Abstract
Background. Chinese herbal medicines reportedly increase efficacy and minimize toxicity of chemotherapy; however, little attention has been paid to how poor study quality can bias outcomes. Methods. We systematically searched MEDLINE, TCMLARS, EMBASE, and Cochrane Library for randomized controlled trials of Chinese herbal medicines combined with fluorouracil-based chemotherapy compared with the same chemotherapy alone. We screened for eligibility, extracted data, and pooled data with random-effects meta-analysis. Outcome measures were survival, toxicity, tumor response, performance status, quality of life, and Cochrane Risk of Bias (ROB) criteria to critically evaluate the quality of reporting in the randomized trials included in the meta-analysis. Results. We found 36 potentially eligible studies, with only 3 (those with low ROB) qualifying for meta-analysis. Two reported chemotherapy-related diarrhea reduced by 57% (relative risk [RR] = 0.43; 95% CI = 0.19-1.01; I2 test for variation in RR due to heterogeneity = 0.0%), with nonsignificant results. Two reported white blood cell toxicity reduced by 66% (RR = 0.34; 95% CI = 0.16-0.72; I2 test for variation in RR due to heterogeneity = 0.0%), with statistically significant results. Stratifying analysis by studies with high versus low ROB, we found substantial overestimation of benefit: Studies with high ROB overestimated by nearly 2-fold reduction of platelet toxicity by Chinese herbal medicines (RR = 0.35, 95% CI = 0.15-0.84 vs RR = 0.65, 95% CI = 0.11-3.92). Studies with high ROB overestimated by nearly 2-fold reduction of vomiting toxicity (RR = 0.45, 95% CI = 0.33-0.61 vs RR = 0.87, 95% CI = 0.48-1.58). And, studies with high ROB overestimated by 21% the reduction in diarrhea toxicity (RR = 0.34, 95% CI = 0.20-0.58 vs RR = 0.43, 95% CI = 0.19-1.01). Studies with high ROB also overestimated by 16% improvement in tumor response (RR = 1.39, 95% CI = 1.18-1.63 vs RR = 1.20; 95% CI = 0.81-1.79). Not accounting for ROB would have exaggerated evidence of benefit and failed to detect nonsignificance of results. Conclusions. In the present analysis, involving 36 studies, 2593 patients, 20 outcomes, 36 medical institutions, and 271 named research authors, 92% of the data points were from studies at high ROB. Given the poor quality of the data in studies identified, it cannot be concluded whether combining Chinese herbs with chemotherapy reduces toxicity of chemotherapy.
Introduction
Colorectal cancer (CRC) is the third most commonly occurring cancer and cause of cancer death for both men and women in the United States population, and represents 9% of all cancer deaths for both men and women. 1 In 2013, an estimated 102,480 new cases were diagnosed. 1 Among colorectal cancer patients, approximately 90% of those diagnosed with localized disease will survive five years, but 5-year survival decreases to 68% if lymph nodes are involved, and to 10% if patients have evidence of metastatic spread at the time they are diagnosed. 2 CRC patients with pre-existing diabetes mellitus have an increased risk of short and long-term mortality, post-operative complications, and 5-year cancer recurrence. 3
First-line Therapies
Surgical resection is essential, and staging with sampling of at least 12 adjacent lymph nodes draining the tumor site provides important prognostic information. 4 While adjuvant radiation therapy was in the past frequently included in treatment regimens, its use is now limited to treating patients who have advanced retroperitoneal tumors and for patients with rectal tumors. 5
The earliest established adjuvant chemotherapy treatment was 5-fluorouracil (5-FU), has been in continuous use since 1957. Important improvements in the efficacy of 5-FU were the Mayo Clinic and Roswell Park protocols,6,7 which improved median survival in patients with metastatic disease to 11 months, compared with 5 months with supportive therapy. 8 Efficacy of 5-FU/leucovorin has been significantly improved by combination with irinotecan and oxaliplatin for metastatic patients.9-12 Another significant new treatment development was the advent of FOLFOX regimens, which by adding oxaliplatin to 5-FU/leucovorin improved response rates and survival. 13 Among metastatic patients, alternating FOLFOX and FOLFIRI protocols has helped achieve even greater survival outcomes when compared to 5-FU alone.14,15 However, more than 70% of patients receiving FOLFOX suffer from thrombocytopenia, and approximately 15% of patients receiving oxaliplatin exhibit a hypersensitivity reaction with 2% of patients having severe reactions. Serious complications of immune-mediated thrombocytopenia may occur if blood count and bleeding symptoms are not monitored after the appearance of oxaliplatin-induced hypersensitivity. 16
Another important addition to treatment options has been the oral 5-FU pro-drug, capecitabine, which is frequently combined with irinotecan and oxaliplatin. 17 Newly discovered genetic variations have helped to better understand variations in treatment success with irinotecan and 5-FU. Among patients treated with irinotecan and 5-FU, carriers of the ABCB1 haplotype not only responded to treatment less frequently, but also had a shorter survival time. 18 Additionally, patients in whom there were genetic variations of the ABCB1 haplotype experienced earlier onset of toxicity and reduced effectiveness of irinotecan and 5-FU. 18
Improved clinical outcomes have been found with the combined treatment of cetuximab, 5-FU/leucovorin and oxaliplatin (FOLFOX6) or irinotecan (FOLFIRI) for patients with the KRAS wild-type gene compared with KRAS mutated mCRC. 19 Newly developed monoclonal antibodies include both bevacizumab, which specifically targets circulating vascular endothelial growth factor (VEGF) and cetuximab, which has an affinity for the epithelial growth factor receptor (EGFR)20,21. Adding the monoclonal antibody bevacizumab to chemotherapy has significantly increased progression-free survival by 17.1%, and overall survival by 8.6%. 22 However, a limiting factor to this protocol is hypertension. Significant increase was shown in progression-free survival of 3.7 and 4.4 months, respectively, in a meta-analysis of bevacizumab in combination with 5-FU/FA, and bevacizumab in combination with irinotecan, fluorouracil and leucovorin (IFL), as first-line treatments of CRC. 23
Herbal Therapies
Preliminary data have been published in cell culture, animal, and human trial studies suggesting Chinese herbal medicine may have an adjunctive role in colorectal cancer therapy. Mechanistic data have been published supporting the plausibility of Chinese herbal medicine having clinical benefit through either improving host defense or directly inhibiting tumor growth.
Inhibition of tumor cell growth and upregulation of apoptosis have been observed in colon cancer cell culture models using crude extracts from Chinese herbal medicines such as Radix Eleutherococcus senticosus, 24 and Ganoderma lucidum. 25 Inhibition of tumor cell growth has also been observed using well-characterized purified extracts, including curcumin (from turmeric root, Rhizoma Curcuma longa), 26 allicin (from garlic, Bulbus Allium sativum),27,28 epigallocatechin gallate (EGCG, from green tea, Folium Thea sinensis),29,30 genistein (from the soybean, Semen Glycine max), 31 and tanshinones (from Radix Salvia miltiorrhiza). 32 Tumor inhibition has also been demonstrated in animal models: honokiol (from Cortex Magnolia officinalis), 33 and triptolide (from Radix Trypterygium wilfordii). 34
Suppression of colon cancer metastasis has been demonstrated with the herbal extract fucoidan (from the seaweeds Kun Bu and Hai Zao, Sargassum), 35 and the herbal combination Pien Tze Huang. 36
Anti-tumor effects of Chinese herbal medicines have also been identified as acting through multiple molecular pathways: inhibition of vascular endothelial growth factor (VEGF) and matrix metalloproteinases with Sargassum, 35 Fas receptor upregulation, and caspase activation, and reduction of mitochondrial membrane potential (MMP, Psim) with Bax protein activation and cytochrome c release with Cortex Morus alba. 37
Improvements in host defense mechanisms have also been observed, which may contribute to improved survival and quality of life. These data include reversal of muscle cell atrophy in cachexia induced by Rhizoma Anemarrhena asphodeloides and Cortex Phellodendron amurense, 38 reduction of gastrointestinal dysfunction following colon surgery achieved by the Chinese herbal combination Da Jian Zhong Tang, 39 reduction of FOLFOX6-related peripheral neuropathy with herbal combination Niu Che Sen Qi Wan, 40 and reduction of post–colorectal surgical time to tolerance of regular diet with the combinations Da Jian Zhong Tang and Gui Zhi Fu Ling Tang. 41 Chinese herbal medicines may derive their reported benefit for colon cancer patients because they are typically used in multi-ingredient combination formulas, thus taking advantage of multiple pathways of therapeutic action.
Prior meta-analysis of randomized trials of Chinese herbal medicine in colorectal cancer has shown a modest increase in 1-year survival (odds ratio [OR] 2.41, 95% CI 1.32-4.41) and 3-year survival (OR 2.40, 95% CI 1.49-3.87), reduction in cancer progression (OR 0.50, 95% CI 0.32-0.77), and improved quality of life (OR 3.43, 95% CI 2.35-5.02). 42 However, the authors did not critically evaluate study quality. In the current article, we sought to critically examine the evidence for effectiveness of Chinese herbal medicines in colon cancer patients, with particular emphasis on study quality. We decided to identify for our systematic search and meta-analysis those published randomized trials using fluorouracil-based chemotherapy in both treatment and control groups because this therapy is a key component of most standard front-line treatment protocols for colorectal cancer.
Materials and Methods
Systematic Search
We conducted systematic searches of TCMLARS (1984 to 2014; www.cintcm.com), PubMed (1966 to 2014), Cochrane Library (1988 to 2014), and the Cochrane Central Register of Controlled Trials (1966 to 2014). We sought all randomized trials in any language, to reduce the risk of language bias seen in previous systematic reviews of Chinese herbal medicine. 43 We sought studies reporting on the use of Chinese herbal medicine combined with fluorouracil-based chemotherapy for colorectal cancer patients compared to chemotherapy alone, and synonyms for each term. We also explored references from bibliographies of identified studies. We first screened titles and abstracts, ordered potentially relevant full-text articles, and subsequently screened those articles prior to data extraction (Figure 1).

Flow diagram. RCT, randomized controlled trial.
Study Eligibility
Eligible studies were randomized controlled trials recruiting patients with colon cancer, with allocation to either Chinese herbal medicines combined with fluorouracil-based combination chemotherapy or the same chemotherapy alone, reporting at least one outcome of interest (survival, toxicity, tumor response, performance status and quality of life), with enough detail to allow calculation of risk ratios. We followed a predefined protocol for our systematic review (protocol not registered), which included the PRISMA Statement guidelines (Supplemental Materials available at http://ict.sagepub.com/content/by/supplemental-data). 44
Data Extraction
Three researchers fluent in both Chinese and English (M.M., H.L., and C.S.) extracted data on treatment details, patient characteristics, study quality and clinical outcomes. We grouped studies by outcome of interest for analysis. Only outcomes with 2 or more studies found were included in quantitative meta-analyses.
Study Quality
We used the Cochrane Collaboration’s Risk of Bias (ROB) criteria to critically evaluate the quality of reporting in the randomized trials included in the meta-analysis, for adequate random sequence allocation, group allocation concealment, participant blinding, completeness of outcome reporting, freedom from selective outcome reporting, and other potential sources of bias. 45 Each of these 7 items on the ROB assessment tool was given a possible score by the assessor of 0 for low, 1 for medium and 2 for high ROB. The total is then combined to give an overall ROB score. Low ROB was assigned to studies with total score 0 to 4, medium ROB with total score 5 to 8, and high ROB with total score of 9 and higher, an approach similar to that used by previous researchers in applying the Cochrane ROB tool. 46 The total possible score was 12, consisting of a range of 0, 1, or 2 for each scored quality item (Table 1).
Risk of Bias (ROB) Scores for Studies Identified: Randomized Trials of Chinese Herbal Medicine Combined With Fluorouracil-Based Chemotherapy, Compared With Chemotherapy Alone for Colon Cancer. a

“Other bias” refers to bias due to problems not covered elsewhere in the Cochrane ROB tool.
Analysis of Outcomes
Survival was defined as the number of patients who died at intervals of 1 year, 2 years, and 3 years following completion of chemotherapy. Probability of failure (death) was calculated by the number of patients in the Chinese herbal medicine plus chemotherapy group, divided by that same number in the chemotherapy-only group. Intention-to-treat analysis was used, treating in the analysis any non-reported patients at follow-up times as having failed. A relative risk of less than one would indicate the Chinese herbal medicine plus fluorouracil-based chemotherapy conferred a survival advantage, compared to the same chemotherapy alone.
Reduction of Chemotherapy Toxicity
Most studies identified in our search used the 5-point World Health Organization (WHO) scale to report severity of chemotherapy-related toxicity. We calculated toxicity reduction in each study as the number of patients reporting severe toxicity (WHO grades 2 or higher), divided by the total number in each group of treatment (WHO grades 0 + 1 + 2 + 3 + 4). The risk reduction was then calculated as toxicity reduction in the herbal medicine plus chemotherapy group, divided by that in the chemotherapy alone group. A relative risk of less than 1 would favor the herbal/chemotherapy combination therapy.
Objective Tumor Response
We calculated tumor response as the total number of patients experiencing complete as well as partial response divided by the total number in each treatment group (complete response, partial response, no change, and progressive disease). The relative risk of tumor response was calculated as the probability of tumor response in the Chinese medicine plus chemotherapy group, divided by the total in the chemotherapy-only group. A relative risk of more than 1 would favor the combination treatment regimen.
Performance Status
All studies reporting performance status used the Karnofsky Performance Scale; most used a 10-point change as a cutoff for worsening or improvement of performance status, a few used a 20-point change as a cutoff. We chose to calculate performance status as a proportion of improved or stable status: (greater than 10 point increase and no change) divided by a total status (no change plus greater than 10-point increase or greater than 10-point decrease). The relative risk of improved or stable status for this meta-analysis included the Chinese medicine plus fluorouracil-based chemotherapy in the numerator, divided by this proportion in the fluorouracil-based chemotherapy treatment group. A relative risk of greater than 1 would support the combination treatment regimen.
Meta-Analysis, Between-study Heterogeneity, and Publication Bias
We performed random-effects meta-analysis using the -metan- command in the Stata software package (Stata Corp, College Station, TX), and the I2 measure to evaluate between-study heterogeneity. 47 The Harbord test was used to evaluate publication bias, 48 and the I2 measure to evaluate between-study heterogeneity. 47
Results
Studies Retrieved
Of the 133 potentially relevant abstracts found, we identified 57 full-text articles for further assessment. These were screened and 21 excluded because the study in question was a diagnostic study (n = 1), 49 duplicate article (n = 1), 50 retrospective study (n = 3),51-53 or meta-analysis (n = 1), 54 or because the study design had no herb + chemo group (n = 1), 55 there were no usable outcomes (n = 8),56-63 other cancers were included in the data reported (n = 5),63-67 or stage was not specified (n = 2).68,69 This yielded 36 studies for our meta-analysis (Figure 1).
In data extraction of these 36 studies, we found data potentially eligible for meta-analysis for reduction of chemotherapy toxicity (anemia, diarrhea, fatigue, kidney toxicity, liver toxicity, neurological toxicity, performance status, platelet toxicity, vomiting, white blood cell [WBC] toxicity) and recurrence, survival, and tumor response. However, of these 36 studies, we found only 3 that qualified for data analysis and reporting on the basis of our predefined eligibility criteria: those studies that (a) had low risk of bias (Cochrane ROB tool), (b) were free of publication bias based on the Harbord test, 48 and (c) had low between-study heterogeneity (Table 1).70-72
These 3 studies provided sufficient data for meta-analysis for the following 6 outcomes: reduction of diarrhea toxicity,71,72 neurotoxicity,71,72 platelet toxicity,71,72 vomiting,71,72 WBC toxicity71,72 (all on WHO scale) and tumor response.70,71 Two of these 3 studies were based on the Chinese herbal medicine Astragalus membranaceus.70,72 Clinical outcomes are reported in Table 2, and each study’s treatment characteristics in Table 3.
Results of Meta-Analyses of Randomized Trials, Chinese Herbal Medicine Combined With Fluorouracil-Based Chemotherapy, Compared With Chemotherapy Alone for Colon Cancer. a
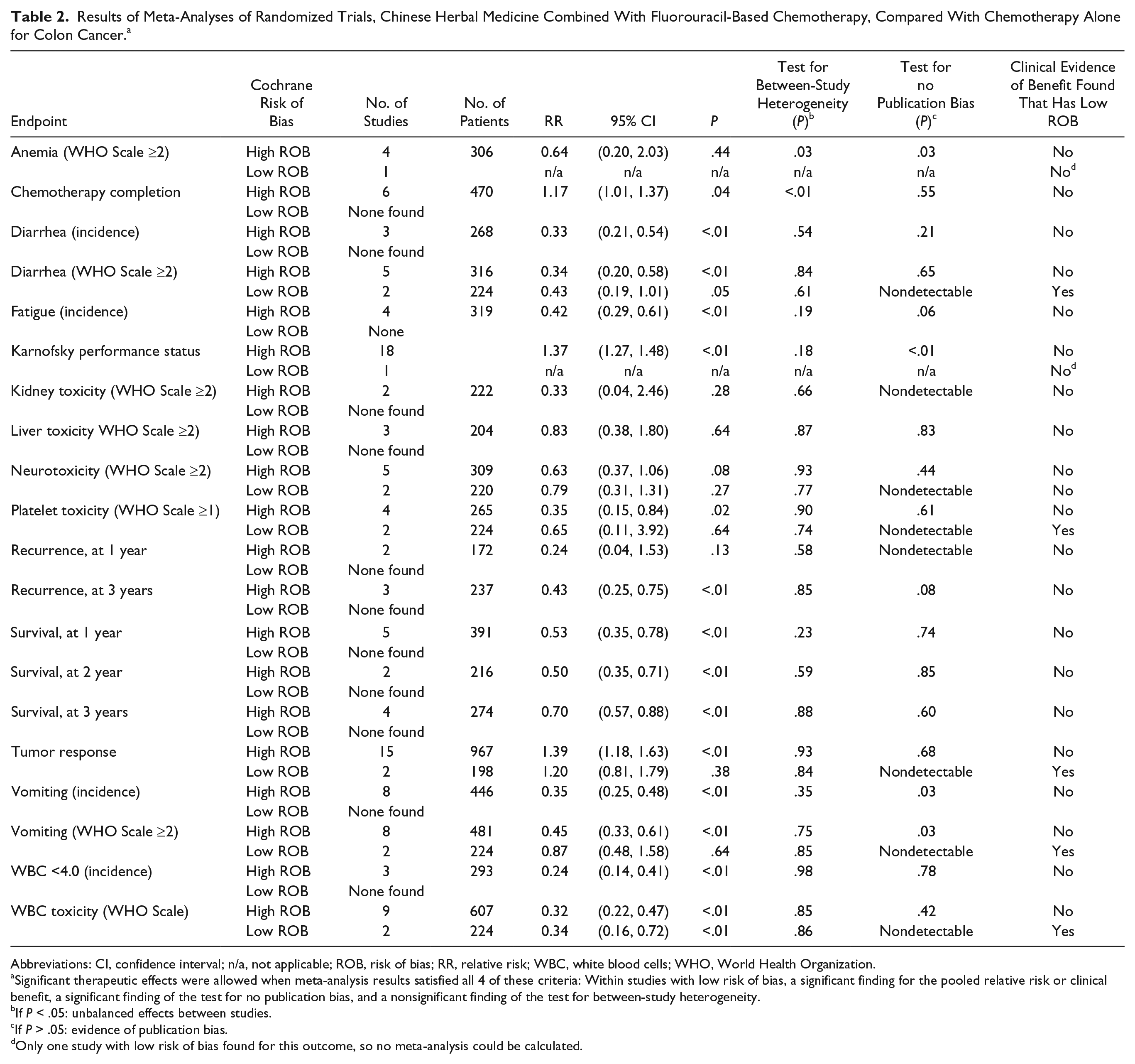
Abbreviations: CI, confidence interval; n/a, not applicable; ROB, risk of bias; RR, relative risk; WBC, white blood cells; WHO, World Health Organization.
Significant therapeutic effects were allowed when meta-analysis results satisfied all 4 of these criteria: Within studies with low risk of bias, a significant finding for the pooled relative risk or clinical benefit, a significant finding of the test for no publication bias, and a nonsignificant finding of the test for between-study heterogeneity.
If P < .05: unbalanced effects between studies.
If P > .05: evidence of publication bias.
Only one study with low risk of bias found for this outcome, so no meta-analysis could be calculated.
Study Characteristics.

For an additional 2 outcomes, there was only 1 study identified per outcome, which made formal meta-analysis not possible: reduction in anemia toxicity (WHO scale), 71 and improvement in Karnofsky Performance Status. 71
Reduction of Diarrhea Toxicity (WHO Scale ≥2)
In meta-analysis, we found 2 high-quality (low risk of bias) studies reporting that addition of Chinese herbal medicine to fluorouracil-based chemotherapy reduced the relative risk of severity of diarrhea toxicity (WHO Scale ≥2) by 57% (relative risk [RR] 0.43; 95% CI 0.19-1.01); however, results were not statistically significant (P = .05; Figure 2),71,72 with P < .05 defined as the upper bound of statistical significance.
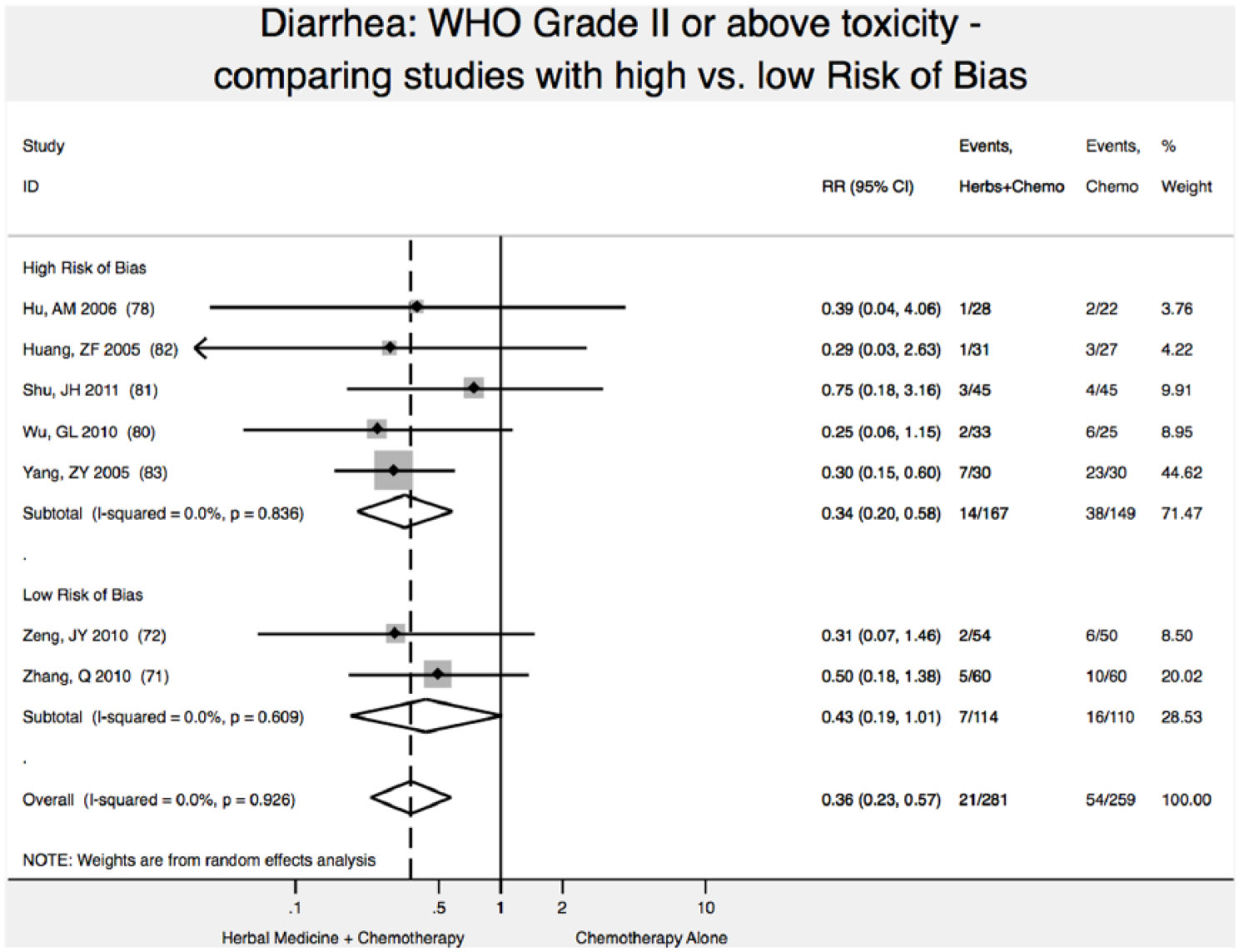
Reduction in diarrhea toxicity.
Reduction of Neurological Toxicity (WHO Scale ≥2)
We found 2 high-quality (low risk of bias) studies reporting that addition of Chinese herbal medicine to fluorouracil-based chemotherapy reduced the relative risk of severity of neurological toxicity (WHO Scale ≥2) by 21% (RR 0.79; 95% CI 0.31-1.31); however, results were not statistically significant (P = .27).71,72
Reduction of Platelet Toxicity (WHO Scale ≥2)
We found 2 high-quality (low risk of bias) studies reporting that addition of Chinese herbal medicine to fluorouracil-based chemotherapy reduced the relative risk of severity of platelet toxicity (WHO Scale ≥2) by 35% (RR 0.65; 95% CI 0.11-3.92), although results were not statistically significant (P = .64; Figure 3).71,72

Reduction in platelet toxicity.
Reduction of Vomiting Toxicity (WHO Scale ≥2)
We found 2 high-quality (low risk of bias) studies reporting that addition of Chinese herbal medicine to fluorouracil-based chemotherapy reduced the relative risk of severity of vomiting toxicity (WHO Scale ≥2) by 13% (RR 0.87; 95% CI 0.48-1.58), although results were not statistically significant (P = .64; Figure 4).71,72

Reduction in vomiting toxicity.
Reduction of WBC Toxicity (WHO Scale ≥2)
We found 2 high-quality (low risk of bias) studies reporting that addition of Chinese herbal medicine to fluorouracil-based chemotherapy reduced the relative risk of severity of WBC toxicity (WHO Scale ≥2) by 66% (RR 0.34; 95% CI 0.16-0.72), with statistically significant results (P < .01; Figure 5).71,72
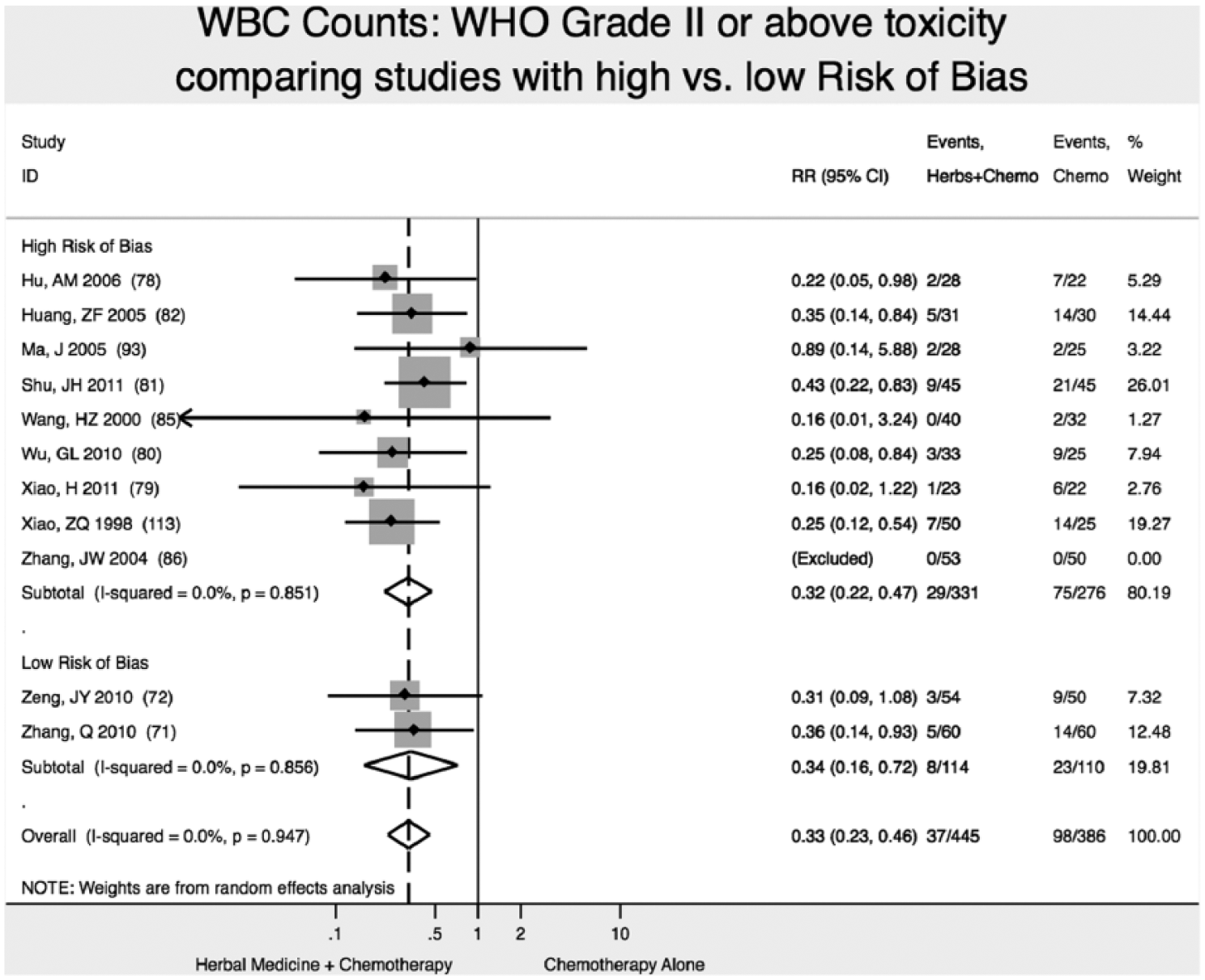
Reduction in white blood cell (WBC) toxicity.
Improvement of Tumor Response (Partial Response + Complete Response)
We found 2 high-quality (low risk of bias) studies reporting that addition of Chinese herbal medicine to fluorouracil-based chemotherapy increase the likelihood of tumor response (partial response or complete response) by 20% (RR 1.20; 95% CI 0.81-1.79), although the results were not statistically significant (P = .38; Figure 6).70,71

Increase in objective tumor response.
Comparing Results of Meta-Analysis of Studies With High Versus Low Risk of Bias
Studies with high ROB overestimated by 21% the reduction in diarrhea toxicity (WHO grade II or higher): RR = 0.34 (95% CI 0.20-0.58) versus RR = 0.43 (95% CI 0.19-1.01). In this analysis, not accounting for study ROB would have failed to detect the nonsignificant result of meta-analysis for reduction of diarrhea toxicity (Figure 2).
Studies with high ROB overestimated by nearly 2-fold the reduction in platelet toxicity (WHO grade II or higher): RR = 0.35 (95% CI 0.15-0.84) versus RR = 0.65 (95% CI 0.11-3.92). In this analysis, not accounting for study ROB would have failed to detect the nonsignificant result of meta-analysis for reduction of platelet toxicity (Figure 3).
Studies with high ROB overestimated by nearly 2-fold the reduction in vomiting toxicity (WHO grade II or higher): RR = 0.45 (95% CI 0.33-0.61), versus RR = 0.87 (95% CI 0.48-1.58). In this analysis, not accounting for study ROB would have failed to detect the nonsignificant result of meta-analysis for reduction of vomiting toxicity (Figure 4).
Studies with high ROB overestimated by 16% the improvement in objective tumor response: RR = 1.39 (95% CI 1.18-1.63) versus RR = 1.20 (95% CI 0.81-1.79). In this analysis, not accounting for study ROB would have failed to detect the nonsignificant result of meta-analysis for improvement in objective tumor response (Figure 5). Statistically significant results on reduction of neurological toxicity were not found in meta-analysis of studies at either high or low ROB.
We found no difference in results of meta-analysis comparing studies with high ROB versus low ROB for reduction of WBC toxicity (Figure 6).
Discussion
Our meta-analysis found very limited published evidence supporting the efficacy of Chinese herbal medicines when used in combination with fluorouracil-based chemotherapy for patients with colon cancer. We found qualified reportable results (those published in articles with low ROB) for only 20% (n = 4) of the 20 outcomes identified: diarrhea, neurological, platelet, vomiting, and WBC toxicity (all on WHO Scale), and objective tumor response. In only 5% (n = 1) of all outcomes analyzed were the results of the analysis both at low ROB and also statistically significant: WBC toxicity.
Study Quality in Articles Assessed
Over the past decade, our team has conducted meta-analyses of Chinese herbal medicines in patients with chronic hepatitis B, 73 advanced non–small cell lung cancer, 74 and hepatocellular carcinoma, 75 initially using the Jadad quality scale. 76 The availability in 2011 of the Cochrane Risk of Bias tool has provided meta-analysts with a clearly-defined set of metrics with which to efficiently evaluate the quality of underlying studies. 45 This tool asks analysts to assign to each study being assessed a 0 to 2 score, that in their judgment the study’s reporting indicates a low ROB, unclear, or high risk. This is assigned in 6 domains: adequate random sequence allocation, group allocation concealment, participant blinding, completeness of outcome reporting, freedom from selective outcome reporting, and other potential sources of bias. This score when totaled is used to assign a low risk (score 0-4), medium (5-8), or high (9-12) risk that the study’s design or outcomes are subject to bias. In effect, it is an indicator of apparent trustworthiness of the reported data.
However, 80% of the evidence (16/20 outcomes) we found has virtually no clinical usefulness, because the articles in which they were published were at high ROB using the Cochrane Risk of Bias tool. 45 Most studies used the outdated clinical outcome for chemotherapy toxicity—the WHO Scale71,72,77-86—a method that is more prone to bias than the 12-year-old CTCAE scale. 87 All studies reporting patient survival and time to recurrence reported these outcomes at fixed time points (1, 2, or 3 years),88-93 a method prone to substantial analytic bias compared to the more modern hazard ratio method. 94 To help understand the broader scientific context within which our results are found, we informally compared our results with the findings of other recently published meta-analyses of Chinese herbal medicines, and found similar issues with study quality and risk of bias.42,95,96
Limitations
Heavy metal contamination in Chinese herbal medicines has been reported.97,98 It is additionally known that heavy metal accumulation can contribute to cancer development, in part due to increased activation of tumor initiation and promotion signaling pathway such as epidermal growth factor receptor (EGFR), phosphatidyl inositol 3-kinase (PI3K), AKT, and mammalian target of rapamycin (mTOR) in carcinogenesis and cancer progression. 99 Therefore, unmeasured confounding may exist in our results due to unknown interaction of heavy metal residues with chemotherapy efficacy.
Most symptomatic outcomes were measured on the WHO Scale, which we have specified in the manuscript. However, several (incidence of diarrhea, fatigue, and vomiting) were not measured using this scale, and in the underlying studies analyzed, it is not clear in the reporting how they were measured, further diminishing the clinical value of our results.
Conclusions
Although these 36 studies involved 2,593 patients, 20 outcomes, 36 medical institutions, and 271 named research authors, unfortunately most of the data points suggesting clinical benefit are of virtually no clinical value due to very poor methodological quality of the studies. Because stratifying our analysis by the articles’ level of risk of bias showed no statistically significant difference in effect size, we suggest that virtually all of these studies to some extent suffer from such risk of bias. Additionally, no studies reported any adverse effects monitoring associated with the use of Chinese herbal medicines, a shortcoming common to many other such RCTs published in China.42,95,96,100 In short, the high frequency of low-quality and/or biased studies of Chinese herbal medicine undermines confidence in the results of published meta-analyses of these trials. This is unfortunate given the wealth of information on combination therapies available from the tradition of Chinese medicine. The solutions to these problems may be found in improved researcher methodology and ethics training, comprehensive clinical trial oversight, and reformed medical journal peer-review and editorial practices.
Footnotes
Acknowledgements
We appreciate the support of Michelle Ching, Daniel Eng, Raleigh Harrell, Andrew Liszt, Nina Ng, Anita Pietrofita, Heather Voborsky, and Carolyn Yeh in conducting background research.
Authors’ Note
Data are available to readers on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
