Abstract
Objective:
To evaluate the effectiveness of Chinese Herbal Medicine (CHM) on leukopenia/neutropenia induced by chemotherapy in adults with colorectal cancer (CRC).
Methods:
Eight electronic databases were searched from their inception to June 2020. Randomized controlled trials with clarified sequence generation were qualified. Two reviewers independently conducted the screening and data extraction. Methodological quality was assessed using the Risk of Bias tool. RevMan 5.4 was applied to the meta-analysis.
Results:
Twenty-seven studies involving 1867 participants were qualified, of which 26 were included in the quantitative synthesis. Meta-analysis showed that CHM significantly reduced the incidence of leukopenia induced by chemotherapy (RR = 0.69; 95% CI 0.59-0.82), as well as the grade 3/4 leukopenia (RR = 0.71; 95% CI 0.55-0.90). Meanwhile,CHM decreased the occurrence of neutropenia (RR = 0.52, 95% CI 0.35-0.77), especially for the grades 3/4 neutropenia (RR = 0.42, 95% CI 0.27-0.64). Twenty-six of the included studies focused on the adverse events related to CHM.
Conclusion:
CHM may relieve neutropenia/leukopenia induced by chemotherapy in adults with colorectal cancer.
Introduction
Neoplasms have become the second leading cause of death from non-communicable diseases worldwide, 1 in which the incidence and mortality of colorectal cancer (CRC) rank third and second respectively. 2 It was estimated that more than 1.8 million new cases of CRC will be diagnosed and approximately 880 000 people would die from it in 2018 alone. Systematic chemotherapy has been playing a prominent role in the treatment of CRC from its development 3 and is recommended to both patients at high-risk of recurrence or metastasis4-7and patients with metastasis8,9 according to the National Comprehensive Cancer Network (NCCN) Guidelines and the Chinese Society of Clinical Oncology (CSCO) Guidelines.
However, adverse effects (AEs) caused by chemotherapy are one of the major problems faced for both patients and practitioners, including leukopenia/neutropenia. The impact of chemo-induced leukopenia/neutropenia shows in categories of severity,10-13 since grade 1/2 leukopenia/neutropenia may recover while grade 3/4 may lead to dose reduction, delay or discontinuation of chemotherapy, which may discount the therapeutic effect of cancer.
Myeloid growth factors (MGF), such as granulocyte colony stimulating factors (G-CSF), or granulocyte macrophage colony-stimulating factor (GM-CSF), are recommended to be used in the prophylactic and treatment of patients at high risk of febrile neutropenia (FN) and the treatment of FN according to NCCN guidelines, since research has provided sufficient evidence for their effectiveness.14-17 However, what cannot be ignored is the toxicity associated with them. Bone pain, myalgia, pyrexia, and asthenia are the most frequent AEs in patients receiving G-CSF,18-20 the incidence of which can range from 9.9% to 47%, from 11.9% to 69.8%, from 12.1% to 55.3%, and from 13.2% to 15.6%, respectively. 18 Besides, gastrointestinal disorders, neutropenia, thrombocytopenia, ineffectiveness have been reported.18,19 Severe AEs, splenic rupture, for instance, might be life-threatening. 21 Thus, the prevention and treatment of leukopenia/neutropenia induced by chemotherapy remains a challenge.
Chinese Herbal Medicine (CHM) has been playing an important role in health care for human beings for more than 2000 years in China. “Treat pre-disease,” meaning prevention of disease before it arises, is one of the most important theories of Chinese Medicine. CHM may play a striking role in the prevention of chemo-induced leukopenia/neutropenia. Results of preliminary studies are questionable due to the small sample sizes and the limitation of study design. Systematic reviews and meta-analyses have been done to investigate the effectiveness of CHM on chemo-related adverse effects.22-25 However, there was no study focusing on chemo-induced leukopenia/neutropenia in patients with CRC. Therefore, we conducted a systematic review to explore the effectiveness of CHM on leukopenia/neutropenia induced by chemotherapy to provide practitioners with reliable information for guiding clinical treatment decisions.
Method
The systematic review and meta-analysis were conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). The protocol has been previously registered in PROSPERO (No. CRD42020177847).
Data Sources and Search Strategies
Two reviewers independently searched 8 electronic databases, including Pubmed, Embase, Web of Science, Cochrane Library, China National Knowledge Infrastructure (CNKI), Wanfang, Chongqing VIP (CQVIP), and China Biology Medicine disc (CBM) from their inception to June 2020 to identify studies that met the inclusion criteria. The language of studies was restricted in English and Chinese. We utilized the medical subject headings “Colorectal Neoplasms,” “Rectal Neoplasms,” or “Colonic Neoplasms” in both Chinese and English databases. Detailed search terms and strategies are shown in Table 1.
PubMed Search Strategy.

Inclusion and Exclusion Criteria for Articles
Inclusion criteria
Studies in accordance with all the following were included.
(1) Participants diagnosed with CRC and receiving chemotherapy, aged ≥18 years, regardless of gender and race.
(2) Intervention of the treatment group was oral CHM alone, with the components of the prescription and treatment courses.
(3) Comparison (control group) untreated or treated with placebo of CHM.
(4) Outcome measurements included the incidence of leukopenia/neutropenia, the incidence of grade 3/4 leukopenia/neutropenia and the absolute count of leukocyte/neutropenia.
(5) Types of studies: randomized controlled trials (RCTs) only.
Exclusion criteria
Studies in accordance with the any of the following were excluded
(1) Information not available on the stage of the tumor, either TNM stage or Duke’s stage.
(2) Chemotherapy scheme (both the agents and the dosage) not clarified.
(3) Intraperitoneal infusion chemotherapy or oral chemotherapy drug alone.
(4) Intervention combined with other types of Chinese Medicine, such as moxibustion, acupuncture, or enema.
(5) Full-text not available.
(6) Sequence generation of randomization not clarified.
Eligibility Screening and Data Extraction
Two authors independently screened the articles for eligibility of trials, according to the criteria demonstrated above. Any inconsistency in this process was resolved by the third party (Yu-Fei Yang, Yun Xu, or Bin He). For the included studies, 2 reviewers extracted the following information: study ID, sequence generation, sample size, mean age, gender ratio, tumor stages, basic chemotherapy regimens, duration of treatment, treatment of the experimental/control groups, and outcomes. For papers with information not available, the correspondence authors were contacted by e-mail.
Risk of Bias of Individual Studies
The risk of bias of individual studies was assessed by 2 reviewers following the Cochrane Handbook for Systematic Review of Interventions, 26 whose main items are: random sequence of generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete data addressed, and selective reporting. Other bias was defined as the imbalance of baseline and for-profit bias.
Data Synthesis
Meta-analysis was performed by RevMan 5.4 software. For continuous data (white blood count and neutrophil count), mean difference (MD) and its 95% confidence intervals (CIs)were used to represent the effect measures, while the relative risk (RR) with 95% CI were applied for the dichotomous outcomes, such as incidence of grade I/II/III/IV leukopenia/neutropenia, incidence of leukopenia/neutropenia, incidence of grade I/II/III/IV/V leukopenia/neutropenia, and incidence of grade III/IV neutropenia.
Heterogeneity was tested with the chi-squared (χ2) test and I2 statistic. A fixed-effects model was used when I2 < 50%, otherwise a random-effects model was applied. We performed subgroup analyses and sensitivity analysis to investigate the factors of heterogeneity and the robustness of the finding, respectively.
Publication Bias
Funnel plots were applied to detect publication bias if there were more than 10 trials included in a meta-analysis.
Results
Literature Screening and Selection
In total, 573 articles were obtained, listed as follows: PubMed, 7; EMBASE, 25; Web of Science, 12; Cochrane Library, 9; CNKI, 80; Wan Fang, 279; VIP, 17; CBM 144. One hundred twenty-two duplicates were removed preliminarily when imported into Endnote X9. After initial screening based on title and abstract, 267 records were eliminated. Further evaluation based on full-text articles removed 157 records. Finally, 27 studies were enrolled in our study, of which 26 RCTs27-52 reported the incidence of leukopenia/neutropenia, while 1 RCT 53 evaluated white blood cell count (WBC) and neutrophil count (NEUT). The quantitative synthesis was conducted with 26 RCTs involving ordinal/dichotomous outcomes. The search results and study selection are displayed in Figure 1.

Flow chart of search results and study selection.
Characteristics of included studies
As shown in Table 2, up to 937 subjects in the treatment groups (T) and 930 subjects in the control groups (C) were enrolled in these RCTs, all of which were published in Chinese. Sample sizes ranged from 30 to 122. Chemotherapy regimens involved OLF, SOX, XELIRI, FOLFOX, FOLFIRI, XELOX (CapeOX) and CPT-11 plus Raltitrexed. The duration of treatment varied from 2 cycles to 6 cycles. Tumor stage was mainly TNM staging II to IV.
Characteristics of Eligible Studies.

Outcomes: (1) incidence of grade I/II/III/IV leukopenia; (2) incidence of grade I/II/III/IV/V leukopenia; (3) incidence of leukopenia; (4) white blood count (x2 ± ‾s); (5) incidence of grade I/II/III/IV neutropenia; (6) incidence of grade I/II/III/IV/V neutropenia; (7) incidence of neutropenia; (8) incidence of grade 0-II/III-IV neutropenia; (9) neutrophil count (x2 ± ‾s).
Risk of Bias Assessment
Figures 2 and 3 indicated the results of the assessment of risk of bias of the qualified studies. All the studies reported the adequate methods of sequence generation. Allocation concealment was mentioned in 1 RCT 29 while others did not. One RCT 29 blinded both participants and personnel, whereas the rest did not mask the doctors and patients. Both the primary outcomes and the secondary outcomes are objective test results, which might be unbiased. We considered that the lack of blinding will not influence the assessment of the outcomes and assessed all the included studies as low risk of bias. Attrition bias was reported in 3 studies.42,44,51 Gao’s research 51 referred to the reasons for withdrawals, which differed between treatment group and control group, while He 44 and Miu et al 42 cleared that there was no “lost to follow-up” or drop-out. All the 27 articles did not refer to the information of the clinical study registration and we cannot determine the reporting bias. All the RCTs referred to no significant difference between treatment group and control group as to baseline information.

Risk of bias graph.

Risk of bias summary.
Primary Outcomes
Incidence of leukopenia
4 RCTs34,38,42,43 reported the incidence of leukopenia, 18 RCTs27,28,30-33,36,37,39-41,44,46,47,49-52 reported the incidence of grade I/II/III/IV leukopenia and 1 RCT 29 reported the incidence of grade I/II/III/IV/V leukopenia. The latter 2 can be made into dichotomous data so that we combined the dichotomous and ordinal outcomes to analyze.
The comparison between treatment groups and control groups of the effectiveness on chemotherapy induced leukopenia is shown in Figure 4. In total, there were 755 and 758 participants involved in the treatment group and control groups, respectively. In view of the high heterogeneity (P < .00001, I2 = 66%) among studies, the random effects model was used for the meta-analysis. The results shows that CHM effectively reduces the incidence of leukopenia induced by chemotherapy with statistical significance (RR = 0.69; 95% CI 0.59-0.82; P < .0001).

Forest plot of treatment group vs control group on the effectiveness of the incidence of leukopenia.
Incidence of neutropenia
In terms of the effectiveness of CHM on chemo-induced neutropenia, 10 RCTs29,32,34,35,37,41,50,52 were included: 277 participants in the treatment groups and 275 in the control groups. Since heterogeneity (P = .0005, I2 = 73%) was observed, a random-effects model was applied to the meta-analysis. In comparison with the control groups, CHM reduced the incidence of chemotherapy-associated neutropenia (RR = 0.71; 95% CI 0.55-0.90; P = .005) (Figure 5).

Forest plot of treatment group vs control group on the effectiveness of the incidence of neutropenia.
Secondary Outcomes
Incidence of grade 3/4 leukopenia
Eighteen RCTs27,28,30-33,36,37,39-41,44,46,47,49-52 reported the incidence of grade I/II/III/IV leukopenia, 1179 participants involved. As is shown, there was insignificant heterogeneity (P = .12, I2 = 31%) among trials, hence a fixed-effects model was applied. The results revealed that patients experiencing grade 3/4 leukopenia induced by chemotherapy may benefit from CHM (RR = 0.52; 95% CI 0.35-0.77; P = .001) (Figure 6.)

Forest plot of treatment group vs control group on the effectiveness of the incidence of grade 3/4 leukopenia.
Incidence of grade 3/4 neutropenia
Grade 3/4 neutropenia was reported in 8 RCTs. A fixed-effects model was utilized for the analysis owing to the insignificant heterogeneity (P = .62, I2 = 0%). The quantitative synthesis shows that CHM is beneficial to those undertaking chemotherapy and subject to grade 3/4 neutropenia (RR = 0.42; 95% CI 0.27-0.64; P < .0001). (Figure 7)

Forest plot of treatment group vs control group on the effectiveness of the incidence of grade 3/4 neutropenia.
WBC and NEUT
Ge et al 53 investigated the changes of WBC and NEUT before and after treatment between the treatment group and control groups. For WBC, there was no statistical difference between the 2 groups at baseline, while WBC in both groups decreased after chemotherapy. However, WBC in CHM group was higher than the control group, suggesting a statistical meaning (P < .01). It was the same with NEUT.
Subgroups and Sensitivity Analysis
To consider the cumulative effect of chemotherapy and the worse physical condition of stage IV patients, we planned subgroup analysis based on the durations of treatment and tumor stage. Meanwhile, post hoc subgroup analyses were performed to investigate the heterogeneity, which grouped the participants according to mean age and chemotherapy regimens, since these 2 may be related to the incidence of AEs of chemotherapy.54-56 As is demonstrated in Table 3, durations of treatment, tumor stage, mean age, or chemotherapy regimen should not be addressed as the sources of heterogeneity.
Subgroup Analysis.
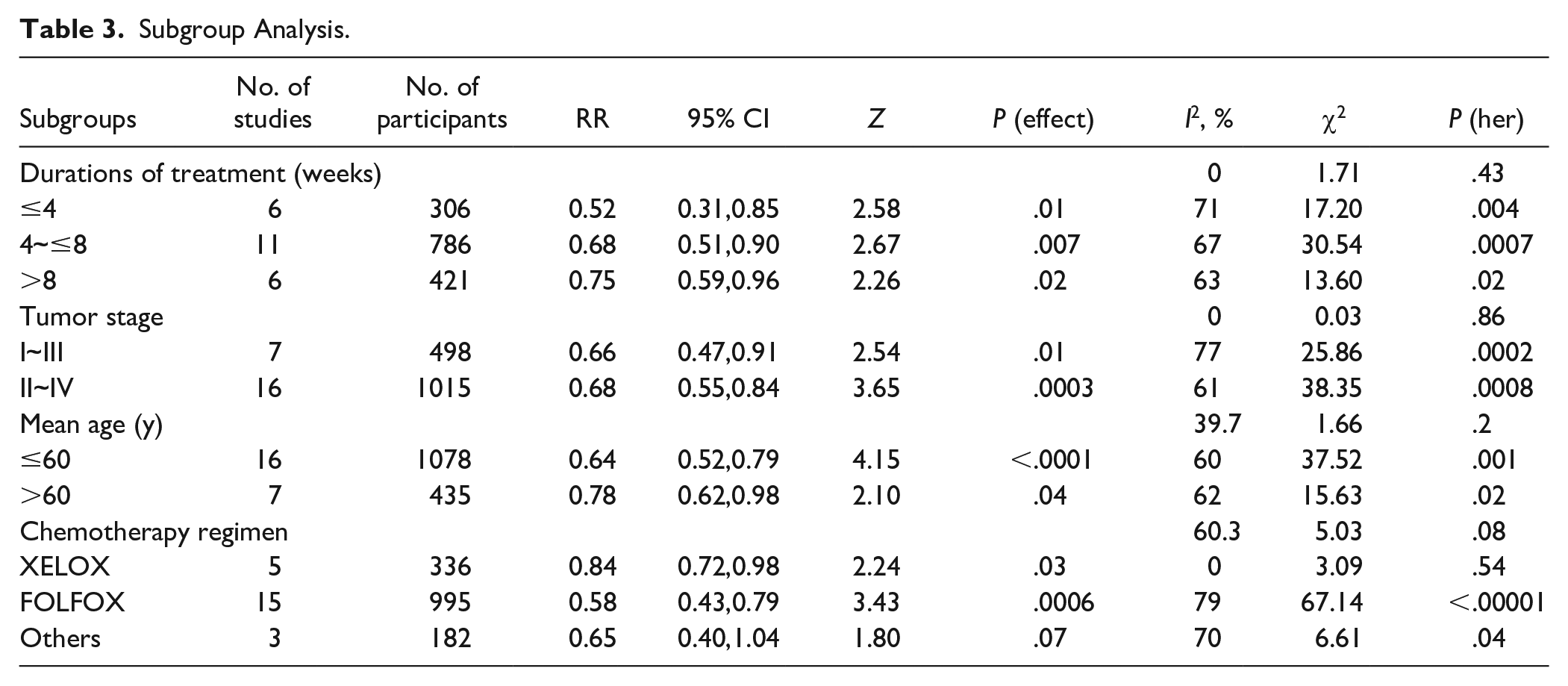
Removing all the studies one by one, sensitivity analysis suggested that RRs were not influenced significantly. Nonetheless, the funnel plots for the publication bias indicating the existence of bias. (Figure 8)

Forest plot for publication bias of incidence of leukopenia.
Discussion
Summary of the Study
Chemotherapy, alone or combined with targeted drugs or radiotherapy, has represented the cornerstone of CRC treatment in past decades. Increasing numbers of clinical studies were designed to seek optimal chemotherapy regimens which were both effective and safe.12,57-62 However, hematotoxicity induced by chemotherapy still poses a threat to patients receiving chemotherapy. In China, CHM is recommended to cancer patients to detoxify chemotherapy. However, there were limitations in the existing evidence and few previous reviews assessed the available trials comprehensively. We conducted a systematic review and meta-analysis to investigate the effectiveness of CHM on chemo-induced leukopenia/neutropenia in adults with CRC regardless of the chemotherapy regimens, which differs from Chen et al’s63,64 meta-analyses, restricting chemotherapy regimens to FOLFOX and oxaliplatin-based regimens, respectively. Actually, chemotherapy regimens can vary in the clinical practice of CRC.
The results revealed that CHM reduces the incidence of leukopenia/neutropenia, as well as grade 3/4 leukopenia/neutropenia induced by chemotherapy, which means that CHM may be helpful for the subjects to improve the completion rate of chemotherapy; to reduce, to some extent, the risk of febrile neutropenia; and to cut down the costs. However, this should be interpreted with caution due to poor quality of the included trials and significant heterogeneity observed in the meta-analysis. Though subgroup analysis was conducted dividing participants into groups on the basis of durations of treatment and tumor stage, neither of them were demonstrated to be influencing factors. Nevertheless, we noted a trend toward the increasing RR among groups of duration ≤4 weeks, 4 weeks < ~ ≤ 8 weeks and >8 weeks, which may be in connection with the cumulative toxic effect of chemotherapy. This indicated us to keep a watchful eye on patients receiving more cycles of chemotherapy in case of leukopenia/neutropenia. Physicians should adjust the prescription and dosage of CHM when it is necessary. Post hoc subgroups analyses were performed to address the sources of heterogeneity, which suggest that neither mean age nor chemotherapy regimen could be causes of heterogeneity. However, a rising tendency of RR is present with increasing mean age. Patients receiving FOLFOX regimen may be more likely to benefit from CHM than those receiving XELOX regimen, although there was no statistically significant difference.
CHM decoctions used were various, such as Sijunzi Decoction,41,52, BaZhen Decoction, 51 Shenling Baizhu Power 50 and self-drafted decoctions based on Sijunzi Decoction and/or Danggui Buxue Decoction. Sijunzi Decoction and Shenling Baizhu Powder are thought to strengthen the spleen while BaZhen Decoction and Danggui Buexue Decoction are composed to tonify qi and replenish blood, which is consistent with the saying in Huangdi Neijing, one of the most famous classics of Traditional Chinese Medicine (TCM), that spleen and stomach are the sources of Qi and Blood.
Common herbals involved are listed as: Radix Astragali seu Hedysari (Huang qi), Rhizoma Atractylodis Macrocephalae (Bai zhu), Poria (Fu ling), Radix Glycyrrhizae (Zhi gan cao), Radix Ginseng (Ren shen), Radix Codonopsis (Dang shen), Semen Coicis (Yi yi ren), Radix Angelicae Sinensis (Dang gui), Radix Paeoniae Alba (Bai shao). Ingredients of Ginseng were found to promote the hemopoiesis via activating JAK-STAT pathway, 65 regulating the differentiation of bone marrow mesenchymal stem cells (BMSCs) by upregulating the expression of RUNX2, 66 inhibiting the aging of BMSCs 67 or inhibiting Sca-1+ HSC/HPC senescence by acting on the SIRT6/NF-κB signal axis. 68 Angelica Sinensis Polysaccharides (ASP) has been demonstrated to improve leukocytes of mice exposed to 5-Fu by boosting response of blood cell to oxidative stress and reversing the imbalance of BMSCs differentiation induced by 5-FU. 69 Constituents of Radix Astragali and Radix Angelicae Sinensis 70 were reported to restore the hematopoietic function of hematopoietic stem cells (HSCs) by alleviating the destruction of HSCs mediated by the immune system. Astragaloside, Calycosin, 71 and Astragalus polysaccharide 72 may have an effect on BMSCs. Atractylode III 73 was found to regulate the adipogenic differentiation of BMSCs.
Moreover, the kidneys are closely associated with hematopoiesis in TCM, as is recorded in Huangdi Neijing, with the kidney generating marrow and dominating bone. Herbals for tonifying the kidney were widely used to preventing or treating chemotherapy induced myelosuppression, such as Radix Rehmanniae Preparata (Shu di huang), Rhizoma Dioscoreae (Shan yao), Fructus Lycii (Gou qi zi), Fructus Ligustri Lucidi (Nv zhen zi), Semen Cuscutae (Tu si zi). It has been demonstrated in vivo that Chinese herbal compound containing Radix Rehmanniae Preparata can attenuate chemotherapy induced leukopenia by promoting cell proliferation and inhibiting apoptosis of bone marrow cells 74 . Zhao’s study 75 uncovered that Heidihuang Bolus increased the production of EPO, IGF-1 and SCF. Rhizoma Dioscoreae promoted the recovery of hematopoietic function of mice affected by cyclophosphamide by upregulating the expression of PCNA, Survivin, MMP-2, and MMP-9 of BMSCs. 76 In summary, herbals for invigorating spleen and tonifying kidney may improve the hematopoietic function of bone marrow via multiple signal pathways.
Limitations
There are some limitations in our study. (1) All the 27 studies included were published in Chinese, and trials were conducted in mainland China. It is unknown whether CHM is applicable for patients in other areas. (2) Most of the RCTs included failed to report the allocation concealment or incomplete outcome data, and none of the 27 RCTs referred to the registration information of the clinical trials, which was of great importance to assess the risk of bias. (3) The presence of substantial heterogeneity may influence the pooled results. (4) Different CHM prescriptions would theoretically differ in terms of efficacy and mechanism, which we failed to measure. (5) The long-term effects of CHM on the disease-free survival (DFS), progression free survival (PFS), or overall survival (OS) were not concerned in our studies.
Implications for Future Research
It is worth noting that 2 elegant RCTs77,78 focusing on the effectiveness of CHM on chemo-induced myelosuppression are being conducted, which are expected to provide strong evidence. It is necessary to update the meta-analysis once the results of these 2 RCTs are published. RCTs are recommended to report the findings following the CONSORT 2010 statement79,80 to ensure the methodological quality. Furthermore, fundamental research is desired to investigate the mechanism of invigorating spleen and kidney CHM on chemo-induced leukopenia/neutropenia in patients with CRC.
Conclusions
In conclusion, CHM may lower the incidence of leukopenia/neutropenia and the incidence of grade 3/4 leukopenia/neutropenia induced by chemotherapy in patients with CRC. However, heterogeneity and publication bias may discount the reliability of our findings.
Footnotes
Acknowledgements
This work was funded by the National Key Research and Development Program of China (no. 2017YFC1700604) and Project of China Academy of Chinese Medical Sciences (no. ZZ11-069). Funding bodies play an economic support role in research design.
Authors’ Contributions
Shao-Hua Yan, Yu-Fei Yang and Yun Xu conceived the study. The protocol was drafted by Shao-Hua Yan and was revised by Yu-Fei Yang and Yun Xu. Shao-Hua Yan and Shuo Feng contributed to structuring the search strategies, and the search was perfomed by Yun-Zi Yan and Mo Tang, Shao-Hua Yan and Yun-Zi Yan completed the literature screening. Yu-Ying Xu and Na Zhao responsible for assessment of the risk of bias, and Yue -Chen and Ming-Kun Yu extracted data from the qualified studies. Any discrepancies were resolved by consulting Yu-Fei Yang, Yun Xu or Bin He. Statistical analysis was performed by Shao-Hua Yan and Shuo Feng, and manuscript was compose by Shao-Hua Yan. Shuo Feng and Bing Pang participated in modification of the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The data used to support the findings of this study are included within the article.
