Abstract
Purpose:
Endometrial cancer is associated with the highest comorbid disease burden of any cancer. The aim of this trial was to assess the feasibility and safety of an allied health intervention during adjuvant treatment.
Methods:
A mixed-methods pilot randomized (2:1) controlled trial with concealed allocation and assessor-blinding. Eligibility criteria: adjuvant endometrial cancer treatment scheduled, disease stage I-IIIC1, ECOG 0-2 and able to perform unsupervised physical activity (PA). Participants received usual care and 8 sessions of weekly, individualized, lifestyle education (diet and PA) with behavior change and social support (intervention group), delivered predominantly by telehealth, or usual care alone. Feasibility outcomes: recruitment and consent rates, decline reasons, program acceptability, intervention adherence and retention.
Results:
22/44 eligible patients (50%, 95%CI: 36%, 64%) were recruited over 10 months (14 intervention, 8 usual care). The recruitment rate was 2.2 patients/month (95%CI: 1.4, 3.3). Patients who declined had too much going on (7/22, 32%) or were not interested (6/22, 27%). Mean (SD) age and BMI were 63.2 years (6.8) and 31.9 kg/m2 (6.7). A majority were FIGO stage I (15/22, 68%) and received vaginal brachytherapy (14/22, 64%). Adherence was high, 11/14 (79%, 95%CI: 52%, 92%) participants attended >70% of scheduled sessions. Retention was 100% (95%CI: 85%, 100%) at 9 weeks, however completion of objective measures was impacted by COVID-19 restrictions. Telehealth and online questionnaires enabled participation. No serious adverse events occurred.
Conclusion:
The intervention was acceptable to participants with high levels of adherence and retention. Trial findings will be used to design a future RCT.
Trial registration:
The trial was registered on www.anzctr.org.au (ACTRN12619000631101) 29/04/2019.
Introduction
Globally in 2018 there were over 380 000 endometrial cancer cases, making it the most common gynecological cancer 1 and disease prevalence and survival are rising. 2 Endometrial cancer is associated with the highest burden of comorbid disease of any form of cancer. 3 Common co-morbidities include hypertension and osteoarthritis. 3 Obesity and poor nutrition are common 4 and are associated with mortality from obesity-related cardiovascular disease. 5 Data from the SEER Program database, of over 40 000 participants, indicate that cardiovascular disease was the leading cause of death in those with endometrial cancer 5 and higher body mass index (BMI, 35+ vs <25) in endometrial cancer survivors is significantly associated with increased mortality (hazard ratio [95%CI] 2.14 [1.08-4.24]). 6
Despite guidelines for nutrition and exercise for people with cancer7,8 and well-established benefits of pre and post-diagnosis physical activity (PA) including improvements in cardiovascular health, health-related quality of life (HRQoL),9,10 fatigue, 11 and survival,12,13; few endometrial cancer patients meet the recommendations (27% nutrition and 12% exercise).4,14 Endometrial cancer survivors report that specific PA and nutrition recommendations were rarely made by oncologists. 15 Participants expressed a desire for monitoring and the need for external motivation to change health behaviors successfully. Identified barriers to successful health behavior change for PA included a lack of motivation, costs associated with gym visits, the weather and emotional eating. 15
There is great potential for low-cost allied health behavior change interventions to improve outcomes which are important in endometrial cancer. Predominantly observational studies demonstrate positive associations between participants who were meeting PA guidelines and HRQoL.4,16 Significant associations are reported between PA, body weight and HRQoL 4 and cancer-related fatigue. 17 Few high-quality studies have assessed the impact of multi-disciplinary allied health interventions in this population and most have been conducted post-treatment completion.14,17-25 Further trials across the treatment continuum are required to strengthen existing evidence. 23
The primary aim of this pilot randomized controlled trial (RCT) was to determine the feasibility, acceptability and safety of a telehealth allied health intervention involving nutrition and physical activity education combined with behavior change and social support, during adjuvant treatment for endometrial cancer.
Methods
Conduct and reporting conform to the CONSORT extension guidelines for randomized pilot and feasibility trials. 26 The intervention components of this trial were reported following the Template for Intervention Description and Replication (TIDiER) guidelines. 27
Trial Population and Recruitment
Eligibility criteria are provided in Table 1. Hospital outpatient clinic lists were screened and eligible patients were provided with a flier regarding the trial from a member of the treating team (medical or radiation oncologist or nurse consultant) during their appointment. Details of patients who expressed an interest in participation were sent by the treating team to the trial staff who subsequently contacted the patient by telephone and provided them with the trial plain language statement and consent forms either by email or mail. Written, informed consent was obtained from all individual participants included in the trial at the start of the baseline assessment appointment. The study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Peter MacCallum Cancer Centre Human Research Ethics Committee on 14th March 2019 (HREC/48033/PMCC-2019).
Trial Eligibility Criteria.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; PA, physical activity.
Study Design
Using a mixed-methods, pilot, parallel group, randomized controlled trial with concealed allocation and assessor blinding, participants were randomized (2 [intervention group]:1[usual care group] allocation) to receive either usual care plus the intervention or usual care alone. The randomization schedule was generated by an independent statistician using permuted random blocks with stratification by adjuvant therapy type (vaginal brachytherapy, pelvic radiotherapy alone, concurrent pelvic radiotherapy and chemotherapy or concurrent pelvic radiotherapy and chemotherapy followed by adjuvant chemotherapy). Randomization was implemented and concealed through the trial database, with an independent database administrator uploading the randomization schedule.
Trial Intervention
Usual care at the trial site did not routinely involve referral to physiotherapy, exercise physiology, nutrition, or social work services during the recruitment period. Participants in each group received usual care support, with no measures taken to control for these services in the usual care group. Nurse consultant usual care includes routine referral to social work or psychology support where indicated, but no access to routine support for weight management or exercise support. In addition to usual care, participants randomized to the intervention group received 8 weeks of weekly individualized PA and nutrition education with behavior change and social support. All intervention sessions were scripted to ensure standardized content. The initial multi-disciplinary session was primarily conducted face-to-face at the hospital, with weekly follow-up conducted via telehealth (video or telephone). The social work assessment was standardized using the Cancer Rehabilitation Evaluation System questionnaire and included screening for social support and social networks. 28 Social work intervention was individualized according to identified needs and referral to support services was made as necessary. In a combined consultation, the dietitian and physiotherapist provided individualized education informed by baseline assessment findings and patient preferences. Education included discussion of the potential consequences of an inadequate diet and inactivity following a diagnosis of endometrial cancer. Recommended nutrition, weight management, PA and associated benefits for people with endometrial cancer were discussed and standard written educational material provided. Participants worked with the health professionals to establish a home-based PA program and develop individualized, patient-centered nutrition and PA-related goals. Participants were educated regarding the benefits of performing PA at a moderate-intensity and guided to use the Borg 29 scale to monitor intensity.
Behavior change strategies to increase and sustain dietary and PA behaviors, based on HealthChange® methodologies, included action plan development, goal setting, and identification of barriers and enablers. 30 Participants were given the option of using a pedometer and a diary to record PA and nutrition details. Participants were also given the option of receiving personalized nutrition and PA motivational text messages. The physiotherapy, nutrition and social work interventionists had 5, 6, and 13 years of oncology rehabilitation experience respectively. Further details, reported according to the TIDiER guidelines, are provided in the Supplemental File (Table S1).
Measures
Assessments were completed for both groups at baseline, 9 weeks and 3 months by 2 research assistants who received a 30-minute training session. Demographic and clinical participant characteristics were collected at baseline. Objective measures were completed in the hospital setting with accelerometers mailed to participants at follow-up assessments. Patient-reported questionnaires were completed in the hospital setting at baseline and electronically via an email survey link or in hard-copy at follow-up. Figure 1 provides an overview of trial assessments.

Trial objective and patient-reported outcomes.
Primary outcomes
Feasibility was assessed as a composite outcome. The primary feasibility outcomes were recruitment (rate) and reasons for ineligibility. Additional feasibility outcomes included the proportion of eligible patients who provided consent, reasons for declining participation, retention at trial follow-up appointments, the number of complete assessments and acceptability to participants. Adherence to the intervention (number of scheduled appointments attended), the delivery mode and the number of external referrals made for social support were also recorded for intervention participants. A priori rates of recruitment, retention and intervention adherence indicating acceptable feasibility were defined as 7 participants/month, <15% loss to follow-up at 9-weeks and ≥70% attendance, respectively. Acceptability to participants was assessed by inviting all participants to complete an individual semi-structured interview by telephone at 3 months. A purpose-designed interview schedule was developed by the research team to gain a deeper understanding of participants’ trial experiences, including completion and delivery format of trial questionnaires and their recommendations for future trials. Intervention participants were additionally asked about their experiences with the various intervention components, including the trial resources and telehealth options.
Safety was assessed by patient-report and medical records and was reported as the number of adverse events occurring related to the trial intervention or assessments. Serious adverse events were defined as those that resulted in death, required hospitalization or prolongation of existing hospitalization, were life threatening or resulted in disability or incapacity.
Secondary effectiveness outcomes
Objective outcomes
Nutrition and physical activity outcomes were assessed objectively. The Patient Generated-Subjective Global Assessment (PG-SGA) includes a subjective assessment of weight-loss, nutritional symptoms, food intake and activity levels and an objective assessment of body composition (fat, muscle stores, and fluid status, scored as “0” = no deficit, “1” = mild deficit, “2” = moderate and “3” = severe). Each component of the PG-SGA is scored between 0 and 4 to provide an overall score (typical scores range from 0 to 35) and category of nutritional status (well-nourished, moderately/suspected malnutrition, and severe malnutrition). 31 Bioelectrical impedance, measured by body composition analysis scales (Tanita Inc., Tokyo, Japan, model T1 SC 330S) was used to calculate fat-free mass index. PA levels were measured by seven-days of accelerometry (Sensewear™ armbands) following each assessment. 32
Patient-reported outcomes
Health-related quality of life (HRQoL) and fatigue were measured using the Functional Assessment of Cancer Therapy-General (FACT-G) and the Functional Assessment of Chronic Illness Therapy—Fatigue Subscale respectively. Dietary behaviors were assessed with the Rapid Eating Assessment for Patients (REAP) questionnaire. PA levels and self-efficacy were reported using the International Physical Activity Questionnaire-Short form (IPAQ-SF) 33 and the Physical Activity Assessment Inventory (PAAI) respectively. The Cancer Rehabilitation Evaluation System (CARES) questionnaire was used to measure the impact of cancer and associated treatments on day-to-day living and patient needs. Further details of the secondary effectiveness outcomes are provided in the Supplementary file.
Statistical Methods
This was a pilot study with a pragmatic sample size and was not powered to assess efficacy. Descriptive statistics (including counts and percentages for nominal and ordinal variables; and means and standard deviations or medians and interquartile ranges for continuous variables) were used to summarize patient demographic, clinical characteristics, secondary outcomes, adverse events, and operational data (eg, number of intervention sessions). Recruitment data were summarized using a rate (95%CI) (Poisson distribution). Compliance with assessments, as well as adherence and retention data were summarized using a proportion (95%CI); the latter estimated using the Wilson method. 34 All quantitative analyses were conducted in R (reference index version 3.5.1). 35
Semi-structured interviews were audio-recorded, transcribed verbatim and analyzed by 2 researchers who independently coded sub-sets of the data and performed an inter-rater process on 10% of the overall data to check consistency of coding and ensure consensus on the initial codes generated. Data were entered and managed using Microsoft Excel for Mac version 16.43. Interpretive description was used as a recommended inductive analysis method for exploring clinical phenomena through patient-based interviews. 36 Data were interpreted in a constant comparative manner to explore emerging concepts and patterns, 37 to generate categories describing participants’ study experiences.
Results
Primary Outcome
Feasibility
Recruitment occurred between May 2019 and March 2020. The flow of participants through the trial is provided in Figure 2. Two-hundred and twelve patients were screened and 44 (21%) were eligible. Key reasons for ineligibility included patients who were not newly diagnosed and were attending the outpatient clinic for follow-up (n = 126, 59%) or those who had stage IV or recurrent disease (n = 30, 14%). Twenty-two of the 44 eligible patients (50%, 95%CI: 36%, 64%) provided informed consent and were recruited to the trial over 10 months (14 intervention group, 8 usual care). The recruitment rate was 2.2 patients/month (95%CI: 1.4, 3.3). Most patients who declined had too much going on (7/22, 32%) or were not interested (6/22, 27%). Table 2 provides baseline participant demographic and clinical characteristics. Descriptive statistics for baseline measures are presented in Table 3. Mean participant age and BMI were 63.2 years (SD = 6.8 years) and 31.9 kg/m2 (SD = 6.7 kg/m2), respectively. The majority of participants were International Federation of Gynecology and Obstetrics (FIGO) stage I (15/22, 68%) and received vaginal brachytherapy alone (14/22, 64%). Retention was 100% (95%CI: 85%, 100%) at 9 weeks and 91% (20/22) at 3 months, however completion of objective nutrition follow-up measures was impacted by COVID-19 government restrictions on face-to-face research assessments at the time. Adherence to the intervention was high with 11/14 (79%, 95%CI: 52%, 92%) participants attending ≥70% of scheduled weekly sessions. Of 14 initial sessions, 7 were conducted via telehealth (2 videoconferencing and 5 telephone) and 7 at the hospital. Eleven intervention group participants (79%) agreed to use the trial pedometer or had their own device to track their physical activity levels throughout the 8-week intervention. Of follow-up sessions 13/88 (15%) were conducted face-to-face at the hospital and 75/88 (85%) by telehealth (61 (81%) telephone, 14 (19%) videoconferencing). Seven participants (50%) returned the trial diary which they had used to record weekly nutrition and physical activity behaviors and goals set. Participants reported being well supported with 3/14 (21%) requiring social work support following initial assessment.

Participant flow through the trial.
Participant Demographic and Clinical Characteristics.
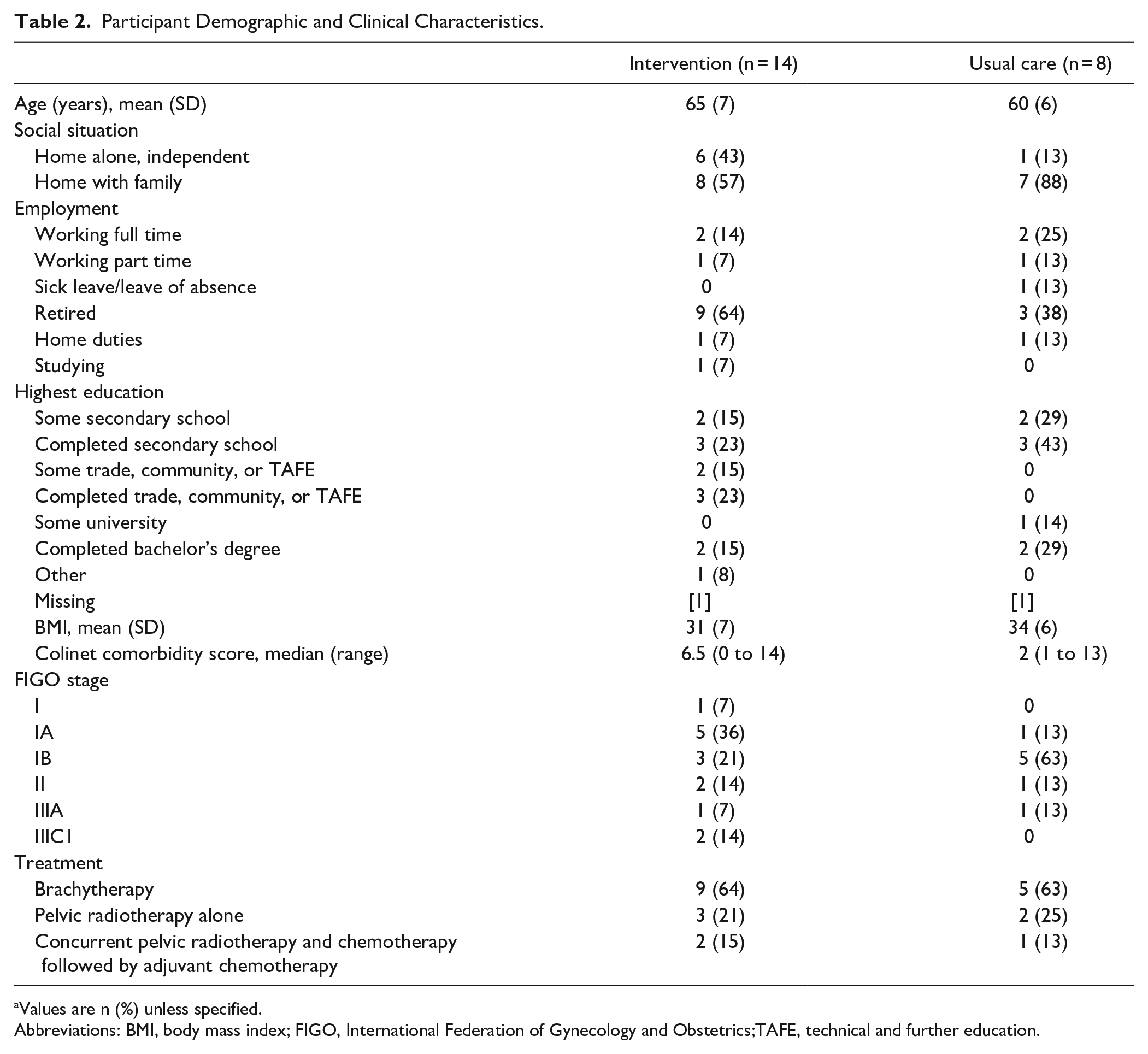
Values are n (%) unless specified.
Abbreviations: BMI, body mass index; FIGO, International Federation of Gynecology and Obstetrics;TAFE, technical and further education.
Baseline Patient-reported and Objective Measures.

Median (interquartile range).
Abbreviations: FFM, fat-free mass; FFMI, fat-free mass index; MET, metabolic equivalent of task; MVPA, moderate-vigorous physical activity; PA, physical activity; PAAI, physical activity assessment inventory; PG-SGA, patient-generated subjective global assessment; REAP, rapid eating assessment profile.
Safety
There were no serious adverse events. One minor adverse event related to trial assessment occurred (skin irritation following accelerometer wear). One intervention participant reported an increase in chronic low back pain as a result of increasing PA intensity. This participant was advised by the trial physiotherapist to continue the PA program but at a lower intensity. Both events resolved without requiring additional management.
Acceptability to participants
Telephone interviews were conducted with 18 participants (12 intervention, 6 usual care) and lasted between 6 and 32 minutes. Six categories were generated describing aspects of participants’ trial experiences (Table 4). From these, 4 themes emerged, which are presented here, and which capture the issues participants consistently and centrally reflected on.
Participant Quotes.

Theme 1
Accessibility enables participation and enhances the study experience.
The opportunity for remote participation was appreciated, which reduced travel burden and made participation more accessible and stress-free. The telehealth and phone consultation options were described as streamlined and easy to navigate. Most participants preferred the opportunity to complete trial questionnaires online, in their own home and at their own pace and time. Participants appreciated the opportunity to coordinate trial appointments with other clinical appointments.
“having the questionnaire when I could do it, when I wanted to do it, you know, I wasn’t told you must do it at x time, you know, so yeah, that to me was . . . I guess it’s the flexibility of the study which was really easy.” (P07) “Well telehealth is telehealth isn’t it and for someone who lives in the country the way I do, three-hour drive from Melbourne, we really want this to work.” (P22)
Theme 2
Participation offers opportunities for receiving and contributing value.
Participants derived value from their participation and reported appreciating the opportunity to contribute value to others. The trial resources such as the diary and activity trackers provided motivation and accountability to stay on track with goals. Participants felt supported and encouraged to solidify existing goals or set new goals. Contact with the trial team provided ongoing support, a sense of caring and being listened to, motivation and encouragement, direction and assistance/advice for questions around diet and exercise and an added sense of accountability and feeling included in something. Many participants reported their motivations for participating in the trial as altruistic and desired to contribute to research and the development of new and improved information and resource.
“And I found the support from [physiotherapist] and from [dietitian] to be invaluable. They were approachable, they obviously had great knowledge in their areas, they always answered the questions I had.” (P22) “I found it was beneficial for me, so therefore I feel it should help many women. I felt encouraged, supported, and above all I participated because I do support this sort of thing and I just hope that it helps other women . . ..” (P04)
Theme 3
Encountering study-related and personal health challenges.
While telehealth participation was reported as the preferred option by most, some felt the technology required involved a steep learning curve. Participants had mixed experiences using the 2 physical activity monitors provided for the study (accelerometers for 1-week assessments and pedometers for motivation throughout intervention). A proportion felt uncertain about the function of the accelerometer and how to use it, and some struggled with unclear instructions for the pedometer and described that it recorded steps inconsistently. The COVID-19 pandemic was reported to impact on trial participation. Usual physical activity routines were disrupted, and pandemic related anxiety and stressors impacted their overall state and wellbeing. A proportion of participants reported having to additionally navigate fluctuating mental health and side effects of their cancer treatments, such as treatment-related fatigue, feeling ill after treatment, rise and fall of energy levels due to steroid medication, mobility and balance issues and variable physical strength.
“that [study assessment] was a little at times inconvenient because of with workload and just meetings and things that I had for work so, yeah. So I found it a little bit hard at times but most of the time it was OK.” (P06)
“when it says what’s your stress levels and that, it’s, and it’s also you’ve got pandemic of COVID.. so it’s an unusual time and, you know,” (P21)
Theme 4
Recommendations.
Participants reported several ways in which the trial could be improved in the future:
Nutrition and exercise: providing recipes for healthy meals and snacks, incorporating mindfulness and relaxation exercises, structured group exercise opportunities with preference for community-based settings close to home;
Participation support: additional social components to increase motivation and accountability;
Trial tools and information capture: more accurate pedometers and tools for capturing information and advice received;
Trial recruitment, information and communication: attention around appropriate timing of recruitment; providing early and comprehensive trial information and additional research staff contacts.
“some kind of guide or even a few little recipes, sort of simple ones . . . just simple healthy alternatives” (P11) “group get-togethers or something like that that people . . . I don’t know whether they want to talk about their circumstances” (P19)
Secondary Effectiveness Outcomes
Secondary effectiveness outcomes are reported in the Supplemental File (Tables S2 and S3). The trial was not powered to detect between-group differences for these outcomes. Objective follow-up measures of nutrition were available for 8 participants (5 intervention and 3 usual care) at 3-months as a result of government restrictions in place due to the COVID-19 pandemic.
Discussion
This pilot RCT of nutrition and PA education with behavior change and social support demonstrated feasibility through participant retention to the trial and intervention adherence. However, a future definitive RCT would require additional sites for recruitment rates to meet our pre-determined levels of feasibility. The intervention was safe, with no serious adverse events occurring and participants perceived the trial to be acceptable.
Previous multi-disciplinary lifestyle interventions in endometrial cancer have targeted weight loss and increasing PA and have been conducted in the post-treatment phase. Such interventions have been undertaken in both hospital,14,17,20,23 home,18,19,22 or mixed21,24,25,38 settings and demonstrate inconsistent results with respect to changes in PA (often not measured objectively), body weight, dietary intake and HRQoL.14,17,19-21 Secondary effectiveness outcomes from this trial are presented for completeness only given the trial was not powered to detect differences between groups. Additionally, for both groups the COVID-19 pandemic and restrictions impacted follow-up measure completion, patterns of PA and HRQoL reported by participants. In keeping with findings from this trial, prior studies report an absence of intervention-related adverse events, however safety is commonly not reported.
The trial uptake by eligible patients of 50% was higher than reported in other lifestyle behavior interventions in this population which report rates of between 10% and 25%.14,17,19-21,25,38 Two single-group trials conducted in the home environment do not report uptake rates.18,22 There are several possible reasons for the improved uptake to this trial. Firstly, interventions initiated closer to the point of diagnosis, representing a potentially teachable moment, 39 may target a timepoint on the cancer treatment continuum where patients are more receptive to making changes to lifestyle behaviors. Interventions to change behaviors closer to diagnosis have been demonstrated to have significant impact; greater use of active coping strategies at diagnosis is associated with reduced all-cause mortality (HR 0.78, P = .04) at 4 to 5 years post endometrial cancer diagnosis. 40 It remains uncertain whether it is best to offer these kinds of interventions during active treatment, or immediately post treatment for treatments given for a limited period of time. Secondly, key medical and nursing staff were actively involved in recruitment. Thirdly, as reported by participants, the use of telehealth and online questionnaires reduced potential barriers to involvement faced by those living a distance from the hospital.
Trial retention was high in contrast to a 6-month single-group trial, which excluded participants receiving active chemotherapy or radiotherapy, in a mixed-cancer sample (n = 99, 58 with endometrial cancer) involving a home-based exercise program for rural participants with a completion rate of only 36%. 22 Similar to our trial where levels of baseline physical activity were high, 68% of participants reported exercising regularly in the past, suggesting selection bias in those who agreed to participate in the trial. Program completion was significantly associated with higher baseline HRQoL and PA levels and findings also suggest that program duration, 6-months compared to the 8-weeks in our trial, may be an important factor impacting on adherence. 22
The trial findings both extend and align with knowledge in the area regarding endometrial cancer participant experiences of nutrition and PA interventions. The majority of previous work reports participant preferences. Survivors express an interest in exercise and feel able to participate, with walking of moderate-intensity commonly preferred.41,42 During or 3 to 6 months after treatment are preferred time points for starting a PA program reported by 49% (49/101) of gynecological cancer survivors within 2 years of diagnosis. 42 Future research should be conducted to compare the effects of interventions delivered at differing timepoints on the treatment continuum. Most gynecologic cancer survivors report being receptive to weight management counseling.19,43 Building on what is known with respect to preferences, participants of this trial reported enablers such as the use of telehealth and online measures, and make key recommendations for modifications in subsequent trials, including group sessions to increase social support.
Several study limitations are worth noting. The generalizability of trial findings is limited given recruitment occurred at a single center. Recruitment bias is likely given few participants required social support and baseline PA levels were relatively high. Participants were excluded from the trial if they had met PA guidelines for aerobic exercise over the preceding 4 weeks at recruitment. Given this time period included post-operative recovery participants were likely to be less active than normal. Future trials should consider excluding participants who report meeting PA aerobic guidelines pre-diagnosis, rather than at recruitment. Stricter eligibility criteria will target the intervention to a less active population, with the greatest potential to benefit from the intervention. Definitive randomized controlled trials are needed to determine the clinical and cost-effectiveness of models of care which are feasible to implement and scale with existing resources, including group sessions with remote monitoring and continued use of telehealth to reduce barriers to accessing services.
Conclusion
This pilot trial demonstrated intervention safety and participant acceptability. Levels of feasibility were above pre-specified thresholds with respect to adherence to the intervention and retention at follow-up outcome assessments. Participant experiences and key recommendations will inform modifications to future intervention design for an appropriately powered RCT with multiple recruitment sites.
Supplemental Material
sj-docx-1-ict-10.1177_15347354211069885 – Supplemental material for ENhAncing Lifestyle Behaviors in EndometriaL CancEr (ENABLE): A Pilot Randomized Controlled Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354211069885 for ENhAncing Lifestyle Behaviors in EndometriaL CancEr (ENABLE): A Pilot Randomized Controlled Trial by Lara Edbrooke, Pearly Khaw, Alison Freimund, Danielle Carpenter, Orla McNally, Lynette Joubert, Jenelle Loeliger, Anya Traill, Karla Gough, Linda Mileshkin and Linda Denehy in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors would like to thank all the trial participants, steering committee members Ms Lesley McGuire and Ms Amanda Appathurai and Dr Sarah-May Blaschke for their contributions.
Author Contributions
LE, PK, AF, DC, OMcN, LJ, JL, AT, LM, and LD secured grant funding.
LE: Conceptualization, Recruitment, Intervention, Data curation, Writing—original draft. PK: Conceptualization, Recruitment, Writing—re- view & editing. AF: Conceptualization, Writing—re- view & editing. DC: Conceptualization, Recruitment, Writing—re- view & editing. OMcN: Conceptualization, Writing—re- view & editing. LJ: Conceptualization, Writing—re- view & editing. JL: Conceptualization, Writing—re- view & editing. AT: Writing—re- view & editing. KG: Conceptualization, Formal analysis, Writing—re- view & editing. LM: Conceptualization, Recruitment, Writing—re- view & editing. LD: Conceptualization, Supervision, Writing —re- view & editing.
Availability of Data and Material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Peter MacCallum Cancer Foundation (grant 1812).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Written consent to publish was obtained from all individual participants included in the study.
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Peter MacCallum Cancer Centre Human Research Ethics Committee on 14th March 2019 (HREC/48033/PMCC-2019).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
