Abstract
In most cancer patients, chemotherapy-induced oral mucositis (OM) is a frequent side effect, leading to low quality of life and delay in therapy. The aim of this study was to evaluate the effects of
Introduction
During chemotherapy and/or radiotherapy, cancer patients suffer from many side effects including oral mucositis (OM), anorexia, and leukopenia. 1 OM is an inflammatory, cytotoxic, and painful side effect of chemotherapy and is characterized by erythema, atrophy, erosion, and ulcers in the oral mucosa. 2 The prevalence of OM is 40% to 70% among cancer patients receiving chemotherapy and radiation. Moreover, the incidence has been reported to be up to 98% when chemotherapy is combined with radiation.1,3 OM often results in treatment plan extension with reduction in drug dosage, discontinuation of the treatment, limitation of the success of cancer chemotherapy, and finally a decrease in survival rate of cancer patients.4,5
There have been many studies on the management of chemotherapy- or radiation-induced OM in cancer patients, such as benzydamine,6,7 corticosteroids,
2
1-palmitoyl-2-linoleoyl-3-acetyl-rac-glycerol,
8
and natural products including herbal mixtures (eg,
In this study, to investigate the effects of
Materials and Methods
Preparation of Onchung-eum Water Extract
Crude herb of
High-Pressure Liquid Chromatography Analyses of Herbal Samples
The high-pressure liquid chromatography (HPLC) system consisted of Alliance 2690 Separation Module (a Waters 996 Photodiode Array Detector and a Millenium32 Chromatography Manager Version 3.2). Nucleosil C18 column (4.0 mm × 250 mm ID, Macherey-Nagel, Germany) was used as the reversed-phase column.
The mobile phase used for the analysis of geniposide, paeoniflorin, ferulic acid, and decursin was 0.1% acetic acid in water (A) and 0.1% acetic acid in acetonitrile (B); the gradient elution was set as follows: 5 minutes, 5% B; 15 minutes, 19% B; 25 minutes, 80% B; and 35 minutes, 100% B. The column temperature was maintained at 25°C. The flow rate was set at 1.0 mL/min and the injection volume was 10 µL. The ultraviolet (UV) detection wavelength was set at 240 nm. The sample (200 mg) for HPLC analysis was extracted with methanol (10 mL) by ultrasonic wave and filtered through a 0.45-µm membrane filter. The levels of geniposide, paeoniflorin, ferulic acid, and decursin were 1.45%, 1.39%, 0.05%, and 0.06%, respectively (Figure 1A and B).

HPLC analysis of each herb. (A) Geniposide, paeoniflorin, ferulic acid, and decursin in Gardeniae fructus, Paeoniae Radix, Cnidii Rhizome, and Angelica Gigantis Radix: (a) standard, (b) sample. (B) Baicalin, baicalein, and wogonin in Scutellariae Radix: (a) standard, (b) sample. (C) Berberine in Phellodendri cortex and Coptidis rhizome: (a) standard, (b) sample.
The mobile phase used for the analysis of baicalin, baicalein, and wogonin was methanol–0.1 M phosphoric acid (600:400, v/v). The column temperature was maintained at 50°C. The flow rate was set at 1.0 mL/min and the injection volume was 10 µL. The UV detection wavelength was set at 280 nm. The sample (200 mg) for HPLC analysis was extracted with methanol (10 mL) by ultrasonic wave and filtered through a 0.45-µm membrane filter. The content of baicalin, baicalein, and wogonin were 4.97%, 0.17% and 0.04%, respectively (Figure 1).
The mobile phase used for the analysis of berberine was 1/15 M monopotassium phosphate–acetonitrile (500:500, v/v) containing 0.1% sodium lauryl sulfate. The column temperature was maintained at 30°C. The flow rate was set at 1.0 mL/min and the injection volume was 10 µL. The UV detection wavelength was set at 340 nm. The sample (200 mg) for HPLC analysis was extracted with methanol (10 mL) by ultrasonic wave and filtered through a 0.45-µm membrane filter. The level of berberine was 1.77% (Figure 1).
Measurement of DPPH Radical Scavenging Activity
The mixtures comprising each concentration of OCE (1, 5, 10, 50, or 100 µg/mL) and 0.1 mM 2,2-diphenyl-1-picrylhydrazyl (DPPH) solution with methanol were incubated in the dark for 30 minutes at room temperature. After 30 minutes, the absorbance at 517 nm was measured for each sample on a microtiter plate reader (Molecular Devices, Sunnyvale, CA). Radical scavenging activity was calculated as follows:

where AB is the absorbance of the blank sample and AT the absorbance of the tested extract solution.
Measurement of Cell Viability and Proliferation
For the cell viability experiment, the human pharyngeal cell line Detroit 562 (ATCC CCL-138) was purchased from American Type Culture Collection (Manassas, VA). The Detroit 562 cell line was cultured in Modified Eagle’s Medium (Gibco-BRL, Grand Island, NY) supplemented with 10% fetal bovine serum (Gibco-BRL, Grand Island, NY), 100 U/mL penicillin, and 100 µg/mL streptomycin at 37°C in a humidified atmosphere of 5% CO2.
The cell proliferation experiment was performed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. For 24 hours, the Detroit 562 cells were starved and simultaneously treated with each concentration of OCE (1, 5, 10, 50, or 100 µg/mL) or with each concentration of OCE plus 10 µM 5-FU. After 48 hours, the medium was removed and the cells were incubated with MTT to measure metabolic activity. To measure metabolic activity, spectrophotometric analysis at 450 nm was performed using a microtiter plate reader (Molecular Devices, LLC, Sunnyvale, CA).
Measurement of ROS Production
ROS production was analyzed according to the manual of the Intracellular ROS Assay Kit (Cell Biolabs, San Diego, CA). Cells were cultured in a 96-well cell culture plate and treated with each concentration of OCE (1, 5, 10, 50, or 100 µg/mL) plus 10 µM 5-FU for 48 hours. Next, the cells pretreated with 100 µM 2′,7′-dichlorofluorescein diacetate (DCFH-DA) were incubated for 60 minutes at 37°C. The cell fluorescence was read on a fluorometric plate reader (Thermo Fisher Scientific, Inc, Waltham, MA) at 480/530 nm. ROS production was determined by comparison with the predetermined DCF standard curve.
Animals and Ethics
Seven-week-old male golden Syrian hamsters (100-110 g of body weight; SLC Inc, Hamamatsu, Japan) were used. The hamsters were housed in a specific pathogen-free environment with a 12-hour light-dark cycle at the Center for Laboratory Animal Care and Use at Kyung Hee University. The animal care and experimental procedures (KHUASP(SE)-12-031) conformed to the Guide for the Care and Use of Laboratory Animals.
Induction of OM
Using a modified protocol based on a previously established method, OM was induced. 14 In brief, all the hamsters received 80 mg/kg 5-FU intraperitoneally (ip) on day 0, followed by 60 mg/kg 5-FU ip on day 2. On days 3 and 4, the cheek pouch of the hamsters was everted and the mucosa was irritated by superficial scratching with the tip of an 18-gauge needle.
Grouping
The hamsters were randomly divided into 6 groups: normal group (vehicle-treated, n = 6), control group (80 mg/kg 5-FU, ip, n = 6), positive control group (0.15% benzydamine HCL, po, n = 6), and experimental groups treated with 3 concentrations of OCE plus 5-FU (100, 500, or 1000 mg/kg OCE plus 80 mg/kg 5-FU, po, n = 6 per group). The allocation of hamsters in all groups was conducted according to the order of hamsters’ body weight at the beginning of the experiment.
5-FU was administrated on days 1 and 2 of the experiment. OCE and benzydamine HCL were administered for 5 days/week for 2 weeks. On a weekly basis, the hamsters were weighed to adjust the gavage volume and monitor their general health.
Histological Evaluation of OM
The cheek pouches were fixed overnight in Bouin’s solution, dehydrated in 70%, 80%, 95%, and 100% ethanol and xylene, and embedded in paraffin. For hematoxylin-eosin (H&E) staining, 5 µm tissue sections were prepared. The sections were deparaffinized and rehydrated in xylene and 70%, 80%, 95%, and 100% ethanol. The sections were overstained with hematoxylin for typically 3 to 5 minutes and the excess stain was rinsed off with deionized water. Next, the sections were destained for a few seconds in acidic alcohol until they were red in color (usually 4-5 dips) and rinsed briefly with deionized water to remove the acid. Hematoxylin-stained slides were rinsed from the last tap water and placed in 70% ethanol for 3 minutes. Slides were placed in eosin for 2 minutes and exposed to 95% and 100% ethanol and xylene, respectively. After H&E staining, slides were mounted with Canada balsam.
TUNEL Assay
Cell death induced by 5-FU was measured on day 14 by using the TUNEL assay (ApopTag, No. S7101, Merck Millipore, Darmstadt, Germany). In brief, the samples were rehydrated and incubated with 20 mg/mL proteinase K for 15 minutes at room temperature after deparaffinizing. Endogenous peroxidases were blocked by treating with 3% (v/v) hydrogen peroxide in phosphate-buffered saline (PBS) for 5 minutes at room temperature. After washing, the sections were incubated in a humidified chamber at 37°C for 1 hour with TdT buffer containing TdT enzyme and reaction buffer. Specimens were incubated for 10 minutes at room temperature with a stop/wash buffer and incubated in the humidified chamber for 30 minutes with anti-digoxigenin peroxidase conjugate at room temperature. After a series of washes with PBS, the slides were covered with peroxidase substrate for color development and washed in 3 changes of dH2O and counter stained in 0.5% (w/v) methyl green for 10 minutes at room temperature. The TUNEL-positive cells were counted (10 fields per slide; ×1000) for statistical comparisons.
Western Blotting
Proteins from homogenized cheek pouches were separated using a nuclear extraction kit following a modification of the manufacturer’s manual (Active & Motif, Carlsbad, CA). Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and western blotting were conducted as previously described. 15 Samples for protein extraction were half of the same cheek pouches used for RNA extractions. Equivalent amount (50 µg) of protein extracts were separated in 10% Tris-glycine gels by SDS-PAGE and transferred to nitrocellulose membranes using 25 mM Tris and 250 mM glycine buffer containing 20% methanol (pH 8.3). Transfer was conducted at a constant voltage of 120 mA for 1 hour. After transfer, the membranes were blocked in PBS containing 0.05% Tween-20 with 5% skim milk for 2 hours at room temperature and incubated with primary antibodies (1:1000) for IL-1β, TNF-α, nuclear factor-κB (NF-κB), and caspase-3 (all from Santa Cruz Biotechnology, Inc, Dallas, TX) in PBS containing 0.05% Tween-20 overnight at 4°C. After overnight incubation, the membranes were rinsed with 1 × PBS thrice and incubated with conjugated goat anti-rabbit IgG (immunoglobulin G) for 1 hour at room temperature, followed by 3 additional washes with 1 × PBS.
Immunohistochemistry
For immunohistochemical staining, the samples were serially sectioned into 5-µm sections. Antigen retrieval was performed using citrate buffer (pH 6.0) preheated in a steamer for 25 minutes. The primary antibodies, sources, dilutions, and incubation times were as follows: IL-1β (1:100), TNF-α (1:100), NF-κB (1:100), and caspase-3 were incubated overnight. The sections were then incubated with diaminobenzidine tetrahydrochloride (Novocastra, Newcastle, UK) and counterstained with hematoxylin. Sections containing normal skin served as positive controls for the immunohistochemical reactions.
Statistical Analysis
For statistical analysis, GraphPad Prism 4.0.3 software (GraphPad Software, Inc, San Diego, CA) was used. All data are presented as the mean with standard error of the mean and 1 way-ANOVA (analysis of variance) with Dunnett’s post hoc test was used for comparison with control group. A
Results
OCE Increases DPPH Radical Scavenging Activity
The DPPH radical scavenging activity of OCE increased to 2.7%, 4.8%, 4.5%, 18.8%, and 29.8% at 1, 5, 10, 50, and 100 µg/mL, respectively. The IC50 value of OCE was 167.7 µg/mL. The activity of the positive control ascorbic acid increased to 22%, 53%, 54%, 59%, and 63% at 1, 5, 10, 50, and 100 µg/mL, respectively. The IC50 value of ascorbic acid was 4.72 µg/mL (Figure 2).

DPPH radical-scavenging activity of
OCE Enhances Proliferation of Human Pharyngeal Cell Line
The cell viability in the OCE-treated groups (10, 50, and 100 µg/mL) significantly increased compared with the normal group (119.4%,

Effect of OCE on the proliferation of human pharyngeal cell. Human pharyngeal cells treated with OCE were incubated in the absence (A) or presence (B) of 10 µm 5-FU at 37°C for 48 hours. Normal is PBS-treated cells. 5-FU is only 5-FU–treated cells. Each column represents the mean ± standard error. * indicates the mean is significantly different from the normal value (*
OCE Decreases Intracellular ROS Production in Human Pharyngeal Cell Line
ROS production in the 5-FU–treated control group was significantly higher than that in the normal group (668.6 vs 16.5,

Effect of OCE on 5-FU–induced ROS production of human pharyngeal cell. Each column or point represents mean ± standard error. * indicates the mean is significantly different from the normal value (**
OCE Protects Histological Damages in Cheek Pouches of Hamsters
A normal arrangement of cellular components in the cheek pouches of hamsters was observed in the normal group (Figure 5A). However, stratum corneum exfoliation, epithelial layer degradation, and ulcers were observed in the control group (Figure 5B). The damaged epithelial layers of positive control (Figure 5C), and 500 and 1000 mg/kg of OCE-treated groups (Figure 5E and F) recovered.

Examination of histological changes in cheek pouches of hamsters with hematoxylin and eosin staining. (A) Normal, (B) control (5-FU), (C) positive control (benzydamine HCl), (D) 100 mg/kg of OCE + 5-FU
OCE Decreases Apoptotic Changes in Cheek Pouches of Hamsters
In the cheek pouches of hamsters of the control group, apoptotic changes significantly increased (Figure 6B) compared with the normal group (Figure 6A). However, in the positive control group (Figure 6C) and 100, 500, and 1000 mg/kg OCE groups treated with 5-FU (Figure 6D-F), the epithelial and granular layers recovered.

TUNEL assay in the cheek pouches of hamsters. (A) Normal, (B) control (5-FU), (C) positive control (benzydamine HCl), (D) 100 mg/kg of OCE + 5-FU
OCE Decreases Expression of IL-1β, TNF-α, NF-κB, and Caspase-3 in Cheek Pouches of Hamsters
Western blotting was performed to determine the effects of OCE on pro-inflammatory cytokines (IL-1β and TNF-α), NF-κB, and caspase-3 expression in 5-FU–induced OM. The expression of NF-κB in the control group increased compared with the normal group (118.34%,

Effects of OCE on the expression of NF-κB, IL-1β, TNF-α, and caspase-3 proteins in cheek pouches of hamsters. (A) Western blots showing the protein level. (B) The western blot bands were quantified by normalization to those from the vehicle-treated groups. Each column represents the mean ± standard error. * indicates the mean is significantly different from the normal value (*
Cleaved caspase-3 protein level in the control group increased compared with the normal group (139.23%,
TNF-α and IL-1β protein levels in the control group increased compared with the normal group (120.38%,
OCE Immunohistochemically Decreases Caspase-3–Positive Cells in Cheek Pouches of Hamsters
In the normal group, caspase-3–positive cells were not observed in the granular layer (Figure 8A-C). However, many caspase-3–positive cells were detected in the control group (Figure 8D-F). In the granular layer of the positive control group, a marked decrease in caspase-3–positive cells was observed (Figure 8G-I). Similarly, in OCE-treated groups, caspase-3–positive cells decreased in the granular layer (Figures 8J-R).
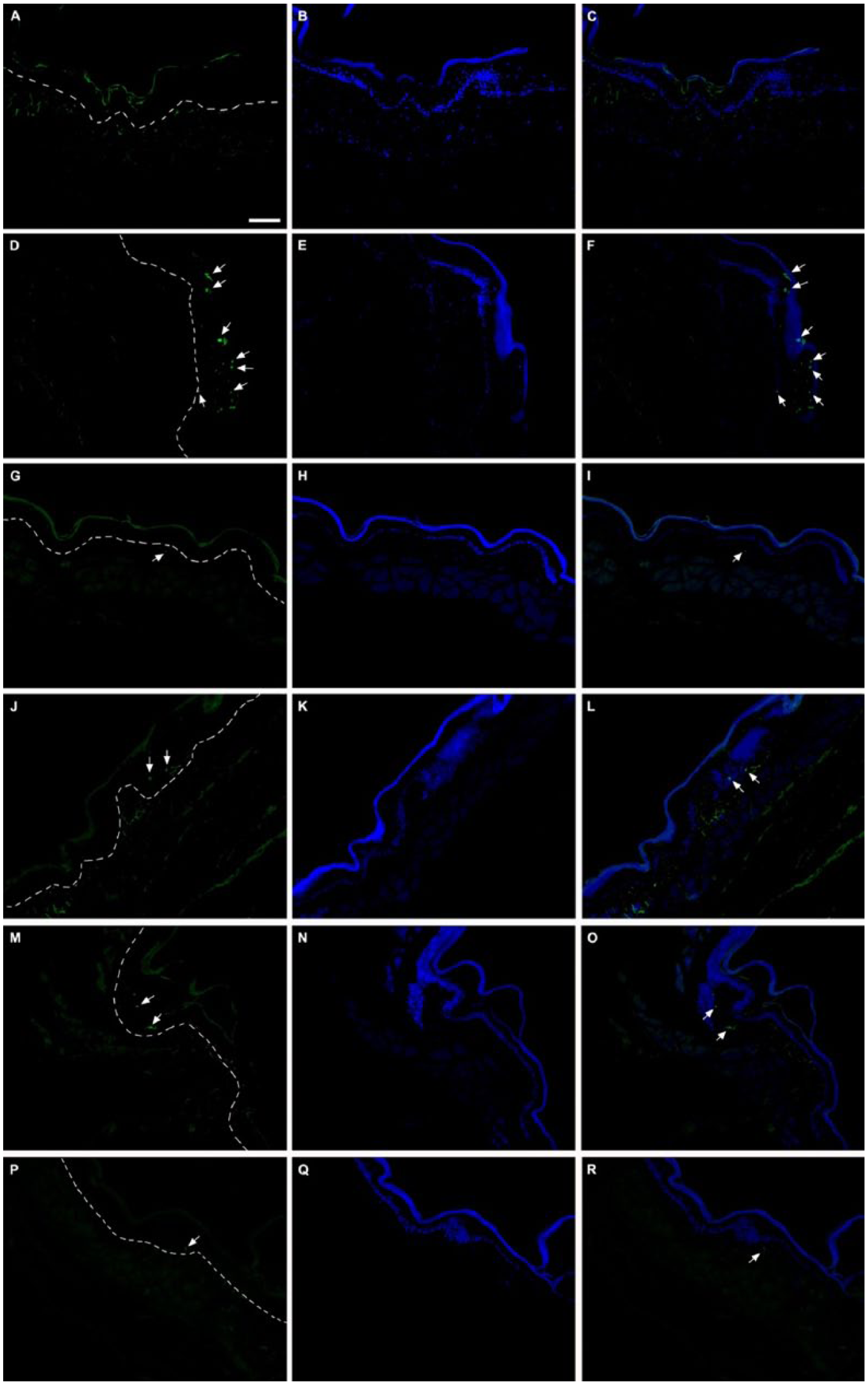
The immunofluorescence analysis of caspase-3 expression in the cheek pouches of hamsters. Caspase-3 was observed as positive form with dots (green color: left line) in the granular layer of buccal mucosa. All samples were counterstained with DAPI (blue color: middle line) and overlay of caspase-3 and DAPI (right line). (A-C) Normal; (D-F) control; (G-I) positive control; (J-L) 100 mg/kg of OCE + 5-FU–treated group; (M-O) 500 mg/kg of OCE + 5-FU
Discussion
Our in vitro study demonstrated that OCE increased DPPH radical scavenging activity and improved 5-FU–induced decrease in human pharyngeal cell viability by decreasing ROS production in cells. In addition, OCE improved 5-FU–induced histological damages and apoptotic changes in the epithelial layer of the cheek pouches in hamsters in vivo. OCE significantly decreased pro-inflammatory cytokines, such as IL-1β and TNF-α, NF-κB, and caspase-3 in the cheek pouches of hamsters. In particular, 1000 mg/kg OCE showed the maximal protective effect against 5-FU–induced OM in the cheek pouches of hamsters.
5-FU is a representative chemotherapeutic agent for breast, colon, and skin cancers, which suppress thymidylate synthase or incorporation of nucleic acid into RNA and DNA, arrests mitotic cell cycle, decreases mitosis and regeneration of cell, and induces inflammation or cell deaths. 16 In particular, OM, a well-known side effect of chemotherapy, in cancer patients usually occurs 4 to 7 days after the initiation of chemotherapy and reaches the maximum severity; in comparison, radiation-induced OM is relatively delayed in initiation. 17 Although spontaneous recovery in OM is observed 2 to 3 weeks after the cessation of chemotherapy, the frequency and toxicity of OM becomes severe with repeated chemotherapies and finally the quality of life decreases due to unbearable pain, discontinuation of chemotherapy, or nutritional imbalance. 18
There are 5 phases in the development of OM as initiation, primary damage response, signal amplification, ulceration, and healing. 19 In the initiation phase, basal epithelium and submucosa directly sustain damages by DNA strand breaks and ROS is produced. 20 In the primary damage response phase, transcription factors, such as p53 and NF-κB, are activated and approximately 200 genes are upregulated. 20 In particular, the expression of NF-κB, a well-known gatekeeper of inflammation pathway in OM, induces pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 and increases the expression of cyclooxygenase-2 in submucosal fibroblasts and endothelial cells.19,21 During signal amplification phase, MMP-1 (matrix metalloproteinases) and MMP-3 are activated by pro-inflammatory cytokines, including TNF-α and IL-1β.20,22 Then, erythema and ulceration develop. In the ulceration phase, mucosal integrity deteriorates, giving rise to painful ulcers. 20 In addition, the expression of pro-apoptotic gene increases and apoptosis occurs. After the cessation of chemotherapy, spontaneous healing of acute OM is observed, which is associated with the proliferation of epithelial cells or hematopoietic recovery. In particular, the regeneration of damaged epithelium is induced by migration, proliferation, and differentiation of epithelium. 23
MTT assay, DPPH radical scavenging activity, and ROS production demonstrated that OCE increased cell viability through its high antioxidant activity. The antioxidant effect may have been an important mechanism that inhibited direct epithelial and mucosal damages during the initiation phase of OM induced by 5-FU. OCE is a mixed herbal prescription of “
In this study, OCE demonstrated significantly inhibited the expression of NF-κB and production of pro-inflammatory cytokines including TNF-α, IL-1β, and IL-6. In previous studies, baicalin (present in
To investigate the anti-apoptotic effect of OCE in terms of NF-κB and caspase-independent apoptotic pathway, an immunoblotting experiment using human pharyngeal cell and hamster cheek pouches treated with 5-FU was conducted. Our TUNEL assay revealed that the number of apoptosis-positive cells in OCE-treated groups markedly decreased in a dose-dependent manner (Figure 6).
Apoptosis is divided into intrinsic and extrinsic pathways according to the control of pro- and anti-apoptotic proteins. 33 In addition, cytochrome C combines with apoptotic protease activation factor 1 and caspase-9 to produce apoptosome and activate caspase-3 to induce apoptosis. 33 To determine the effect of OCE on intrinsic apoptotic signal pathway caused by various stimulants, such as DNA damage, cytokines, or ROS, the expression of caspase 3 was investigated by western blotting and immunofluorescence analysis (Figures 7 and 8). The expression of cleaved caspase-3 (higher levels in apoptotic signal pathway) in OCE-treated groups significantly decreased compared with 5-FU–induced control group, which is comparable with the effect of benzydamine-treated group as a standard treatment group. This indicates that OCE exhibits protective effect against 5-FU–induced apoptotic changes in the oral mucosa, at least at the protein level.
OCE showed beneficial effects on oral ulcers in Behcet’s disease, recurrent aphthous ulcers, psoriasis vulgaris, neurodermatitis, and skin pruritus.11,13 OCE and its components showed positive effects in skin regeneration and as antiwrinkle, whitening, and skin moisturizing agent in previous in vitro and in vivo studies.12,34 Thus, in this study, it was hypothesized that OCE could improve chemotherapy-induced OM. Although the markedly protective effect of OCE against epithelial damage and ulcers in the cheek pouches of hamster was identified based on the observation of histological findings and immunohistochemical staining in this experiment, the evaluation of histological findings was not a quantitative method and there is no established assessment for histological damage. Therefore, further research for the establishment of quantitative method on histological findings is required.
Benzydamine, a positive control in this study, has been used for reducing chemotherapy- or radiation-induced OM and the associated pain across multiple studies.6,7,35 In addition, the use of benzydamine in cancer patients treated with moderate radiation dose was reported to reduce the rate of erythema and ulceration and the use of analgesics significantly. 36 In this study, the effects of OCE, particularly at high concentrations, was also comparable with that of benzydamine in terms of antioxidative and protective effects against histological damages, apoptotic changes, pro-inflammatory cytokines, and transcription factors of OM models. Thus, OCE may be applicable to the entire phase of 5-FU-induced OM including the prevention of OM.
According to general clinical usage of
Conclusions
The present study demonstrated that OCE improved cell viability and showed protective effects against oxidative stimulants. Furthermore, OCE inhibited pro-inflammatory cytokines and apoptosis in the oral mucosa. Thus, OCE could be a potential candidate originating from natural products for the treatment of OM, which is a frequent side effect in cancer patients receiving chemotherapy. However, further details about their molecular mechanisms and pharmacokinetics will be required for clinical application.
Footnotes
Authors’ Note
This research was based on the data for the doctoral degree course of Jayoung Oh.
Author Contributions
Jayoung Oh and Jae-Woo Park were the major contributors in writing the manuscript. Jinsung Kim and Mun Seog Chang conceptualized the project. Jayoung Oh and Mun Seog Chang investigated the experiment. Seok-Jae Ko, Jayoung Oh, and Mun Seog Chang analyzed the data and played a role in writing the draft. Jinsung Kim was the project leader. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
