Abstract
Objective
Gemcitabine plus cisplatin is the standard adjuvant regimen for postoperative management of muscle-invasive upper tract urothelial carcinoma. This study investigated whether adding tislelizumab to gemcitabine plus cisplatin improves outcomes in patients with muscle-invasive upper tract urothelial carcinoma.
Methods
We conducted a retrospective, single-center cohort analysis of consecutive patients who underwent laparoscopic radical nephroureterectomy (April 2020 to April 2025) and received adjuvant gemcitabine plus cisplatin or tislelizumab combined with gemcitabine plus cisplatin within 90 days postsurgery (≥3 cycles). No patients were lost to follow-up. The observed outcomes were disease-free survival, distant metastasis-free survival, and adverse events.
Results
Among 67 patients (37, tislelizumab combined with gemcitabine plus cisplatin group; 30, gemcitabine plus cisplatin group), the median follow-up duration was 15.6 months. Median disease-free survival was not reached in the tislelizumab combined with gemcitabine plus cisplatin group compared with 27.1 months in the gemcitabine plus cisplatin group. Tislelizumab combined with gemcitabine plus cisplatin significantly improved disease-free survival (hazard ratio = 0.441, p = 0.035) and distant metastasis-free survival (hazard ratio = 0.389, p = 0.036). Adverse events were comparable between groups, with no severe immune-related toxicities or treatment-related deaths.
Conclusions
Adjuvant tislelizumab combined with gemcitabine plus cisplatin demonstrated superior disease-free survival and distant metastasis-free survival compared with gemcitabine plus cisplatin alone, with acceptable safety, supporting further investigation in patients with muscle-invasive upper tract urothelial carcinoma.
Keywords
Background
Upper tract urothelial carcinoma (UTUC) is a relatively rare malignancy, accounting for only 5%–10% of all urothelial carcinomas (UC). With the development of diagnostic technologies, this proportion has been increasing. Despite its relative rarity, UTUC is associated with aggressive biological behavior. The thin muscular layer of the renal pelvis and ureter facilitates early tumor infiltration, resulting in a high proportion of patients presenting with muscle-invasive disease at diagnosis.1,2 Even after radical nephroureterectomy (RNU), more than half of patients with muscle-invasive or lymph node–positive disease experience disease recurrence or distant metastasis, leading to unfavorable long-term outcomes.3,4 Population-based studies have reported that patients with pathologic tumor stage (pT) 2 (pT2)–pT3 have a 5-year survival rate of less than 50%, whereas those with pT4 disease face even poorer outcomes, with survival rates falling below 10%. 5 Given these challenges, adjuvant therapy following radical surgery plays a critical role in improving prognosis. Gemcitabine plus cisplatin (GC) has emerged as the standard adjuvant regimen for UTUC, demonstrating significant efficacy in prolonging disease-free survival (DFS) with manageable toxicity profiles.6–8 However, resistance to chemotherapy and early disease progression remain major challenges, 9 and chemotherapy alone appears insufficient. Therefore, exploring additional adjuvant therapeutic options is necessary to further improve the prognosis of patients with UTUC. In recent years, immunotherapy has made considerable progress in the treatment of UTUC. The CheckMate-274 trial demonstrated that adjuvant nivolumab significantly prolonged DFS in patients with high-risk muscle-invasive UC, highlighting the role of immune checkpoint inhibitors (ICIs) in the adjuvant setting.10,11 These findings have stimulated interest in exploring combination regimens that integrate immunotherapy with conventional chemotherapy to enhance clinical outcomes.
Tislelizumab is a novel humanized immunoglobulin G4 (IgG4) monoclonal antibody, similar to nivolumab, and has demonstrated favorable clinical efficacy and safety.12,13 Compared with other programmed death 1 (PD-1) inhibitors, tislelizumab exhibits higher affinity and binding specificity for PD-1, which may result in a longer therapeutic effect. 14 Based on these characteristics, tislelizumab has been widely used in the treatment of various cancers, including esophageal cancer, 15 lung cancer,16,17 and bladder cancer, 18 and has achieved promising clinical efficacy. The combination of tislelizumab and chemotherapy has also demonstrated favorable outcomes in the treatment of several cancers.19–21 Previous studies have reported that tislelizumab combined with gemcitabine plus cisplatin (TGC) significantly improved the prognosis of patients with locally advanced UC. 22 Based on the promising clinical efficacy demonstrated by TGC therapy in UC, this study aimed to evaluate the efficacy and safety of TGC as adjuvant therapy in patients with muscle-invasive UTUC and to provide guidance for the selection of future clinical adjuvant therapy strategies.
Methods
Study design
We conducted a single-center, retrospective cohort study of consecutive patients who underwent laparoscopic RNU at the Affiliated Hospital of Xuzhou Medical University between April 2020 and April 2025. Patients were selected consecutively from our prospective UTUC database, and no random or selective sampling was applied. Inclusion criteria were as follows: (a) age ≥18 years; (b) pathologically confirmed muscle-invasive UTUC (pT2–pT4, any lymph node status (N), and distant metastasis status (M) 0 (M0)); (c) residual tumor status after surgery (R) 0 (R0) resection; and (d) initiation of adjuvant GC or TGC within 90 days after surgery. No patients received neoadjuvant therapy. During RNU, all radiologically suspicious or grossly enlarged lymph nodes were excised. Eligible participants had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 and a pretreatment glomerular filtration rate (GFR) ≥50 mL/min. All patients completed at least three cycles of adjuvant treatment. Patients were allocated to either the TGC or GC group based on the regimen they actually received. The choice between GC and TGC was made jointly by the attending urologist and the patient and was primarily driven by reimbursement availability (local tislelizumab coverage since March 2021) and economic considerations. Written informed consent was obtained from all patients. The institutional ethics committee approved the protocol (Approval Number: XYFY2023-KL156-01). This retrospective cohort study was conducted in accordance with the Declaration of Helsinki, as revised in 2024. All patient identifiers were removed before analysis, and no details that could identify individuals are presented. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 23
Procedures
Adjuvant therapy was administered in 21-day cycles. Patients in the GC group received 1000 mg/m2 gemcitabine on days 1 and 8 along with 70 mg/m2 cisplatin on day 2. Patients in the TGC group received the same chemotherapy schedule with the addition of 200 mg tislelizumab on day 7. Both regimens were administered for ≥3 cycles, and no maintenance immunotherapy was administered subsequently. Precycle hematology and renal function were evaluated in all cases. Adverse events were prospectively recorded during clinic visits and graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE), version 4.03.
All patients underwent regular follow-up after surgery every 3 months for the first 2 years, every 6 months in the third year, and annually after that. During follow-up, treatment-related adverse events (TRAEs) were evaluated and graded. Tumor recurrence or distant metastasis was assessed using chest, abdominal, and pelvic computed tomography (CT); positron emission tomography/CT (PET/CT); or cystoscopy examinations during routine follow-up. Pathological staging was confirmed by two urological pathologists. Imaging-based recurrence was independently assessed by two radiologists using a blinded method, and a third reviewer was consulted in cases of disagreement.
Lymph node management
Extended lymph node dissection was not performed; only clinically suspicious nodes were excised. Overall, 58.2% (39/67) of patients had atleast one lymph node removed.
Outcomes
The primary observed outcome of this study was DFS in patients in both groups, defined as the time from surgery to the first recurrence, metastasis (including bladder recurrence), or death from any cause, whichever occurred first. Recurrence and metastasis were determined through imaging, cystoscopy, and pathological examination. Secondary observed outcomes included distant metastasis-free survival (DMFS) and recorded TRAEs.
Bias control
To reduce selection bias, patients were included consecutively based on stringent inclusion and exclusion criteria. Imaging endpoints were determined using a blinded method, and multivariable Cox regression analysis with prespecified covariates was performed to reduce confounding bias.
Statistical analysis
Fisher’s exact test or the chi-square test was used to evaluate intergroup differences. Kaplan–Meier (K–M) curves were used to analyze DFS and DMFS in patients in both groups, and differences were evaluated using the log-rank test. Cox regression analysis was performed for univariable and multivariable analyses. A two-sided p <0.05 was considered statistically significant. All statistical analyses were conducted using Statistical Package for Social Sciences (SPSS) version 26.0. Because of the retrospective nature of the study, all p values were exploratory and should be considered descriptive rather than confirmatory.
Results
Clinical characteristics of patients
A total of 67 patients met the inclusion criteria (37 TGC and 30 GC), and none were lost to follow-up (Figure 1). The sample size in this retrospective design was determined by the actual number of patients admitted over the past 5 years. The calculation showed that when 27 DFS events were observed, the statistical power was 68%. Pathological staging revealed pT2 disease in 22 patients (32.8%), pT3 in 41 (61.1%), and pT4 in 4 (5.9%). Renal function (GFR ≥ 50 mL/min) was confirmed before every adjuvant cycle in all patients. There were no statistically significant differences in baseline characteristics (age, sex, ECOG status, smoking status, hematuria status, tumor size, tumor grade, pT, lymph node metastasis, tumor location, number of comorbidities, and GFR) between patients in both groups (Table 1).

Patient screening flowchart.
Clinical characteristics of the two groups of patients.

-: Fisher exact test; ECOG: Eastern Cooperative Oncology Group; GC: gemcitabine plus cisplatin; GFR: glomerular filtration rate; pN+: lymph node involvement; pN0: no lymph node involvement; TGC: tislelizumab combined with gemcitabine plus cisplatin; χ2: Chi-square test.
Efficacy and safety
At the time of analysis, the median follow-up duration was 16.9 months (interquartile range (IQR): 8.1–26.5) in the TGC group and 12.8 months (IQR: 8.7–27.2) in the GC group. No patients were lost to follow-up or withdrew consent. By April 2025, 27 disease-related events had been recorded. Recurrence or metastasis occurred in 10 (27.0%) patients in the TGC group compared to 17 (56.7%) patients in the GC group. At a median follow-up of 15.6 months (IQR: 8.7–27.1), the median DFS was 27.1 months (95% confidence intervals (CI): 11.5–42.7) in the GC group, whereas it had not been reached in the TGC group (Figure 2). TGC significantly prolonged DFS compared with GC alone (hazard ratio (HR) = 0.441, 95% CI: 0.201–0.965; p = 0.035). The 3-year DFS rates were 46.7% (95% CI: 28.3–65.1) with GC and 72.9% (95% CI: 57.8–87.9) with TGC, yielding an absolute risk reduction of approximately 26 events per 100 patients treated. Similarly, DMFS was significantly improved with TGC (HR = 0.389, 95% CI: 0.155–0.978; p = 0.036) (Figure 3).

Comparison of disease-free survival (DFS) between patients in the TGC group and the GC group. GC: gemcitabine plus cisplatin; TGC: tislelizumab combined with gemcitabine plus cisplatin.

Comparison of distant metastasis-free survival (DMFS) between patients in the TGC group and the GC group. GC: gemcitabine plus cisplatin; TGC: tislelizumab combined with gemcitabine plus cisplatin.
Univariable and multivariable Cox regression models identified TGC as an independent predictor of both DFS and DMFS (Tables 2 and 3). Subgroup analyses confirmed consistent benefit across all predefined strata (Figures 4 and 5).
Univariable and multivariable Cox regression analyses for disease-free survival.

CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; GFR: estimated glomerular filtration rate; HR: hazard ratios; pN+: lymph node involvement; pN0: no lymph node involvement; TGC: tislelizumab combined with gemcitabine plus cisplatin; GC: gemcitabine plus cisplatin; inf: infinity.
Univariable and multivariable Cox regression analyses for distant metastasis-free survival.

CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; GFR: estimated glomerular filtration rate; HR: hazard ratios; pN+: lymph node involvement; pN0: no lymph node involvement; TGC: tislelizumab combined with gemcitabine plus cisplatin; GC: gemcitabine plus cisplatin; Inf: infinity.
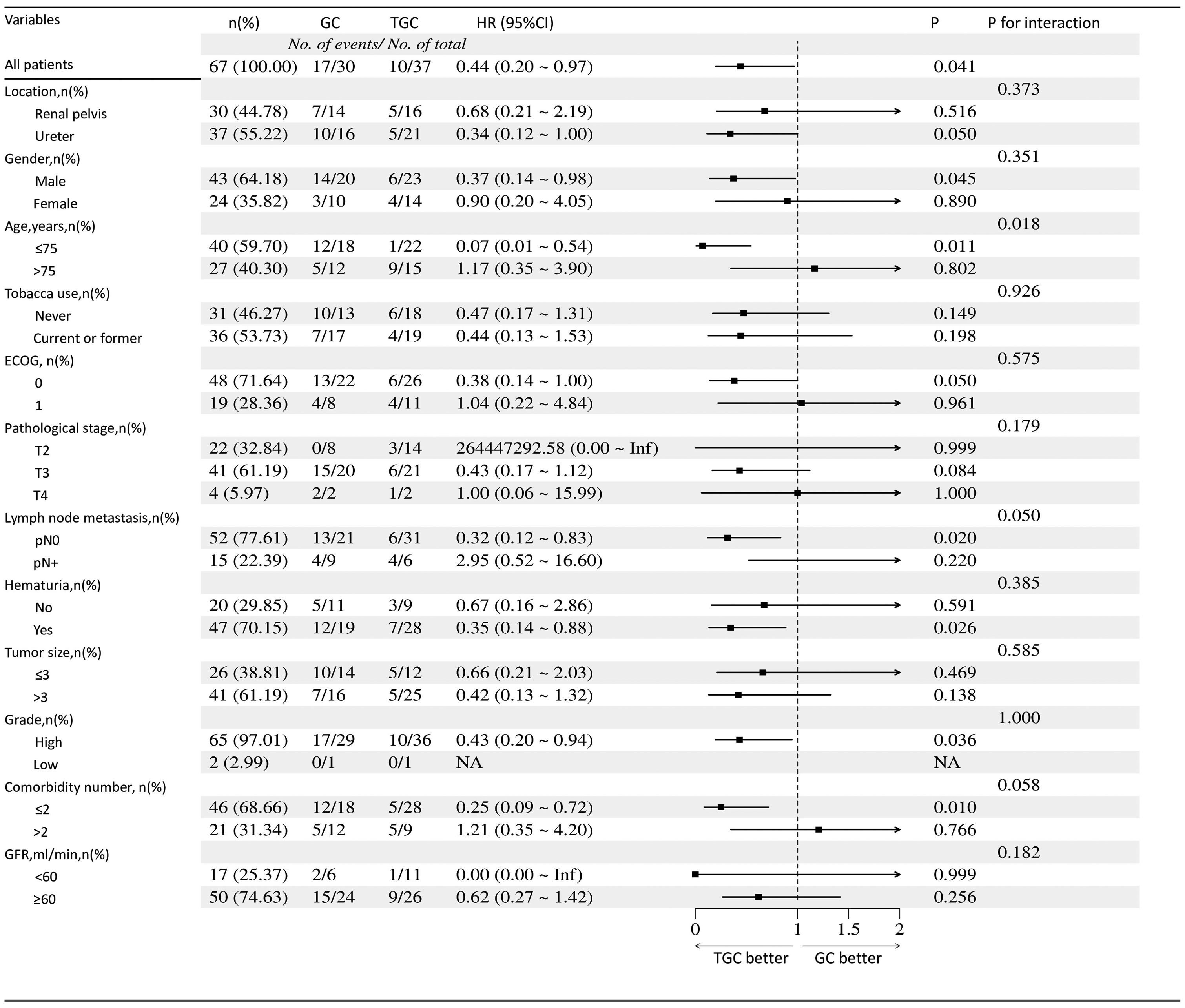
Forest plot of subgroup analyses for disease-free survival (DFS).

Forest plot of subgroup analyses for distant metastasis-free survival (DMFS).
Among the 37 patients in the TGC group, 36 (97.3%) experienced TRAEs, including 5 (13.5%) cases of grade 3 adverse events. Among the 30 patients receiving GC chemotherapy, 28 (93.3%) experienced TRAEs, including 3 (10.0%) cases of grade 3 adverse events. There were no treatment-related deaths. The incidence of adverse events observed in this study was generally consistent between the two groups and included leukopenia, anemia, nausea/vomiting, diarrhea, pruritus, and fatigue. These findings are consistent with those of previous studies reporting adverse events associated with chemotherapy, and no new adverse reactions were observed. Notably, some studies have reported that hypothyroidism is a common adverse event associated with immunotherapy.24–26 In this study, regular thyroid function tests were conducted in patients receiving TGC therapy, and no patients with treatment-related hypothyroidism were observed.
Discussion
At present, a large body of evidence indicates that platinum-based chemotherapy is the standard adjuvant treatment for postoperative patients with UTUC.27–29 Among these regimens, GC is widely used in patients with postoperative UTUC because of its favorable clinical efficacy and tolerability. The phase 3 POUT trial, which included 261 patients with UTUC with pT2–pT4 N0–N3 M0 or pTany N1–N3 M0 disease, showed that at a median follow-up of 30.3 months, adjuvant chemotherapy significantly improved DFS and DMFS, reducing the relative risk of disease recurrence or death by 55%. 30 The study also suggested that combination strategies integrating adjuvant chemotherapy with novel agents warrant further investigation to determine whether such regimens can further improve outcomes in patients with UTUC. On this basis, we conducted the present study to evaluate the efficacy of TGC combination therapy.
Our results indicated that adding tislelizumab to GC chemotherapy significantly improved DFS and DMFS in patients with UTUC, which may be attributed to the synergistic effect of platinum-based chemotherapy and PD-1 inhibitors, thereby enhancing clinical efficacy. 31 Importantly, the incidence and severity of TRAEs were comparable between the two groups, and no new safety signals were observed, highlighting the tolerability of the TGC regimen.
In addition, this study found that advanced age was an independent risk factor affecting DFS and DMFS in patients, and T stage was an independent risk factor affecting DFS. These findings are consistent with those of previous studies. A retrospective study involving 776 patients who underwent UTUC surgery showed that patients aged ≥70 years undergoing RNU had poorer prognosis than younger patients. 32 Another retrospective study involving 401 patients undergoing RNU showed that a higher pT was an independent risk factor affecting survival outcomes.33–35 Although the sample size in the present study was relatively small, our findings are consistent with those reported in the aforementioned studies.
This study has several limitations. First, PD-L1 status was available in only 10.8% of patients in the TGC group, and the absence of maintenance immunotherapy leaves the optimal treatment duration unclear. However, even short-term PD-1 blockade may induce durable systemic immunity, and our findings warrant prospective validation. As a biomarker for assessing the response to ICIs, PD-L1 can, to some extent, predict the prognosis of patients receiving PD-1 inhibitor therapy.36,37 Based on patients’ economic conditions and personal willingness, only 4 (10.8%) patients in the TGC group underwent PD-L1 expression evaluation. Therefore, subgroup analysis of patients with PD-L1 expression levels ≥1% was not performed. Nevertheless, the CheckMate-274 trial demonstrated a DFS benefit with adjuvant nivolumab irrespective of PD-L1 level, 10 suggesting that our findings are unlikely to be driven solely by PD-L1–high tumors. Future studies should evaluate PD-L1 expression status in patients with UTUC to exclude the potential influence of higher PD-L1 expression, as patients with elevated PD-L1 levels may achieve greater clinical benefit from treatment, thereby affecting study outcomes. In addition, the relationship between PD-L1 expression status and patient prognosis requires further clarification. Second, because UTUC is relatively rare and tislelizumab is a novel PD-1 inhibitor with a relatively short clinical application history, the number of patients included in this study was relatively small. This limitation may have introduced bias in the statistical analysis and ultimately affected the representativeness and accuracy of the results. Moreover, the median follow-up of 15.6 months remains immature for adjuvant immunotherapy trials; therefore, multicenter studies with larger sample sizes are required to validate these findings. Third, treatment assignment was based on physician–patient shared decision-making rather than randomization, and propensity score matching was not performed. Therefore, residual selection bias cannot be fully excluded. In addition, studies have shown that the standard surgical treatment for patients with UTUC does not routinely include formal lymph node dissection, and the survival benefit of abdominal extended lymph node dissection (ELND)remains controversial.38–41 Therefore, patients in this study did not undergo routine lymph node dissection during surgery; only abnormal lymph nodes identified by direct visualization or preoperative imaging were removed (39 of 67). This approach may have resulted in some lymph node–positive patients being overlooked, potentially affecting the final study outcomes.
At present, TGC combination therapy has been used as a novel adjuvant treatment in various types of cancer,42–45 including muscle-invasive bladder cancer (MIBC), gastric cancer, and lung cancer, and has demonstrated favorable clinical efficacy. As a closely related malignant tumor, the promising clinical efficacy of TGC combination therapy as a neoadjuvant treatment in MIBC may also be applicable to patients with muscle-invasive UTUC. A study involving 57 patients showed that the GC regimen as neoadjuvant chemotherapy can improve overall survival (OS) and progression-free survival (PFS) in patients; 46 however, TGC therapy has not yet been used as a neoadjuvant treatment for UTUC. Further studies are warranted to clarify the reliability of TGC therapy as a neoadjuvant treatment. Meanwhile, DFS has certain limitations as an alternative indicator for OS in patients with UTUC undergoing radical surgery. In future studies, OS should be evaluated over longer follow-up periods to further validate the findings of the present study.
Conclusion
In this retrospective cohort, adjuvant TGC was associated with potentially improved DFS and DMFS compared with GC alone; however, confirmation in larger prospective trials is warranted.
Footnotes
Acknowledgments
Not applicable.
Author contributions
The study was designed by JZ and YX, who also collected the data, carried out the statistical analysis, and drafted the final manuscript. DC, HZ, and WZ assisted with the study design and manuscript preparation. All authors contributed to the article and approved the submitted version.
Consent for publication
Not applicable.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that they have no competing interests.
Funding
This work was supported by the Nantong University Clinical Medicine Special Scientific Research Fund Project (2024LY006) and the Xuzhou Science and Technology Project (KC23341).
