Abstract
Oral microbiota may be associated with serious local or systemic medical conditions resulting from chemotherapy. This study was conducted to evaluate the changes in the oral microbiota following the initiation of chemotherapy in patients with hematopoietic malignancies and to identify the characteristics of the oral microbiota associated with oral mucositis. Oral samples were collected from 57 patients with hematopoietic malignancies at 2 time points: before the start of chemotherapy and 8 to 20 days after the start of chemotherapy, when chemotherapy-induced oral mucositis often occurs, and 16S rRNA metagenomic analyses were performed. Comparative and linear discriminant analysis effect size (LEfSe) analyses were used to determine the characteristic bacterial groups before and after the initiation of chemotherapy and in those who developed oral mucositis. The alpha and beta diversities of oral microbiota before and after the initiation of chemotherapy differed significantly (operational taxonomic unit index,
Introduction
There are far more microorganisms on our body surface constituting the microbiota than the number of cells constituting our bodies. 1 Recent advances in microbial research have revealed that symbiotic relationships between commensal microbiota and the host are involved in various diseases and health conditions. Alterations in the gastrointestinal microbiota caused by treatments of malignancies, including chemotherapy, have been shown to play an important role in response to treatment and susceptibility to adverse effects.2-4 Microbiota in the oral cavity, which represents the second largest population of commensal microorganisms after the gut microbiota, may also influence chemotherapy-induced adverse effects 5 ; however, chemotherapy-induced changes in the oral microbiota are not well understood.
Approximately 700 species of bacteria have been identified in the oral cavity, and the number of bacteria per gram of plaque wet weight is 1011, comparable to the number of bacteria in 1 g of feces. If plaque control is poor, the number of bacteria increases further. Specific oral bacterial species such as
Chemotherapy for malignant tumors has long been known to alter the oral bacterial environment.13,14 However, studies using sensitive, high-throughput methods to characterize the oral microbiota during chemotherapy are lacking. Recent advances in next-generation sequencing and bioinformatics technologies that can easily collect and analyze a large amount of sequence data, hundreds to thousands of times larger than conventional methods can do, have made it possible to comprehensively evaluate the composition of microorganisms, including those that cannot be detected using conventional culture methods or specific evaluation methods for specific bacteria. The oral environment of patients with hematopoietic malignancies is remarkably altered by chemotherapy, causing adverse events such as oral dryness and oral mucositis. 15 As oral microbiota may be associated with these adverse events,16-18 it is necessary to elucidate the changes in oral microbiota caused by chemotherapy in patients with hematopoietic tumors in detail.
In the present study, we comprehensively evaluated the changes in oral microbiota associated with the start of chemotherapy in patients with hematopoietic malignancies via the metagenomic analysis of 16S ribosomal RNA (16S rRNA). In addition, the characteristics of the oral microbiota associated with oral mucositis were assessed.
Materials and Methods
Participants
The present study was conducted in accordance with the tenets of the Declaration of Helsinki and its latest amendments and was approved by the Ethics Committee of Osaka Medical and Pharmaceutical University (approval no. 2145) and Chugoku Central Hospital of the Mutual Aid Association of Public School Teachers (approval no. 1905-02). Written informed consent was obtained from all participants.
The study involved 57 patients with hematopoietic tumors, namely, 14 patients with acute myelogenous leukemia, 30 patients with malignant lymphoma (ML), 3 patients with acute lymphocytic leukemia (AML), 8 patients with multiple myeloma (MM), and 2 patients with myelodysplastic syndromes. The patients were diagnosed at the Department of Hematology of Chugoku Central after being referred from the Department of Dental Surgery for Perioperative Oral Function Management between June 2019 and October 2020. Patients diagnosed with hematologic malignancies and receiving chemotherapy for the first time were included in the study; there were no exclusion criteria. Table 1 shows the baseline characteristics of the study population, including the types of malignancies and chemotherapies. Metadata associated with the study participants are shown in Supplemental Table 1.
Baseline Characteristics of the Study Population (n = 57).

Oral Sample Collection and Oral Examination
Oral samples from the participants were collected twice: before and after the start of chemotherapy. Samples from the post-chemotherapy group were collected 8 to 20 days after chemotherapy initiation as it is known that oral mucositis appears more frequently in the first or second week after the start of chemotherapy. 19 The period between the start of chemotherapy and sample collection for each patient is shown in Supplemental Table 1.
The previously described oral rinse method was used for sample collection. 10 Briefly, the participants were instructed to gargle 5 mL of isotonic sodium chloride solution (Fuso Pharmaceutical, Osaka, Japan) for 10 seconds and then spit the solution into sterile tubes. All samples were immediately frozen. After collecting the oral samples, the oral conditions were examined. Oral mucositis status was assessed at oral sample collection after the start of chemotherapy. After the start of chemotherapy, 6/57 patients had oral mucositis of grade 2 or higher, as assessed based on the Common Terminology Criteria for Adverse Events (CTCAE) version 4.0.
DNA Extraction, 16S rRNA Sequencing, and Taxonomic Classification
DNA extraction, 16S rRNA sequencing, and taxonomic classification were performed as described previously, 10 with some modifications. Briefly, the oral samples were homogenized with glass beads using a homogenizer (Disruptor Genie; Scientific Industries, Bohemia, NY, USA). Bacterial genomic DNA from the homogenized samples was extracted using GENE PREP STAR PI-480 (Kurabo Industries Ltd., Osaka, Japan), according to the manufacturer’s instructions. The V1–V2 region of the 16S rRNA gene was amplified, and each library was prepared following the 16S metagenomic sequencing library preparation protocol (Illumina, San Diego, CA, USA). DNA was sequenced in 500 cycles using MiSeq Reagent Kit v2 (Illumina). An average of 36 653 sequence reads with 250-bp paired-ends were denoised and quality-filtered using DADA2 in Quantitative Insights Into Microbial Ecology 2 (QIIME2) version 2020.02. After denoising, amplicon sequence variants were generated and regarded as 100% operational taxonomic units (OTUs). A rarefaction minimum depth cut-off was selected at 10 000, which retained all samples for downstream analysis. Each OTU was assigned against the curated Greengenes 13_8 reference database.
Statistical Analysis
Statistical analysis was performed by modifying some of the methods used in our previous study. 11 The within-patient alpha diversity of bacterial communities was assessed using the observed OTU and Shannon’s indices. The Kruskal–Wallis test was used for comparisons between the groups. Between-patient beta diversity was assessed based on unweighted and weighted UniFrac distance metrics. 20 To visualize global differences in the microbiota structure determined using the UniFrac analysis, we performed principal coordinate analysis (PCoA). The significance of compositional differences between the groups was assessed using permutational multivariate analysis of variance (PERMANOVA). QIIME2 software was used for these analyses.
The linear discriminant analysis (LDA) effect size (LEfSe) algorithm was used to determine the differentially abundant genera between the groups. 21 All analyses were performed with the α parameter of the LEfSe analysis for pairwise tests set to .05 and the threshold of the logarithmic score for the LDA set to 3.0. The changes in the abundance of each bacterial genus before and after treatment were evaluated using a corresponding analysis of variance (RM ANOVA). All analyses were performed using R ver. 4.0.3 (2020-10-10; R Foundation for Statistical Computing, Vienna, Austria).
Results
Microbiota Composition
Oral bacteria with a relative abundance of at least 0.1% in both the before and after chemotherapy initiation groups (hereinafter, before and after groups, respectively) were classified into 13 phyla, 19 classes, 38 orders, 74 families, and 145 genera. At the phylum level, the predominant bacteria (>1% of the total sequences in either group) were Firmicutes and Bacteroidetes, followed by Actinobacteria, Proteobacteria, and Fusobacteria, comprising 95.54% and 95.88% of the salivary microbiota before and after starting chemotherapy, respectively (Figure 1A). At the genus level, the before group was characterized by 119 genera, of which 17 were absent from the after group, whereas the after group included 128 genera, of which 26 were absent from the before group. A total of 37 genera were present in at least 50% of the patients in both groups, with 23 genera being common to both. The predominant bacteria at the genus level (>10% of the total sequences in either group) included

Taxonomic composition of oral microbiota before and after the start of chemotherapy. The vertical bar plot shows the relative abundance of bacterial phyla (A) and genera (B).
Differences in Diversity Before and After the Start of Chemotherapy
Analysis of alpha diversity showed that the abundance and evenness of oral bacteria decreased significantly in the after group compared with those in the before group (OTU index,

Alpha and beta diversities before and after the start of chemotherapy. (A) Alpha diversity (operational taxonomic unit [OTU] and Shannon’s indices) of before (green) and after (red) chemotherapy initiation groups is shown as box-and-whisker plots. The
Patients were divided into 2 groups according to age (over and under 65 years), denture use, and periodontal status (mild and severe). Differences in the oral microbiota before and after the start of chemotherapy were compared. Based on the unweighted and weighted UniFrac distances, there were no differences observed between the groups in terms of the changes in the oral microbiota before and after the start of chemotherapy (Supplemental Figure S1A-C). Furthermore, examining whether the extent of change before and after the start of chemotherapy differed by disease revealed that the change was significantly smaller in the ML and MM groups compared with that in the AML group (
Oral Bacteria with Different Abundances Before and After the Start of Chemotherapy
Oral bacteria with different abundances before and after the start of chemotherapy were identified using the LEfSe analysis. The cladogram in Figure 3A represents the taxa that differed significantly between the groups in a taxonomic hierarchy from phylum to genus. At the genus level,

Bacterial abundance before and after chemotherapy initiation, assessed based on linear discriminant analysis (LDA) effect size. (A) Cladograms of differentially abundant bacterial taxa in the before (green) and after chemotherapy initiation (red) groups. The central point represents the root of the tree (bacteria), and each ring represents the next lower taxonomic level (phylum to genus: D1, phylum; D3, class; D3, order; D4, family; D5, genus). The diameter of each circle represents the relative abundance of the taxon. (B) Histogram of the LDA scores for differentially abundant bacterial taxa at the genus level between the before (green) and after (red) chemotherapy initiation groups. LDA scores ≥3.0 are shown.
Changes in the Abundance of Specific Bacteria Before and After the Start of Chemotherapy
Twenty-five bacterial genera showed significant changes in their abundance before and after the start of chemotherapy (RM ANOVA test,
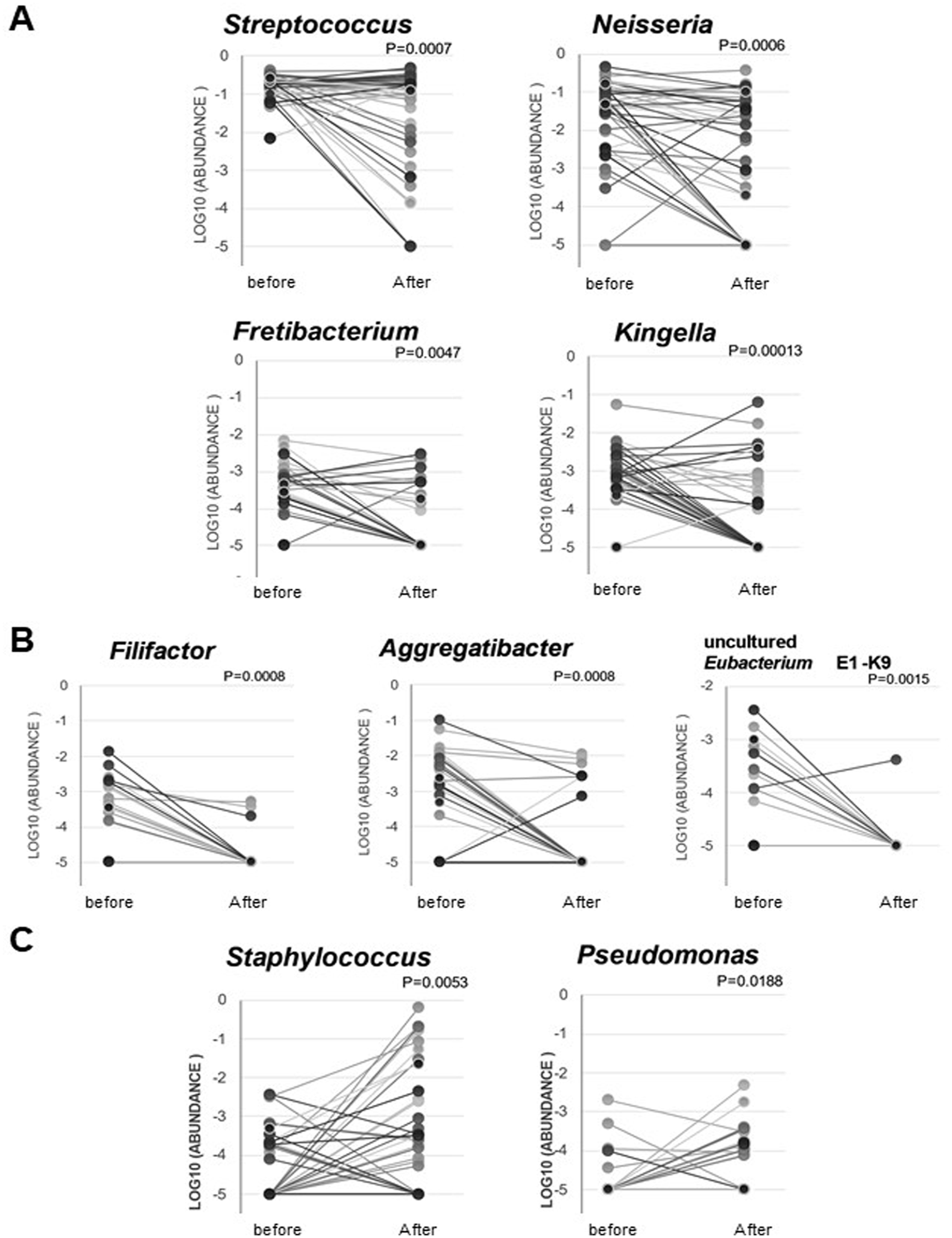
Changes in the abundance of specific bacteria before and after the start of chemotherapy. (A) Changes in the 4 representative bacterial genera whose abundance changed significantly before and after the start of chemotherapy. The
Oral Microbiota Associated with the Development of Oral Mucositis
Bacterial composition before and after chemotherapy initiation was compared between the group of patients who developed oral mucositis on days 8to 20 after chemotherapy initiation (OM) and those who did not (NonOM). The number of days between the start of chemotherapy and sample collection ranged from 8 to 20 days, and information on individual patients is presented in Supplemental Table 1. The change in microbiota composition before and after the start of chemotherapy was not different between the OM and NonOM groups. Before chemotherapy initiation, the OM group had a lower OTU index than did the NonOM group (
The LEfSe analysis showed that before the start of chemotherapy, the OM group had a lower abundance of

Bacterial abundance of the oral mucositis and non-oral mucositis groups, assessed based on linear discriminant analysis (LDA) effect size. Histogram of the LDA scores for differentially abundant bacterial taxa at the genus level between the groups of patients who developed oral mucositis after chemotherapy initiation (OM, red) and those who did not (NonOM, green) (A) before and (B) after the start of chemotherapy. LDA scores ≥3.0 are shown.
Discussion
In the present study, we characterized the changes in the oral microbiota composition associated with the initiation of chemotherapy for hematopoietic malignancies using 16S rRNA metagenomic analysis. The overall diversity of the oral microbiota decreased, and the bacterial composition changed after the start of chemotherapy. A decrease in the diversity of oral microbiota due to chemotherapy for hematopoietic malignancies has been reported in children with leukemia 22 and in patients undergoing autologous hematopoietic stem cell transplantation, 23 consistent with the results of our study. Furthermore, recent reports have shown that changes in the gut 5 and oral microbiota during chemotherapy for leukemia are associated with the risk of infection during treatment.24-26 Therefore, changes in the oral microbiota composition associated with chemotherapy for hematopoietic malignancies need to be investigated more comprehensively in relation to adverse events.
In the present study, the abundances of the predominant oral commensals
Chemotherapy for patients with hematopoietic malignancies causes oral-related adverse events, such as oral mucositis, cheilitis, xerostomia, and taste disorders.29,30 Oral mucositis can cause severe pain, eating disorders, and sleep disturbances, which may affect the completion of chemotherapy.
19
However, the causes and pathogenesis of oral mucositis remain unclear.
15
The microbiota has been reported to play an important role in chemotherapy-induced mucosal damage in the digestive tract.31,32 The characteristics of the oral microbiota involved in chemotherapy-induced oral mucositis have recently been reported.17,33 In the present study, 6 of the 57 patients developed oral mucositis 8 to 20 days after the start of chemotherapy, and we identified the characteristic oral microbiota in these 6 patients. Patients who developed oral mucositis had a higher abundance of
There are some limitations associated with our study’s results. First, the participants in this study varied in the type of malignancy. Moreover, even for the same disease, the types of chemotherapy varied. As the type of malignancy and the type of chemotherapy may affect the changes in the oral microbiota, we consider that another study is warranted in the future to investigate the differences in the oral microbiota based on these factors. Second, in the present study, we could not evaluate the effect of antimicrobial exposure on the oral microbiota. Antimicrobial exposure changes gut microbiota composition, whereas oral microbiota is relatively stable 25 ; therefore, this should be investigated in future studies by assessing more cases. Finally, the age range of the patients in this study was wide, from 16 to 87 years old. As age is a factor that affects the oral microbiota, since the dietary and oral habits and oral health behaviors vary with age, it is necessary to consider age in future research. In addition, as the patients were under oral management from the time of oral sample collection before the start of chemotherapy, we cannot rule out the possibility that this may have influenced the results of our study. Furthermore, we did not investigate background diseases or conditions, and their influence cannot be thus excluded. Although our study approach is limited in its ability to investigate all the confounding factors, we consider the present study to be a preliminary study. The present study provides a basis for further research on oral microbiota changes following the initiation of chemotherapy in patients with hematopoietic malignancies and to identify the characteristics of the oral microbiota associated with oral mucositis.
In the future, further evaluation of the effects of chemotherapy on oral microbiota will be possible by studying local factors such as salivary flow and systemic factors such as the degree of myelosuppression caused by chemotherapy, as well as other factors such as the type of medication and other treatments that may affect oral microbiota, in a more homogeneous cohort.
Conclusions
We characterized the changes in the oral microbiota associated with the initiation of chemotherapy in patients with hematopoietic tumors. In addition, we characterized the oral microbiota associated with oral mucositis. The results of this study suggest that focusing on the oral microbiota is crucial for understanding the pathogenesis of chemotherapy-associated adverse events such as oral mucositis. These findings will contribute notably toward predicting, preventing, and ameliorating adverse events in patients with cancer undergoing chemotherapy.
Supplemental Material
sj-tif-2-ict-10.1177_15347354231159309 – Supplemental material for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies
Supplemental material, sj-tif-2-ict-10.1177_15347354231159309 for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies by Michi Omori, Nahoko Kato-Kogoe, Shoichi Sakaguchi, Eri Komori, Kazuya Inoue, Kayoko Yamamoto, Wataru Hamada, Tomoyoshi Hayase, Tomoyuki Tano, Shota Nakamura, Takashi Nakano, Hidenori Une and Takaaki Ueno in Integrative Cancer Therapies
Supplemental Material
sj-tif-3-ict-10.1177_15347354231159309 – Supplemental material for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies
Supplemental material, sj-tif-3-ict-10.1177_15347354231159309 for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies by Michi Omori, Nahoko Kato-Kogoe, Shoichi Sakaguchi, Eri Komori, Kazuya Inoue, Kayoko Yamamoto, Wataru Hamada, Tomoyoshi Hayase, Tomoyuki Tano, Shota Nakamura, Takashi Nakano, Hidenori Une and Takaaki Ueno in Integrative Cancer Therapies
Supplemental Material
sj-tif-4-ict-10.1177_15347354231159309 – Supplemental material for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies
Supplemental material, sj-tif-4-ict-10.1177_15347354231159309 for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies by Michi Omori, Nahoko Kato-Kogoe, Shoichi Sakaguchi, Eri Komori, Kazuya Inoue, Kayoko Yamamoto, Wataru Hamada, Tomoyoshi Hayase, Tomoyuki Tano, Shota Nakamura, Takashi Nakano, Hidenori Une and Takaaki Ueno in Integrative Cancer Therapies
Supplemental Material
sj-xlsx-1-ict-10.1177_15347354231159309 – Supplemental material for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies
Supplemental material, sj-xlsx-1-ict-10.1177_15347354231159309 for Characterization of Oral Microbiota Following Chemotherapy in Patients With Hematopoietic Malignancies by Michi Omori, Nahoko Kato-Kogoe, Shoichi Sakaguchi, Eri Komori, Kazuya Inoue, Kayoko Yamamoto, Wataru Hamada, Tomoyoshi Hayase, Tomoyuki Tano, Shota Nakamura, Takashi Nakano, Hidenori Une and Takaaki Ueno in Integrative Cancer Therapies
Footnotes
Acknowledgements
We gratefully acknowledge Cykinso Inc. for their support with the microbiome analysis in this research.
Authors’ Note
This work was partly presented at the 65th Congress of the Japanese Society of Oral and Maxillofacial Surgeons last November 13 to 15, 2020.
Author Contributions
Conceptualization and design of the study: all authors. Material preparation, data collection, and analysis: Michi Omori, Nahoko Kato-Kogoe, Shoichi Sakaguchi, Eri Komori, Kazuya Inoue, Kayoko Yamamoto, Tomoyoshi Hayase, and Tomoyuki Tano. Writing – original draft: Komori. Writing – review and editing: all authors. All authors have read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by the Private University Research Branding Project (2017-2019) of the Ministry of Education, Culture, Sports, Science, and Technology and the Japan Society for the Promotion of Science KAKENHI (Grant Numbers JP20K18713, JP20K09932, and JP20K18521).
Ethical Approval
All procedures conducted in studies involving human participants were performed in accordance with the ethical standards of the Ethics Committee of Osaka Medical College, approval no. 2145, Chugoku Central Hospital of the Mutual Aid Association of Public School Teachers, approval no. 1905-02, and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
