In this study, total scores are added with the final score ranging from 0 to 45. Subjects completed VAS consisting of 100-mm line to measure oral pain with descriptors of severity (no pain, most severe pain) and ability to swallow (no trouble in swallowing, cannot swallow anything at all).
Results and Discussion
This pilot study shows encouraging results in the prevention of symptomatic radiation- and chemotherapy-induced oral mucositis. No unexpected adverse side effects were reported by either treatment group. There were no issues of treatment compliance observed in the DPP-treated group. Table 2 shows that DPP administration before treatment almost prevented mucositis. The difference between the mean OMAS in the DPP-treated group and the control group was statistically significant during the evaluation period. The difference between the means was analyzed using the
Mean OMAS: DPP-Treated Group Versus Control Group.

Abbreviations: OMAS, Oral Mucositis Assessment Scale; DPP, date palm pollen.
Confidence level 95%.
Mean Oral Pain Level: DPP-Treated Group Versus Control Group.
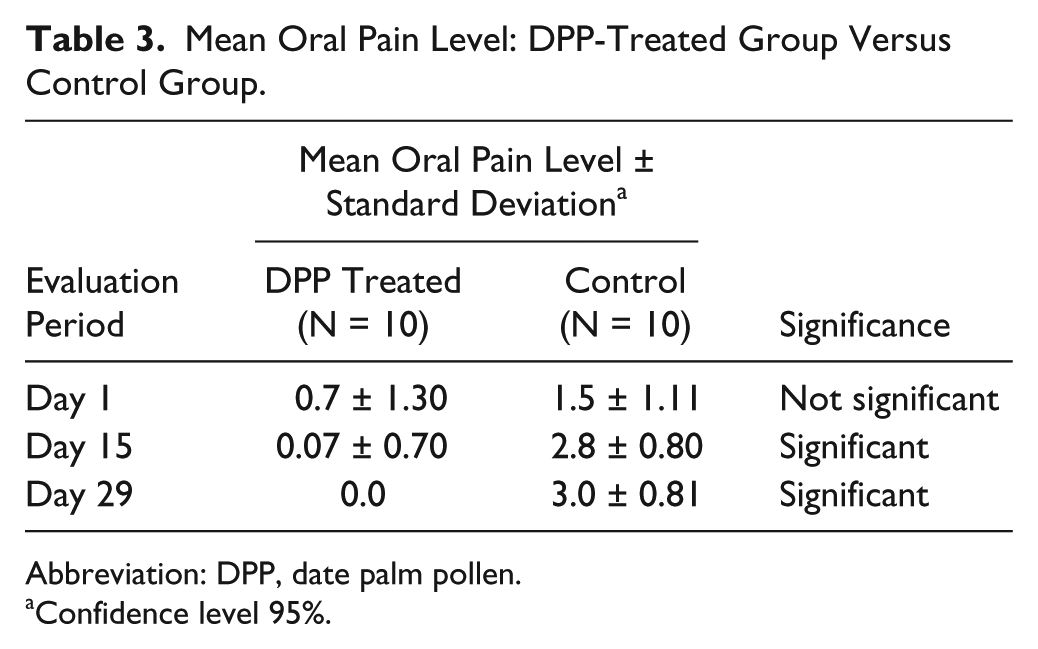
Abbreviation: DPP, date palm pollen.
Confidence level 95%.
Mean Impact on Swallowing: DPP-Treated Group Versus Control Group.

Abbreviation: DPP, date palm pollen.
Confidence level 95%.

Erythema and ulcers in chemotherapy- and radiation-induced oral mucositis observed at day 15 in the control group and not seen in the DPP-treated group: (A) Control subject no. 1; (B) DPP treatment subject no. 2.
The results obtained suggest that there is a significant reduction in severity and incidence of mucositis during treatment with DPP. The exact mechanism of action is unknown. Various plant derived agents such as genistein, curcumin, epigallocatechin, gallate, resveratrol, indole-3 carbinol, and proanthocyanidin have shown to be able to affect the efficacy of traditional chemotherapeutic agents.26,27 The resulting benefits of the intake of DPP may not be due only to its antioxidant constituents. As a nutritional supplement, DPP contains natural lipids, which act as moisturizing agents; estrone and estradiol, which improve microcirculation; rutin, which modulates and combats DNA damage; and triterpenoids, which act as anti-inflammatory and antibacterial agents. 23 The complex synergy between these phytochemicals in this natural microcapsule of the DPP cell may have been responsible for the observed protection. It is unlikely that the same effect would occur if these constituents were administered individually.
Conclusion
This pilot study proves that the male reproductive cell of the date palm is effective in the protection of mucous membranes from chemotherapy- and radiotherapy-induced mucositis. Protection from mucosal injury could be due to the synergistic actions of the phytochemical constituents of DPP. DPP is a nutritional supplement that holds promise in the therapeutic intervention for oral mucositis complicating chemotherapy and/or radiotherapy. Other gastrointestinal functional disorders such as irritable bowel syndrome and ulcerative colitis could benefit from DPP administration.
These results on their own are not definitive, but the promising findings should stimulate further research to clarify the potential benefits of DPP bioactive phytochemicals in the prevention and treatment of chemotherapy- and radiation-induced oral mucositis. Larger placebo controlled randomized trials are needed.
Footnotes
Acknowledgements
Thanks are due to Amr Shaaban MD, Resident at the Department of Clinical Oncology - Medical Research Institute, Alexandria University and Mohamed Ebeid B.Pharm for their technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A grant was received from ClinAvenir LLC, Los Altos, California, to cover costs for materials and supplies for this investigation.
