Abstract
Treatment fidelity is the use of methodological strategies to monitor and enhance reliability and validity of behavioral intervention trials. Despite availability of guidelines and checklists, treatment fidelity remains underreported, hindering evaluation, interpretation, and cross-study comparisons. Treatment fidelity is particularly important for music interventions given the inherent complexity of musical stimuli and flexibility required for tailored delivery. The purpose of this paper is to define and describe treatment fidelity strategies for our trial of a music-based play intervention for young children with cancer and parents grounded in the NIH Behavior Change Consortium Treatment Fidelity Recommendations. We report strategies for all 5 areas: study design, training providers, delivery of treatment, receipt of treatment, and enactment of treatment skills. We also discuss 4 challenges our team encountered, including: (1) standardizing live music delivery, (2) defining boundaries for tailored intervention delivery, (3) managing extended time between participants, and (4) minimizing risk for bias. This paper expands on current fidelity literature and may provide a working model for other investigators examining dyadic and/or active music interventions.
Introduction
Treatment fidelity is important to the design and conduct of rigorous clinical research. In 2004, the National Institutes of Health Behavior Change Consortium (NIH BCC) defined treatment fidelity as “. . .methodological strategies used to monitor and enhance the reliability and validity of behavioral interventions. [Treatment fidelity] also refers to the methodological practices used to ensure that a research study reliably and validly tests a clinical intervention.” 1 These practices help to ensure that changes seen post-treatment can be attributed to the intervention under investigation rather than external factors and reduce the chances of Type I and II errors. 2 To encourage uptake and use of these recommendations, consortium authors published a validated treatment fidelity checklist for use in trial design and evaluation, 3 with an updated checklist 2 published in 2011.
Despite NIH BCC guidelines and checklist availability, treatment fidelity continues to be under-reported in published research.1-3 Prior reviews indicate that publishing authors often report details about treatment delivery, while other areas such as treatment receipt and enactment are rarely reported.4,5 In addition, there is inconsistent use of standardized NIH BCC treatment fidelity definitions and reporting categories, which contributes to these observed inconsistencies in reporting. 5 The resulting absence and inconsistency of reporting make evaluation, interpretation, and cross-study comparisons difficult.
Treatment fidelity is especially important for music interventions given the inherent complexity of musical stimuli6,7 and flexibility required for delivery of tailored interventions. 8 A scoping review of music intervention reporting quality revealed that only 20% of identified studies described strategies to ensure consistent treatment delivery across participants and providers—emphasizing the need for publications that detail selection and use of treatment fidelity strategies for music intervention trials. 9 To date, there have been 3 publications detailing NIH BCC guideline implementation in music intervention trials.6,10,11 Implementation of the NIH BCC recommendations requires that investigative teams interpret, define, and select strategies that consider the unique context, attributes, and design features of their trial. As such, published manuscripts describing selection and implementation of specific fidelity strategies help other investigators increase rigor in their own trials.
The purpose of this paper is to define and describe treatment fidelity strategies being used in our multisite randomized controlled trial of a music-based play intervention for young children with cancer and their parents (R01NR019190). This manuscript expands on current fidelity literature and may provide a working model for other studies investigating dyadic and/or active music interventions.
Overview of the BIO-MUSE Trial
The Biologic Mechanisms and Dosing of Active Music Engagement (BIO-MUSE) trial is a 2-group single-blind randomized controlled trial examining the use of music play to manage distress and improve health outcomes in young children with Acute Lymphoblastic Leukemia (ALL) and their parents (R01NR019190). Children and one parent (enrolled as dyads) are stratified and randomized in blocks of 4 to the Active Music Engagement (AME) intervention or Audio-Storybooks (ASB) attention control condition. Child/parent dyads are stratified by child age (preschoolers 3-5 years.; school-agers 6-8 years.), site (Riley Hospital for Children at Indiana University Health, Indianapolis, Indiana; Ann & Robert H. Lurie Children’s Hospital, Chicago, Illinois; Children’s Mercy Hospital, Kansas City, Missouri), and ALL risk level (standard risk; high risk). Both the intervention and attention control conditions are standardized, and each group receives one 30-minute AME or ASB session during weekly outpatient clinic visits for the duration of the consolidation phase of ALL treatment (4 sessions for standard risk participants; 8 sessions for high-risk participants). The purpose of the BIO-MUSE trial is to examine biological mechanisms of effect and explore dose-response relationships of AME on child/parent stress during the consolidation phase of ALL treatment.
Methods
In this paper we describe treatment fidelity strategies specific to the BIO-MUSE trial and their grounding in the NIH BCC Treatment Fidelity Recommendations. 1 Table 1 provides definitions for each of the 5 NIH BCC Treatment Fidelity components. We then provide an overview for each component and rationale for selected strategies.
NIH BCC Components and Definitions.

Area 1: Study Design Strategies
Treatment fidelity strategies specific to study design help ensure that the investigative team can adequately test their proposed hypotheses in relation to underlying theory and clinical processes. Selected strategies are unique to each study and increase rigor by ensuring that: (1) interventions are congruent with relevant theory and clinical practice; (2) dosing is consistent within and across study conditions; and (3) implementation setbacks are minimized. 1 Table 2 summarizes the goals of treatment fidelity specific to study design, with corresponding strategies for the BIO-MUSE trial. In the sections that follow, we elaborate on our selection of study-specific strategies.
Treatment Fidelity Strategies for Study Design.

Goal 1: Ensure Interventions are Congruent With Relevant Theory and Clinical Experience
In theory-driven research design, the selected theory informs 3 aspects of study design: 1) selection of intervention content (ie, essential elements), 2) specified relationship between independent and dependent variables, and 3) selection of outcome measures. The NIH BCC guidelines specify that the theoretical model underlying the intervention must be clearly described, with subsequent checklists created to support implementation.1-3
Theory-informed essential content
In previous publications, we have outlined how the AME intervention aligns with the Contextual Support Model of Music Therapy (CSM-MT 12 ) which is grounded in Self-Determination Theory. 13 The CSM-MT explains how music can be used to create a supportive environment by offering optimal levels of structure, autonomy support, and relationship support. 15
In brief, we know that attributes of the environment play an important role in whether an individual perceives a situation to be stressful or benign, and how they respond behaviorally to that situation (engagement vs disaffection).12,16-22 Engagement is a necessary precursor to learning and enactment of positive coping strategies, such as the use of music play to manage cancer-related stress.18-21,23 The AME intervention was designed to mitigate qualities of stressful environments, like cancer treatment.12,17,18,24 Stressful environments are unpredictable, constrain autonomy, and often lack or strain social support. In contrast, supportive environments are structured, provide opportunities for choice/control, and offer relationship support. Through tailored delivery of shared music experiences, the AME intervention creates a supportive environment using familiar music play (structure), child-directed experiences (autonomy support), and supportive child/parent interaction (relationship support).12,16,17 Supportive environments lead to higher engagement and subsequent acquisition and use of positive coping strategies.12,16,17,19-21 Music play is particularly viable for children ages 3 to 8 years because young children cope with stressful experiences through play, 25 and music can offer a sense of normalcy through shared, meaningful experiences that focus on healthy aspects of the child and family.26-28 Table 3 shows the relationship of essential AME intervention content to CSM-MT theoretical principles.
Active Music Engagement Intervention Components and Theoretical Principles.*.

Table reprinted with permission in accordance with creative commons open access license “Attribution-NonCommercial 4.0 International” (CC BY-NC 4.0) http://creativecommons.org/licenses/by-nc/4.0/ for the following publication: Russ KA, Holochwost SJ, Perkins SM, et al. Cortisol as an acute stress biomarker in young hematopoietic cell transplant patients/caregivers: active music engagement protocol. JACM. 2020;26(5):424- 434.
Attention control design
We have previously established the ASB condition as a feasible and acceptable control condition for patients and families.12,16,17 The ASB condition controls for (1) audio-visual stimulation; (2) presence of a trained provider; and (3) shared activity between parent and child.
Conceptual framework and measures selection
Our conceptual framework is based on Robb’s Contextual Support Model of Music Therapy,12,18 Self-Determination Theory, 13 and further informed by Kazak and Baxt’s Pediatric Medical Traumatic Stress Model, 29 which provides a useful heuristic for understanding short- and long-term consequences of pediatric cancer treatment for parents and their children. These theories, and the resulting model, directly informed our selection of covariates, mediators, and outcome measures.
Goal 2: Ensure Same Treatment Dose Within and Across Conditions
Our BIO-MUSE protocol specifies condition frequency, duration, and schedule for participants in both conditions. Regardless of condition, children with standard risk ALL receive 4 weekly sessions during the consolidation phase of treatment, whereas children with high risk ALL receive 8 weekly sessions. The number of sessions is based on Children’s Oncology Group (COG) approved treatment protocols, which vary based on ALL risk level. The decision to see participants for the full duration of their treatment is grounded in ethical and practical considerations, including justice and non-maleficence. Providing support for the full treatment course for standard risk participants and for only half of the treatment course for high-risk participants would be inadequate and potentially harmful.
The NIH BCC guidelines recommend equivalent dosage between study conditions. In previous AME trials, the AME and ASB conditions were delivered in an in-patient setting and had an equal duration (45 minutes, with 30 minutes of music or stories). The current trial takes place in the outpatient clinic setting. To accommodate clinic flow and patient needs we needed to limit session duration to 30 minutes or less. As such, total session duration for AME sessions is 30 minutes total (5 minutes collaborative goal setting; 20 minutes of music-play; 5 minutes educational content). Total session duration for ASB is 20 minutes (5 minutes set-up; 15 minutes audio-storybooks—the length of one storybook). Although total session length is not equivalent, the amount of audio-visual stimulation is similar across groups, with the additional 10 minutes in AME attributed to assessment and educational activities that are unique to that condition. In addition to in-person sessions, participants take home a music-play or audio-storybooks kit to encourage between-session use of the condition-related activities.
We monitor dose (frequency, duration) through provider-completed field notes that include the session date, session begin/end times, and parent-reported kit use between sessions. Providers also audio- or video-record sessions which provides an external validity check for data reported in the provider field note. We also perform self and external quality assurance monitoring which are explained in greater detail below (see Area 2—Goal 2: Ensure Provider Skill Acquisition).
Goal 3: Plan for Implementation Setbacks
The BIO-MUSE protocol specifies plans for a variety of possible implementation setbacks. Examples include training multiple music therapists and data collectors at each site to minimize the impact of absence/illness or staff turnover and providing paper copies of measures to data collectors in the event of technology failure. Our team also meets weekly to discuss ongoing participants and any issues with protocol implementation, which are then addressed with input from the principal investigator (PI), site PIs, and core project manager.
Area 2: Training Providers
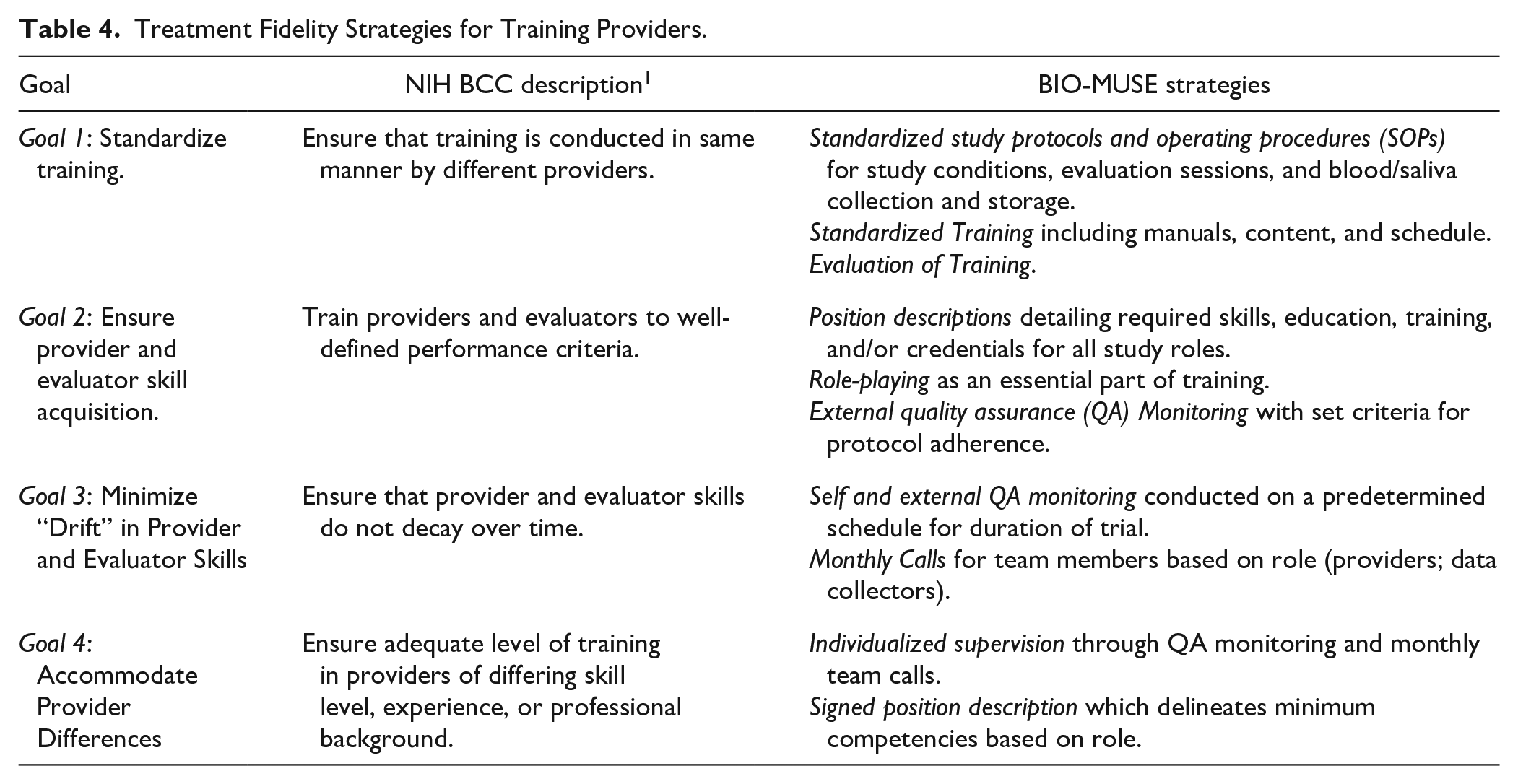
The use of standardized training programs helps to ensure consistent delivery of study protocols across providers and sites. These strategies are also important to address differences in providers’ educational training, clinical background, and experience. Table 4 summarizes provider training goals and corresponding strategies for the BIO-MUSE trial.
Treatment Fidelity Strategies for Training Providers.
Goal 1: Standardize Training
Our providers and data collectors are trained on condition-specific protocols and SOPs during training sessions led by the PI and core project manager. Training session agendas, content, and protocols are standardized to ensure consistent training over the course of the trial. We have training manuals specific to each role including providers, site PIs, and data collectors. All team members receive the same 1 hour of core content including the study overview, theoretical framework, clinical overview of ALL treatment, responsibilities and communication, identification and reporting of adverse events, online database file management procedures, and strategies to minimize bias when discussing the study. Provider and data collector manuals also include standardized protocols for delivering study conditions or conducting data collection. Site PIs have additional SOP requirements for providing study oversight including Single IRB requirements, recruitment, informed consent, billing, and coordination of study activities. Total initial training time varies based on role (4 hours for data collectors; 6 hours for site PIs; 10 hours for providers). At the conclusion of training, we ask team members to complete evaluations to improve future training sessions.
Goal 2: Ensure Provider Skill Acquisition
Our providers are board-certified music therapists who have a minimum of a bachelor’s degree in music therapy and experience working in pediatric medical settings. Our data collectors are hospital employees or research assistants in a variety of roles including clinical research assistants, nurse practitioners, and undergraduate students. We have position descriptions for all study-related roles, specifying minimum requirements for education, training, and experience, as well as competencies, responsibilities, and skills needed to fulfill the role. The position description establishes the minimum skills required and communicates expectations. All team members receive a role-specific position description at the start of their training and sign the position statement agreement after initial training is complete.
Role-play is an essential feature of our training which helps to ensure skill acquisition. We also use self and external QA monitoring to establish whether team members are delivering essential elements of the study protocol with predetermined thresholds for initial and sustained protocol adherence. During QA monitoring, providers (and data collectors) listen to their audio-recorded sessions and fill out session-specific checklists listing essential content that should be delivered for a specified session. An external monitor also listens to sessions and completes the same session-specific checklist. The threshold for initial protocol adherence is >80% of all QA items during the last session of a provider’s second AME participant, during the last session of a provider’s first ASB participant, or during the last session of a data collector’s first participant. We monitor 2 AME participants to establish initial adherence due to the increased complexity of AME sessions. External QA monitors schedule individual meetings to discuss any missed items or challenges observed during monitoring. If a provider or data collector does not meet the initial adherence threshold by the indicated timeline, self and external QA monitoring continues until the threshold of 80% or more of QA items has been met.
Goal 3: Minimize “Drift” in Provider Skills
Once providers and data collectors achieve the initial adherence threshold, we continue to engage in self and external QA procedures using a priori monitoring schedules to minimize drift. The schedule for sustained monitoring specifies completion of self and external monitoring for every fifth participant (or 60 days, whichever comes first). All sessions are audio-recorded to allow for unscheduled monitoring as needed. In addition, all providers and data collectors attend monthly role-specific meetings to discuss study progress, protocol implementation, and ongoing participants. Meetings are essential to communicating study updates, building a collaborative team environment, addressing common challenges, sharing solutions, and promoting protocol adherence over time.
Goal 4: Accommodate Provider Differences
Providers receive individualized instruction during training and individualized mentoring through ongoing quality assurance monitoring. External QA monitoring facilitates identification of any provider differences that might require additional training and monitoring. The signed position description (described above) also helps to ensure that providers meet minimum specified requirements (education, certification, experience) and agree to deliver study conditions or conduct data collections sessions according to study protocols.
Area 3: Delivery of Treatment
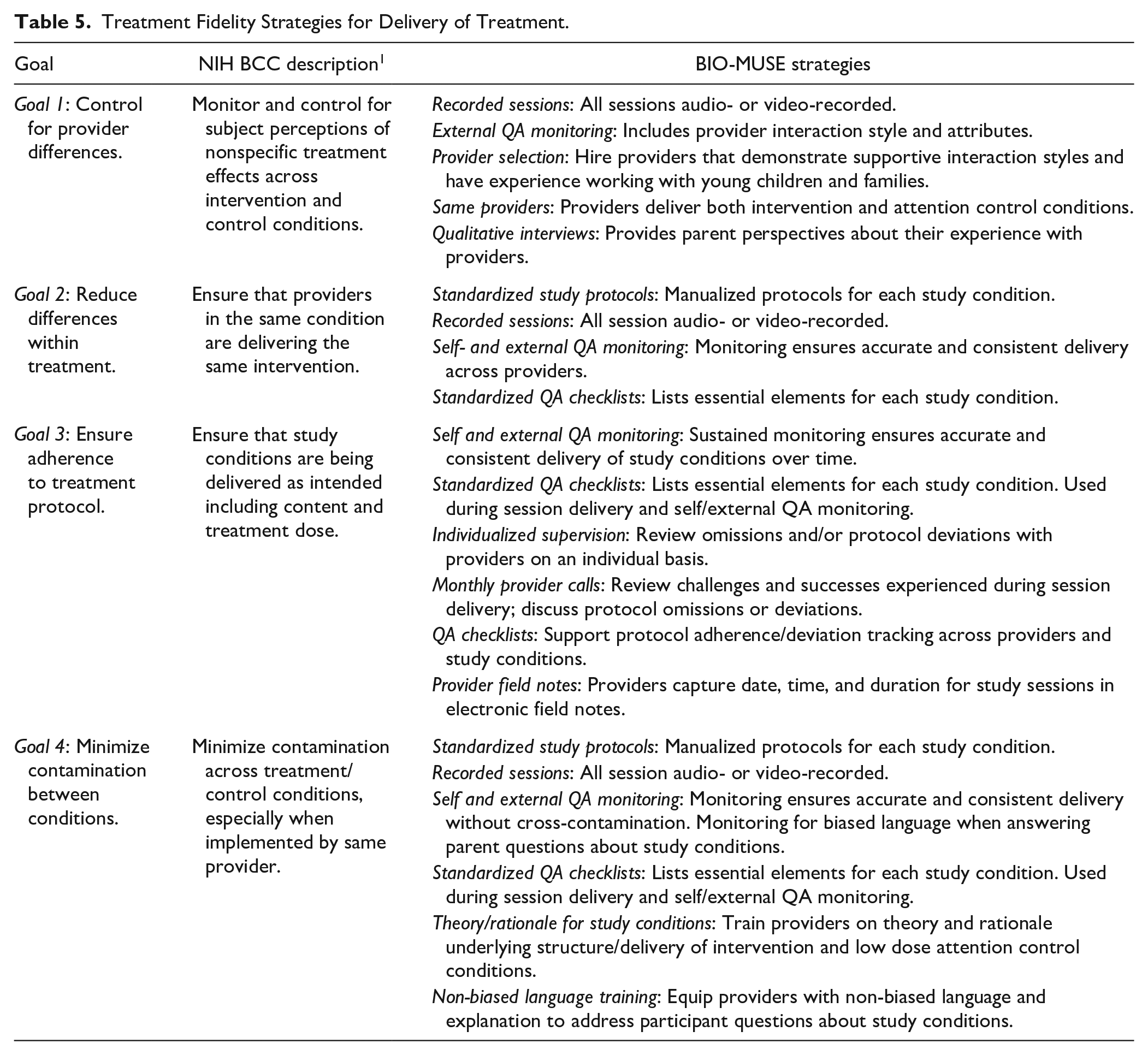
Treatment delivery strategies help ensure that study conditions are delivered as intended. Primary strategies include the use of standardized treatment protocols and sustained monitoring of provider delivery during the trial, with additional strategies to reduce within-treatment differences and contamination between study conditions. Table 5 summarizes treatment delivery goals and corresponding strategies for the BIO-MUSE trial.
Treatment Fidelity Strategies for Delivery of Treatment.
Goal 1: Control for Provider Differences
We use several strategies to monitor and control for nonspecific treatment effects that may result from provider differences in terms of their perceived warmth or credibility. As described earlier, all provider sessions are audio- or video-recorded to facilitate QA monitoring. In addition to protocol adherence, external monitors also look at variations in provider delivery. When hiring study personnel, we look for providers with experience working with young children and parents in a hospital setting, and who demonstrate a supportive interaction style that embodies inclusivity and cultural sensitivity. In addition, we use the same providers across study conditions to help control for therapist attributes and conduct qualitative interviews with parents that include questions about their experience with the provider.
Goal 2: Reduce Differences Within Treatment
BIO-MUSE providers use manualized intervention protocols and session-specific checklists during condition delivery. The session checklists contain suggested language for information delivery, reminders about specific intervention content, and a step-by-step guide to session flow. Checklists help ensure consistent and complete delivery of content identified as essential to each study condition. Differences within treatment are also monitored during self and external QA monitoring.
Goal 3: Ensure Adherence to Treatment Protocol
Protocol adherence is monitored through our previously described QA monitoring procedures and session-specific checklists. The external QA monitor reviews any omissions and/or protocol deviations with providers on an individual basis. Self and external QA checklists are computerized and completed in our Research Electronic Data Capture (REDCap 30 ) study database (which is compliant with the Health Insurance Portability and Accountability Act, or HIPAA). This feature allows us to generate real-time reports and easily track protocol adherence across sessions and providers. Providers also complete a study field note for each session that includes treatment dose information (ie, session date; session start/end times). The fieldnote is a part of our REDCap study database allowing us to generate reports to identify protocol deviations related to timing and duration of intervention delivery.
Goal 4: Minimize Contamination Between Conditions
We elected to use the same providers for intervention and attention control conditions. This was done to help control for provider differences across conditions, but this also creates opportunities for contamination (ie, essential elements of intervention appearing in attention control sessions) and risk of providers introducing bias that favors the intervention. In addition to sustained and rigorous QA monitoring, our providers receive training on the theories and scientific rationale that informed the content, structure, and delivery of the intervention and attention control conditions. Provider training also includes scenarios where unintended bias might occur and how to address participant questions about the study or their assigned condition. For example, when describing our study, we share that we are interested in learning how play activities like stories and music might be helpful during cancer treatment—positioning AME and ASB as equal experiences.
Area 4: Receipt of Treatment
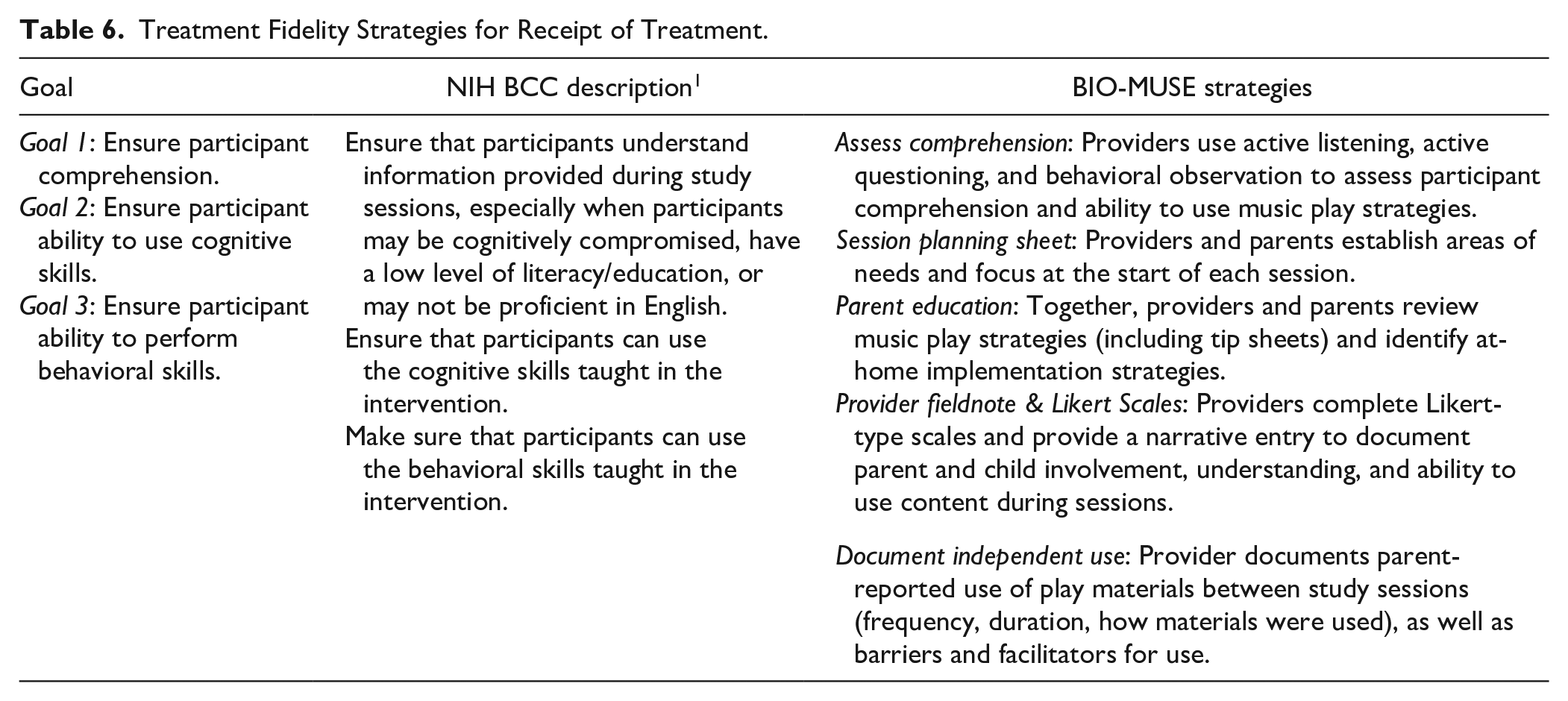
Fidelity of treatment receipt involves strategies to ensure that the treatment delivered is received by the participant. 2 Receipt involves participants’ understanding and their ability to use and demonstrate skills learned in treatment or control conditions. Limited understanding or ability to perform skills presented during treatment and control sessions will interfere with evaluation of treatment effects. As such, strategies for treatment receipt center on assessing comprehension and ability to use treatment-related skills. Table 6 summarizes treatment receipt goals and corresponding strategies for the BIO-MUSE trial.
Treatment Fidelity Strategies for Receipt of Treatment.
Goals 1 to 3: Ensure Participant Comprehension, Ability to Use Cognitive Skills and Ability to Perform Behavioral Skills
We train providers to use active listening, active questioning, and behavioral observation to assess parent and child comprehension of intervention content. At the start of each session, providers work with parents to identify areas of need for their child through verbal dialog and use of a planning sheet. This supports parent-identification of needs for their child and positions the provider as a collaborator. At the close of sessions, providers (1) reference standardized tip sheets; (2) engage parents in a reflective dialog about what worked during the session (based on planning sheet conversation); and (3) identify strategies the parent and child can try using at home or in the hospital. Parents are given copies of tip sheets to reinforce concepts and children are given a music-play kit to encourage enactment of music play strategies outside of study sessions. After sessions, providers complete an electronic fieldnote that includes Likert-type scales to capture parent and child engagement and a narrative entry detailing behavioral observations, participant questions, and any events that may influence parent/child understanding and use of music play strategies (eg, session interruptions, presence of other clinic staff).
Area 5: Enactment of Treatment Skills
Enactment of treatment skills involves strategies to assess, monitor, and improve participants’ ability to perform and use treatment-related skills (cognitive and behavioral) in settings beyond study sessions. Borelli 2 describes enactment as “. . .an important addition to the treatment fidelity model because a distinction is made between what is actually taught (treatment delivery), what is learned (treatment receipt) and what is actually used (enactment).” Table 7 summarizes enactment of treatment goals and corresponding strategies for the BIO-MUSE trial.
Treatment Fidelity Strategies for Enactment of Treatment Skills.

Goals 1 to 2. Ensure Participant Use of Cognitive and Behavioral Skills
As mentioned under Area 4, providers use parent education materials (ie, standardized tip sheets) and engage parents in reflective dialog about music play strategies, what works for their child, and ways to use the play materials at home (or in other settings) to manage stress. This individualized and cooperative approach encourages active reflection and use of materials outside sessions. Children are given a music play kit to further encourage use of music play strategies to manage distress at home or in the hospital. Music-play kits include recorded music, instruments, activity cards, and materials used during provider-directed sessions. The kits were developed and refined during initial pilot testing to establish their acceptability and use. 17 At the close of each session providers encourage self-directed use of play materials (although it is never framed as a requirement). Providers monitor and document use of materials between study sessions (frequency, duration, how materials were used), along with any barriers and facilitators for use. Finally, we conduct qualitative interviews with parents at study conclusion. Interviews provide more detailed descriptions about parent and child use of music-play strategies and materials outside study sessions, and identification of facilitators and barriers to use.
Discussion
The NIH BCC guidelines were developed to improve the design and rigor of complex behavioral interventions. 1 Music intervention trials are especially complex due to the compositional features of music stimuli (rhythm, tempo, pitch, harmonic structure, timbre may affect outcomes differentially), variety of music experiences (active music making, music listening, songwriting), and other factors unique to music interventions.6,7 Here we describe some of the greatest challenges our team has encountered in both design and early implementation of fidelity strategies for the BIO-MUSE trial including: (1) standardizing live music delivery; (2) defining boundaries for tailored intervention delivery; (3) managing extended time between participants; and (4) minimizing risk for bias.
Standardizing Live Music Delivery
In contrast to recorded music listening, the use of live music introduces greater potential for variability in delivery across study participants and providers. In the BIO-MUSE trial we use pre-composed music activities with embedded improvised content that is tailored to meet the unique needs of the child and parent. To reduce variability, we selected and composed age-appropriate activities for use during sessions, specified strategies to facilitate improvised content, and trained providers on theoretical principles to guide tailored delivery. To facilitate learning and consistent “performance” of the music activities, we provide sound recordings, music notation, and detailed descriptions for each activity in our protocols. During training, activities are modeled using live demonstration and example videos and practiced to establish competency. AME activities also use an improvisational approach to parent/child music-making. To help standardize and support this improvisational approach we use specific strategies to structure and support child/parent musical interactions. For example, we use “call and response” formats, steady rhythms for grounding, narrate parent/child actions through improvised lyrics, and rhythm matching.
Defining Boundaries for Tailored Intervention Delivery
Tailored interventions have been defined as those that rely on clinical judgment and adapt treatment to meet individual needs.31,32 The AME intervention is tailored in 3 ways. First, the intervention uses collaborative goal setting at the start of sessions. This provides focus for activity selection and tailored use of music to meet identified goals. Second, the intervention includes a range of activities to support variations in child energy and distress while supporting child autonomy through meaningful choices. The pacing, physical activity requirements, and qualities of the music activity can be adjusted to match immediate needs and modulated over time to achieve desired goals. Finally, therapists are trained to use theoretical principles of Self-Determination Theory 13 and the CSM-MT 12 to guide clinical decision making as they tailor the intervention (Table 3).
Determining how much flexibility can occur in a standardized protocol is challenging. 32 Consistent with recommendations from O’Malley and Qualls, 32 we specify a basic structure for sessions including duration with recommendations for time spent in collaborative goal setting, active music-making, and parent education. Time spent in each area may vary based on parent or child needs. Variations are monitored and documented.
To build therapist skills in tailored delivery, O’Malley and Qualls32 recommend support and expert consultation during trial implementation. For our team, this involves sustained individualized QA monitoring from a board-certified music therapist experienced in the protocol and monthly provider meetings led by the PI. Individual and group meetings often focus on therapists’ decision-making processes as they tailor the AME experience and “defining boundaries” of the intervention. These conversations, along with session-specific fidelity checklists, help solidify theory-informed delivery and increased confidence with the tailoring process.
Managing Extended Time Between Participants
An additional challenge is managing extended time between participant accruals and the resulting need for increased self and external QA monitoring. 11 The incidence rate of ALL is relatively low, 33 affecting how frequently our providers deliver study protocols and results in the need for more frequent QA monitoring. This places greater demands on therapists’ time and increases costs for compensation of therapists and external QA monitors.1,2,10,11,32 This also affects perceived burden by providers and study personnel that can lead to burnout. Our core group of music therapists have published on their experience of QA monitoring and its value for their own professional development. 34 However, it has been essential to engage in sustained conversations about the importance of QA monitoring and explore strategies to reduce burden.
Minimizing Risk for Bias
We identified potential risk for bias in how members of the healthcare team talk about our trial, sometimes referring to it as the music study. To address this concern, we refer to our study as the “Music & Stories Trial” in our brochures and during conversations with hematology/oncology professionals, clinical staff, patients, and families. In addition, we provide ongoing staff in-services that include information about the trial and recommended language to reduce bias when talking with families. Members of our study team are also trained to present and talk about both conditions using non-biased language during informed consent and conversations with medical staff and families.
In closing, we recommend investigative teams use the NIH BCC guidelines, along with related checklists, to guide the design and selection of treatment fidelity strategies during study conceptualization.1-3 We also recommend that investigators use published Reporting Guidelines for Music-based Intervention Trials to inform standardization of music experiences. 7 The reporting guidelines discuss attributes of music interventions that should be specified and reported in music trials—making it an informative resource as investigators work to specify essential features and standardize delivery of both live and recorded music interventions. Treatment fidelity is essential to research integrity. Though treatment fidelity requires time and added resources, the cost of not engaging in treatment fidelity strategies is far greater. 2 At the time of publication, we found a limited number of publications explicating treatment fidelity strategies for music intervention trials. Dissemination, implementation, and transparency about trial-specific fidelity strategies are important to advancing the field, and we hope our trial may provide a working model for other investigators examining dyadic and active music interventions.
Footnotes
Authors’ Note
Amanda K. Henley is also affiliated to Purdue School of Engineering and Technology, Indianapolis, IN, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The BIO-MUSE trial is supported by the National Institutes of Health-National Institute of Nursing Research R01NR019190 (Robb, PI).
