Abstract
In this study, we investigated the synergistic effect of co-administration of osimertinib and HAD-B1 using gefitinib-resistant non-small cell lung cancer cells, HCC827-GR. HAD-B1 is composed of 4 natural drugs, Panax Notoginseng Radix, Panax ginseng C. A. Meyer, Cordyceps militaris, and Boswellia carterii Birdwood, and has been reported to have therapeutic effects on patients with advanced non-small cell lung cancer in several studies. Resistance to gefitinib in HCC827 cells was acquired through MET activity. Co-treatment with osimertinib and HAD-B1 reduced the cell viability of HCC827-GR cells. In addition, phosphorylation of MET and ERK were effectively suppressed for HCC827-GR cells. And, compared to when osimertinib and HAD-B1 were administered alone, cell proliferation was significantly inhibited and apoptosis was effectively induced when osimertinib and HAD-B1 were co-administered to HCC827-GR cells. We found that the synergistic effect of osimertinib and HAD-B1 combination therapy resulted in cancer cell death and cell cycle arrest by targeting the ERK and mTOR signaling pathways. In conclusion, this study confirmed that the combination of osimertinib, a third-generation anticancer drug, and HAD-B1, a natural anticancer drug, had a potentially synergistic effect on non-small cell lung cancer resistant to EGFR-targeted anticancer drugs.
Introduction
Lung cancer is the most frequent cancer worldwide and is also responsible for the highest worldwide cancer-related deaths. 1 Because of the lack of early detection, most lung cancers are discovered in the late stages of distant metastasis and local tumor invasion, with 5 year survival rates ranging from 4% to 17%, depending on stage and region. 2 Lung cancer is histologically classified into 2 major categories: non-small cell lung carcinoma (NSCLC), accounting for nearly 85% of all lung cancer, and small cell lung carcinoma (SCLC), which accounts for the remaining 15%. 3 Adenocarcinoma is among the most common NSCLC histological subtypes. 4 Epidermal growth factor receptor (EGFR) is a protein that plays a key role in regulating cell growth and differentiation. It receives signals from outside the cell and activates several biochemical pathways inside the cell, a process that mobilizes various signaling systems such as the PI3K-AKT-mTOR and RAS-RAF-MEK-ERK pathways. 5 The mTOR is a kinase, which constitutes 2 distinct complexes, mTORC1 and mTORC2, regulating several physiological processes. By phosphorylating S6K1, mTORC1 promotes protein translation and cell growth. And mTORC2 activates the AKT pathway to control metabolism, survival, and cell mobility. 6 NSCLC of adenocarcinoma histology is reportedly associated with the EGFR oncogene mutations in approximately 40% to 48% of Asian patients and 14% to 19% of Western patients. 7 The most common mutation associated with these cancers is the exon 21 change of arginine to leucine at amino acid 858 (L858R) mutation or exon 19 deletion (ex19del). 8 With the advancement of targeted therapies, EGFR tyrosine kinase inhibitors (EGFR-TKIs) have become a standard treatment for patients with EGFR-mutant advanced NSCLC. 9
Studies have indicated that EGFR-TKIs significantly increase overall survival (OS) and progression-free survival (PFS). 10 Several phase III clinical trials have consistently demonstrated first-generation (gefitinib, erlotinib, and icotinib) and second-generation (afatinib) EGFR-TKIs. EGFR-TKIs are more effective for treating advanced NSCLC with EGFR mutations. Ex19del and L858R are the most responsive mutations observed in NSCLC patients, indicating a favorable response to treatment with gefitinib and afatinib.11 -14 Despite the vigorous clinical action exerted by EGFR-TKIs, most patients develop resistance after a mean of 9 to 15 months. Acquired genetic variations in EGFR have been linked to EGFR TKI resistance. The mutation EGFR T790M in exon 20 is the governing resistance mechanism in approximately 50% of patients treated with first- and second-generation TKIs.15,16 To overcome T790M resistance, a third-generation EGFR-TKI (osimertinib) is being developed. Third-generation TKI shows superior clinical effectiveness in patients with EGFR-positive and EGFR T790M mutations; however, drug resistance is unavoidable, and tumor heterogeneity determines resistance diversity.17,18 Acquired resistance has become a clinical problem. Various resistance mechanisms have been described, including MET amplification, EGFR C797S mutations, and small-cell transformation. 19 As a result, adjuvant drugs may be used to overcome resistance in combination with EGFR-TKIs.
HangAmDan-B1 (HAD-B1) is a traditional Korean prescription consisting of 4 natural products: Panax Notoginseng Radix, Panax ginseng C. A. Meyer, Cordyceps militaris, and Boswellia carterii Birdwood.20,21 Previous preclinical studies discovered that HAD-B1 has an anti-cancer property by inhibiting cell proliferation in lung cancer xenograft animal trials using A549 cells as well as an effect of inducing apoptosis and halting the cell cycle in A549 cells with cisplatin resistance. 22 In this study, we analyzed the synergistic effect of HAD-B1 and osimertinib on gefitinib-resistant HCC827-GR, NSCLC cell lines. 23
Materials and Methods
Preparation of Osimertinib and HAD-B1 Extract
Osimertinib was obtained from LC Laboratories (Woburn, MA, USA). HAD-B1 is a mixed herbal extract contains Panax notoginseng radix and Panax ginseng C. A. Meyer, Cordyceps militaris, and Boswellia carterii birdwood (Table 1). It was produced by Daehan Biopharm (Gyeonggi, Korea) and supplied by EWCC (Daejeon Korean Medicine Hospital, Daejeon University, Korea). The HAD-B1 stock was prepared by extracting HAD-B1 powder with 1 mg of powder in 10 mL of DW at room temperature. The extract was then centrifuged at 1000 × g for 30 minutes and filtered and applied to the C18 column and eluted using acetonitrile mixed with DW. Supplemental Figure 1 shows the results of HPLC of HAD-B1 fractions. 24
Ingredients of HangAmDan-B1.

One 645 mg tablet of HAD-B1 contains 1.270 mg ginsenoside Rb1, 1.110 mg notoginsenoside R1, 0.610 mg ginsenoside Rg1, 0.058 mg β-boswellic acid, 0.320 mg cordycepin, and 0.031 mg α-boswellic acid.
Cell Culture
HCC827-GR cell lines was donated from Ildong Pharmaceutical, Korea and the cells were cultured in Roswell Park Memorial Institute 1640 (RPMI 1640; Welgene, Inc. Daejeon, Korea) containing 10% fetal bovine serum (FBS, Welgene Inc.). Cells were maintained at 37°C in a humidified atmosphere containing 5% CO2. RRID number of the cell line is CVCL_S703.
Cell Viability Assay
Cell viability was determined using a CCK-8 assay. HCC827-GR cells (3000 cells/100 μL/well) were seeded into 96-well plates and incubated at 37°C in a CO2 incubator. Osimertinib and HAD-B1 were diluted in RPMI medium, and 100 μL was added to each well. Some cells were treated with HAD-B1 plus osimertinib. The cells were then incubated at 37°C in a CO2 incubator for 72 hours. The CCK-8 reagent (10 μL; Dongin Biotech Co., Ltd. Seoul, Republic of Korea) was added to each well, after which the wells were incubated at 37°C in a CO2 incubator for 2 hours. The absorbance of the solution was measured at a wavelength of 450 nm using a microplate reader (Spectramax ID3, Molecular Device, USA).
Western Blot Analysis
HCC827-GR cells were incubated with osimertinib (0.1 μM) and/or HAD-B1 (1 mg/mL) for 12 hours at 37°C. Some cells were treated with HAD-B1 plus osimertinib. The cells were harvested and lysed on ice in a lysis buffer containing phosphatase inhibitor (1X) and protease inhibitor (1X) for 30 minutes. Lysates containing 15 μg of protein were loaded into each well of a 4% to 20% gradient gel (Mini-PROREAN® TGX Gels; Bio-Rad Pacific Ltd.) and separated by gel electrophoresis. The gels were soaked in transfer buffer (30 mM glycine, 16 mM Tris-HCl, and 20% methanol), and the proteins in the gels were transferred to polyvinylidene difluoride membranes (Bio-Rad). Nonspecific binding sites were blocked using nonfat dry milk in phosphate-buffered saline with Tween (PBST; 137 mM NaCl, 27 mM KCl, 100 mM Na2HPO4, 20 mM KH2PO4, 0.05% Tween 20, pH 7.4) at room temperature. The polyvinylidene difluoride (PVDF) membranes were then incubated with different primary antibodies against pEGFR (1:1000), EGFR (1:1000), p-MET (1:1000), MET (1:1000), p-ERK1/2 (1:1000), ERK1/2 (1:1000), p-S6 (1:3000), S6 (1:3000), and GAPDH (1:5000) diluted in 1% BSA in TBST at 4 Secondary antibodies against pEGFR (anti-rabbit at 1:3000), EGFR (anti-rabbit at 1:3000), p-MET (anti-rabbit at 1:3000), p-ERK1/2 (anti-rabbit at 1:3000), ERK1/2 (anti-rabbit at 1:3000), p-S6, S6 (anti-rabbit at 1:3000), MET (anti-mouse at 1:3000), and GAPDH (anti-rabbit at 1:10 000) were applied to the membranes. An enhanced chemiluminescence western blotting detection kit was used to detect the protein bands on the membranes. The signals were developed using X-ray films. The intensities of the protein bands were quantified using ImageJ software and normalized to the intensity of GAPDH band.
RRID number of the cell line is CVCL_S703. And the catalog numbers for antibodies and kits are as follows: Anti-pERK1/2 (9101S, cell signaling technology, MA, USA), Anti-ERK1/2 (9102S, cell signaling technology, MA, USA), Anti-pS6 (2211S, cell signaling technology, MA, USA), Anti-S6(2217S, cell signaling technology, MA, USA), Anti-GAPDH (2118S, cell signaling technology, MA, USA), Muse Annexin V, Dead cell kit (MCH100105, Luminex, USA).
Apoptosis Assay
The HCC827-GR cell apoptosis assay was performed using the Muse™ Annexin V & Dead Cell Kit (Luminex, MCH100105). HCC827-GR cells were treated with osimertinib or HAD-B1 for 24 or 48 hours, respectively. Some cells were treated with HAD-B1 plus osimertinib. Total cells from each group were harvested, and the cell pellets were suspended in RPMI1640 medium containing 10% FBS mixed with Muse Annexin V and Dead Cell reagent. After 20 minutes of incubation at room temperature in the dark, optical density was measured using a Muse Cell Analyzer (Merck Millipore, Bedford, MA, USA).
Cell Cycle Analysis
The HCC827-GR cell cycle was analyzed using a Muse Cell Cycle Kit (Merck Millipore). HCC827-GR cells were treated with osimertinib and/or HAD-B1 for 24 and 48hours, respectively. Some cells were treated with HAD-B1 plus osimertinib. Total cells were harvested, and cell pellets were suspended in RPMI1640 media containing 10% FBS. Ethanol (70%) was cooled on ice and then added to the cells. After 3hours incubation at −20°C, the cells were washed with ice-cold PBS and treated with 200 μL of Muse Cell Cycle reagent (Millipore Corp., Bedford, MA, USA). After 30 minutes of incubation at room temperature in the dark, the cell suspension was analyzed using a Muse Cell Analyzer.
Statistical Analysis
All data are represented as the mean ± standard deviation, and statistical comparisons were performed using Student’s t-test. Statistical analyses were performed using Microsoft Excel® Office 365 (Microsoft Corporation, Redmond, WA, USA) and Graphpad Prism v8. Statistical significance was set at P < .05. difference. Values represent mean ± standard error of mean.
Results
A Combination of HAD-B1 and Osimertinib Decreased the Cell Viability of HCC827-GR
Based on previous studies demonstrating the anti-tumor effect of HAD-B1, we examined the effects of a combination of osimertinib and HAD-B1 on the growth of HCC827-GR cells. 20 A CCK-8 cell viability assay was carried out to confirm the synergistic, additive, or antagonistic effects of osimertinib and HAD-B1 using the non-constant ratio drug combination method proposed by Chou and Talalay.25,26 The half-maximal inhibition of HCC827-GR cell viability in the monotherapy groups, osimertinib or HAD-B1 alone, was 0.6830 μM or 0.6697 mg/mL, respectively (Figure 1A). The combination index (CI) value based on the cell viability analysis data showed that the combined treatment had a synergistic effect with a CI value of less than 1 only at high concentrations of HAD-B1 at both 0.1 and 0.5 μM osimertinib concentrations (Figure 1B, Table 2).

Growth inhibitory effects of HangAmDan-B1 (HAD-B1) and osimertinib on HCC827-GR cell lines. HCC827-GR cells were treated with increasing concentrations of (A) osimertinib and (B) HAD-B1 for 72 hours. (B) Combined treatment with osimertinib (0.1 or 0.5 μM) and HAD-B1. Data shown are the mean ± SDs, and statistical analyses were performed using Student’s t-test.
Combination Indexa (CI) Analysis of HangAmDan-B1 (HAD-B1) Combined With Osimertinib at a Non-Constant Ratio in HCC827-GR Cells.

CI is widely used to quantify drug synergism based on the Chou-Talalay multiple-drug effect equation.26,27 CI values were determined for each concentration of osimertinib, HAD-B1, and their combination in cell proliferation assays using CompuSyn (ComboSyn, Inc. Paramus, NJ). CI < 1 indicates synergism, CI = 1.0 designates an additive effect, and CI > 1.0 designates antagonism. 26
Inhibition of MET Amplification and EGFR Phosphorylation by the Combined Treatment of HAD-B1 and Osimertinib
HCC827-GR cells showed enhanced EGFR phosphorylation and enhanced expression in MET. 28 Immunoblot analysis was performed to assess whether combined treatment of HAD-B1 with osimertinib showed inhibition of MET amplification and phosphorylation. Treatment with osimertinib alone decreased pEGFR and increased EGFR expression but did not affect the phosphorylation of ERK or MET amplification compared with empty vehicle-treated control cells. Treatment with osimertinib and HAD-B1 synergistically inhibits MET phosphorylation, amplification, and ERK phosphorylation. In addition, combined treatment with osimertinib and HAD-B1 reduced the phosphorylation of ERK1/2 and S6 in the mTOR pathway (Figure 2). These data indicate that the inhibitory effect of HAD-B1 on HCC827-GR cell proliferation was due to the inhibition of the ERK and mTOR signaling pathways, resulting from the reduction of both S6 phosphorylation and MET amplification.
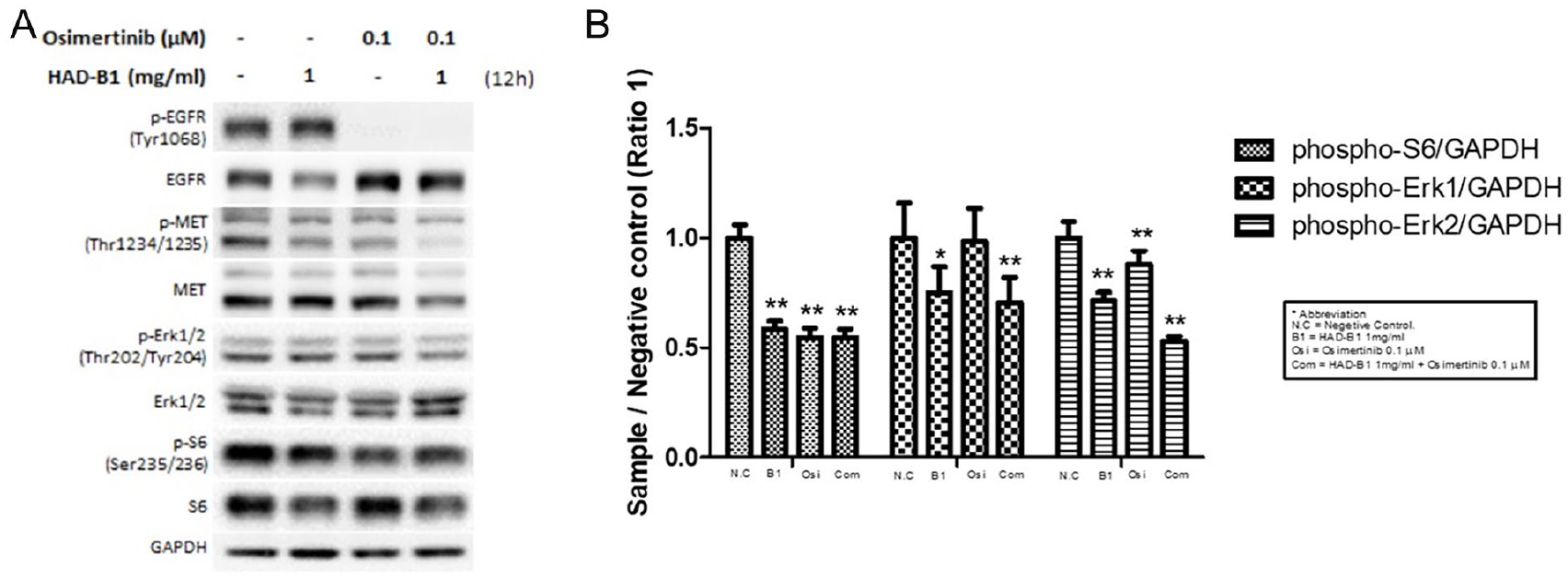
Inhibitory effects of HangAmDan-B1 (HAD-B1) and osimertinib on MET and ERK signaling. (A) Western blot analysis of cell lysates from HCC827-GR cells treated with HAD-B1 and osimertinib. HCC827-GR cells were treated with osimertinib (0.1 μM) or HAD-B1 at the indicated concentrations for 12 hours. (B) Densitometric analysis of the expression level of phospho-ERK1/2 and phospho-S6. **P < .01 compared to negative control cells. Statistical analysis was performed with Graphpad Prism v8.
Induction of Apoptosis in HCC827-GR Cell by HAD-B1 and Osimertinib
We demonstrated that HAD-B1 regulated the cell cycle and induced apoptosis in H1975 cells, an EGFR-L858R/T790M double mutation NSCLC cells. 29 To confirm whether the inhibitory effect of HAD-B1 and osimertinib on tumor cell proliferation was due to cell apoptosis, we carried out a cell apoptosis assay using Muse Annexin V and Dead Cell Kit. HCC827-GR cells were treated with HAD-B1 (1.0 mg/mL) or osimertinib (0.1 or 0.5 μM). And cells were also treated with a combination of the two. Live and apoptotic cells were analyzed using the Muse system after treatment with HAD-B1 or osimertinib. The early apoptotic cells were significantly increased by 55.60% and 43.90% in the cells treated with HAD-B1 (1.0 mg/mL) and osimertinib (0.1 or 0.5 μM) compared to the cells treated with osimertinib or HAD-B1 alone (Figure 3, Table 3). The total apoptosis was significantly higher in the HAD-B1- and osimertinib-treated groups than in the vehicle-treated control group.

Effects of HangAmDan-B1 (HAD-B1) and osimertinib on apoptosis. HCC827-GR cells were treated with osimertinib and HAD-B1 for 48 hours. Apoptosis was assessed using annexin staining and a Mini Flow Cytometry Muse Cell Analyzer. Shown is a representative data set of three independent experiments: (A) FACS analysis of HCC-827GR cells treated with HAD-B1 and osimertinib; (B) Apoptosis analysis of the cells treated with HAD-B1 and osimertinib.
Effects of HangAmDan-B1 (HAD-B1) and Osimertinib on Apoptosis.

P < .05, **P < .01, ****P < .0001 versus negative control cells.
Cell Cycle Arrest in HCC827-GR Cells Treated With HAD-B1 and Osimertinib
To confirm that the induction of apoptosis in HCC827 cells treated with HAD-B1 and osimertinib was caused by cell cycle arrest, cell cycle analysis was performed using a Muse Cell Analyzer (Millipore Corp., Bedford, MA, USA). When HCC827-GR cells were treated with HAD-B1 (1.0 mg/m L) or osimertinib (0.1 or 0.5 μM), a significant increase in the G0/G1 phase was observed in cells treated with the combination of HAD-B1 and osimertinib in comparison with the HAD-B1- or osimertinib-treated groups (Figure 4). The combined treatment with HAD-B1 and osimertinib resulted in 54.4% and 53.3% of G0/G1 phase cells, respectively, compared with osimertinib-treated cells (42.2% and 44.6%) or HAD-B1-treated cells (37.6%) at 48 hours (Figure 4). Consequently, these findings demonstrate that the combined treatment of HAD-B1 with osimertinib increased the accumulation of cells in the G0/G1 phase in HCC827-GR cells, followed by stimulation of cell apoptosis.

Effect of HangAmDan-B1 (HAD-B1) and osimertinib on cell cycle. The HCC827-GR cells were treated with osimertinib and/or HAD-B1 for 48 hours. Cell cycle distribution was analyzed using a Muse Cell Cycle Kit (Merck Millipore, Billerica, MA, USA). Shown is a representative data set of three independent experiments.
The Effect of Active Ingredients of HAD-B1 on the Cell Proliferation of HCC827-GR Cells
To further determine the antiproliferative effect of HAD-B1 on HCC827-GR cells, we examined whether the active constituents of HAD-B1 inhibited cell proliferation and intracellular signaling. Based on the cell viability assay data, ginsenoside Rb1, notoginsenoside R1, ginsenoside Rg1, β-boswellic acid, cordycepin, and α-boswellic acid suppressed the growth of the HCC-GR cells in a dose-dependent manner. The half-maximal inhibition of HCC827-GR cell viability was observed at 193 μM for α-boswellic acid, 66.05 μM for β-boswellic acid, and 86.27 μM for cordycepin (Figure 5). These data demonstrated that the inhibitory effect of HAD-B1 combined with osimertinib on the growth of HCC827 cells was consistent with a synergistic effect of the active components of HAD-B1 and osimertinib.

Inhibitory effect of HangAmDan-B1 (HAD-B1) on the proliferation of HCC827-GR cells. The HAD-B1 components were added to the cells at different concentrations for 72 hours.
HAD-B1 Active Ingredients Inhibit MET Amplification and Phosphorylation
Immunoblot analysis was performed to assess whether HAD-B1 components inhibited MET amplification. Beta-boswellic acid inhibited MET phosphorylation at the concentration of 50 μM. Notoginsenoside R1 also reduced S6 phosphorylation (Figure 6A and B). Cordycepin also inhibited the phosphorylation of MET and S6 (Figure 6C and D). These data indicate that the inhibitory effect of HAD-B1 on HCC827-GR cell proliferation was due to reduced MET and S6 phosphorylation by the different active components of HAD-B1. A schematic representation of the anti-cancer mechanism of HAD-B1 is shown in Figure 7.

Inhibitory effect of HangAmDan-B1 (HAD-B1) components on MET signaling. Western blot analysis of cell lysates from HCC827-GR cells treated with HAD-B1 components. HCC827-GR cells were treated with HAD-B1 at the indicated doses for 12 hours: (A) Western blot analysis of the cells treated with Notoginsenoside R1, a-boswellic acid, or b-boswellic acid, respectively; (B) Phosphorylation pattern of MET and S6 in the cells treated with Notoginsenoside R1, a-boswellic acid, or b-boswellic acid, respectively; (C) Western blot analysis of the cells treated with Cordysepin, Ginsenoside Rb1, or Ginsenoside Rg1, respectively; (D) Phosphorylation pattern of MET and S6 in the cells treated with Cordysepin, Ginsenoside Rb1, or Ginsenoside Rg1, respectively.

Molecular mechanisms underlying the actions of HangAmDan-B1 (HAD-B1) and osimertinib. Osimertinib is an inhibitor of receptor tyrosine kinases, including EGFR and HER2. The inhibitory effect of HAD-B1 on HCC827-GR cell proliferation occurred through the reduction of MET phosphorylation and amplification. The synergistic effect of HAD-B1 and osimertinib inhibit the ERK and mTOR signaling pathways, leading to cell cycle arrest and apoptosis in cancer cells.
Discussion
Osimertinib is a third-generation EGFR-TKI that selectively blocks an activated EGFR mutation with T790M resistance. 30 Patients treated with osimertinib have a high response rate and a median PFS of about 10 months.17,31 Despite the high objective response rate and improved survival outcomes in patients with EGFR mutations treated with EGFR-TKIs, complete responses are rare, and resistance typically develops after an average of 8 to 18 months of treatment. 10 Resistance mechanisms such as MET amplification, EGFR C797S mutations, and small cell transformation have all been reported. 19 The acquired EGFR C797S mutation has been confirmed as an osimertinib-resistant mechanism in about 40% in one study. 32 However, the remaining TKI resistance mechanisms are largely unknown. 33 Therefore, to overcome the limitations of osimertinib treatment, additional drugs capable of overcoming EGFR-TKI resistance must be developed.
HAD-B1 is an anti-cancer drug developed to treat lung cancer. It includes 4 natural products that inhibit proliferation of lung cancer cells. A previous study showed that HAD-B1 inhibited the growth of solid tumors in an A549 cell xenograft mouse model compared to vehicle or cisplatin-treated controls. 20 The 3-dimensional high-performance liquid chromatography (HPLC) analysis of HAD-B1 revealed the presence of 6 important compounds, including R1, Rb1, Rg1, cordycepin, α-boswellic acid, and β-boswellic acid. 34
HAD-B1 combined with osimertinib treatment inhibited cell proliferation more effectively than osimertinib-alone treatment (Figure 1). Furthermore, the combination of osimertinib and HAD-B1 effectively induced apoptosis and induced cell cycle arrest. Combined treatment of HAD-B1 with osimertinib increased the accumulation of cells in the G0/G1 phase in HCC827-GR cells, followed by the stimulation of cell apoptosis (Figures 3 and 4). HAD-B1 induces apoptosis in cancer cells by affecting the cell cycle signaling system.
ERK1/2 is a serine/threonine protein kinase belong to the mitogen-activated protein kinase (MAPK) family. ERK1/2 phosphorylation inhibits apoptosis and controls cell proliferation, migration, and gene expression. In contrast, ERK1/2 inhibition promotes apoptosis. MTOR phosphorylates S6K, and S6K phosphorylates S6 to increase cell growth, gene transcription, and cell proliferation in the mTOR pathway. Since HCC827-GR cells expressed more MET than parental HCC827 cells,28,35 phosphorylation of ERK and S6 through the MET signaling pathway persisted, even when EGFR activation was inhibited by osimertinib treatment. However, when MET was simultaneously inhibited by HAD-B1 treatment, phosphorylation of ERK and S6 was reduced (Figures 2 and 7).
HAD-B1 has been shown to have potential antiproliferative effects in NSCLC cells, regardless of EGFR-TKI resistance. In a previous study, treatment of HAD-B1 on cisplatin-resistant A549 lung cancer cells showed anti-cancer effects, and a molecular mechanism study showed that apoptosis was controlled by promoting Bax genes and inhibiting STAT3, as in A549 cells. HAD-B1 has also indicated anti-cancer effects on H1975 EGFR-T790M/L858R double mutation lung cancer cells with solid tumor growth and molecular mechanism. 36 In addition, HAD-B1 induces apoptosis of cancer cells by targeting the cell cycle of H1975 lung cancer cells. 29 Also, in our previous study, HAD-B1 administration in an acute toxicity study in SD rats caused neither adverse effects nor mortality in clinical signs. 34 Furthermore, safety and pharmacokinetics were confirmed in a study of HAD-B1 in healthy males. 37 Ginsenoside Rb1, notoginsenoside R1, ginsenoside Rg1, β-boswellic acid, cordycepin, and α-boswellic acid suppressed the growth of HCC-GR cells. In addition, α-boswellic acid inhibited MET phosphorylation. Notoginsenoside R1 reduced S6 phosphorylation, whereas cordycepin inhibited MET and S6 phosphorylation. The inhibitory effect of HAD-B1 combined with osimertinib on the growth of HCC827 cells was attributed to the synergistic effect of the active components of HAD-B1 and osimertinib (Figures 5 and 6).
Combining the above results, HAD-B1 showed an anti-cancer effect when used alone and a greater effect when combined with osimertinib. The types and coverage of targeted therapies for NSCLC are also increasing. Simultaneously, research on the molecular genetic mechanisms of carcinogenesis and the development of targeted therapeutic agents are steadily being carried out. Currently, the third-generation EGFR-TKI osimertinib has been developed and is being used as a first-line treatment for patients with NSCLC. Although osimertinib has excellent clinical efficacy, the development of drug resistance is inevitable, and tumor heterogeneity determines the variability of resistance. Cancer cells develop resistance to therapy, resulting in severe symptoms and recurrence. As a result, the unique antitumor effect of HAD-B1 will be beneficial in overcoming EGFR-TKI resistance.38,39 In conclusion, it was confirmed that when osimertinib and HAD-B1 are used together, results consistent with a synergistic effect can be produced compared with when used alone.
Our results suggest that HAD-B1 could be a potential treatment option for patients with NSCLC who develop TKI resistance by inhibiting MET phosphorylation and amplification. Combination therapy with HAD-B1 and osimertinib is expected to have a synergistic effect in the treatment of patients with gefitinib-resistant NSCLC.
Collectively, several lines of in vitro experimental evidence suggest that combined treatment with HAD-B1 and osimertinib is more effective than osimertinib monotherapy in terms of EGFR-TKI-resistant tumor cell proliferation. Several pathways involved in the anti-cancer effect of HAD-B1 on HCC827-GR cells were identified in this study, and the roles of additional important pathways need to be identified. Additionally, since in vivo experiments were not conducted, there are limitations in confirming synergic effects. Therefore further studies are required to confirm its efficacy and safety.
Conclusion
We conclude that combining HAD-B1 and osimertinib caused cell cycle arrest and apoptosis in HCC-827-GR cells by suppressing the ERK and mTOR signaling pathways via inhibition of MET phosphorylation, amplification. The inhibitory effect of HAD-B1 combined with osimertinib on HCC827-GR cell proliferation may be attributable to the different active components (Cordycepin, α-boswellic acid, and β-boswellic acid) of HAD-B1. These results suggest that HAD-B1 may be an option for treating NSCLC that develops resistance to EGFR-TKIs.
Supplemental Material
sj-pptx-1-ict-10.1177_15347354241307006 – Supplemental material for Synergistic Effect of HAD-B1 and Osimertinib Against Gefitinib Resistant HCC827 Non-Small Cell Lung Cancer Cells
Supplemental material, sj-pptx-1-ict-10.1177_15347354241307006 for Synergistic Effect of HAD-B1 and Osimertinib Against Gefitinib Resistant HCC827 Non-Small Cell Lung Cancer Cells by Eun-Ju Ko, Eun-Bin Kwag, Ji-Hye Park, Sung-Hyuk Cho, So-Jung Park, Mi-Kyung Jung, In-Cheol Kang and Hwa-Seung Yoo in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI19C1046 and HI22C1936). We thank Ildong Pharmaceutical for donating HCC827GR cells.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
