Abstract
Keywords
Introduction and Objectives
Consistent and topical information about the frequency and patterns of use of complementary and alternative medicine (CAM) in cancer patients is essential to assess its implications for oncological care as well as to develop evidence-based concepts for research that have the potential to inform continuing education and regulation in the CAM field.1-4
However, the only review that attempted to systematically summarize data on the prevalence of CAM use in cancer patients was published in 1998 and included 26 surveys of cancer patients, either adults or children, from 13 countries and found an average prevalence of CAM use of 31%, with rates ranging from 14% up to 64%. The wide range of prevalence of CAM use was not explained by variation among countries or change over time; in fact, the authors concluded that methodological factors like lack of specificity and inconsistent definitions of CAM, possible selection and recall bias, and the lack of a standardized series of questions were the reasons for the heterogeneity in the prevalence estimates. 5 Since the publication of this systematic review, a large number of surveys on the use of CAM in cancer patients have been published. Although many reports and narrative reviews from these survey data alleged an increase in the prevalence of CAM use in cancer patients over the past years6-11 and considerable differences among European countries, 12 a comprehensive review that systematically summarizes and appraises this literature is absent. Therefore, we felt the need to provide a systematic and critical review on the use of CAM in cancer patients and to offer recommendations for future research. Our systematic review addresses several research questions:
How many patients with cancer use CAM?
Which factors contribute to the variation in prevalence rates of CAM use?
Do the prevalence rates of CAM use change over time?
Are there differences in prevalence rates among countries or continents?
To what extent are prevalence rates influenced by survey methods?
Methods
For the purpose of this review, we decided to adopt
Literature Search and Eligibility Criteria
We searched MEDLINE (1966 to January 2009), AMED (1985 to January 2009), and Embase (1991 to January 2009) and the bibliographies of all located articles and the authors’ own files. Search terms included subject heading terms (eg, complementary therapies; medicine, traditional; questionnaires; health behavior) along with a large number of free text terms (eg, cancer, complementary medicine, alternative medicine, unorthodox medicine, unproven, unconventional, surveys, or interview). The results were then combined with subject heading cancer terms (eg, neoplasms or cancer). All studies were checked for double publications. To be included in this review, surveys had to report original data of at least 100 adult cancer patients from the following countries: Australia, Canada, all European countries (including Israel and Turkey), New Zealand, and the United States (including Hawaii). Only quantitative survey data assessing how many cancer patients used CAM were included in this review. Surveys restricting their definition to a single CAM treatment, published only in abstract form, and conducted at study sites that predominantly offer CAM were excluded. No language restrictions were made.
Data Extraction and Assessment
Data on survey methods, participants, procedures, and results were extracted by means of a predefined comprehensive extraction form and independently by 2 reviewers. Data from articles in languages other than German and English were extracted with the help of a native speaker. A criteria list for assessment of methodological quality was developed and amended during the review process according to the STROBE guideline and another guideline published by Kelley et al.13,14 The list comprised 4 categories (sampling, participants, procedures of data collection, and outcomes), with 16 items in all. All discrepancies were solved through discussion among the reviewers. Instead of using arbitrary measures of quality (ie, quality scores), the actual effect of key methodological characteristics on results was tested. Extracted study data were entered into a relational database management system (MS Access 2000) and double-checked for entry errors.
Data Synthesis
To compare data, surveys were grouped into (1) 3 continents—Australia/NZ (surveys from Australia and New Zealand), Europe (ie, surveys from all European countries including Israel and Turkey), and North America (surveys from Canada and the United States, including Hawaii); (2) 5 European regions—Benelux (surveys from the Netherlands), German-speaking (Austria, Germany, and Switzerland), Great Britain (United Kingdom), Mediterranean (France, Israel, Italy, Spain, and Turkey), and Scandinavia (Denmark, Sweden, and Norway); (3) 3 periods in which surveys were conducted—<1990 (data collection before 1990), 1990-1999 (data collections between 1990 and 1999), and ≥2000 (data collection in or after 2000).
The metaanalysis had 2 main outcomes: the proportion of respondents who reported using CAM (1) at the time of the survey (ie, point prevalence) or (2) during a period of time prior to the survey (ie, period prevalence). For each outcome, we calculated the standard error of the proportion (SE = sqrt [(π × (1 − π)/n], where π is the proportion of CAM users and n the number of participants in a survey) or derived the standard error from reported 95% confidence intervals. A further aim was to explore how the prevalence of CAM use and its variation among countries and over time was influenced by methodological factors and/or survey characteristics.
Mean pooled proportion sizes and corresponding confidence intervals were calculated with a random effects metaanalysis using the method of DerSimonian and Laird
15
. Heterogeneity was quantified by calculating the
To avoid a biasing effect of multiple outcomes within the same survey, all metaanalyses were conducted using only 1 prevalence outcome per study. That means, if studies reported both point and period prevalence of CAM use, the prevalence estimates were analyzed separately; if studies reported more than 1 estimate of period prevalence (eg, CAM use during the antecedent 3 or 12 months), the estimate with the longest time recall was used for analyses.
The regression model first tested the combined effect of methodological factors measured by binary variables (interview vs self-administration, list with prespecified CAM presented vs open questions, certain CAM excluded [eg, spiritual therapies] vs no exclusion of CAM). Then, the effect of several survey characteristics was tested: surveys conducted before 2000 versus in or after 2000 (the groups <1990 and 1990-1999 were collapsed and compared with ≥2000) and surveys conducted in North America versus Europe and Australia/NZ versus Europe. To avoid overfitting, each study characteristic was tested independently of the others. Questions on other outcomes of possible interest where too diverse or the number of surveys reporting on it in sufficient quality was too low to allow meaningful metaanalysis. All statistical analyses were run on STATA (version 11; StataCorp LP, College Station, TX) using the “metan” and “metareg” commands.
Results
Flow of Included Surveys
This review includes 152 surveys, and the flow of studies is shown in Figure 1.

Selection of relevant publications, reasons for exclusion
Survey Characteristics
Table 1 lists the 18 countries in which the included surveys were carried out and gives an overview of the time periods of data collection and extent of study populations of the respective surveys. There was an uneven distribution of countries, states, or provinces in which surveys were carried out: In Canada, the majority of surveys came from Ontario; in the United States, from north-eastern and midwestern states (data not shown); and in Europe from German-speaking countries (Austria, Germany, and Switzerland). The time period in which the surveys were published spanned nearly 30 years (1979 to 2008) with 76% of surveys (n = 116) being published between 1999 and 2008. Three quarters of surveys (n = 111, 73%) reported the time period in which the survey data were collected. The mean time span between collection of survey data and publication was 3 years (standard deviation ± 2). If authors did not report the dates of data collection (n = 41), it was estimated by assuming the above-mentioned difference of 3 years between the survey and its publication. In all, 11% (n = 16) of the included surveys were carried out in the 1980s or before, 45% (n = 69) in the 1990s, and 44% (n = 67) between 2000 and 2006 (data not shown). Overall, 65 000 female and male cancer patients participated in the surveys. Sample sizes ranged from 10086,101,129 to 5047, 119 with a median number of 231. Detailed characteristics of the included studies are listed in an online-only supplemental table (Supplement Table 1).
Overview of the Studies, Year of Publication and Data Collection, and Extent of Study Population, by Countries, Ordered Alphabetically

Methodological Assessment
Key variables related to methodological quality are listed in Table 2. The majority of surveys had small sample sizes and nonprobability samplings. Only a few studies had sampling frames representative of large target populations. Although 68% (n = 104) of surveys reported response rates, detailed information on those who had not participated or responded was uncommon. Only a minority of authors examined the prevalence of CAM use by a longitudinal investigation of the same study population. Pretesting of survey instruments or training of interviewers was infrequently reported. Two thirds of the surveys (67%, n = 102) did not describe the exact period of time to which the questions concerning CAM use pertained, or the period of time varied highly among the included individuals. A few studies reported prevalence estimates of CAM use for different time recalls. We had planned to systematically address the question regarding how the term
Key Variables of the Included Surveys, Grouped by Continents

Abbreviations: NZ, New Zealand; NP, nonparticipants; NR, nonreponders; CAM, complementary and alternative medicine.
Hospitals, practices, and local registries.
At least basic information on NP and/or NR.
Proportions in these rows were calculated using the number of surveys in which data collection were done with interviews or questionnaires as denominators.
Training of interviewers reported.
Pilot testing of questionnaire reported.
Participants were provided with predefined checklists of CAM therapies.
At the time of the survey (ie, current use).
During previous 3/6/12 months.
Since diagnosis/unclear period of time/not reported.
Quantitative Data Analysis
Current use of CAM
A total of 39 surveys reported the proportion of patients using CAM therapies at the time of the survey (ie, point prevalence). The proportions ranged from 9% to 88%, and the pooled proportion was 40% (95% CI = 33%-47%), with significant heterogeneity (
Past use of CAM
In all, 115 surveys reported the proportion of patients having used CAM during a time period previous to the survey (ie, period prevalence). The proportions ranged from 6% to 91%, and the pooled proportion was 43% (95% CI = 38%-48%;
Difference between current and past use of CAM
The difference between the pooled means of point and period prevalences of CAM use was not significant (
Three authors compared the use of CAM during different time periods: Paltiel et al 74 found that 35% of patients used CAM during 3 months prior to the survey, whereas 51% had used it since their cancer diagnosis. Rees et al 110 reported a difference of 10% in the prevalence of CAM use when the recall period spanned 12 months (22%) compared with when the recall related to the time period since the cancer was diagnosed (32%), which spanned a mean of 4.4 years. Shakeel et al 117 found a difference of 24% in the prevalence rates of CAM use when the recall period spanned 12 months (26%) compared with when the recall related to the lifetime period (50%).
International differences in CAM use
There were considerable differences between countries in the pooled proportions of patients using CAM (Figure 2), with the highest proportions in the United States (50%; 95% CI = 40%-60%; n = 36) and the lowest in Italy (22%; 95% CI = 10%-35%, n = 2) and the Netherlands (22%; 95% CI = 9%-37%, n = 2). Grouped into continents, pooled proportions were higher in North America compared with Australia/NZ or Europe (Table 3). The 13% difference in the period prevalence of CAM use between North America and Europe was statistically significant (

Forrest plot of period prevalence of complementary and alternative medicine use in different countries: diamonds show the pooled estimates and their 95% confidence intervals, vertical dotted line shows the pooled weighted estimate
Pooled Prevalences of CAM Use by Continents
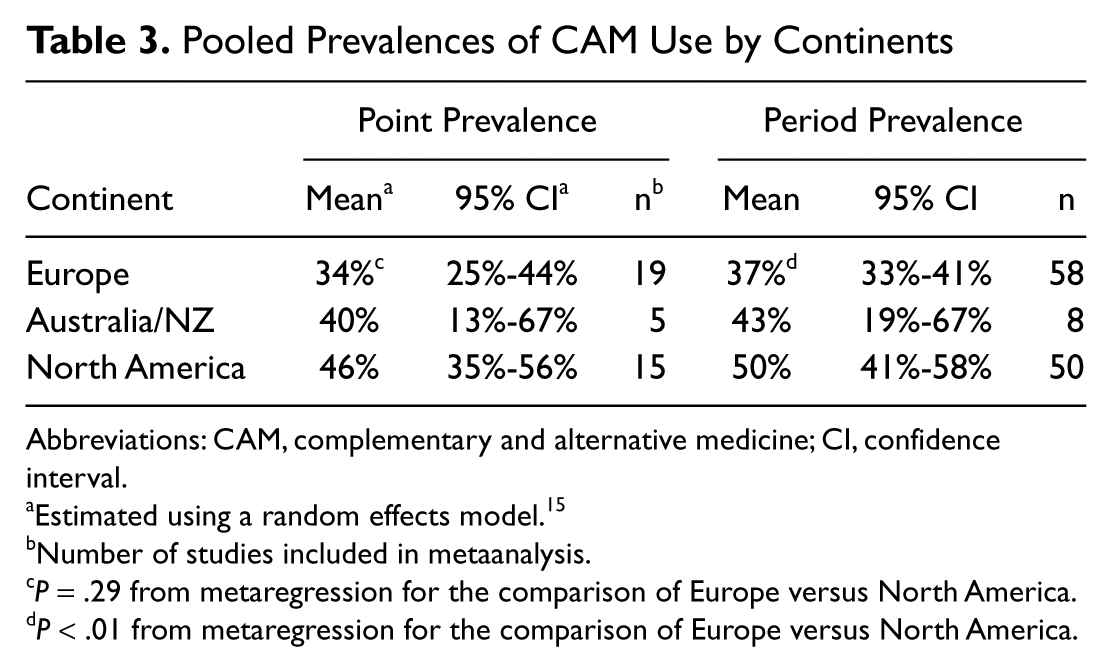
Abbreviations: CAM, complementary and alternative medicine; CI, confidence interval.
Estimated using a random effects model.15
Number of studies included in metaanalysis.

Forrest plot of period prevalence of complementary and alternative medicine use in different European regions: diamonds show the pooled estimates and their 95% confidence intervals, vertical dotted line shows the pooled weighted estimate
Variation over time in CAM use
To explore the possible change in the prevalence of CAM use over time, surveys were grouped according to the time period in which they were conducted. Surveys that were conducted in the 1970s or 1980s reported an average proportion of patients using CAM at the time of the investigation of 25% (95% CI = 12%-38%), compared with 32% or 49% if it was conducted in the 1990s or after 1999, respectively (Table 4). The 24% increase in CAM use between <1990 and ≥2000 was statistically significant (
Pooled Prevalences of CAM Use by Periods of Data Collection a

Abbreviations: CAM, complementary and alternative medicine; CI, confidence interval.
The table shows parameters of an initial model including 3 methodological variables (top 4 rows) and 2 additional variables (period of data collection and continents) for sample characteristic, entered one at a time in the basic model. All variables are categorical.
Estimated using a random effects model. 15
Number of studies included in metaanalysis.
Methodological variables influencing prevalence
Three of the methodological variables listed in Table 2 explained the largest portion of the variance among proportions of CAM use (Table 5). Higher proportions of patients using CAM over a period of time (ie, period prevalence) were reported in surveys presenting lists with CAM from which participants could choose (+19%;
Metaregression on Period Prevalence (Model I) and Point Prevalence (Model II) of CAM Use

Abbreviations: CAM, complementary and alternative medicine; NZ, New Zealand; CI, confidence interval.
The constant term describes the pooled estimate (ie, proportion of patients using CAM) for studies having the following characteristics: no CAM list presented, questionnaire based, and certain CAM excluded from the underlying definition of CAM.
Estimates indicate the extent of increase (+) or decrease (−) in CAM use rates when respondents fall in the first category of the respective variable.
If the model was run with point prevalence of CAM, the 3 methodological variables explained less variance among the proportions (Table 5 [model II]). Higher proportions of patients using CAM at the time of the survey (ie, point prevalence) were reported in surveys presenting CAM lists (+17%;
Discussion
This is the first metaanalysis of a body of literature consisting of surveys spanning nearly 30 years that ask cancer patients about their use of CAM. On average, about 40% of cancer patients reported current or previous use of CAM therapies. There were considerable differences in the pooled prevalence estimates between the countries and continents with higher rates of CAM use in North America compared with Australia/NZ and Europe. There was some evidence that over the past 30 years and especially after 2000, the proportion of cancer patients using CAM has increased. The high degree of heterogeneity in CAM use indicated that the estimates in individual studies differed more from each other than one would expect from chance alone. Nearly a third of the between-surveys variance in effect size was explained by 3 basic methodological factors: whether the participants were interviewed face-to-face or had filled out questionnaires, whether participants were provided with predefined lists of different CAM treatments or had to recall them without being prompted, and whether surveys restricted CAM use to certain CAM categories or treatments or not. Once these factors were controlled for, surveys conducted in 2000 or later appeared to yield higher rates of CAM use than surveys from before 2000, but no larger differences were found between continents.
How reliable are these results? And what can they tell us actually about the frequency of CAM use in cancer patients?
The procedure adopted to standardize data in the review and in the single surveys clearly has limitations that might affect the interpretation of results. In particular, the frequently reported percentage of respondents that recall at least 1 incident of CAM use is a very rough measure of overall CAM use because respondents might have used a single CAM treatment only once. In this case, the frequencies reported in these surveys would clearly overestimate the relevance of CAM use in cancer patients. However, other studies showed that about a third of CAM users are usually committed to it and use more than 1 CAM treatment.53,166
Survey participants reported a higher rate of CAM use when they chose CAM treatments from a presented list rather than with free recall. This result is consistent with the findings of a recent systematic review that investigated the effect of questionnaire design on the recall of pharmacological treatments. 167 It found that the frequency of use varied between 5% and 40%, depending on whether lists with drug names or similar memory aids were parts of self-administered questionnaires.
Mailed or handed-out surveys yielded, on average, lower percentages of CAM use than interview-based surveys. A recent study that compared the effects of questionnaires with interview-based data collection in surveys found that the mode of data collection considerably affected the pattern of reporting 168 (eg, persons who were interviewed had a lower rate of missing responses and used the “don’t know” option less often). This points to the fact that a trained, motivating interviewer can increase item response rates, clarify ambiguous questions, and jog respondents’ memories for aiding recall of events and behavior. However, the accuracy of self-reports on CAM use in face-to-face interviews might be biased by social desirability. Studies suggest that respondents give more positive and socially desirable responses in interview surveys than in self-administration.168,169 One could even suspect that respondents assume interviewers to be in favor of CAM and act accordingly. However, one could also argue that within a setting of conventional oncology, respondents are probably less willing to reveal CAM use fearing that this might be regarded as undesirable. Robinson et al 170 reviewed the disclosure of CAM use to physicians and found that the main reasons for not disclosing were concerns about negative responses by the physicians. The differences concerning self-reports of patients on their use of CAM could explain why the effect of the parameter, “Interview yes vs no”—that is, interview or questionnaire-based assessment of CAM use—was not robust over the multivariate models of our metaanalysis (Table 5). Therefore, the influence of different methods of assessment on the extent of CAM use would need to be confirmed by independent studies before being conclusively accepted.
Surveys that restricted CAM use to certain categories or treatments yielded lower prevalence estimates than surveys that did not. This is exemplified by the study from Abu Realh et al,
120
who confined their definition of CAM use to mind-body approaches, counseling, and attendance to self-help groups and found that 12% of respondents were “CAM users.” Accordingly, studies with broad definitions of CAM use tended to inflate estimates: for example, many of the included surveys with very high usage rates had integrated prayer and exercise defined as CAM, and the usage rate would have been approximately halved if these therapies had not been included. We planned to systematically address the question of how different definitions for the term
The findings of 2 studies included in this review could further illustrate this problem: Turkish cancer outpatients who had used indigenous herbal remedies did not regard these remedies as CAM 98 ; in contrast, participants of Indo-Asian ethnic backgrounds in a sample of Canadian outpatients with head and neck cancer classified treatments indigenous to their native regions as CAM. 41 These findings correspond to those of a large survey, in which the authors identified ethnic-specific CAM modalities within a diverse California population and concluded that “evaluation of CAM use in ethnically diverse populations should recognize ethnic-specific modalities and variation across ethnicity.”281
Our analysis indicated further that respondents in samples drawn from German-speaking countries reported CAM use more frequently than respondents in other European countries. This could be explained by the fact that homeopathy, anthroposophic medicine, and naturopathy have their origins in German-speaking countries, and hence, many home remedies and self-medication practices are rooted in these health care approaches.
Once methodological differences were controlled for, cross-study comparisons suggested an increase in the prevalence of CAM use in cancer patients over the past 30 years, especially after 2000. This could be explained by a combination of expanded level and quality of evidence, research and training in the field of CAM, and rising awareness among health professionals and the public. 173 Furthermore, there is evidence from a nationally representative survey that the use of CAM in other fields of medicine had also increased. 174 Our findings are supported by those of 2 included surveys: Boon et al44,54 compared 2 random samples of Canadian women with breast cancer in 1998 and in 2005 and found a 15% increase in the use of CAM (67% in 1998 and 82% in 2005). In our metaanalysis the increase of CAM use was seen only in the multivariate model using point prevalences of CAM use. This could be explained by the fact that the variable “period of data collection” was dichotomized (before vs after 2000) for metaregression, and many surveys that were carried out after 2000 had time recalls concerning CAM use of up to several years that reached back to before 2000.
The lack of statistical significance for the effect of several factors in the regression models is not strong evidence against their relevance because the high between-survey variance caused by methodological factors limited the power of the analysis (ie, the regression had to control for 3 methodological factors before testing any other effect). However, it suggests that such differences need to be explored at the study level, with surveys designed adequately to compare groups.
A variable that survey authors in this field should give more attention to is the time period to which the question about CAM use pertains. The included surveys provided prevalence estimates covering highly variable periods of time, ranging from the date of the survey interview up to several years. More than two thirds of the surveys did not describe the exact period of time to which the questions pertained, or the time period—for instance, the time since cancer diagnosis—varied highly among the included individuals. Given the fact that the use of CAM—being part of a patient’s coping strategy—is a dynamic behavior, any assessment of this behavior should take into account its time dependence to ensure both accuracy and comparability of estimates of its prevalence. Finally, it is not to be forgotten that the recall of a behavior may also change over time.
These limitations notwithstanding, there was some evidence that proportions increased with the length of the time recall and were higher when participants were asked about their use during a time period than when they were asked about their use at the time of the survey. The findings from Risberg’s longitudinal study illustrate this influence. In a study population followed up for several years, the survey showed an ~20% prevalence of CAM in the repeatedly performed cross-sectional interviews and a cumulated 5-year period prevalence of 45%. 87
Conclusions and Recommendations for Future Research
In conclusion, over the past 30 years, many surveys have asked cancer patients about their use of CAM, and the differences in their results are largely a result of differences in their methods. However, there was a significant portion of unexplained heterogeneity in the prevalence rates probably related to patient- and disease-related factors, which were not only too diverse but were also often incompletely reported to allow meaningful metaanalysis. Only by carefully controlling for these factors can the effects of country and time be studied in detail. Therefore, there appears to be little scope for smaller descriptive surveys, unless they adopt high-quality and standard methodologies. On the other hand, there is a certain scope for longitudinal studies aimed at identifying patterns of CAM use during the so-called “cancer journey.”
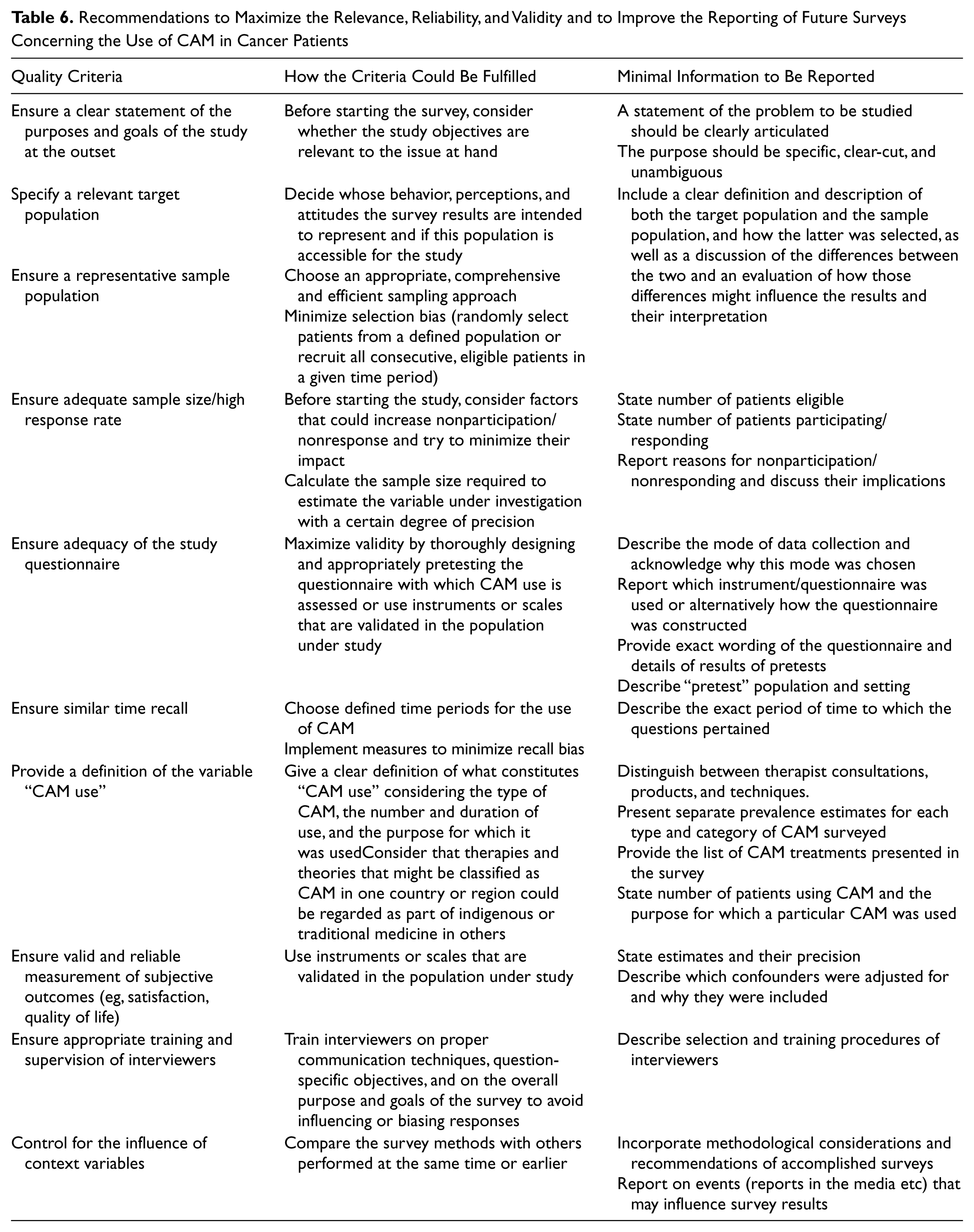
Because numerous surveys failed to provide sufficient information for us to assess their performance on many of the methodological indicators, we compiled a number of suggestions for how future surveys of CAM use in cancer patients could maximize their validity as well as some minimal reporting standards that authors should observe, and journal editors should enforce, when publishing work in this area.13,175 As summarized in Table 6, such improvements in the quality of the conduct and reporting of future trials should be feasible without incurring much additional costs or requiring substantially more journal space when publishing results.
Recommendations to Maximize the Relevance, Reliability, and Validity and to Improve the Reporting of Future Surveys Concerning the Use of CAM in Cancer Patients
Overall, rates of CAM use found in this review are lower than what many CAM protagonists usually claim but consistent with the more conservative estimates of the few existing narrative reviews in this field. However, the findings suggest that, on average, half of all cancer patients use CAM and that this proportion has increased over the past years. These results highlight the need for clear strategies of how to further deal with this prevalent health-related behavior of cancer patients.
Footnotes
Acknowledgements
The authors thank Gudrun Greim, Lars Krueger, and Elke Wolf for their help with extracting data from studies in Spanish and Scandinavian languages. Many thanks are also extended to Corina Guethlin and Michael Wiesand for carefully editing the manuscript and to Christine Fink and Birgit Kraus for coding the references.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The review was funded by grants from the FP5 project “Concerted Action for Complementary and Alternative Medicine Assessment in the Cancer Field (CAM-Cancer)”; Quality of Life and Management of Living Resources programme, European Commission [QLRT- 2001- 00786]; and the AG Biologische Krebstherapie, Deutsche Krebshilfe [70-301].
