Abstract
Pancreatic cancer is one of the most lethal malignancies with very limited treatment option. In the effort of enhancing the effect of the conventional chemotherapeutic drug gemcitabine against pancreatic cancer, we investigated in vitro and in vivo the anticancer effect of a β-carboline-enriched extract from the plant Rauwolfia vomitoria (Rau), either alone or in combination with gemcitabine, in preclinical pancreatic cancer models. Rau induced apoptosis in pancreatic cancer cells in a concentration-dependent manner, and completely inhibited colony formation of PANC-1 cells in soft agar. The combination of Rau and gemcitabine had synergistic effect in inhibiting cell growth with dose reduction effect for gemcitabine. In an orthotopic pancreatic cancer mouse model, PANC-1 tumor growth was significantly suppressed by Rau treatment. Metastasis was inhibited by Rau. Adding Rau to gemcitabine treatment reduced tumor burden and metastatic potential in the gemcitabine non-responsive tumor. These data suggest that Rau possesses anti–pancreatic cancer activity and could improve effect of gemcitabine.
Abbreviations
CI, Combination Index; DRI, Dose-reduction index; Gem, Gemcitabine; H&E, Haematoxylin-eosin; i.p., intraperitoneal; MTT, 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide; PARP, poly-ADP ribose polymerase; PI, Propedium Iodine; Rau, Rauwolfia vomitoria extract.
Introduction
With estimates of 43 920 new cases and 37 390 deaths in the United States in 2012 (>100 deaths per day), pancreatic cancer has the highest fatality rate among cancers.1-3 This is likely because of disappointing treatment efficacy in addition to late detection and fulminant disease course.4,5 The 1- and 5-year overall survival rates are 26% and 6%, respectively, which remain unchanged for the recent 30 years. 2 More than half of pancreatic cancer patients are diagnosed at a distant stage, for whom 5-year survival is only 2%. 1 Gemcitabine as a single agent remains to be the first-line therapy of metastatic pancreatic cancer. Gemcitabine provides benefit at early stages of the disease; however, it has little impact on median overall survival for patients with locally advanced or metastatic disease, who comprise the majority of cases.6-8 Recent clinical trials adding agents to gemcitabine had statistical significance, but are not really meaningful for patients.9-15 A new gemcitabine-free regimen FOLFIRINOX combining 5-fluoruoracel, leucovorin, irinotecan, and oxaliplatin provided a 5-month survival benefit over gemcitabine. 16 However, this regimen added adverse effects. Lack of effective therapeutic options, lack of adjuvant therapy, significant side effects with existing chemotherapies, and radiation therapies or their combinations remain major problems in the treatment of pancreatic cancer.
Natural products have long been proven a bountiful resource for bioactive anticancer agents. One of the advantages of natural products is their low toxicity compared with conventional chemo-drugs. Combination of natural compounds to standard chemotherapeutic drugs may exert additive or synergistic effects in killing cancer cells, which would in turn allow lower and safer doses to be used. Herbal preparation of Rauwolfia vomitoria, a tropical shrub in the family Apocynaceae, is a traditional folk medicine in Africa used to treat a variety of ailments, including hypertension,17-20 fever,21,22 general weakness,23(pp75-76),24(pp174-176) gastrointestinal diseases,25,26 liver diseases, 27 psychosis,28-30 pain, 29 and cancers. 31 This herbal extract is rich in bioactive compounds. Reserpine, a drug for the control of high blood pressure and for the relief of psychotic symptoms, was isolated from R vomitoria.32-34 Extracts from the root bark of this plant, from which reserpine has been removed, are enriched with β-carboline alkaloids.35,36 β-Carboline alkaloids have been previously reported to demonstrate many bioactivities, including antitumor effects.31,37-39 Here, we evaluated the effect of an extract from the root of R vomitoria in the treatment of pancreatic cancer, either used alone or in combination with gemcitabine.
Materials and Methods
Experimental Materials, Cell Lines, and Viability Assay
Rauwolfia vomitoria extract (Rau) was provided by Natural Source International (New York, NY). Rau and gemcitabine (Sigma, St Louis, MO) were prepared in sterile water and stored at −20°C.
Human pancreatic cancer cell lines PANC-1, MIA PaCa-2, AsPC-1, HPAF-II, and BxPC-3 were obtained from the American Type Culture Collection (Manassas, VA). Immortalized human lung epithelial cells MRC-5 were provided by Dr Sittampalam at the University of Kansas Medical Center, and were used as a comparison to cancer cells. All the cells were cultured at 37°C in 5% CO2/95% air in recommended growth media containing 10% fetal calf serum.
Cells were assessed for viability by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay at 72 hours of treatment. Cells in exponential growth phase were exposed to serial dilutions of Rau, gemcitabine, or the combination of the 2, for 72 hours. Then cells were changed into fresh media containing MTT and were incubated for 4 hours. The colorimetric MTT assay assessed relative proliferation, based on the ability of living, but not dead cells to reduce MTT to formazan.40,41 Cells did not reach plateau phase during the incubation period. Fifty percent inhibitory concentration (IC50) was defined as the concentration of drug that inhibited cell growth by 50% relative to the untreated control. Pilot experiments for each cell line were performed to optimize cell density and assay duration and to center drug dilution series approximately on the IC50.
Anchorage-Independent Colony Formation Assay
Anchorage-independent colony formation assay in soft agar was utilized to determine long-term survival of tumorigenic cancer cells after treatment. In 6-well plates, PANC-1 cells (5000 cells per well) were seeded in the upper layer containing 0.5% agar, Dulbecco’s modified Eagle medium, and 10% fetal bovine serum, with or without 400 µg/mL Rau. The solid agar base (lower layer) contained 0.75% agar and the complete growth medium, with or without 400 μg/ml Rau. Cells were incubated for 20 days. Colonies were visualized by crystal violet staining and counted.
Apoptosis Detection by Flow Cytometry
Cells were exposed to various concentrations of Rau for 48 hours. Cells were washed in phosphate-buffered saline, resuspended in binding buffer, and subjected to fluorescein isothiocyanate conjugate (FITC)-conjugated annexin V and propidium iodide (PI) staining according to the manufacturer’s protocol (BD Biosciences, San Jose, CA). Cells were analyzed by flow cytometry. Annexin V positive and annexin V–PI double positive cells were identified as apoptotic cells, whereas PI single positive cells were identified as necrotic cells.
Western Blot
Forty micrograms of protein were loaded for sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Primary and secondary antibodies were from Cell Signaling Technology Inc (Danvers, MA): rabbit anti-poly-(ADP-ribose)-polymerase (PARP) (1:2000), rabbit anti-caspase-3 (1:1000), rabbit anti-capase-8 (1:1000), mouse anti-β-actin (1:1000), and goat anti-rabbit or anti-mouse IgG (1:5000). Blots were developed using immobilon chemiluminescent substrate (Thermo Scientific, Waltham, MA).
Intraperitoneal Pancreatic Cancer Mouse Model
Through an intrapancreas surgical procedure, PANC-1 cells were orthotopically implanted into the pancreas of nude mice at 3 × 105/mouse. To allow in vivo imaging, PANC-1 cells were transfected with luciferase gene prior to inoculation. Luciferin was given intraperitoneally each time prior to imaging to luminance the tumor cells. Ten days after tumor cells were implanted, mice were imaged and grouped to 8 mice per group based on tumor burden. Then treatment began with intraperitoneal injection of gemcitabine (Gem; 20 mg/kg, every 4 days), Rau (20 or 50 mg/kg daily), the respective combination of Gem and Rau, and saline as control. After 48 days of treatment, mice were euthanized. All tumor lesions in the peritoneal cavity were collected and weighted. Metastasis lesions were assessed and counted. Major organs such as liver, kidney, and spleen were fixed in 4% formaldehyde and subjected to histological analysis for any damage due to potential drug toxicity.
Data Analysis
MTT data were normalized to their corresponding untreated controls for each condition (drug, cell type) and were expressed as percentage viability. Dose reduction index (DRI) values for gemcitabine were calculated by the equation DRIICx = (DGem/DGem+Rau), where DGem is the dose of gemcitabine alone required to produce an ICx level of cytotoxicity, and the divisor DGem+Rau is the dose of gemcitabine needed to produce the same ICx level of cytotoxicity when it is combined with Rau (at a given molar ratio). DRIGem is defined with respect to gemcitabine. SPSS15.0 was used for additional statistical analysis.
Results
Effect of Rauwolfia vomitoria Extract (Rau) Against Pancreatic Cancer Cells
Human pancreatic cancer cell lines PANC-1, MIA PaCa-2, AsPC-1, HPAF-II, and BxPC-3 were compared to an immortalized non-cancerous epithelial cell line MRC-5 for sensitivity to Rau. The dose–response curves showed that cancer cells were more sensitive to Rau treatment than the normal cells MRC-5 (Figure 1A). The cancer cells had IC50 values ranging from 83 to 283 µg/mL. The noncancerous cell MRC-5 had IC50 of 553 µg/mL, more than 2-fold higher than those of the cancer cells.

Cytotoxicity of Rauwolfia vomitoria (Rau) in normal cells and pancreatic cancer cells. (A) Dose–response curves of normal and pancreatic cancer cells to Rau. Pancreatic cancer cells PANC-1, MIA PaCa-2, AsPC-1, HPAF-II, and BxPC-3 were exposed to serial concentrations of Rau for 48 hrs, and cell viabilities were detected by MTT assay. An immortalized noncancerous epithelial cell MCR-5 was subjected to the same treatment. IC50 was defined as the concentration of drug that inhibited cell growth by 50% relative to the untreated control. (B) Colony formation of PANC-1 cells in soft agar with and without Rau treatment. Five thousand PANC-1 cells per well in 6-well plate were either treated with 400 μg/mL Rau (Rau) or untreated (Control). All values are expressed as mean ± standard deviation of 3 independent experiments.
Colony formation in soft agar was used to assess the survival of tumorigenic cancer cells in a longer time, which has been positively correlated to in vivo tumorigenicity of the cancer cells in animal models.42,43 The PANC-1 cancer cells formed colonies at a rate of 12% when untreated. Rau at 400 µg/mL completely inhibited formation of colonies of PANC-1 cells in soft agar (Figure 1B), indicating no survival of tumorigenic cancer cells with this treatment.
To assess Rau-induced death pathway, annexin V/PI straining was performed to detect apoptosis versus necrosis in PANC-1 cells treated with Rau. Data from flow cytometry demonstrated a predominant apoptotic cell death was induced by Rau treatment. With 0, 50, 100, 200, and 400 µg/mL Rau treatment, the percentage of apoptotic cells were 9.5%, 25.9%, 73%, 80.5%, and 94%, respectively (Figure 2A). The induction of apoptosis was clearly dependent on the concentration of Rau and was the major form of cell death induced by Rau. Necrosis (cells that were PI positive only) contributed only 4% to 19% of total cell death across all the different Rau concentrations. Consistent with this finding, Western blot analysis detected extensive cleavage of caspase-8, caspase-3, and PARP in Rau-treated PANC-1 cells in a dose- and time-dependent manner (Figure 2B).

Apoptosis in PANC-1 cells induced by Rauwolfia vomitoria (Rau). (A) Flow cytometry detection of apoptotic cells. PANC-1 cells were treated with Rau at indicated concentrations. At 48 hours of treatment cells were subjected to fluorescein isothiocyanate conjugate (FITC)-affiliated annexin V and propidium iodine (PI) double staining and flow cytometry. Cells in Q2 and Q4 were identified as apoptotic cells, and cells in Q1 (PI positive only) were identified as necrotic cells. Percentage of apoptotic cells in each treatment was quantified and shown in the bar graph. (B) Cleavage of caspase-8, caspase-3, and poly-ADP ribose polymerase (PARP) in PANC-1 cells treated with Rau. Cells were treated with Rau at different concentrations (left panel) and for different time (right panel) as indicated. The cleavage of caspase-8, caspase-3, and PARP were detected by Western blots.
Synergistic Effect of Rau in Combination With Gemcitabine Against Pancreatic Cancer Cells
After determining the dose–response relationships for Rau (Figure 1A), the dose–response relationships for Gem cytotoxicity were established in PANC-1, MIA PaCa-2, AsPC-1, HPAF-II, and BxPC-3 cells (Figure 3A, dotted lines). These cells demonstrated a spectrum of resistance to Gem. Chou–Talalay’s constant ratio design 44 was used to systematically examine combination dose–response relationships between Gem and Rau. The molar ratio of Gem:Rau was chosen as IC50Gem:IC50Rau. Combination data were presented in terms of Gem concentration. Despite the different inherent sensitivity of these cell lines to Gem, the results unambiguously showed an enhanced cytotoxicity with the curves in Gem + Rau combinations for all cell lines compared with the corresponding curves with Gem alone (Figure 3A).

Combination effect of Rauwolfia vomitoria (Rau) and gemcitabine against pancreatic cancer cells. (A) Dose–response curves of pancreatic cancer cells to gemcitabine (Gem), or gemcitabine and Rau combination (Gem + Rau). Cells were treated with gemcitabine (Gem, dotted line) and the combination of gemcitabine and Rau (Gem + Rau, solid line) for 72 hours to obtain optimum effect with gemcitabine. The combination took the molar ratio of IC50Rau: IC50Gem, and was plotted against gemcitabine concentration. (B) Dose reduction index (DRI, right panel) across the fraction affected (fa) for gemcitabine when Rau was combined.
To quantitatively assess whether gemcitabine effect was enhanced by pairing with Rau, the DRI for Gem was calculated. The DRI indicates decrease in Gem concentration when combined with Rau than that of gemcitabine alone to achieve the same cytotoxic effect. As shown in Figure 3B, DRI was >1 for all cell lines, and across the aimed cytotoxic effect of 60% to 90% (fraction affected, or fa). Depending on cell lines and the aimed level of effect, DRI can reach 100 to 10 000 (Figure 3B). These data unequivocally support the conclusion that the concentration of Gem can be decreased to produce an equitoxic effect on pancreatic cancer cells when Rau is combined.
In Vivo Tumor Inhibitory Effect of Rau Either Alone or in Combination With Gemcitabine
An orthotopic pancreatic cancer mouse model was used to evaluate the effect of Rau and gemcitabine plus Rau (Gem + Rau) treatment. Compared with the subcutaneous model, this model better mimics clinical condition of human pancreatic cancer, because the local environment for pancreatic cancer development was represented.
Ten days after PANC-1 cells were inoculated; treatment was carried out as described in the Materials and Methods section. Longitudinal tumor progress was monitored through live animal images. Representative images are shown in Figure 4A. As the tumors did not respond to gemcitabine, Rau at either 20 mg/kg or 50 mg/kg provided observable inhibition in tumor growth. The combination of Gem + Rau at both doses also inhibited tumor progression. When tumor burden was quantified by photon flux, the results were consistent with the observation (Figure 4B). Rau treatment groups at both doses had less tumor burden than the control group and gemcitabine-treated group at the end of treatment. The combination groups showed less tumor burden than the control and gemcitabine groups; however, these were not different from the Rau treatment alone groups. Notably, there were 2 mice in the Gem + Rau50 group that had complete tumor regression, an effect that had not been seen in any other treatment groups.

Live animal imaging. PANC-1 cells were orthotopically implanted into the pancreas of nude mice (3.0 × 105/mouse). After 10 day of tumor inoculation, treatment began (day 0) with indicated doses in the Materials and Methods section. To allow in vivo imaging, PANC-1 cells were transfected with luciferase gene and verified for stable expression of luciferase before inoculation. At imaging, 150 mg/kg luciferin was given intraperitoneally to luminance tumor cells. (A) Representative images from each group at day 0, day 24, and day 45. (B) Longitudinal tumor growth shown by quantification of all images in each treatment group. Tumor burden were represented by average total photon flux in each mouse; n = 8 for each group. *P < .05 versus control group, #P < .05 versus gemcitabine-treated group.
Necropsy confirmed the imaging results at the end of the treatment (Figure 5). Gemcitabine at the used dose did not provide any inhibition in tumor weight. Rau alone decreased tumor weight by 53% at the daily dose of 20 mg/kg, and 46% at the daily dose of 50 mg/kg, compared with saline-treated control. By combining Rau with gemcitabine, the decreases in tumor weight were 56% at both Rau doses. The improvement in tumor inhibition provided by the combination was significant compared with gemcitabine alone; however, it was not significant compared with Rau treatment alone (Figure 5A).
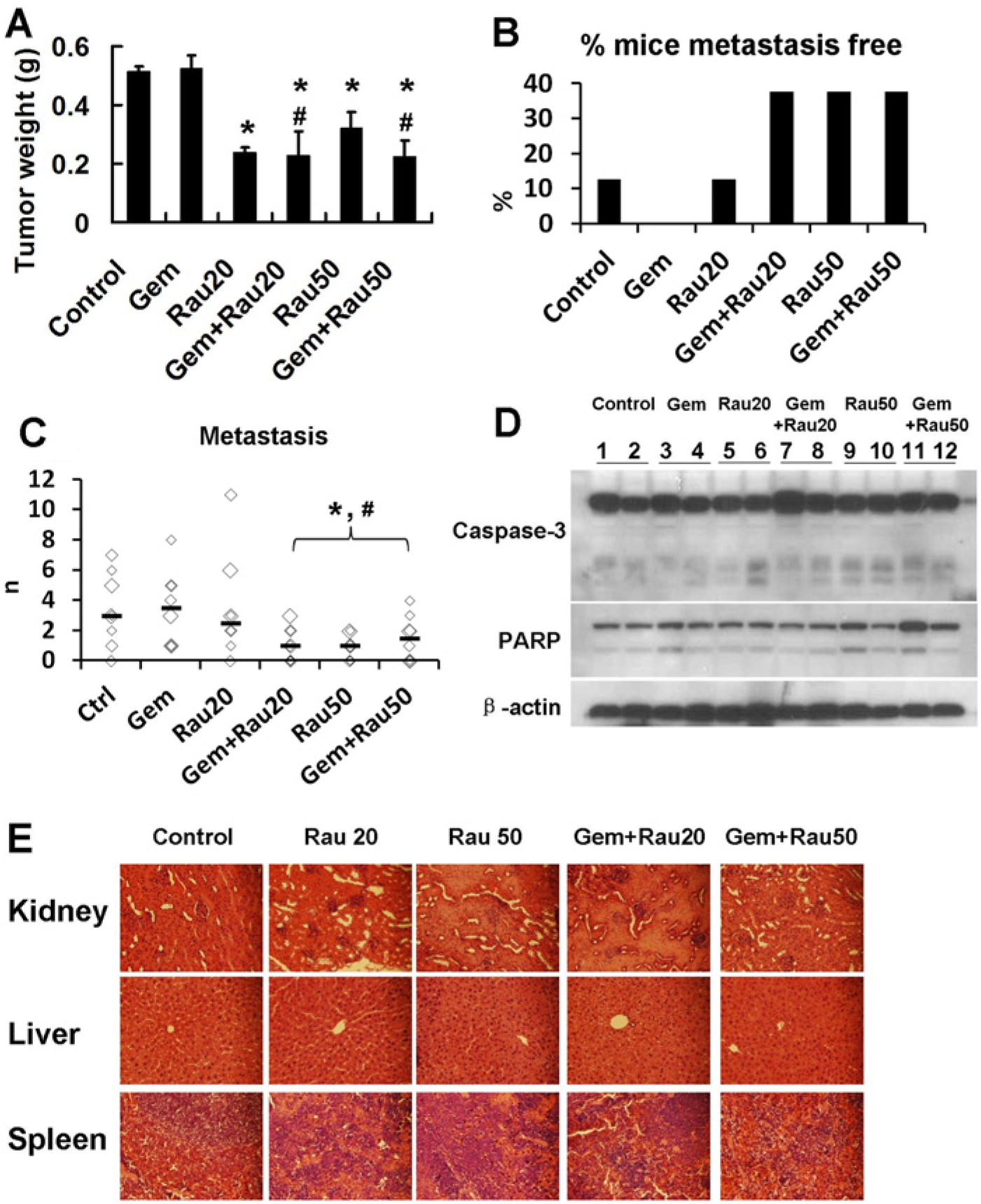
Inhibition of tumor weight and metastasis by Rauwolfia vomitoria (Rau) and the combination of Rau and gemcitabine in vivo. Mice bearing orthotopic PANC-1 pancreatic tumors were treated for 48 days. Tumor weight and metastasis were assessed at the end of treatment. (A) Tumor weight shown as mean ± standard error of the mean (mean ± SEM). (B) Percentages of mice that did not form metastasis. (C) Number of metastatic lesions in each mouse that had metastasis. The black bars indicate the median number of metastasis in each group. (D) Cleavage of capase-3 and poly-ADP ribose polymerase (PARP) in tumor samples from different treatment groups. (E) Hematoxylin and eosin staining of major organs from different treatment group (400× magnitudes). Kidney, liver, and spleen were collected from each treatment group and fixed in 4% formaldehyde, and later subjected to histological analysis. *P < .05 relative to control group; #P < .05 relative to gemcitabine (Gem)-treated group.
On assessing tumor metastasis, 12% mice in the control group (saline-treated) and in the Rau 20 mg/kg group did not form metastasis, whereas all mice in gemcitabine group formed metastasis. The percentage of metastasis free mice increased to 40% with both Rau 50 mg/kg treatment and Gem + Rau, suggesting that Rau provided benefit in reducing metastatic potential while gemcitabine did not (Figure 5B). Number of metastasis lesions was also decreased with Rau 50 mg/kg treatment, or the combination treatments of gemcitabine and Rau (Figure 5C). As gemcitabine did not provide any benefit in inhibiting metastasis, Rau or Gem + Rau combinations significantly improved the effect versus gemcitabine alone.
Proteins were extracted from tumor samples of the treated and control mice. Western blot analysis showed cleavage of caspase-3 and PARP in Rau and Gem + Rau treatment groups at all the doses used (Figure 5D). These results confirmed the in vitro data that Rau induced apoptosis in tumor cells.
None of the mice demonstrated observable toxicity associated with the treatments. At the end of the experiments, major organs (kidney, liver, and spleen) were subjected to hematoxylin and eosin staining and pathological analysis. No tissue damage was detected in the treatment groups, and there were no significant differences between the control group and treated groups (Figure 5E). These data demonstrated that Rau at the doses used, either alone or combined with gemcitabine, was of low toxicity.
Discussion
Gemcitabine as the first line of pancreatic cancer care failed to provide impact in the median survival for patients with locally advanced or metastatic pancreatic cancer.14,45,46 Our study had consistent observation that gemcitabine at a commonly used dose in mice did not produce inhibition in tumor burden or metastasis in our animal model. Quite in contrast, the extract of R vomitoria (Rau) exhibited significant inhibition in PANC-1 tumors, and ended with >50% tumor inhibition and reduced chance of metastasis and numbers of metastasis lesion formed. Consistent with the in vitro dose reduction effect for gemcitabine, the combination of Rau and gemcitabine had better effect than gemcitabine in vivo. However, the combination did not make a real difference than Rau-alone treatment in vivo, although Gem + Rau20 showed a tendency of less metastasis than Rau20 alone. This is likely because the tumors did not respond to gemcitabine treatment in the in vivo experiment. The gemcitabine nonresponsive tumor still responded to Rau treatment. These results greatly raise the potential for using R vomitoria in pancreatic cancer treatment, alone or in addition to gemcitabine.
Moreover, our data showed that Rau had relatively low toxicity toward normal cells. The low toxicity was evident in mice treated with Rau where major organ toxicities were absent. Although pathological changes were also absent in gemcitabine treated mice at our used dose, gemcitabine did not show any efficacy. By the dose reduction effect Rau exhibited, Rau could allow lower doses of gemcitabine while achieving an equivalent efficacy than seen with higher gemcitabine doses alone. This may allow decrease of toxicity associated with chemotherapy.
While potential benefit was suggested by our study, mechanism(s) of Rau-induced anticancer effect warrant further study. It has been reported that Rau induced DNA damage and cell cycle inhibition of prostate cancer cells. 31 Activation of these pathways could lead to apoptosis, which is a powerful tumor-suppressive pathway preventing the uncontrolled proliferation of cancerous cells and potentially depleting stem-like and progenitor cancer cell pools.47,48 Our study showed that Rau mainly induced apoptosis both in vitro and in vivo as shown by flow cytometry and the cleavage of caspase-8, caspase-3, and PARP. Caspase-8 is generally regarded as the initiator caspase for the extrinsic apoptotic pathway. Once it was activated by Rau treatment, the downstream cascade of execution caspases were activated, such as capases-3. Caspase-3 in turn cleaved cytoskeletal and nuclear proteins such as PARP. PARP plays a critical role in the maintenance of DNA integrity and its expression has been associated with overall prognosis in cancer. 49 Our data suggest that by activating this apoptotic pathway, Rau completely inhibited the in vitro tumorigenic capacity of pancreatic cancer cells in soft agar. However, as this plant preparation contains a complex mixture of natural compounds, there is potential to affect multiple molecular targets and pathways that lead to cell death.
The β-carboline-enriched R vomitoria extract could contain compounds that possess potent anticancer activity. The data presented herein are part of the initial step in identifying of the anticancer activity of Rau. Active components could be isolated and developed for optimizing efficacy, toxicity, and other profiles that could lead to anticancer drug development.
Conclusion
Taken together, our data demonstrate anti–pancreatic cancer activity of R vomitoria extract in vitro and in vivo. Continued investigation is needed for the use of this plant extract in the treatment of pancreatic cancer.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Beljanski Foundation.
