Abstract
Propolis is a resinous beehive product that has a wide range of biological activities, namely antimicrobial, antioxidant, and anti-inflammatory properties. Propolis is collected by the bees from plant resin and exudates to protect hives and maintain hive homeostasis. The aim of the present systematic scoping review is to explore the potential and suitability of propolis as an adjunctive treatment in breast cancers, based on the latest available experimental evidence (2012-2021). After applying the exclusion criteria, a total of 83 research publications were identified and retrieved from Scopus, Web of Science, and Pubmed. Several relevant key themes identified from the included studies were cytotoxicity, synergistic/combination treatment, improvement in bioavailability, human clinical trials, and others. A majority of the studies identified were still in the in vitro and in vivo stages. Nonetheless, we managed to identify 4 human clinical trials that demonstrated the successful use of propolis in alleviating side effects of chemotherapy and radiotherapy while increasing the quality of life of breast cancer patients, with minimal adverse effects. In conclusion, propolis, as an adjunctive treatment, may have therapeutic benefits in alleviating symptoms related to breast cancers. However, further clinical trials, preferably with higher number of participants/subjects/patients, are urgently needed.
Introduction
The burdensome nature of cancers is considered to be a challenging issue in modern healthcare and scientific discussions. Many epidemiological studies, psychosocial impact investigations, and economic burden analyses delineate the impact of cancers, not only on the medical and healthcare fields, but also its extensive influence on other important social structures.1,2 As an example, a report by National Cancer Institute in 2021 evaluated the US national economic burden for cancer care and found breast cancers were ranked first for top net economic burden with the total out-of-pocket cost and indirect expenditure of US$3.1 billion and $1.1 billion, respectively. 3 Additionally, a recent GLOBOCAN report had placed breast cancers as the most commonly diagnosed malignancy worldwide with more than 2.2 million new cases in 2020, accounting for 11.7% incidence of cancer worldwide, surpassing lung cancer rates, and also ranked first in term of cancer-related mortality in females. Statistically, breast cancer is found in 1 in 4 diagnosed cancer cases and 1 in 6 cancer-related deaths among women, which are remarkably alarming considering the changes of global pattern toward heavier cancer burden in upcoming years.4-6
Furthermore, the improvement in breast cancer management should be considered since the emerging issues of treatment-resistant breast cancers in certain subtypes notably the triple-negative breast cancer (TNBC). The therapeutic approaches often consist of a plethora of molecular/physiological targets, ranging from endocrine-based regiments, cytotoxic chemotherapy, radiotherapy, the targeted therapies such as the development of combination treatments.7-10 In addition, the anti-endocrine treatment is the principal approach in managing steroid-receptor positive breast cancers (ER+/PR+). However, the resistance toward these hormonal approaches due to genetic and/or epigenetic mutations may alter the ER gene expressions of the breast cancers and subsequently activates alternative signaling pathways to prevent estrogen blocking; treatment resistance will therefore eventually develop.10-12 The resistance issues toward conventional treatments illustrate the need to establish other therapeutic approaches, such as novel alternative or adjunctive treatments.
Although the earliest known written records of cancers were obtained from the ancient Egyptians’ manuscripts circa 1500 to 1600 BCE, and possibly were based on much older records; some notable advancements of cancer treatment options were only apparent after the early 20th century. Throughout the entire human history, the utilization of natural remedies to alleviate numerous diseases is unquestionably important and arguably safe to be implemented, and can be adapted to the current modern treatment strategies.13,14 Natural products have long been shown to be promising sources of anti-cancer compounds and/or therapeutics, for example, the Catharanthus roseus-derived vincristine and Taxus brevifolia-derived paclitaxel. 15 Propolis, or bee glue, with its diverse range of chemical compounds, has frequently been demonstrated to exhibit various biological activities, including immunomodulatory, anti-inflammatory, and anti-cancer properties.16-18 The aim of the present scoping review is to analyze the potential use and suitability of propolis as a supportive or adjunctive therapeutic substance for breast cancer management.
Methods
The present systematic review was performed in accordance with the guidelines provided by Peters et al 19 and Munn et al. 20 The guiding question was as follows: Can propolis be used as an adjunctive therapy in breast cancers? Two independent reviewers (F. Z. and K. C.) performed the search for articles dated January 2012 up to December 2021. The databases searched were Scopus, Pubmed, and Web of Science. Supplemental Table S1 shows the terms used in the search process. We intentionally did not include the terms that describe individual bioactive compounds of propolis such as caffeic acid phenethyl ester (CAPE), quercetin, galangin, kaempferol, and so on, as we focused on propolis as a whole. However, if during the search and screening process we encountered relevant studies that described the individual propolis bioactive compounds and met the inclusion criteria, we included them in the final list of the included studies. Only articles that were written in English were included. All articles that describe the potential use of propolis in treating breast cancers were selected; in vitro, in silico, animal models, and human clinical trials. However, we excluded any article that describes the use of synthetic derivatives of propolis bioactive compounds. The studies were recorded in Mendeley and the duplicates subsequently removed. We also excluded review articles as they might impart biases.
Subsequently, 2 reviewers (F. Z. and K. C.) assessed the search results independently. The articles were screened based on the titles, keywords, abstracts, and full texts. The articles that did not fit in the guiding question and the set criteria were then removed. If any disagreement arose on the eligibility of a particular article, the disagreement was resolved through discussion with another reviewer (D. H.). The following data were subsequently tabulated in Microsoft Excel: geographic locations of the propolis source and types of bees, types of propolis extract and/or propolis bioactive compounds, types of study, concentration of the propolis extract and/or bioactive compounds, outcome of the study, and references. The reviewers subsequently categorized the included studies based on the objectives of the studies into the appropriate themes.
Characteristics of the Studies
There were 307 scientific articles found in the initial search. The articles were initially screened based on the titles and abstracts. Further screening based on the full texts resulted in 83 articles. Figure 1 illustrates the screening process. Table 1 summarizes the themes, types of study, types of propolis extract and/or bioactive compounds, the measured outcome of the included studies, and the references. Figure 2 summarizes the characteristics of the included studies: percentages of the types of extract, the percentages of the study types, themes, and types of bees identified in the included studies.

The screening process of the studies adapted from Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA).
The Summary of the Included Studies Demonstrating the Potential Use of Propolis in Breast Cancers.
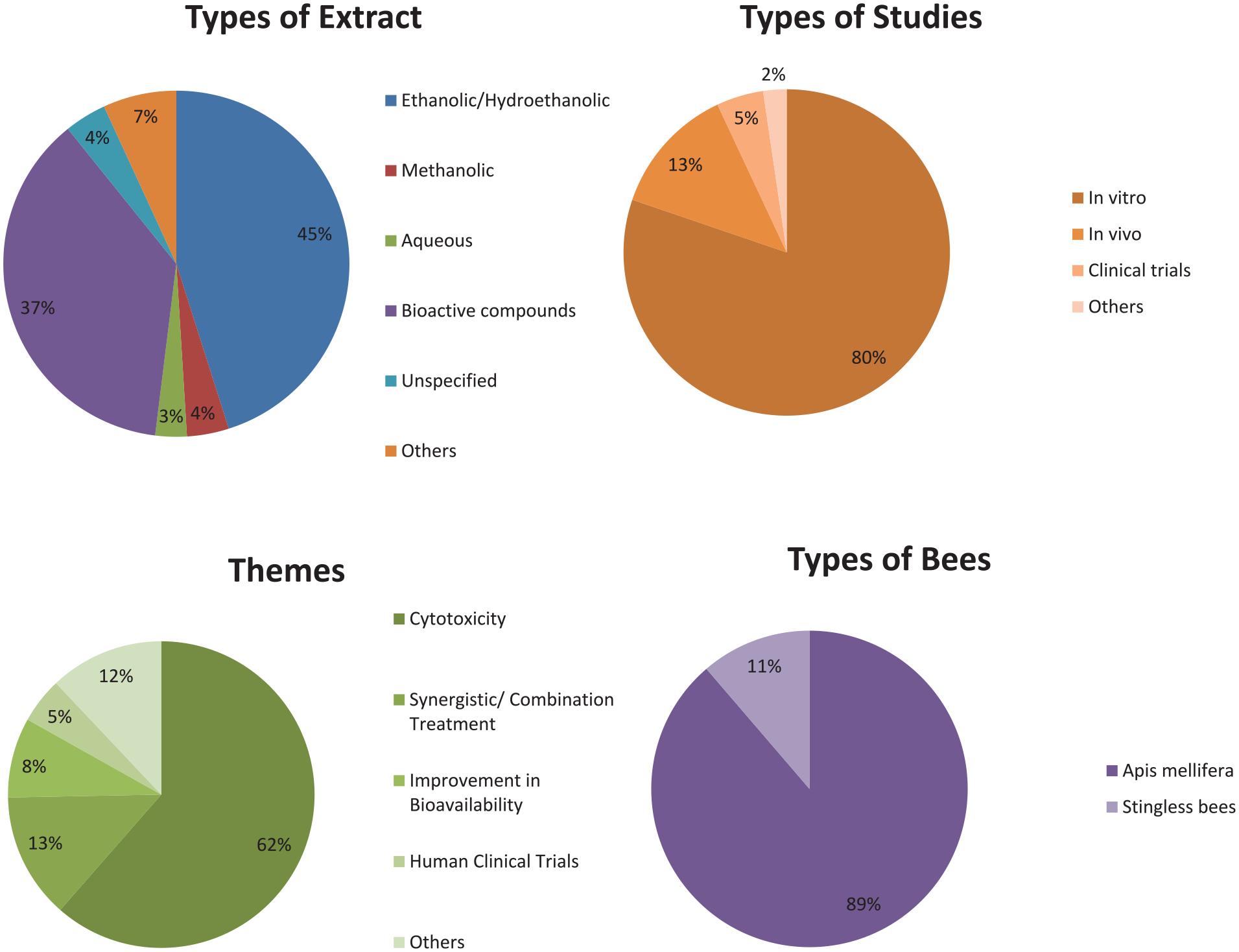
Characteristics of the included studies. (A) Types of extract. (B) Types of studies. (C) Themes. (D) Types of bees.
The largest body of experimental evidence in the included studies was cytotoxicity against breast cancers (61%), followed by synergistic/combination treatment (13%), improvement of bioavailability (8%), human clinical trials (5%), and others (12%). In terms of types of study, in vitro studies were the largest category at 80%, followed by animal models/in vivo (13%), human clinical trials (5%), and others (2%). The majority of the included studies identified the types of extract, geographical locations of the propolis source, and the bioactive compounds. Only 4% of the included studies did not identify the types of extract and/or the geographical locations where the raw propolis was sourced. All percentages were rounded to the nearest whole number. A majority of the studies utilize propolis sourced from Apis mellifera (89%) whereas 11% of the studies use propolis from stingless bees.
Cytotoxicity Against Breast Cancer Cells
Propolis has been shown to be cytotoxic against a wide range of breast cancer cells, namely BT-20, BT-474, BT-549, SKBR-3, MCF-7, MDA-MB-231, MDA-MB-436, T47D, Hs578T, and so on (Table 1). The mechanisms of action of propolis and its bioactive compounds with regards to breast cancer cytotoxicity have been extensively elucidated by various studies. The main mode of action appears to be through inducing apoptosis. Kamiya et al 22 demonstrated that ethanolic extracts of propolis and its bioactive compound, caffeic acid phenethyl ester (CAPE) induced apoptosis of MCF-7 breast cancer cells by upregulating caspase-3 activity, DNA fragmentation, and CCAAT/enhancer-binding protein homologous protein (CHOP) expression. Propolis also promotes mitochondrial dysfunction and endoplasmic reticulum stress. In addition, propolis induces apoptosis in MCF-7 and MDA-MB-231 cancer cells by upregulating the expression of Annexin A7 (ANXA7), reactive oxygen species (ROS) level, and NF-κB p65 level, while simultaneously reducing the mitochondrial membrane potential.31,45 More importantly, these studies demonstrate that propolis extracts have little cytotoxicity on normal human umbilical vein endothelial cells (HUVECs) and normal fibroblast cells.31,45
In addition, Chang et al demonstrated that propolis inhibited lipopolysaccharide (LPS)-stimulated MDA-MB-231 cell proliferation by inducing apoptosis through the upregulation of caspase 3 and poly (ADP-ribose) polymerase (PARP). They also showed that propolis induced autophagy by increasing the expression of LC3-II and reducing the expression of p62 level. Furthermore, propolis downregulates the inflammatory TLR4 signaling pathway molecules such as TLR4, MyD88, IRAK4, TRIF, and NF-κB p65. 42 This study reported contradictory results compared to studies by Asgharpour et al 45 and Xuan et al 31 where propolis was shown to promote inflammatory and oxidative stresses on the cancer cells whereby apoptosis was consequently induced. This suggests propolis may act differentially depending upon types of cancer and probably their stages .
Moreover, Frión-Herrera et al showed that propolis affected the expression of apoptosis-related genes in PI3K/Akt and ERK1/2 pathways, namely TP53, CASP3, BAX, and P21. They also found that propolis induced mitochondrial dysfunction and lactate dehydrogenase release indicating ROS-associated necrosis in MDA MB-231cancer cells. 104 Li et al demonstrated that propolis was able to inhibit the proliferation of MDA-MB-231 cells by targeting key enzymes of glycolysis, namely glycolysis-hexokinase 2 (HK2), phosphofructokinase (PFK), pyruvate kinase muscle isozyme M2 (PKM2), and lactate dehydrogenase A (LDHA), in an inflammatory microenvironment. There is also evidence that propolis affects immune response in the presence of breast cancers. 66 Kusnul et al 96 reported that propolis significantly reduced the relative number of CD4+, CD25+, FoxP3+ regulatory T cells expressing IL-10 and TGF-β in mice with breast cancer. The suppression of IL-10, which is an immunosuppressive cytokine, is thought to be beneficial in treating cancers.
Furthermore, Seyhan et al investigated propolis extracts from 3 countries, China, Argentina, and Turkey, and found that cytotoxicity against 3 cancer cell lines namely, MCF-7, SK-BR-3, and MDA-MB-231 did not correlate with the total phenolics and/or flavonoids but rather with the diversity of the phenolics and/or flavonoids, suggesting that the biological activities of propolis are due to the synergy of the bioactive compounds. 51 Nevertheless, several propolis-derived bioactive compounds, such as chrysin, caffeic acid phenethyl ester (CAPE), caffeic acid, quercetin, hesperidin, apigenin, naringin, myricetin, luteolin, galangin, artepillin C, pinocembrin, baccharin, cardanol, α-amyrin, and mangiferonic acid have been shown to have anti-breast cancer activities (Table 1).
Chrysin, a propolis bioactive compound, inhibits HDAC8 and significantly increases the expression of p21 (waf1/cip1) in breast cancer cells, leading to apoptosis. 24 Chrysin also inhibits the hypoxia-induced STAT3 tyrosine phosphorylation leading to the significant reduction of hypoxia survival rate of 4T1 breast cancer cells. 105 In addition, CAPE can reduce the malignancy of MDA-MB-231 cells by inducing changes in breast cancer stem cell characteristics such as inhibition of self-renewal, progenitor formation, and clonal growth, and reduction of CD44 content. 90 CAPE also has cytotoxicity activity against breast cancer cells through various mechanisms such as by promoting mitochondrial dysfunction and endoplasmic reticulum stress, 22 inducing cell cycle arrest in the in S, G0/G1, and G2/M phase,24,39 promoting the accumulation of acetylated histone proteins (epigenetic effects), downregulating the expression of estrogen receptor and progesterone receptor, 26 inducing autophagy and downregulating TLR4 signaling pathway molecules, 42 interfering with and inhibiting the voltage-gated sodium channels, 38 inhibiting mitochondrial oxygen consumption rate and mammosphere formation, 43 and inducing oxidative stress by promoting endothelial nitric oxide synthase (eNOS) and inducible nitric oxide synthase (iNOS) levels. 58
Buahorm et al demonstrated that a phenolic lipid cardanol, isolated from Thai propolis caused BT-474 cell apoptosis by inducing cell cycle arrest at the G1 subphase and cell death at late apoptosis stage and modulating the expression of genes related to apoptosis: upregulating the expression of DR5 and Bcl-2 (apoptosis regulator) and downregulating the expression of Mcl-1, MADD, and c-FLIPP. They also found that cardanol modulated the expression of genes related to cell division: it increased the expression of p21, E2FI, p21 p-ERK, p-JNK, and p-p38 and decreased the expression of cyclin D, cyclin D1, cyclin E, CDK4, and CDK2, resulting in the failure to progress from the G1 to the S subphase. 34 Moreover, other propolis-derived compounds such as genistein, luteolin, benzoic acid, quercetin, and vanillic acid, are shown, in silico and in vitro, to interfere with cancer-associated targets such as CYP1A1, CYP19A1, ESR1, NOS3, CASP3, and AKT1. 70
Combination Effects of Propolis With Other Anti-Cancer Treatments
In addition to affecting the cancer cells directly, propolis has also been demonstrated to work synergistically with other compounds. Tan and Hayati found that propolis decelerated the growth of mammary tumor in mice. However, the combination of propolis extract and mangostin had a more pronounced effect. The combination of propolis and mangostin significantly reduced the expression of Wnt2, FAK, and HIF-1α, when compared to propolis or mangostin alone. 75 Oršolić et al showed that propolis enhanced the tumor-inhibiting effect of cisplatin and improved the survivability of mice with Ehrlich ascites tumor (murine breast carcinoma). They also found that propolis increased the cytotoxic activity of macrophages to tumor cells and sensitivity of tumor cells to hyperthermal intraperitoneal chemotherapy (HIPEC). Interestingly, propolis also reduced cisplatin toxicity to normal cells. 81
Propolis has synergistic activity with doxorubicin, a standard drug for breast cancer. Alsherbiny et al 80 demonstrated that propolis significantly improved the proliferation inhibitory effect of doxorubicin in MCF-7 cells in a dose-dependent manner. Propolis also upregulated the expression of catalase, HTRA2/Omi, FADD, and TRAIL-associated DR5 and DR4 which significantly enhanced the cytotoxicity of doxorubicin in MCF-7 cells. They also found the differential expression in 21 proteins in the combination treatment compared to single treatments of either propolis or doxorubicin. The differentially expressed proteins were associated with TP53/ATM-regulated non-homologous end-joining pathway and double-strand breaks repairs, recruitment of overexpressed BRCA1, and the suppression of RIF1 encoded proteins. Perhaps more importantly, there was an overexpression of UPF2 in the combination treatment, indicating that it could potentially treat doxorubicin resistance-associated long non-coding RNA and the subsequent metastasis of the MCF7 cells. The study also elucidated the potential protective effect of propolis against the side effects of doxorubicin by reversing doxorubicin-mediated necrosis. 80
Moreover, propolis has synergistic activities with beneficial lactic acid bacterium Lactobacillus acidophilus LA-5. Onur et al demonstrated that propolis extract, L. acidophilus LA-5, and the combination of both treatments inhibited the tumor volumes by 59.16%, 28.29%, and 63.39%, respectively, when given to mice with murine breast carcinoma 4 T1. Propolis extract and the combination treatment upregulated the ConA-, LPS-, and PHA-induced splenocyte proliferation. Additionally, the combination treatment stimulated IFN-γ production. 82
Propolis bioactive compounds have also been shown to improve breast cancer therapies. CAPE improves the efficacy of radiotherapy by sensitizing breast cancer cells through impairing DNA damage repair mechanisms in cancer cells. 72 CAPE also works synergistically with tamoxifen, a selective estrogen receptor modulator, by significantly downregulating the levels of Bcl-2 and beclin-1, and endothelial growth factor and consequently inducing apoptosis. Notably, the combination of tamoxifen and CAPE increased the life span of the tumor-bearing mice compared to tamoxifen or CAPE alone. 72 Maasomi et al 76 demonstrated that chrysin acted synergistically with silibinin, a bioactive compound of Silybum marianum, in inducing cytotoxicity of T47D breast cancer cells through enhancing the downregulation of the expression of telomerase reverse transcriptase and cyclin D1.
Potential Improvement of Delivery Through Encapsulation
During the search process, we also found several interesting studies exploring the methods to improve the bioavailability of propolis and its bioactive compounds through encapsulation. Hasan et al demonstrated that by encapsulating propolis extract into nanoparticles, the effective concentration, to reduce the tumor size, heal tumor-associated wounds, and eliminate cancer cells of mammary gland tumors in rats, was significantly reduced from 233 to 32 µg/mL. However, in this particular study, it was not clear what encapsulation materials were used. 88 Kapare et al encapsulated ethanolic extract of propolis with poly (ε-caprolactone), a biodegradable polymer, into ~190 nm particles. They found that the concentration of the encapsulated propolis required for total growth inhibition of MCF-7 cancer cells in a designated time period was reduced by 33.06%, compared to non-encapsulated propolis. Furthermore, the solubility and sustained drug release were also enhanced. 89
However, Sherif et al reported a negative effect of nano-encapsulation of propolis in terms of cytotoxicity efficacy. It was shown that encapsulation using 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) liposomes actually increased the IC50 against MCF-7 breast cancer cells of hexane extract of propolis from ~222.4to 333.3 µg/mL. More significantly, the encapsulation using 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC) liposomes fully removed the cytotoxic effect of the propolis extract against MCF-7 cells. 87 These studies illustrate the need to extensively investigate the types of materials in the preparation of the propolis-infused nanoparticles/nanoencapsulation.
Additionally, several studies reported the improved biological activities of propolis bioactive compounds through encapsulation and/or other molecular complexes. Gamma-cyclodextrin appears to be a promising molecule for propolis bioactive compound complex formation. Wadhwa et al demonstrated that γ-cyclodextrin greatly enhanced the heat stability, chemical stability, and oxidative stability of CAPE. Gamma-cyclodextrin did not reduce the anti-cancer properties of CAPE. 86 Furthermore, nano-encapsulation of CAPE with sucrose fatty acid esters appears to enhance anti-cancer properties of CAPE. Guan et al found that nano-encapsulation of CAPE with sucrose fatty acid esters, polyethylene glycol, and/or thymol as co-surfactants enhanced the storage stability. The encapsulation also further enhanced the antioxidant and cytotoxicity of CAPE against MCF-7 cells. The authors postulated that the enhanced biological activities were due to better dispersion of nano-encapsulated CAPE in the aqueous solution compared to free CAPE. 83 A similar trend is also observed for another propolis bioactive compound, chrysin. Encapsulation using chitosan, poly (D,L-lactic-co-glycolic acid), and polyvinyl alcohol result in enhanced cytotoxicity against breast cancer cells.84,85
Human Clinical Trials
The most important thing for the development for any therapeutic is the translation of pre-clinical data to the application in humans. Propolis has been extensively studied for decades and therapeutically used for thousands of years as folk medicine. However, there is still relatively limited human clinical data, especially in the sphere of the treatment of cancers. In the present scoping review, we managed to identify 3 groups of researchers that performed human clinical trials investigating the effect of propolis in breast cancer patients.
Ebeid et al investigated the potential protective effect of propolis in breast cancer patients who were undergoing chemotherapy and radiotherapy. The total patients included in the clinical trial were 135 females who were divided into 3 groups. Group I (control group) consisted of 45 healthy females who were age and menopausal status-matched to the cancer patients in the subsequent groups. Group II consisted of 45 breast cancer patients who received chemotherapy followed by radiation therapy. Group III consisted of 45 breast cancer patients who received chemotherapy followed by radiation therapy and 400 mg propolis extract, 3 times daily for 10 consecutive days before radiotherapy and during the course of radiotherapy, and 10 days after completing the radiotherapy session. It was found that propolis alleviated the negative impact associated with radiotherapy in breast cancer patients, namely the increase in Comet tail parameters (Tail length, % Tail DNA, Tail moment) in peripheral blood mononuclear cells, and serum malonaldehyde (MDA). Propolis also prevented the decrease in total antioxidant capacity, hemoglobin (Hb) concentration, white blood cells (WBCs), and platelets counts associated with radiotherapy. More importantly, patients supplemented with propolis had significantly longer median disease free survival time. No adverse effect linked to propolis consumption was reported in this study. However, the study did not describe the randomization of the patients and any adverse effect experienced by the patients. Furthermore, the phytochemical analyses and the source of the propolis extract used in the study were not reported. 99
In addition, Piredda et al investigated the effect of propolis consumption in reducing the incidence of oral mucositis in breast cancer patients receiving chemotherapy, in a pilot randomized controlled trial with 60 patients. The intervention trial was carried out in the first chemotherapy cycle and lasted for 15 days. The included patients were randomized into a control group and an intervention group. The control group received the treatment of mouth rinsing with sodium bicarbonate 3 times a day, whereas the intervention group received the exact treatment of mouth rinsing with sodium bicarbonate 3 times a day in addition to being instructed to consume tablets of a dry extract of propolis, 2 to 3 times/day between meals. The propolis tablets (80 mg/tablet) were supplied by Natur Farma S.A.S. and they contained 8% to 12% galangin. The total daily number of tablets consumed by the patients was calculated according to patients’ body weight and ranged from 8 to 10 mg/kg BW/day of propolis. Incidence of oral mucositis was then evaluated at days 5, 10, 15, and 21. The patients were also followed up at the end of each chemotherapy cycle over a 6-month period. The incidence of oral mucositis was measured in accordance to the National Cancer Institute Scale (NCI-CTCAE) version 4.0. The patients in the intervention group recorded no incidence of oral mucositis that was more severe than grade G1, whereas in the control arm, 13.3% of patients had G2 and 3.3% of the patients had severe oral mucositis of grade G3. However, compliance with the propolis therapy was not completed for 6 patients due to emesis, suspected allergy, and complaints of the consumption many other oral drugs. In addition, the blinding of assessors could not be achieved as they were also involved in evaluating participants’ compliance with the treatments. 101
Moreover, a group of Iranian researchers investigated the effect of propolis on the antioxidant, inflammation, nutritional status, and quality of life of breast cancer patients treated with chemotherapy in a randomized, double-blind, placebo-controlled trial.102,103 Propolis used in the study was collected from the bee hives in the Kurdistan province, Iran. The harvested raw propolis was put in the water bath. Wood and paint particles were subsequently removed from the raw propolis. The relatively pure propolis gum was subsequently subjected to liquid nitrogen and crushed. The powdered propolis was then obtained. The resulting propolis powder was placed in gelatin capsules as 250 mg doses. Placebo capsules with the similar look, shape, and size were also prepared. No phytochemical analysis was carried out. A total of 50 patients with newly diagnosed breast cancers were included: 26 patients in the intervention arm and 24 patients in the placebo arm. The patients in the intervention arm were instructed to consume the 250 mg propolis capsules twice a day with breakfast and lunch for the duration of chemotherapy (3 months). The intervention started 1 week before the chemotherapy. The patients in the placebo arm received the exact same treatment. They reported no side effect and propolis in the study was well tolerated.
During the course of the study, they found that the patients in the placebo arm had a significant increase in the serum pro-inflammatory cytokines, namely TNF-α and IL-2, and oxidative stress marker protein carbonyl. Conversely, the propolis arm patients did not record any significant increase in pro-inflammatory and oxidative markers. In addition, the intervention group patients had a statistically significant reduction in serum prooxidant-antioxidant balance (PAB), whereas the placebo arm patients did not. 102 The same group of researchers also found that the patients in the intervention arm tended to have improvements in their quality of life, assessed using EORTC QLQ30 questionnaire. At the end of the 3 months of intervention, the patients in the propolis arm had increased energy intake and significant improvements especially in terms of emotional functioning and global quality of life, relative to the patients in the placebo arm. Interestingly, they also found the patients in the placebo group had increased incidence of financial difficulties. 103 These sets of human clinical trials appeared to support the use of propolis as an adjunctive nutraceutical in breast cancer patients. However, larger clinical trials are needed to confirm the therapeutic benefit of propolis in clinical settings.
General Discussion and Future Direction
The present systematic review found that most experimental studies investigating the potential therapeutic use of propolis in breast cancers were in vitro studies, followed by in vivo, in silico, and clinical trials. Majority of the studies demonstrated cytotoxicity activity of propolis and its bioactive compounds against various breast cancer cells. Some studies also investigated the potential synergistic activity of propolis with other therapeutics and more importantly, 3 sets of human clinical trials were identified with no serious adverse event recorded.
Figure 3 illustrates the potential mechanisms of action of propolis against breast cancers summarized in accordance to the studies in the present systematic review. Propolis induces cytotoxicity in breast cancer cells (in vitro and in vivo) through various mechanisms, namely apoptosis, cell cycle arrest, glycolysis inhibition, mitochondrial dysfunction, oxidative stress promotion, and immunomodulatory and inflammation pathways. Propolis induces apoptosis and oxidative stress by inducing Caspase-3, ANXA7, PARP, DR5, Bcl-2, DNA fragmentation, iNOS, and eNOS levels while downregulating Mcl-1, MADD, c-FLIPP. In addition, cell cycle arrest is promoted by propolis through the upregulation of p21, E2FI, p21 p-ERK, p-JNK, and p-p38 and the downregulation of cyclin D, cyclin D1, cyclin E, CDK4, and CDK2. Propolis also inhibits glycolysis by downregulating the activity of glycolysis-hexokinase 2 (HK2), phosphofructokinase (PFK), pyruvate kinase muscle isozyme M2 (PKM2), and lactate dehydrogenase A (LDHA). Propolis negatively impacts the mitochondrial functions of the breast cancer cells by affecting membrane potential and oxygen consumption. Additionally, propolis also works through immune system and inflammation pathway modulation such as CD4+ CD25+ FoxP3+ regulatory T-cells expressing IL-10, NF-κB, TLR4, MYD88, IRAK4, and TRIF.

Summary of the mechanisms of action of propolis against breast cancer cells based on in vitro and in vivo studies.
The therapeutic benefits of propolis have also been observed in breast cancer patients. In these pilot clinical studies, propolis appears to reduce the negative impact of chemotherapy, such as the reduction in the incidence of oral mucositis, inflammation, and oxidative stress. Propolis also appears to maintain the quality of life the breast cancer patients. Furthermore, propolis reduces the adverse effect of radiotherapy, namely DNA damage, while maintaining total antioxidant capacity, hemoglobin (Hb) concentration, white blood cells (WBCs), and platelet counts of the breast cancer patients subjected to radiotherapy (Figure 4). More importantly, these clinical trials reported minimal adverse effect with regards to the consumption of propolis. Perhaps not surprisingly since propolis has been used therapeutically as traditional/folk medicine for thousands of years in many civilizations. 106 However, these clinical trials should be considered preliminary and future research with larger number of participants needs to be conducted.
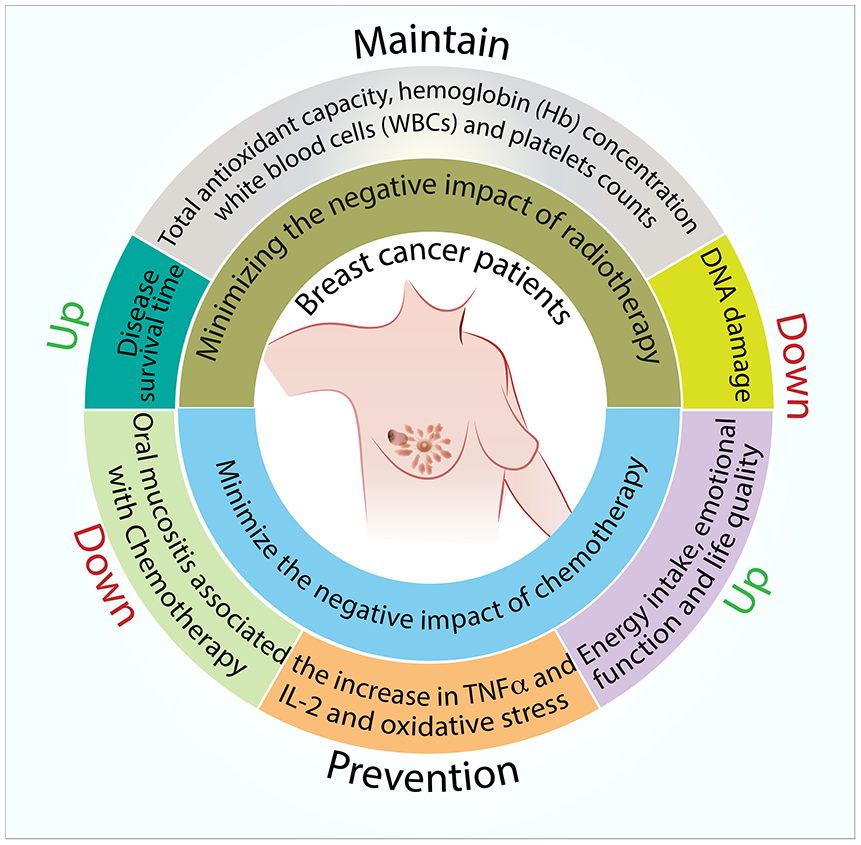
Summary of proposed areas of research on potential clinical benefits of propolis in breast cancer.
In conclusion, the present systematic review demonstrates that propolis may be a useful therapeutic substance to be used as an adjunctive therapy for treating breast cancers. However, more human clinical trials are needed to find the optimum therapeutic concentrations and further explore its potential.
Study Limitations
In the present review, the authors adopted a comprehensive and systematic search strategy in order to objectively fulfill the aim of the study. A broad range of studies from all fields of science and technology was collected and analyzed. The reviewers limited the search to studies that were published in the last 10 years, to provide coverage of the latest experimental evidence in the field. However, the reviewers only assessed and included English language articles, which could potentially lead to missing studies from non-English databases, as it is apparent most studies originated from non-English speaking countries. In addition, the reviewers did not perform a meta-analysis as it is not appropriate due to the heterogeneity of the included studies.
Supplemental Material
sj-docx-1-ict-10.1177_15347354221096868 – Supplemental material for The Potential Use of Propolis as an Adjunctive Therapy in Breast Cancers
Supplemental material, sj-docx-1-ict-10.1177_15347354221096868 for The Potential Use of Propolis as an Adjunctive Therapy in Breast Cancers by Dedy Hermansyah, Felix Zulhendri, Conrad O. Perera, Naufal N. Firsty, Kavita Chandrasekaran, Rizky Abdulah, Herry Herman and Ronny Lesmana in Integrative Cancer Therapies
Footnotes
Acknowledgements
We are thankful to Fuad Bahram, PhD for the production Figures 3 and ![]() .
.
Author Contributions
Conceptualization, Methodology, Writing—original draft: D.H., F.Z., N.N.F., C.O.P., K.C. Supervision, Writing—review and editing: R.A., H.H., R.L. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kebun Efi produces propolis extracts of the Indonesian stingless bees. All other authors declare no competing financial interests and no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no external funding. If accepted, the APC is funded by Universitas Sumatera Utara.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
