Abstract
Background:
Lung cancer, a leading cause of mortality, is often complicated by severe treatment-related side effects. Traditional herbal medicine (THM), particularly Fuzheng-Based Qi and Yin Therapy (FBQYT), has emerged as a promising adjuvant therapy to mitigate these effects and enhance immune response. This meta-analysis evaluates the efficacy and safety of THM as an adjunct to palliative chemotherapy.
Methods:
A systematic search was conducted in PubMed, EMBASE, ScienceON, RISS, KMBASE, OASIS, Cochrane Library, KISS, CNKI, and CiNii up to April 2025. Outcomes included tumor response, quality of life (QoL), adverse drug reactions (ADRs), and tumor markers. Eighty randomized controlled trials (RCTs) involving stage III/IV lung cancer patients were analyzed using Review Manager Software 5.4.1.
Results:
Combining chemotherapy with THM significantly improved the objective response rate (ORR, RR 1.41) and disease control rate (DCR, RR 1.20). It also enhanced quality of life, increasing Karnofsky Performance Status (KPS) score (MD 8.53) and KPS improvement (RR 1.51), alongside improvements in Quality of Life Questionnaire-Core 30 (QLQ-C30) total score (MD 13.05). Tumor markers and most ADRs were reduced, except for neutropenia, liver injury, and diarrhea (P > .05). Survival rates, progression-free survival (PFS), and median survival time (MST) were higher in the THM group.
Conclusion:
Integrating THM with palliative chemotherapy enhances tumor response, QoL, and survival rates while reducing adverse effects. Further rigorous RCTs are needed to confirm these findings and address methodological limitations.
Registration: PROSPERO CRD 42024523418.
Keywords
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide, accounting for 18.7% of all cancer deaths in 2022. 1 It is also the most commonly diagnosed cancer, broadly classified into non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). NSCLC accounts for approximately 80% of cases and includes subtypes such as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. In contrast, SCLC accounts for 10% to 15% of cases. 1 Notably, early-stage NSCLC often presents with non-specific symptoms, and the limited sensitivity of current screening methods frequently leads to diagnosis at an advanced stage. 2 Treatment strategies for lung cancer, including chemotherapy, are selected based on the type of cancer, stage of the disease, and the patient’s overall health status. For NSCLC, treatment options vary depending on the stage of progression. In addition to chemotherapy and immunotherapy, localized treatments such as surgery and radiation therapy are also considered. 3
Chemotherapy for advanced NSCLC (stage III or IV) is a systemic treatment that targets rapidly dividing cancer cells. Common chemotherapy regimens include platinum-based agents such as cisplatin and carboplatin, as well as non-platinum agents such as paclitaxel and docetaxel. However, these drugs are often associated with systemic side effects such as nausea, fatigue, and leukopenia. 4 Immunotherapy, which enhances the immune system’s ability to target cancer cells, utilizes immune checkpoint inhibitors such as pembrolizumab and nivolumab. These agents block immune evasion mechanisms, allowing the immune system to target the tumor more effectively. However, immunotherapy can trigger immune-related adverse events, such as dermatitis, colitis, and pneumonitis. 5
In cases of advanced or terminal lung cancer where curative treatment is not feasible, palliative chemotherapy is implemented to improve the quality of life and alleviate symptoms for cancer patients. This treatment aims to reduce tumor size, slow the progression of cancer, and extend survival. However, chemotherapy can also cause adverse effects on normal tissue cells, leading to undesirable reactions such as gastrointestinal disturbances, bone marrow suppression, and other side effects. 6
To address the adverse effects of chemotherapy and improve therapeutic outcomes, traditional herbal medicine (THM) has gained attention as a complementary and alternative treatment for cancer. THM is widely used alongside chemotherapy, particularly in palliative care for terminal-stage lung cancer patients. Recent studies have shown that in cases of unresectable stage III and IV lung cancer, patients treated with THM in combination with chemotherapy experienced several benefits. These benefits included tumor size reduction, symptom improvement, enhanced quality of life, and fewer chemotherapy-related side effects compared to patients receiving chemotherapy alone. 7 Additionally, patients in the integrative treatment group demonstrated extended survival time and overall better therapeutic responses. 8 These findings suggest that THM may play a valuable role in enhancing the effectiveness and tolerability of chemotherapy in advanced lung cancer.
In this study, we introduce the term Fuzheng-Based Qi and Yin Therapy (FBQYT) to describe the THM regimen grounded in the principle of Fuzheng (supporting and reinforcing the body’s vital energy). This approach encompasses therapies aimed at “tonifying qi and nourishing yin” to address deficiencies and enhance overall vitality. The introduction of FBQYT provides a concise and precise term for referring to this therapeutic strategy throughout the study. 9 This principle is particularly relevant in the context of terminal lung cancer, where patients often exhibit both qi and yin deficiency as a result of the advanced disease stage and the adverse effects of chemotherapy, such as fluid imbalance and malnutrition. 10
Patients with a combined deficiency of Qi and Yin typically present with fatigue, shortness of breath, night sweating, dry mouth and throat, and a persistent dry cough with scanty sputum. 9 These symptoms reflect a complex pattern of constitutional weakness, internal dryness, and impaired warming function, commonly observed in advanced stages of lung cancer or following intensive anticancer treatments. 7
As the disease progresses and tumors metastasize, the depletion of qi, blood, yin, and yang becomes increasingly severe. In these cases, THM formulations grounded in the “Fuzheng” approach, including tonifying qi and nourishing yin, are frequently employed. Modern research has demonstrated that “fuzheng” therapy can inhibit tumor progression, alleviate chemotherapy-related side effects, and enhance patients’ quality of life.
This study aims to evaluate the efficacy and safety of integrating fuzheng-based therapies, particularly those focused on tonifying qi and nourishing yin, with palliative chemotherapy for unresectable advanced lung cancer. By assessing their effects on tumor reduction and survival, we aim to provide clinical evidence supporting the incorporation of THM into contemporary cancer treatment.
Methods
Study Design and Eligibility Criteria
This meta-analysis aimed to evaluate the efficacy and safety of integrating the FBQYT method of Traditional Asian Medicine (TAM) with palliative chemotherapy in patients with lung cancer. The study adhered to the PRISMA guidelines and was registered with PROSPERO (CRD42024523418).
Eligible studies included randomized controlled trials (RCTs) involving patients with stage III or IV lung cancer who were ineligible for surgical resection and were receiving palliative chemotherapy. Most trials employed standard platinum-based doublet chemotherapy, typically cisplatin or carboplatin in combination with paclitaxel or gemcitabine as widely recommended for advanced NSCLC. A few trials also included targeted therapies such as EGFR-TKIs; however, their limited number precluded meaningful subgroup analysis. Participants were assigned to 2 groups: one receiving a combination of TAM and palliative chemotherapy, and the other receiving palliative chemotherapy alone.
FBQYT-based interventions generally consisted of herbal decoctions containing key components such as Astragalus membranaceus, Atractylodes macrocephala, Poria cocos, and Ophiopogon japonicus. These formulations were typically administered orally as water-based decoctions, usually twice daily for durations ranging from 4 to 12 weeks, with total daily dosages between 100 and 300 g. The detailed compositions, dosages, and preparation methods of commonly used prescriptions are summarized in Supplemental Tables S1 to S3.
To ensure methodological rigor, non-randomized studies, observational designs, case reports, and studies with incomplete or inconsistent data were excluded. Meta-analysis was conducted only when at least 2 studies demonstrated sufficient homogeneity in participants, interventions, comparators, and outcomes based on predefined protocol criteria.
Identification Criteria for FBQYT Therapies
In this study, therapies were classified as FBQYT only when the original article explicitly stated that the intervention aimed to implement a “Fuzheng” strategy, or to “tonify qi” or “nourish yin.” Inclusion was strictly based on the authors’ theoretical description rather than on herbal composition alone. For example, even if a formulation contained frequently used herbs such as Astragalus membranaceus, Atractylodes macrocephala, or Poria cocos, it was not considered FBQYT unless the study clearly articulated its alignment with the Fuzheng principle or a qi/yin deficiency pattern. Two independent reviewers assessed each study’s textual description to determine whether the therapeutic intent met the FBQYT framework. Discrepancies were resolved through discussion, ensuring consistent application of this definition across all included trials.
Search Strategy
The literature search was conducted using international databases, including PubMed, EMBASE, ScienceON, RISS, KMBASE, OASIS, Cochrane Library, KISS, CNKI, and CiNii. Studies published in English, Chinese, Japanese, and Korean were included. The search period extended up to April 2025. A detailed search strategy for the PubMed database is provided in Table S4 of the Supplemental Material. The search was conducted exclusively using electronic sources.
Study Selection
Search results were compiled using EndNote version 20, and duplicates were removed. Two independent reviewers (KJL and JHK) screened titles and abstracts to exclude studies that did not meet the inclusion criteria. Full texts of potentially eligible articles were assessed, and inclusion decisions were finalized through mutual agreement. In cases of disagreement, a third reviewer (DHK) resolved the issue. All reasons for study exclusion were documented.
Data Extraction
Data extraction was performed independently by 2 reviewers (KJL and JHK). Extracted data included the first author, study period, publication year, participant characteristics, comparisons, details of interventions in both groups, outcomes, and reported adverse events. In cases of missing or unclear data, the reviewers consulted each other and contacted the corresponding author via email for clarification.
Quality Assessment
To evaluate the methodological quality of the included studies, we used the Cochrane Risk of Bias (ROB) tool. 11 Two independent reviewers (KJL and JHK) assessed the risk of bias across 7 domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other potential sources of bias. Disagreements were resolved through discussion with a third reviewer (DHK).
Additionally, we assessed the overall certainty of the evidence using the GRADE system, which classifies the quality of evidence into 4 levels: high, moderate, low, and very low. By default, randomized controlled trials were considered high-quality evidence but could be downgraded based on serious limitations such as risk of bias, inconsistency, indirectness, imprecision, or publication bias. Conversely, evidence could be upgraded in cases of large effect sizes or the presence of a dose-response gradient. A GRADE evidence profile was developed for each outcome, providing a transparent and systematic assessment of the strength of evidence and highlighting areas where further research may be warranted.
Outcome Measures
The primary outcome of this study was the objective response rate (ORR), defined as the proportion of patients who achieved either a complete response (CR) or partial response (PR). Secondary outcomes included the disease control rate (DCR), quality of life (QoL), adverse drug reactions (ADRs), tumor marker levels, and long-term survival outcomes, such as progression-free survival (PFS), median survival time (MST), and 1 to 3-year survival rates. These outcomes were selected to provide a comprehensive evaluation of the clinical efficacy and safety profile of the integrative therapeutic approach.
Statistical Analysis
We used Review Manager Software 5.4.1 to conduct the meta-analyses. 12 For dichotomous outcomes, we calculated risk ratios (RRs) with 95% confidence intervals (CIs), and for continuous outcomes measured on the same scale, mean differences (MDs) with 95% CIs were used. Heterogeneity was assessed using Higgins’ I2 statistic and the Chi-squared (Chi2) test. An I2 value greater than 50% was considered to indicate substantial heterogeneity. In such cases, we performed subgroup analyses based on predefined variables such as chemotherapy regimen, type of herbal medicine, and population characteristics. We also conducted sensitivity analyses by excluding studies with a high risk of bias or small sample sizes to assess the robustness of the results. Since substantial heterogeneity was anticipated due to variations in herbal composition, treatment duration, and chemotherapy regimens, a random-effects model was applied throughout all analyses regardless of I2 values.
Frequency Analysis
To identify the predominant herbal interventions used in the treatment of lung cancer, we conducted a frequency analysis of the included studies. This analysis aimed to determine the most commonly used herbs by counting the frequency of their occurrences across all included interventions.
Network Analysis Method
Based on the frequency analysis results, we selected herbs with an occurrence frequency ≥4.81, representing the average across all listed herbs. For each herbal medicine (HM) in the systematic review, pairwise combinations of constituent herbs were generated and their frequencies calculated. Only combinations involving the selected high-frequency herbs were retained for network analysis. The extracted herb-pair data were input into Cytoscape software (version 3.10.2) to construct a visualized network representing co-occurrence and connectivity patterns among the herbs. In the resulting network, circular nodes represented individual herbs, while edges represented co-occurrences between herbs within each HM. Node size reflected the frequency of herb use, with larger nodes indicating more commonly used herbs. Edge thickness (weight) corresponded to the frequency of herb-pair co-occurrence, reflecting the strength of association.
To enhance interpretability, the yFiles Circular Layout was applied to arrange nodes in a circular pattern, and the Prefuse Force Directed Layout was used to emphasize stronger associations by drawing related herbs closer together. Herbs located near the center of the network were regarded as more influential due to their higher frequency and connectivity, indicating their key roles in the overall herbal therapeutic framework.
Results
Study Selection
An initial search across 10 electronic databases yielded 2924 records. After removing 701 duplicates, 2223 records remained for title and abstract screening. Of these, 1589 were excluded based on title and abstract review, leaving 634 articles for full-text assessment. Upon applying the eligibility criteria, 553 studies were excluded for the following reasons: 92 did not target the appropriate population, 115 were not randomized controlled trials or were study protocols, 41 involved inappropriate interventions, and 305 reported outcomes that did not meet inclusion criteria. Consequently, 81 randomized controlled trials were included in the final analysis (Figure 1).

PRISMA flowchart.
Study Characteristics
This meta-analysis includes 81 randomized controlled trials published up to 2025, involving a total of 5955 patients diagnosed with lung cancer at TNM stages III or IV.13 -93 The experimental groups consisted of patients who received palliative chemotherapy in combination with FBQYT, a form of TAM, as an adjunctive treatment. All patients were inoperable and not eligible for surgical resection. The control groups received standard chemotherapy regimens, including GP, DP, EP, and others (Table 1). A total of 101 chemotherapy regimens were reported across the 81 trials. The most frequently used combinations were NP (31.7%), GP (31.7%), TP (23.2%), and DP (13.4%). Less frequently used regimens, such as EP, PC, and Apatinib, were reported only once and were collectively categorized as “Others” (Supplemental Table S5). The treatment durations ranged from 1 week to 10 years. The primary outcomes assessed were the ORR and DCR. ORR, comprising complete and partial responses, measures the effectiveness of treatment in reducing tumor size. DCR includes complete response, partial response, and stable disease, providing a broader assessment of treatment efficacy. Secondary outcomes included QoL, ADRs, tumor marker levels, and survival rates. These outcomes offer a comprehensive evaluation of both the clinical efficacy and safety of combining FBQYT with chemotherapy in lung cancer treatment.
Characteristics of Included Trials.

Abbreviation: NP, Vinorelbine, Cisplatin; TP, Paclitaxel, Cisplatin; GP, Gemcitabine, Cisplatin; DP, Docetaxel, Cisplatin; MVP, Mitomycin, Vinblastine, Cisplatin; CE, Cyclophosphamide, Epirubicin; CAP, Cyclophosphamide, Adriamycin, Prednisone; MAF, Methotrexate, Doxorubicin, 5-Fluorourac; EP, Etoposide, Cisplatin; DCCRT, Definitive Concurrent Chemoradiotherap.
Risk of Bias in Included Trials
According to the Cochrane Risk of Bias tool, the included trials generally exhibited a moderate to unclear risk of bias due to limited methodological reporting (Figure 2). Most studies adequately reported random sequence generation; however, 2 trials36,78 were rated as high risk in this domain. Allocation concealment was not reported in 32 trials,14,18,22,25,27 -29,31,33,35,36,39 -41,48 -50,52,53,57,62,64,66,67,74,78,85,87,90 -93 resulting in 31 being judged as unclear risk and 1 as high risk. 36 Blinding of participants, personnel, and outcome assessors was generally not feasible, as TAM was administered exclusively to the intervention group. However, 3 studies20,32,86 employed a placebo and thus allowed for partial blinding. Incomplete outcome data was rated as unclear in 19 studies16,20,29,35,45,47,49,50,61,71,72,76,79 -82,86,87,89 due to imbalance in participant numbers, and as high risk in 12 studies13,19,21,22,26,36,44,55,64,67,69,78due to unexplained differences involving more than 4 participants. Selective reporting bias was generally judged as unclear; however, 8 studies13,17,20,30,43,74,86,93 were rated as low risk due to the availability of study protocols and ethical approval. Other biases were identified in 3 studies: 1 as high risk 45 and 2 as unclear27,60 due to reporting errors and insufficient methodological detail (Figure 3).
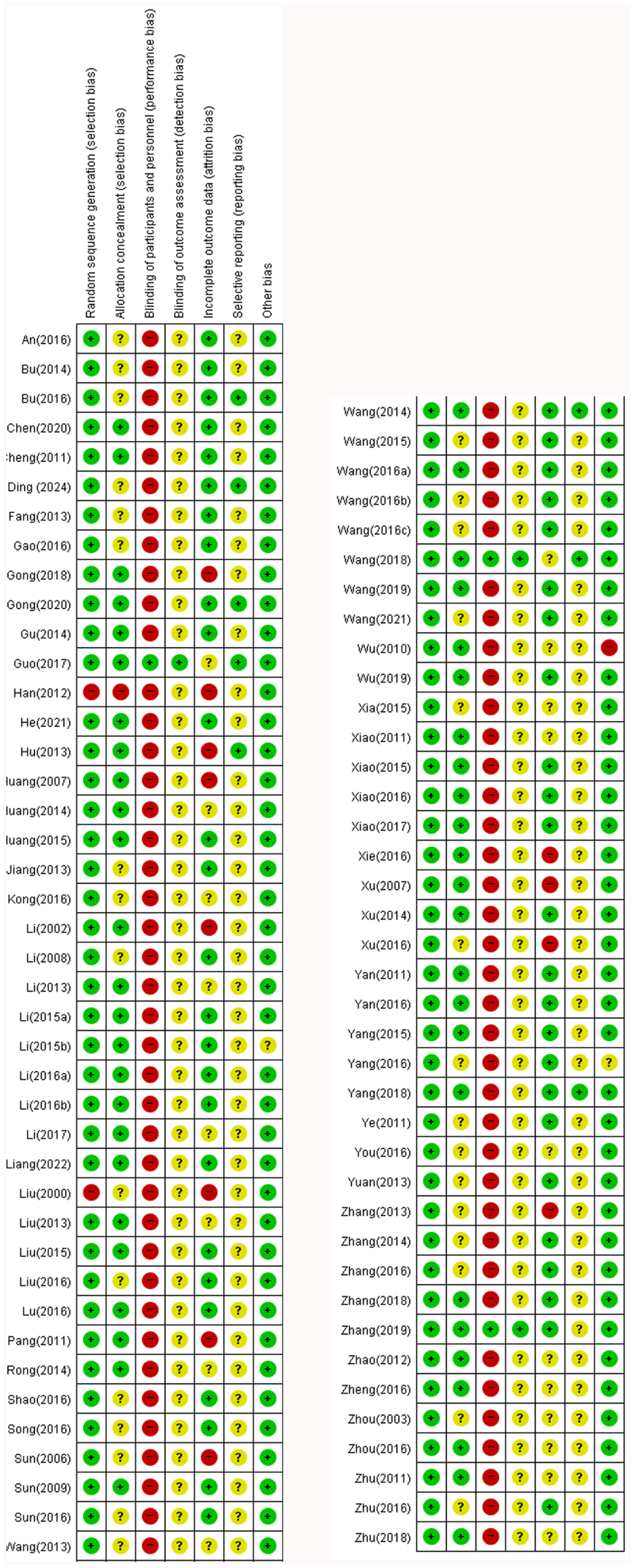
Risk of bias summary.

Risk of bias graph.
Primary Outcome
Tumor Response Assessment
A comprehensive review of 46 and 44 studies demonstrated a statistically significant increase in both ORR13 -16,18,19,22,24,26,30 -34,36,38,40,42,43,45,46,49,50,52,54 -56,62,63,66 -68,71,73 -76,78,79,84,85,87 -89,91,93 and DCR13 -16,18,19,22,24,26,30 -32,34,36 -38,40,42,43,45,46,48 -50,52,54 -56,62,63,66 -68,71,73 -76,78,79,84,85,89,91 for patients receiving a combination of THM and chemotherapy compared to those receiving chemotherapy alone. The results showed a relative risk (RR; 1.41, 95% CI [1.30, 1.52], I2 = 0%, n = 3294) for ORR, and a RR (1.20, 95% CI [1.15, 1.25], I2 = 26%, n = 3213) for DCR (Figures 4 and 5).

ORR of FBQYT method THM + chemotherapy.

DCR of FBQYT method THM + chemotherapy.
Secondary Outcomes
Quality of Life
The Karnofsky Performance Status (KPS) score, based on 21 studies23,25,26,28,32,37,48,55 -57,59,60,64,66,72,79,82,83,91 -93 and KPS improvement was based on 23 studies13,16,22,33,34,40 ,41,43,46,47 ,49,52,65,67 ,69,73,75 ,77,83,85-87,89 were assessed, showing a significantly higher enhancement in the group receiving both THM and chemotherapy compared to the chemotherapy-only group. The KPS score was (MD 8.53), and the KPS improvement was (MD 1.51). Both outcomes were statistically significant (P < .05; Figures 6 and 7). For the FACT-L score, as reported in 3 studies20,24,70 no significant improvement was observed in Physical Well-Being (PWB, MD 4.68), Social Well-Being (SWB, MD 1.30, 3 trials), Emotional Well-Being (EWB, MD 2.78, 3 trials), Functional Well-Being (FWB, MD 7.38, 3 trials), and Lung Cancer Subscale (LCS, MD 8.28, 3 trials), Total score (MD 16.70, 2 trials)20,24 with P > .05 for all subscales. In the QLQ-C30 functional scales, based on 5 studies,15,29,61,68,90 significant improvement was seen in Physical Functioning (PF, MD 5.17, 5 trials), Role Functioning (RF, MD 8.95, 5 trials), Emotional Functioning (EF, MD 8.85, 5 trials), and Social Functioning (SF, MD 5.87, 4 trials),15,29,61,90 Total (MD 13.05, 3 trials)15,29,61 (P < .05), while Cognitive Functioning (CF, MD 5.56, 5 trials) showed no significant improvement (P > .05). In the symptom scales, based on 3 studies,15,29,61 significant improvement was observed in Fatigue (MD −17.79), Nausea and Vomiting (MD −8.02), Dyspnea (MD −7.11), Insomnia (MD −12.06), Appetite Loss (MD −13.04), and Constipation (MD −6.74) (P < .05), while Pain (MD −4.39), Diarrhea (MD −2.84), and Financial Difficulties (MD −2.96) showed no significant improvement (P > .05; Table 2).

KPS Score of FBQYT method THM + chemotherapy.

KPS improvement rate of FBQYT method THM + chemotherapy.
QLQ-C30 and FACT-L of FBQYT Methods THM + Chemotherapy.

P < .05, **P < .01, ***P < .001.
Tumor Marker Level
In the THM plus chemotherapy group, there was a greater decrease in tumor markers compared to the chemotherapy-alone group. Specifically, CEA levels (MD −7.34, 8 trials),16,21,42,45,46,51,91,93 CYFRA21-1 levels (MD −1.55, 5 trials),16,21,42,45,46 and VEGF levels (MD −17.71, 6 trials)19,21,38,45,49,51 decreased. All of these reductions were statistically significant, with P < .05 for all (Table 3).
Tumor Markers of FBQYT Methods THM + Chemotherapy.

P < .05, ***P < .001.
Adverse Drug Reactions Assessment
The findings from the meta-analysis revealed that the group receiving both THM and chemotherapy had a reduced likelihood of experiencing adverse drug reactions compared to the chemotherapy-only group. Significant reductions were observed in the incidence of myelosuppression (RR 0.46, 6 trials),27,35,45,58,65,66 leukocyte reduction (RR 0.59, 28 trials),13,14,16,21,23,26 -28,31,33,38 -40,42,47,49 -52,54,56 -58,67,81,84,86,90 platelet reduction (RR 0.51, 20 trials),16,21,23,26 -28,31,33,38 -40,47,49,52,57,58,67,79,81,90 hemoglobin reduction (RR 0.51, 14 trials),16,26,28,33,38,39,42,47,49,51,52,57,84,86 renal injury (RR 0.33, 2 trials),16,28 nausea and vomiting (RR 0.46, 20 trials),17,21,23,25,26,31 -33,40,51 -54,56,57,61,67,79,81,90 neurotoxicity (RR 0.48, 5 trials),52,54,57,71,84 and gastrointestinal symptoms (RR 0.52, 14 trials),14,16,17,30,34,38,42,45,49,50,58,65,66,84 all with P < .05. No significant differences were found for neutropenia (RR 0.38, 4 trials),27,47,52,86 liver injury (RR 0.76, 10 trials),16,21,28,32,42,51,52,57,90,93 and diarrhea (RR 0.55, 6 trials),17,30,32,51,56,93 with P > .05 for these adverse effects. All reductions, except for liver injury, diarrhea, and neutropenia, were statistically significant (Table 4). However, THM-specific adverse events were rarely reported or not clearly distinguished from general chemotherapy-related ADRs in the included studies. Therefore, a separate quantitative analysis of ADRs directly attributable to THM was not feasible.
Adverse Drug Reactions of FBQYT Methods THM + Chemotherapy.

P < .01, ***P < .001.
Survival Rate
The meta-analysis revealed that the combination of THM and chemotherapy significantly improved survival outcomes. Specifically, there was a marked increase in the 1-year survival rate (RR 1.27, 9 trials, Figure 8),15,16,18,44,48,55,72,80,87 the 2-year survival rate (RR 1.64, 3 trials,Figure 9),18,72,87 and the 3-year survival rate (RR 1.82, 3 trials, Figure 10).44,72,87 Furthermore, the combination therapy significantly enhanced progression-free survival (PFS; RR 2.84, 5 trials, Figure 11)26,29,30,32,60 and extended median survival time (MST) by 3.24 months (MD 3.24 months, 8 trials, Figure 12).16,18,26,30,44,45,72,80 All these improvements were statistically significant (P < .05 for all outcomes), underscoring the effectiveness of combining THM with chemotherapy in improving long-term survival and patient outcomes.

One-year survival rate of FBQYT method THM + chemotherapy.
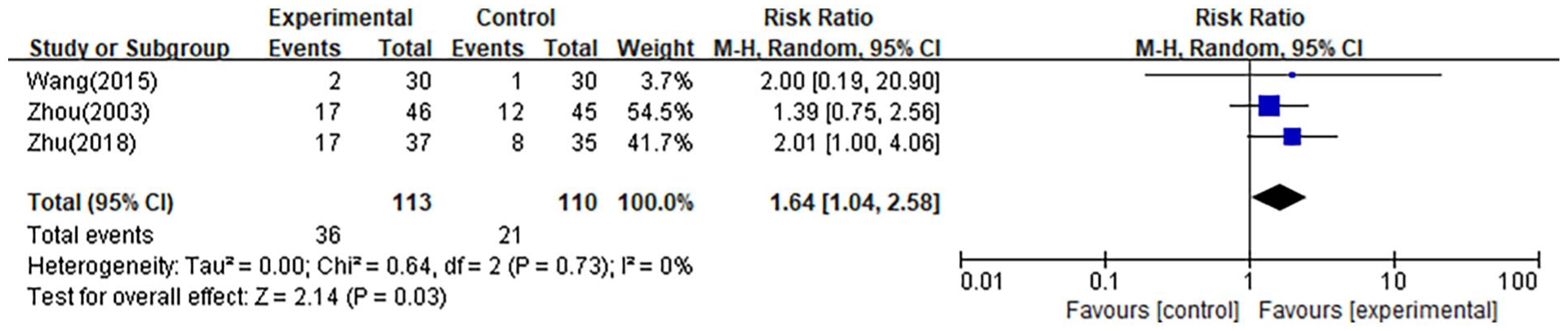
Two-year survival rate of FBQYT method THM + chemotherapy.

Three-year survival rate of FBQYT method THM + chemotherapy.

Progression-Free Survival of FBQYT method THM + chemotherapy.

Median Survival Time (Months) of FBQYT method THM + chemotherapy.
Publication Bias
To investigate publication bias, funnel plots were created using RR, and MD values extracted from trials that measured ORR, KPS score, and 1-year survival rate. The funnel plot analysis for the ORR study exhibited a symmetrical shape (Figure 13), indicating a balanced distribution of studies across a range of effect sizes and their corresponding precision measures. This symmetry suggests the absence of significant publication bias or small study effects, reinforcing the reliability and robustness of the study’s findings regarding ORR. However, the funnel plots generated based on the KPS score and 1-year survival rate displayed an asymmetrical distribution (Figures 14 and 15), suggesting the potential presence of publication bias in these analyses.

Funnel plot assessing publication bias (ORR in 45 trials).

Funnel plot assessing publication bias (KPS score in 20 trials).
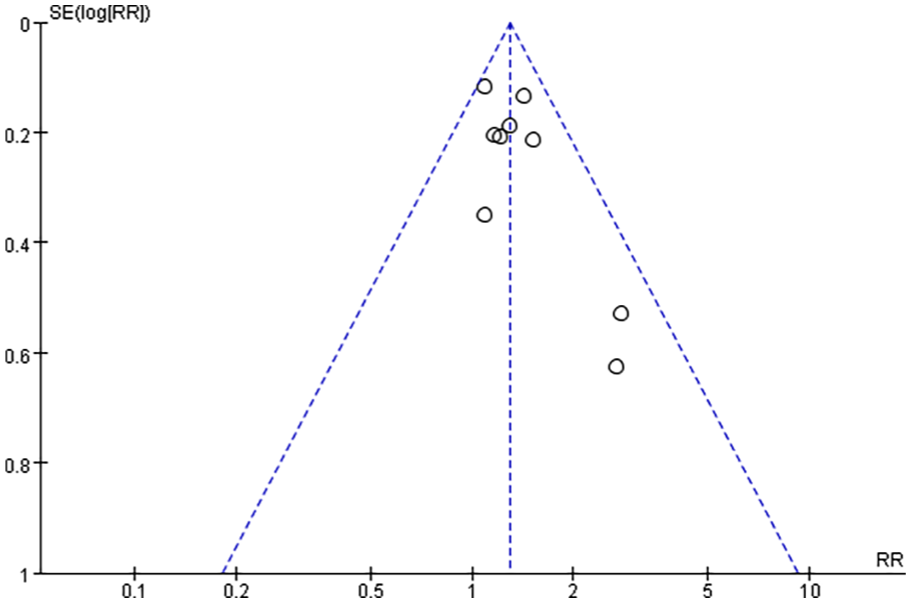
Funnel plot assessing publication bias (1-year survival rate in 9 trials).
Quality of Evidence Assessment
In evaluating tumor response, the ORR and DCR were analyzed across 45 and 44 RCTs, respectively. The QoL outcomes were assessed using the KPS score (20 RCTs), KPS improvement rate (23 RCTs), FACT-L (3 RCTs), and QLQ-C30 (5 RCTs). Additionally, tumor markers (CEA, CYFRA21-1, VEGF), 1 to 3-year survival rates, PFS, MST, and adverse drug reactions including myelosuppression, leukocyte decrease, neutropenia, platelet reduction, hemoglobin decrease, liver and renal injury, nausea, vomiting, diarrhea, nerve infections, and gastrointestinal symptoms were all analyzed. Most outcomes were assessed with “Moderate” certainty. However, PFS and MST were evaluated with high certainty. QLQ-C30 subscales for PF, CF, SF, pain, and dyspnea were rated as low, while diarrhea was rated as very low. The main reasons for downgrading the quality of evidence for certain outcomes were inconsistencies, particularly when 1 or more mean results deviated in the opposite direction, and imprecision, especially when CIs crossed into non-significant ranges or when the total sample size was less than 400. Detailed outcomes, effects, and absolute effects are available in the Supplemental Materials (Table 5).
A Summary of Comprehensive Assessment.
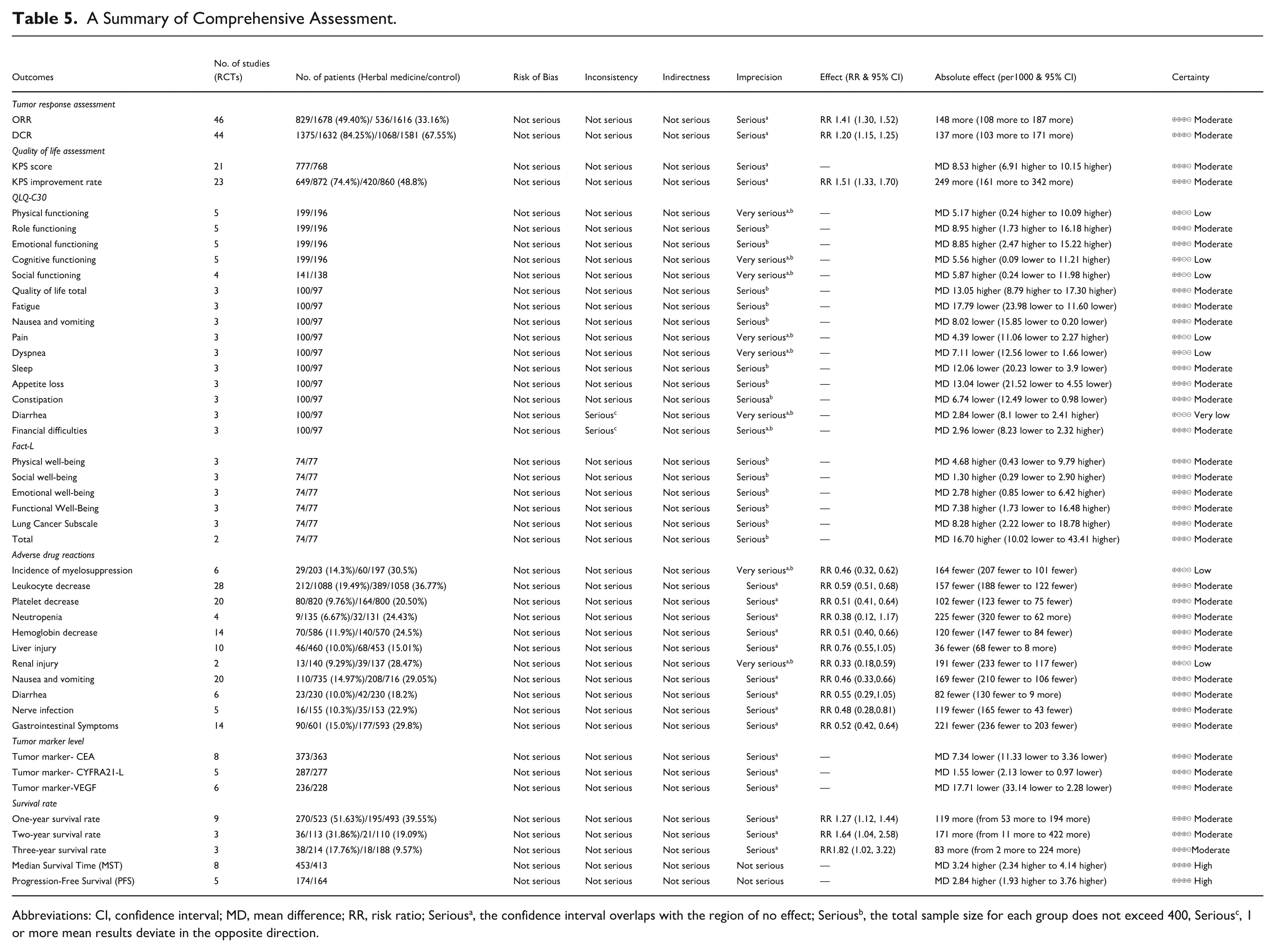
Abbreviations: CI, confidence interval; MD, mean difference; RR, risk ratio; Seriousa, the confidence interval overlaps with the region of no effect; Seriousb, the total sample size for each group does not exceed 400, Seriousc, 1 or more mean results deviate in the opposite direction.
Frequency Analysis
A total of 192 HMs were identified from the selected literature, with each prescription demonstrating enhanced anti-cancer effectiveness and reduced side effects when combined with palliative chemotherapy. Among these, 80 herbs were classified as high-frequency, each used more than 3 times (median ≥ 2), accounting for 42.71% of the total. The top 10 most frequently used herbs included Astragalus membranaceus (82.72%), Atractylodes macrocephala (44.44%), Ophiopogon japonicus (each 39.51%), Poria cocos (38.27%), Glycyrrhiza uralensis (32.10%), Pseudostellaria heterophylla (27.16%), Codonopsis pilosula (25.93%), Glehnia littoralis and Citrus reticulata (each 24.69%), as well as Pinellia ternata (23.46%). Among herbs used more than 10 times, 37.8% were classified as Tonifying Qi or Nourishing Yin, followed by Heat-Clearing Herbs (24.4%), Blood-Invigorating Herbs (9.8%), Phlegm-Resolving Herbs (6.1%), and Qi-Regulating Herbs (4.88%). Additionally, Astringent Herbs and External Application Herbs accounted for 3.66% each, while Digestive Herbs contributed 2.44%. The remaining 7.32%, categorized as “others,” consisted of Water-Dampness Draining Herbs, Blood-Tonifying Herbs, Cough-Stopping Herbs, Dampness-Transforming Herbs, Surface-Relieving Herbs, and Calm Spirit Herbs, each contributing 1.22% 94 (Table 6 & Figure 16). Additionally, 54 high-frequency families, each used more than 5 times (mean frequency ≥ 4.81), comprised 27.83% of the total. Network analysis revealed 5283 herbal pair combinations, with Astragalus membranaceus being the most frequently used herb (67 times), and the Astragalus membranaceus-Atractylodes macrocephala pair being the most common combination (31 times). Core herbs extracted from the network showed high centrality (Figure 17A and B). These findings are summarized and categorized based on the Fuzheng principle and Qi-Yin classification (Supplemental Table S1–3). In the centrality analysis, we ranked the herbs by sorting the Average Shortest Path Length from lowest to highest and the other 3 centrality metrics from highest to lowest. The herbs with the highest centrality based on Average Shortest Path Length were Astragali Radix, Atractylodis Macrocephalae Rhizoma, Poria, Ophiopogonis Radix, Glycyrrhiza uralensis, Pseudostellariae Radix, Codonopsis Radix, Glehniae Radix, Citri Reticulatae Pericarpium, and Pinelliae Rhizoma (Table 7).
Herbal Medicine Frequencies in Lung Cancer Therapy.

Total prescription numbers, n = 81

Herb categories distribution.

(A) Visualized networks of herbs (Circular layout): The above figure displays the network connectivity of 53 herbs in a circular layout. The size of the circle increases as the herb is used more frequently, and the thickness of the line between the hubs indicates the frequency of co-occurrence. (B) Visualized networks of herbs (Force-directed layout in core): While the circular layout allows to see the relationship between herbs at a glance, it has the limitation of making it difficult to identify clusters. To overcome this, the force-directed layout introduces the concept of electromagnetic force based on co-occurrence. It suggests that the core hubs are in the center. The primary clusters are Astragali Radix and Atractylodis Macrocephalae Rhizoma.
Centrality Analysis of 10 Core Herbs.

Discussion
This meta-analysis systematically reviewed and analyzed 81 studies involving 5955 participants to evaluate the efficacy and safety of combining THM with chemotherapy in treating lung cancer. A comprehensive assessment of the included studies was conducted using the RoB tool, which facilitated the evaluation of potential biases in these trials. The analysis revealed that the trials demonstrated an unclear risk of bias, primarily due to deficiencies in methodological reporting. Specifically, many trials did not provide sufficient details on allocation concealment, and blinding of participants was challenging due to the nature of the intervention with THM. Additionally, detection bias was unclear in many studies, and reporting bias was difficult to assess due to the lack of protocol registration. Despite including a large number of studies, the overall methodological quality varied. To mitigate this, we applied strict eligibility criteria and emphasized clarity in outcome definitions. These observations highlight the need to prioritize methodological rigor over quantity in future research.
The RoB assessments highlight the necessity for more rigorously designed RCTs in this field to bolster the evidence base. To ensure clarity and consistency in evaluating treatment outcomes, studies focusing on various syndrome types, including Qi or Yin deficiency, were included. Additionally, studies incorporating the concept of Fuzheng, even without explicitly mentioning Qi or Yin deficiency, were also included. Studies that did not align with these criteria were excluded. Furthermore, studies using interventions such as radiotherapy or injectable treatments, as well as those involving combined treatments of herbal medicine and acupuncture, were excluded.
Primary outcomes demonstrated a positive link between THM and tumor response, with both ORR and DCR showing statistically significant improvements. Secondary outcomes revealed that patients receiving both THM and chemotherapy mostly showed improved QoL scores. However, the FACT-L scores and QLQ-C30 subscales, including CF, SF, Pain, Diarrhea, and Financial Difficulties, did not show significant improvement, which may be attributed to the subjective nature of these patient-reported tools and a small number of studies included. In contrast, improvements in quality of life were observed through third-party evaluations such as KPS, which confirmed an improvement in patients’ quality of life. Although the FACT-L and QLQ-C30 did not show significant improvements in some subscales, their established reliability and effectiveness as quality-of-life indicators remain valuable, as demonstrated in previous studies.7,61
Additionally, the combination of THM and chemotherapy resulted in fewer adverse drug reactions and reduced tumor marker levels compared to chemotherapy alone. However, adverse drug reactions such as neutropenia, liver injury, and diarrhea, which are closely associated with certain chemotherapy agents, may be more difficult to alleviate through adjunctive therapies like THM due to the inherent mechanisms of these drugs. The relatively small sample size for studies focusing on these specific side effects may have also limited the ability to demonstrate statistically significant improvements. Nevertheless, positive outcomes observed in other cancer studies, 95 alongside favorable results in other indicators within our study, suggest that the integration of THM with chemotherapy may offer effective management of chemotherapy-related adverse reactions.
Most included studies did not report adverse events attributable solely to THM, limiting our ability to evaluate its independent safety profile. Nevertheless, the combination therapy demonstrated improved long-term outcomes, including 1 to 3-year survival rates, prolonged PFS, and extended MST. Although subgroup analysis by herbal category was not feasible due to heterogeneous compositions and inconsistent reporting across studies, subgroup analyses based on chemotherapy type (eg, NP, GP, TP) were conducted for ORR, DCR, KPS score, and KPS improvement. This review focused exclusively on NSCLC, and studies involving SCLC were excluded based on our predefined eligibility criteria, as the rapid progression of SCLC limits the feasibility of integrating herbal therapies.
Based on these positive results, our study may provide new insights into the global challenge of treating lung cancer, which is complicated by various risk factors and has a significant impact on patients quality of life. Lung cancer is the leading cause of cancer-related deaths globally, outpacing other types of cancer in mortality rates. The treatment regimen for NSCLC, which accounts for about 85% of lung cancer cases, typically includes surgery, radiation, and chemotherapy. 1 However, many patients present with advanced-stage disease, making surgical intervention infeasible. Chemotherapy remains the primary treatment for these patients, often complemented by targeted therapies and immunotherapies. Despite significant therapeutic advancements, the side effects of chemotherapy, such as myelosuppression gastrointestinal disturbances, and decreased quality of life, continue to pose major challenges. 4
Recently, interest has increased in integrating THM with conventional chemotherapy. THM, consisting of various herbal formulations long used in TAM for health support and disease treatment, aims to enhance chemotherapy’s effectiveness, reduce side effects, and improve patients’ overall quality of life.7,8 One of the key therapeutic methods in THM is the FBQYT approach, which is centered on tonifying Qi and nourishing Yin. In line with this approach, the RCTs analyzed for ORR and DCR research frequently used herbs like Astragalus membranaceus, Poria cocos, Atractylodes macrocephala, Ophiopogon japonicus, and Glycyrrhiza uralensis. Zhang et al’s study 96 demonstrated that such decoctions alleviate symptoms such as cough and phlegm, enhance immune function, and improve quality of life while reducing side effects. When combined with palliative chemotherapy and EGFR-TKIs (erlotinib, gefitinib, osimertinib), the FBQYT method decoction has shown potential to enhance treatment efficacy by inhibiting cancer cell proliferation and inducing apoptosis. Similarly, studies by Liu et al 97 on BuZhongYiQi decoction and Cui et al 98 on ShaShenMaiDong decoction, which also include these herbs, found they inhibit cancer growth and aid in overcoming drug resistance. This effect is attributed to Astragalus’s ability to boost immune function by increasing cAMP levels, enhancing leukocyte and macrophage activity, stabilizing red blood cells, and promoting DNA synthesis in regenerating liver cells. Atractylodes and Glycyrrhiza promote T cell growth and maturation, improving immune response. Poria cocos reverses TNF-β-induced EMT and reduces E-cadherin downregulation and Vimentin upregulation, Downregulates GPX4 and SLC7A11, Ferroptosis inhibitor (Fer-1) reverses the effects. 99 Glehnia root contains sterols with anti-tumor, anti-inflammatory, and immune-regulating properties, while Ophiopogon root contains isoflavones that inhibit lung cancer cell growth. 100 Furthermore, chemotherapeutic agents such as docetaxel and cisplatin were found to enhance these effects by promoting apoptosis and suppressing the tumor microenvironment. Thus, the combination of the FBQYT method of traditional herbal medicines with chemotherapy appears to enhance therapeutic efficacy while reducing common side effects of cancer therapies. The therapeutic efficacy of FBQYT-based integrative therapy may be partially explained by its underlying biological mechanisms. Many of the included herbs, such as Astragalus membranaceus and Glycyrrhiza uralensis, are known to enhance immune function by promoting lymphocyte activity, cytokine regulation, and macrophage activation. These immunomodulatory effects may contribute to improved tumor control and resilience during chemotherapy. Additionally, herbs like Poria cocos and Atractylodes macrocephala have demonstrated properties in reducing inflammation, mitigating oxidative stress, and reversing EMT, which may help in reducing chemotherapy-induced toxicity and inhibiting tumor metastasis. Some ingredients also influence ferroptosis-related pathways, suggesting a possible mechanism in enhancing cell death of resistant tumor cells. Collectively, these mechanisms support the observed clinical benefits and provide biological plausibility for integrating THM with chemotherapy.
In this study, we confirmed the frequent use of herbs such as Astragali Radix, Atractylodis Macrocephalae Rhizoma, Ophiopogonis Radix, Poria, and Glycyrrhiza uralensis. Notably, 37.8% of herbs used more than 10 times were classified as tonifying qi or nourishing yin herbs in traditional Chinese medicine. These findings align with previous research that also reported the frequent use of these herbs. 101 Although the inclusion criteria required explicit reference to Fuzheng principles including strategies such as tonifying qi or nourishing yin rather than herb composition alone, it is noteworthy that the central herbs identified via network analysis overlapped significantly with commonly cited Fuzheng components. This alignment reinforces the validity of the classification while also suggesting a degree of convergence in clinical practice.
Despite the evident benefits of combining THM with palliative chemotherapy for lung cancer, there remain knowledge gaps in THM’s specifics, such as discerning essential components and probing potential interactions between herbal medicines and standard treatments. This study, focusing on FBQYT traditional herbal medicines, provides a clearer evaluation of their effects. Previous studies and recent meta-analyses have primarily focused on immune functions within a specific timeframe, lacking a more comprehensive evaluation. In contrast, this study conducted an extensive meta-analysis and provided a thorough evaluation, covering a broader range of clinical outcomes. This comprehensive approach can be particularly helpful to practitioners who treat lung cancer patients with palliative chemotherapy, offering them more effective and targeted herbal treatment options. Integrating THM into palliative chemotherapy regimens for lung cancer offers a promising strategy to enhance treatment outcomes and improve patient quality of life.
Limitations
This study has several limitations. Firstly, although we performed subgroup analyses based on chemotherapy type (eg, NP, GP, TP, DP) for ORR, DCR, KPS score, and KPS improvement, subgroup analyses based on cancer subtype or herbal treatment category were not feasible due to the limited number of trials per group and inconsistent reporting. Although the frequency of THM categories was analyzed, their heterogeneous compositions precluded meaningful subgroup comparisons. Moreover, variations in THM formulations and treatment durations across studies may have affected the outcomes but were not amenable to detailed analysis due to limited reporting. Blinding was inherently infeasible since THM was administered solely to the intervention group, potentially introducing placebo effects, especially in subjective outcomes like quality of life. Most included studies did not distinguish between THM-related and chemotherapy-related adverse events, limiting our ability to assess the safety of THM independently. Overall survival (OS) was reported in only 1 study, precluding its inclusion in the meta-analysis due to inconsistent availability. The funnel plot indicated potential publication bias, and many studies had small sample sizes, limiting the generalizability of the findings. Future studies should adopt more standardized reporting, larger sample sizes, and clearer outcome definitions to validate the clinical value of FBQYT-based integrative therapy.
Conclusions
This systematic review and meta-analysis indicates that the combination of palliative chemotherapy and the FBQYT method of THM yields promising therapeutic benefits in lung cancer. The integrative therapy significantly improves tumor response, quality of life, and survival outcomes, while also reducing the incidence of common chemotherapy-related adverse effects, including myelosuppression, leukopenia, thrombocytopenia, anemia, renal injury, nausea and vomiting, gastrointestinal symptoms, and neurotoxicity. Based on current evidence, this approach appears most suitable for stage III or IV NSCLC patients, particularly those with Qi and Yin deficiency. Herbal therapies are typically administered concurrently with chemotherapy over a period of 4 to 12 weeks. However, due to the moderate-to-unclear risk of bias in many of the included studies, further well-designed randomized controlled trials with standardized interventions, placebo controls, and rigorous methodological quality are warranted to validate these findings and support their clinical application.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251373045 – Supplemental material for Efficacy of Herbal Medicine Therapy Combined With Palliative Chemotherapy in Unresectable Advanced NSCLC: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ict-10.1177_15347354251373045 for Efficacy of Herbal Medicine Therapy Combined With Palliative Chemotherapy in Unresectable Advanced NSCLC: A Systematic Review and Meta-Analysis by Keon-Jun Lee, Jong-Ha Kwon, Dong-Hyeon Kim, Mi-Sun Park, Ye-Jin Jin, Ji-Hwan Kim, Hwa-Seung Yoo and So-Jung Park in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors thank all researchers and supporters involved in this study.
Ethical Considerations
No ethical approval was required as this study did not involve human participants or laboratory animals
Author Contributions
KJL and JHK contributed to study conception and design, and drafted the original manuscript. JHK, DHK, and MSP performed data collection, extraction, and assessment. KJL, JHK, HSY, and JHKim were involved in manuscript writing and critical revision. KJL, JHK, and DHK contributed to writing review and editing. JHK, HSY, and SJP were responsible for methodology and critical revision. SJP supervised the project, managed administration, and acquired funding. All authors reviewed and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant number: RS-2023-KH139460)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data and materials included in this study are included in this published article and supplementary material.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
