Abstract
Introduction. Patients with advanced-stage lung cancer face poor survival and experience co-occurring chronic physical and psychosocial symptoms. Despite several years of research in exercise oncology, few exercise studies have targeted advanced lung cancer patients undergoing chemotherapy. The aim of the present study was to investigate the benefits of a 6-week supervised group exercise intervention and to outline the effect on aerobic capacity, strength, health-related quality of life (HRQoL), anxiety, and depression. Methods. VO2peak was assessed using an incremental exercise test. Muscle strength was measured with one repetition maximum test (1RM). HRQoL, anxiety, and depression were assessed using Functional Assessment of Cancer Therapy–Lung (FACT-L) scale and the Hospital Anxiety and Depression Scale (HADS). Results. One hundred and forthteen patients with advanced stage lung cancer were recruited. Forty-three patients dropped out. No serious adverse events were reported. Exercise adherence in the group training was 68%. Improvements in VO2peak (P < .001) and 6-minute walk distance (P < .001) and muscle strength measurements (P < .05) were seen. There was a reduction in anxiety level (P = .0007) and improvement in the emotional well-being parameter (FACT-L) but no statistically significant changes in HRQoL were observed. Conclusion. The results of the present study show that during a 6-week hospital-based supervised, structured, and group-based exercise program, patients with advanced-stage lung cancer (NSCLC IIIb-IV, ED-SCLC) improve their physical capacity (VO2peak, 1RM), functional capacity, anxiety level, and emotional well-being, but not their overall HRQoL. A randomized controlled trial testing the intervention including 216 patients is currently being carried out.
Keywords
Introduction
For patients with advanced-stage lung cancer (non–small cell lung cancer [NSCLC] stage IIIb-IV; extensive-stage disease small cell lung cancer [SCLC-ED]), the median survival after diagnosis is 10 to 13 months. 1 Despite improved diagnostic and treatment methods and increased focus on supportive therapy, survival has not changed significantly over the last decade.2,3
Consequently, a natural question to ask is whether physical exercise interventions can benefit patients with advanced-stage lung cancer. In 2009, Temel et al were the first to perform a study addressing this question. 4 The intervention comprised 8 weeks of aerobic exercise and weight training twice weekly. Due to low adherence (completion rate 44%) and few significant results (lung cancer symptoms, elbow extension), the authors concluded that a less intensive, community-based, or briefer exercise intervention may be more feasible in this population.
Since then, 4 feasibility and/or pilot studies,5 -8 including a study from this research group, have examined various (eg, duration, intensity, type) supervised exercise interventions in this population. These studies, however, have enrolled small populations (n range from 24 to 46) and have dropout rates of 20.7% to 56% and present data on few patients (n = 11-31). All the studies5 -8 found improvements in physical and functional capacity but failed to show improvements in overall HRQoL, anxiety, or depression. These results represent 164 patients with advanced-stage lung cancer, but only data on 118 patients were evaluated. Consequently, no exercise recommendations can be issued at present, despite the fact that none of the studies reported serious adverse events, though smaller adverse events were reported.
Patients with advanced-stage cancer generally have a greater need for supportive care, due to poorer physical functioning, a greater symptom burden, and higher levels of distress, anxiety, and depression than other cancer patients.9 -14 Moreover, physical activity and relaxation training have been shown to relieve side effects and symptoms in cancer patients with advanced disease undergoing chemotherapy.15 -17
Patients with advanced-stage lung cancer face poor survival and experience co-occurring chronic physical and psychosocial symptoms. The nature of symptoms calls for complex interventions designed to address these symptoms, which is why our group designed a multimodal supervised group intervention combined with a home-based walking program that was tested in a feasibility study.6,18,19 Due to low adherence in the home-based intervention and lack of energy and motivation toward the home-based component, 18 this part was removed in this phase II study. This observation was in line with our previous research, stating that Danish cancer patients have preferences for exercise organized in groups with peers.20,21 Therefore, high adherence rate to the group exercise component (73%) and indication of promising results within some aspects of physical and psychosocial outcomes formed the basis for the present phase II study including a larger population.
We hypothesized that patients with advanced-stage lung cancer, irrespective of symptoms and undergoing chemotherapy, could maintain their physical and functional capacity. The aim of the present phase II study was therefore to investigate the benefits of a 6-week supervised and structured multimodal group exercise intervention in patients with advanced-stage lung cancer undergoing chemotherapy and to outline the benefits on aerobic capacity, muscle strength, HRQoL, anxiety, and depression.
Materials and Methods
This was a prospective study that used a 1-group design, and outcomes were assessed at baseline and 6 weeks.
The subjects were over 18 years of age; had a World Health Organization performance status of 0, 1, or 2 with stage IIIb-IV NSCLC and SCLC-ED; and were undergoing chemotherapy after referral from the Department of Oncology, Rigshospitalet, University of Copenhagen. Exclusion criteria were the following: brain or bone metastases; prolonged bone marrow suppression; anticoagulant treatment; symptomatic heart disease, including congestive heart failure, arrhythmia, or myocardial infarction diagnosed within the last 3 months; and inability to provide informed consent. A clinical nurse specialist (JL) carried out the screening procedure for inclusion/exclusion.
The study was approved by the Danish Data Protection Agency (File No. 2008-41-2279) and by the Regional Ethics Committee of the Capital Region (Case No. HA-2008-06). All included patients provided signed informed consent.
Prior to participating in each physical training session and before the physiological tests, a clinical nurse specialist (JL) screened each patient.6,22 If one of the following criteria were met, the patient was prohibited from exercising/being tested on that day: diastolic blood pressure <45 or >95; heart rate (HR) at rest >115/min; temperature >38°C; respiratory rate at rest >30/min; infection requiring treatment; fresh bleeding; and total leukocyte count <1.0 × 109/L or platelets <50 × 109/L. Physical tests and the HRQoL assessment were performed at baseline and after 6 weeks of training.
Aerobic capacity (VO2peak) was measured using a stationary cycle ergometer (MONARK Ergomedic 839E) Watt (W) max test. The test started with a burden of 10 to 70 W and increased thereafter by 5 to 10 W. The test was complete when the patient could no longer maintain a momentum of 60 RPM or if the patient felt discomfort (eg, developed sudden paleness, dizziness, sudden changes in heart rhythm, or developed a cold sweat). The burden achieved (maximum power output [MPO]) was used to calculate the estimated VO2peak = 0.16+ (0.0117 × MPO) and was expressed in liters per minute. 23 This test has been used before in the same patient population undergoing chemotherapy 6 and in other studies of cancer patients undergoing chemotherapy performed by this group.6,15
Muscle strength was measured by one repetition maximum (1RM) tests using a Technogym that included a leg press (lower extremity), chest press (pectoral muscles), lateral machine (latissimus dorsi), leg extension (quadriceps femoris), abdominal crunch (rectus abdominis), and lower back press (erector spinae). The 1RM test has been found to be a reliable assessment for measuring upper and lower extremity strength. 24
Functional capacity was measured by a 6-minute walk test (6MWD). The 6MWD test was performed twice in a 20-m corridor according to ATS/ERS guidelines. 25 The 6MWT, which has demonstrated good reliability and validity in chronic obstructive pulmonary disease (COPD) patients, 26 has also been used in several studies to demonstrate functionality in patients with lung cancer.7,27
With regard to lung capacity, forced expiratory volume in 1 second (FEV1) was measured using a spirometer (piko-6, Ferraris Respiratory), which has demonstrated good reliability and validity in COPD patients. 28
FACT-General and FACT-L were used to evaluate HRQoL and cancer-related symptoms. The reliability and validity of FACT-L has been documented in patients with lung cancer. 29
Demographic data were collected using questionnaires and patient files.
Anxiety and depression were measured using HADS, which is a 14-item questionnaire comprising 2 scales covering anxiety (HADS-A) and depression (HADS-D). HADS-A (7 items) measures generalized autonomic anxiety and indicates physiological and emotional states characterized by high muscle tension and strong feelings of subconscious and uncontrollable fear or anger. HADS-D (7 items) measures anhedonia, understood as a complete lack of pleasure or the capacity to experience it. Each item is scored on a 4-point Likert-type scale. 30
The intervention comprised supervised group training (physical training and relaxation) carried out in groups of 10 to 12 patients twice weekly. Each session lasted 1.5 hours and was supervised by a research physiotherapist (MQ). The training comprised warmup exercises, strength and fitness training, and stretching. Warmup exercises consisted of 10 minutes of stationary cycling, adjusted to 60% to 90% of the patient’s maximum HR. Strength training was carried out using the 6 Technogym machines described above. The practical aim of the strength training was to complete 3 series of 5 to 8 sets, with 70% to 90% of 1RM. The exercises were specifically selected to involve the largest possible number of muscle groups in the least number of exercises.
To ensure progression in the strength training, each patient was taught how to carry out the 1RM test, using each machine once every other week, after which their program would be adjusted. Cardiovascular training was carried out as interval training on stationery bicycles. Intensity was equivalent to 85% to 95% of each patient’s maximum HR and lasted approximately 10 to 15 minutes. All patients were wearing HR monitors. After the training session, 5 to 10 minutes were dedicated to stretching the large muscle groups to increase agility. Following each training session, 15 to 20 minutes of progressive relaxation was performed. The intervention was based on 3 key principles: (a) early initiation of an intervention during cancer treatment; (b) EXercise/physical activity; and (c) Patient ACTivation (EEX-ACT).31,32
Statistical Analysis
Baseline values of the study population were compared to baseline values in the subgroup (dropout) for which measurements were not obtained at follow-up, for each of the variables: aerobic capacity, muscle strength, functional capacity, lung capacity, and for each subscale of the FACT and HADS instruments. Independent-samples t tests were used for this purpose.
The variables are reported as means and standard deviations (SD) at baseline and at follow-up, respectively.
The effect of the intervention is reported as change scores with corresponding 95% confidence intervals (CI) and the groups (completer and dropout) were compared using independent-samples t tests. The effect of the intervention was examined using a linear mixed model taking into account the effect of gender, age, marital status, smoking, and cancer stage.
Effect sizes were estimated using Cohen’s guidelines, whereby a value of 0.2 denotes a small, 0.5 a medium, and 0.8 a large effect size. 33 Effect size was calculated by the mean difference divided by the pooled SD and the root mean square error estimated from the general linear model.
Results
From October 2008 to January 2012, 713 patients with inoperable NSCLC and SCLC were screened for eligibility (Figure 1); 344 patients were excluded, leaving 369 eligible patients.

Flow chart over eligible lung cancer patients.
With 114 patients included in the study, the recruitment rate was 30.8% (57 females, 57 males; median age 66; Table 1). The patients were undergoing concurrent systemic treatment (Table 2). Patients (n = 29) who were included in the feasibility study 6 were also included in this study. All 114 patients completed the baseline testing; however, 43 dropped out (37.7%) and did not perform the 6-week test point due to disease progression (n = 10), lack of energy (n = 12), or because they did not wish to participate in the training (n = 21). They did not differ demographically (Table 1) or regarding treatment (Table 2) from the patients included in the analyses. Thus, for most of our analyses, there were results available from 71 patients. Only 70 patients were available for the analysis of anxiety, depression, and the FACT variables.
Baseline Demographic Characteristics of the Population (N = 114).

Concurrent Systemic Antineoplastic Treatment in Patients With Inoperable Lung Cancer (N = 114).

Abbreviations: NSCLC, non–small cell lung cancer; SCLC, small cell lung cancer.
Appendix S1 provides complete details for the dropout analysis comparing patients who dropped out and the patients who completed the study (all supplementary materials are available online at http://ict.sagepub.com/content/by/supplemental-data).
Baseline comparison of patients who dropped out and the patients who completed the study revealed significant differences in 6MWD in favor of the completing group (67.1 m; 95% CI [7.0; 127.2]), which was also true for the dropout group for anxiety (2.53; 95% CI [0.83; 4.23]) and emotional well-being (−2.77; 95% CI [−4.760; −0.77]).
Physical Capacity and Functional Capacity
Table 3 shows the results of aerobic capacity (VO2peak, 1RM) and functional capacity after the 6-week program. There was a significant increase in aerobic capacity, VO2peak (P < .0001, effect size 0.22) and functional capacity, 6MWD (P < .0001, effect size 0.27). There was a significant improvement in strength: leg press (P < .0001, effect size 0.46); chest press (P < .0001, effect size 0.35); lateral machine (P = .0063, effect size 0.13); leg extension (P < .0001, effect size 0.31); abdominal crunch (P < .0001, effect size 0.47); and lower back press (P < .0001, effect size 0.36).
Aerobic Capacity (VO2peak, 1RM), Functional Capacity (6MWD), and Muscle Strength Before and After the 6-Week Intervention (N = 71).

Abbreviations: 1RM, one repetition maximum test; 6MWD, 6-minute walk test; SD, standard deviation; CI, confidence interval; FEV1, forced expiratory volume in 1 second.
Anxiety and Depression
There was a statistically significant reduction in anxiety score as shown in table 5 (P = .0075, effect size 0.21) from baseline to 6 weeks with a reduced anxiety score of −0.9 points. There was no significant reduction in depression (P = .0755, effect size 0.16).
Health-Related Quality of Life
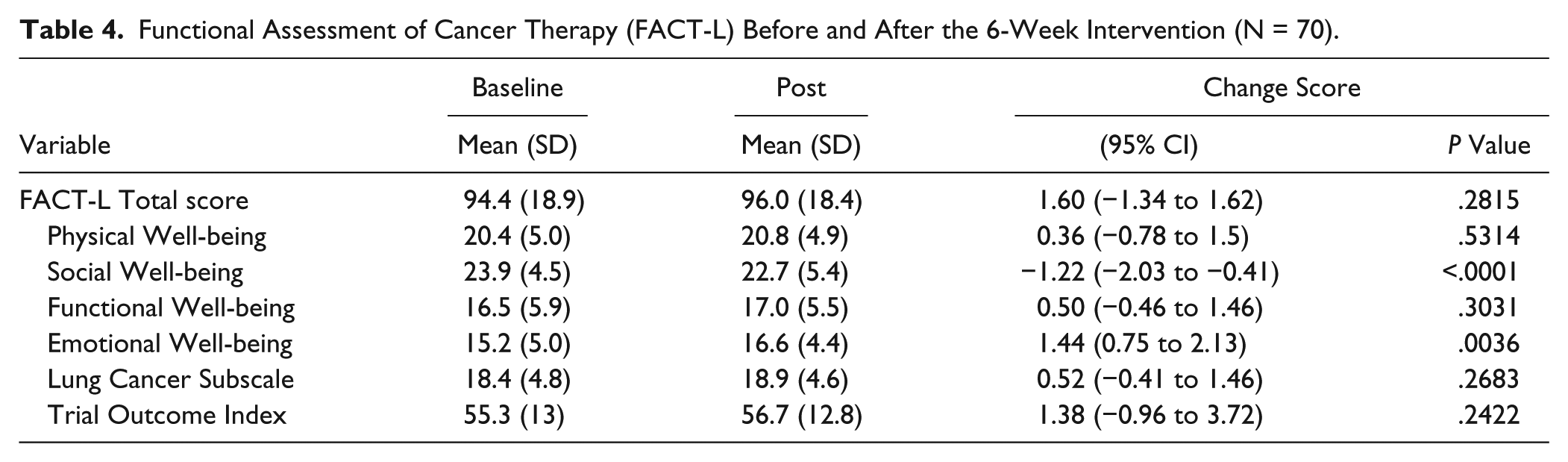
Table 4 presents the HRQoL results. There was a significant change in the emotional well-being parameter (P < .0001, effect size 0.29) when comparing baseline to the 6-week evaluation and a significant decrease in social well-being (P = .0036, effect size 0.24). There was no significant improvement or decline, however, in general HRQoL, fatigue, or on the FACT-L subscales (ie, physical well-being, functional well-being, social/family well-being, and lung cancer subscale).
Functional Assessment of Cancer Therapy (FACT-L) Before and After the 6-Week Intervention (N = 70).
Hospital Anxiety and Depression Scale (HADS) Before and After the 6-Week Intervention (N = 70).

Adverse Events
No serious adverse events were reported, but during the prescreening process before the supervised training, 10 patients were excluded from the physical training component (1 to 2 exercise sessions out of 12) due to fever, dizziness, pain, and bodily discomfort.
Adjusted Analyses
Additional analyses adjusted for the effect of gender, age, marital status, smoking, and cancer stage yielded comparable results. Appendix S2 provides the details of these adjusted analyses.
Discussion
Patients with advanced-stage lung cancer will, due to disease progression and chemotherapeutic cumulative effects, experience a decrease in physical and functional capacity34,35 throughout the course of their disease and, as a result of this, experience poor HRQoL. Therefore, a positive response from this intervention would be maintenance of physical and functional capacity. In support of our hypothesis, the intervention proved beneficial with a small effect size <0.3 on aerobic capacity (VO2peak), muscle strength (1RM), and functional capacity (6MWD). We also found increased emotional well-being (FACT-L) and reduction in social well-being (FACT-L) and anxiety (HADS).
In the present study, we found a significant increase (P < .0001) in VO2peak (primary outcome) after 6 weeks of training (Table 3), which is in line with the study by Hwang et al, 5 which also achieved a significant improvement (P = .001) in VO2peak in a randomized study with 24 locally advanced and advanced-stage lung cancer patients (NSCLC, stage IIIa-IV). 5 The intervention, which lasted 8 weeks, took place 3 times a week with 30- to 40-minute exercise sessions that included a 10-minute warmup and a 5-minute cool-down phase, all supervised by a physiotherapist. These 2 studies used similar interventions that incorporated VO2peak training (range) that is likely to affect VO2peak in advanced-stage lung cancer. The clinical significance of an improved VO2peak is not known for inoperable lung cancer patients, but VO2peak has been demonstrated to be an independent predictor of mortality in a wide range of adult noncancer populations.36,37
The present study’s significant reduction in anxiety (HADS) and improvement in emotional well-being (FACT-L) should be seen in light of the fact that all the included patients with lung cancer were terminally ill and had an expected 5-year survival rate of <15%. 1 Patients often reported an escalation of anxiety when experiencing somatic symptoms like nausea, fatigue, or pain, which may be functionally limiting and difficult to interpret. 38 A possible explanation for the change in anxiety and emotional well-being could be that the patients in this study increased their aerobic capacity (VO2peak, 1RM) and functional capacity (6MWD) significantly. It must be assumed that patients who consent to participate in an intervention of this nature expect to improve their fitness, strength, and functioning. Once this expectation has been fulfilled, patients may change their attitude toward their own situation. This is supported by a qualitative interview survey of the patient population in this study. 18 Another possible explanation for this increase may be the effect of the relaxation exercises, which in addition to the effect on the overall HRQoL have also been shown to have an effect on emotional well-being. This effect is emphasized in a systematic review of randomized controlled trials of psychological and/or lifestyle interventions for adults with COPD that concluded that complex psychological and/or lifestyle interventions that include an exercise component significantly reduce symptoms of depression and anxiety. 39
In contrast to the significant reduction in anxiety and improvement in emotional well-being in the present study, we found a reduction in social well-being. One possible explanation for this rather contradictory finding is the effect resulting from being in a group with other patients with lung cancer, with the same prognosis as one’s self. When patients experience a shared understanding with other patients about their cancer, it may stand in contrast to the extent to which family and friends accept their situation. This is supported by a qualitative interview survey of the patient population in this study. 18
We did not find any significant improvement in overall HRQoL (FACT-L total score), which is consistent with similar exercise intervention studies.4,5,7,8 Being diagnosed with advanced-stage lung cancer and exposed to chemotherapy disrupts the patient’s life, which affects physiological and psychological functioning and contributes to negative effects on their HRQoL.
The strengths of this study are the use of well-validated objective tests (indirect VO2peak, 1RM, 6MWD), the anxiety and depression scales (HADS-A and HADS-D), the psychometric testing (FACT-L), and the dropout analyses. Pujol et al 40 and Jones et al 41 point out the importance of using the right tests to measure VO2peak. Our study employed the indirect Watt max test, which has been validated by comparison to a direct watt max test and a high correlation was found. 15 Independent qualitative research also shows that cancer patients receiving chemotherapy experience greater confidence when performing the Watt max test, the 1RM test, and in subsequent workouts. 20 In our study, we chose to maintain high training intensity, as this has been shown to be effective in a heterogeneous group of cancer patients undergoing chemotherapy when they exercise 3 times a week for 6 weeks. 15
The results of this study are based on 71 out of 114 enrolled lung cancer patients, which means generalization of its results may be limited. The results demonstrate the effect on this selected group of lung cancer patients. To determine the effect of the intervention, it should be tested in a randomized controlled study. Another limitation of this study is selection bias. All patients in the department who met the inclusion criteria were screened and then offered participation in the project. Thus, it cannot be ruled out that those who participated in the training had a higher motivation to participate than those who did not wish to participate (N = 255). In addition, lung cancer patients with brain metastases, bone metastases, and a WHO performance status worse than two were excluded.
A methodological weakness of the study is the fact that the professionals in charge of the physical training also collected the data. Entering and analysis of data were carried out by staff not involved in the study.
Conclusion
The results of the present phase II study show that lung cancer patients with advanced disease (NSCLC, stage IIIb-IV, and SCLC-ED) improve their aerobic capacity (VO2peak, strength), functional capacity, anxiety level, and emotional well-being during a 6-week hospital-based, supervised, structured, and group-based exercise program, but not their HRQoL. A randomized controlled trial including 216 patients testing the intervention is currently being carried out.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Center for Integrated Rehabilitation of Cancer Patients (CIRE), which was established by and receives support from the Danish Cancer Society and the Novo Nordisk Foundation. The study was also supported by a grant from Roche A/S.
