Abstract
Introduction
A best case series (BCS) review process has been used at the US National Cancer Institute (NCI) to assess the available case report documentation of unconventional cancer approaches. The review process dates back at least to the review of documentation of patients using Laetrile in the mid-1970s.
1
Today, it continues to be used at the NCI to assess the case report documentation of all unconventional cancer therapies submitted for review. In 1990, the Office of Technology Assessment of the US Congress published a report on unconventional cancer treatment. It recommended that NCI provide technical assistance for the compilation of such case series, and allow access to their review. As a result, from 1991 to 1997, NCI’s Cancer Therapy Evaluation Program of the Division of Cancer Treatment and Diagnosis (DCTD) oversaw the NCI BCS Program to (
In 1998, the management of the NCI BCS Program transitioned to the auspices of the Office of Cancer Complementary and Alternative Medicine (OCCAM). Currently, it is administered by the Case Review and Intramural Science Program (CRISP) of the OCCAM. The program strives to receive and evaluate case reports from unconventional practitioners around the world. What makes the program unique is that it uses the same rigorous scientific methods employed in evaluating treatment responses with conventional medicine. As such, the BCS is a distinctive program that provides an opportunity for unconventional practitioners to submit their “best cases” for an independent, retrospective review and validation of medical records, imaging, and pathology. Specifically, the case reports include information from primary source documents confirming the diagnosis, treatment, and outcome (tumor shrinkage or stability).2,3 However, since the cases are selected by the practitioner and represent the “best” outcomes from a larger group of patients,
Over the past 10 years, about 82 cases have been submitted from South Asia through the NCI BCS Program. The modalities of treatment range from homeopathy to ayurvedic and Tibetan medicine. This article highlights the cases of 4 Indian practitioners who have treated patients with unconventional approaches and successfully submitted a BCS for review at NCI OCCAM. These cases were selected because the practitioners provided the required information to complete the NCI BCS review process.
Ayurvedic medicine, also called ayurveda, originated in India several thousand years ago. The term
The alternative medical system of homeopathy was developed in Germany at the end of the 18th century. Supporters of homeopathy point to 2 unconventional theories: “like cures like”—the notion that a disease can be cured by a substance that produces similar symptoms in healthy people; and “law of minimum dose”—the notion that the lower the dose of the medication, the greater its effectiveness. Many homeopathic remedies are so diluted that no molecules of the original substance remain. Treatments are “individualized” or tailored to each person—it is not uncommon for different people with the same condition to receive different treatments. 5
Materials and Methods
NCI OCCAM solicits for case reports from cancer complementary and alternative medicine (CAM) practitioners through advertisement at CAM conferences, on the OCCAM Web site, in medical journals, and in magazines. Health care practitioners interested in compiling and presenting a BCS to the Program for review may contact staff of the CRISP via post, fax, or e-mail. These practitioners are invited to submit case report summaries of patients treated with a particular CAM therapy, subsequent to obtaining consent from the patients. The cases are then reviewed to see if they meet the NCI BCS Program eligibility criteria.
There must be a definitive diagnosis of cancer documented through tissue biopsy or fine needle aspiration, or in the case of some leukemias and a few other cancer types, appropriate blood markers.
There must be an identifiable disease at the start of the alternative regimen and documentation of disease response that can be followed radiographically, or through other validated biomarkers of tumor response. Radiological measurement of the tumor(s) before, during, and after completion of treatment is required for solid tumors.
There must not be any other treatments that would confound the interpretation of the outcome data. Specifically, the patient should not have received concurrent treatment(s) with known therapeutic potential, for example, chemotherapy or radiation therapy. There should be sufficient time (4 weeks) between the end of any conventional anticancer therapy and the beginning of an alternative therapy, to minimize the probability that a response was due to the conventional therapy.
There must be primary source documentation (medical records) that the patient received the specific unconventional therapies described. Furthermore, the dates of all interventions and change in the size and character of the tumor during interventions must be clearly documented in the medical records.
For each case report summary that satisfies the NCI BCS criteria, the practitioner is asked to submit the primary source documentation (ie, the physician’s notes and patient’s pathology and radiology reports).
After a comprehensive review of all materials presented, the CRISP program designates each case as “persuasive” (P), “supportive” (S), or “not evaluable” (NE). P cases have a pathologic confirmation of cancer, evidence of tumor regression, absence of confounders, and confirmation that the patient used the unconventional therapy in question. S cases meet all program criteria except that the tumor response is stable at best. Additionally, in some S cases, there is inadequate radiographic follow-up but an unexpectedly long survival. In some instances, the pathology reports were available, but the microscopic slides of the pathology specimen were not available for review by National Institutes of Health (NIH). Cases defined as NE are either missing essential primary source information or have confounders that make it difficult to assess the case objectively. For example, there may not be evidence of pathologic diagnosis of cancer, or the patient may have had concurrent chemotherapy while taking the CAM treatment. Detailed information about the NCI BCS program can be found on the OCCAM Web site (http://www.cancer.gov/cam/best_case_series_program.html). 6
The postreview process has varied over the years, but currently all findings are summarized, and those cases that indicate evidence of tumor regression or stability are sent to an “expert panel” for a secondary review. The expert panel consists of oncologists within NCI, as well as those in academia and clinical practice. Their role is to review the case series after the BCS Program has validated the information and make a recommendation to OCCAM regarding the utility of future NCI-initiated research (preclinical or clinical). In previous years, promising case series were presented to the Drug Development Group (DDG) within the NCI’s Division of Cancer Treatment and Diagnosis or the Cancer Advisory Panel for Complementary and Alternative Medicine (CAPCAM).
Finally, a BCS review and outcome summary is generated and shared with the submitting practitioners. Herein, we present a sample of several cases that were submitted by CAM practitioners resident in India to the NCI BCS Program and underwent the entire review process.
Results
Table 1 provides a concise overview of the 4 case series submissions to the NCI BCS Program from health care practitioners in India that underwent the complete review process. A total of 68 case reports were submitted and reviewed, representing the unique homeopathic or ayurvedic approach of the 4 practitioners. Overall, 60% of the patients who visited these 4 practitioners did not have any prior conventional treatment. Specifically, 64%, 90%, 70%, and 15% of patients treated with Homeopathic Therapy I, Homeopathic Therapy II, Ayurvedic Therapy I, and Ayurvedic Therapy II, respectively, did not have prior conventional therapy. Of the total of 68 cases, 32 (47%) were labeled as either S or P. The majority (53%) were NE. This was due to a variety of factors, such as missing primary source information (ie, pathology slides or medical imaging studies) or a confounding variable such as recent (<4 weeks) or concurrent chemotherapy. The P and S cases constituted 25% and 22%, respectively (Table 2).
Indian NCI Best Case Series CAM Practitioner Profile.
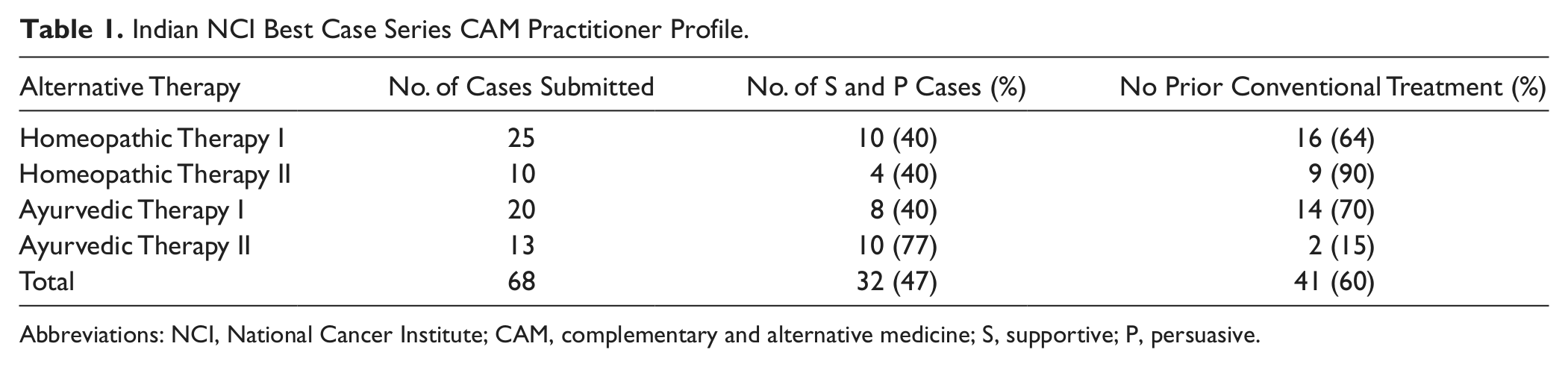
Abbreviations: NCI, National Cancer Institute; CAM, complementary and alternative medicine; S, supportive; P, persuasive.
Indian NCI Best Case Series CAM practitioner outcomes.
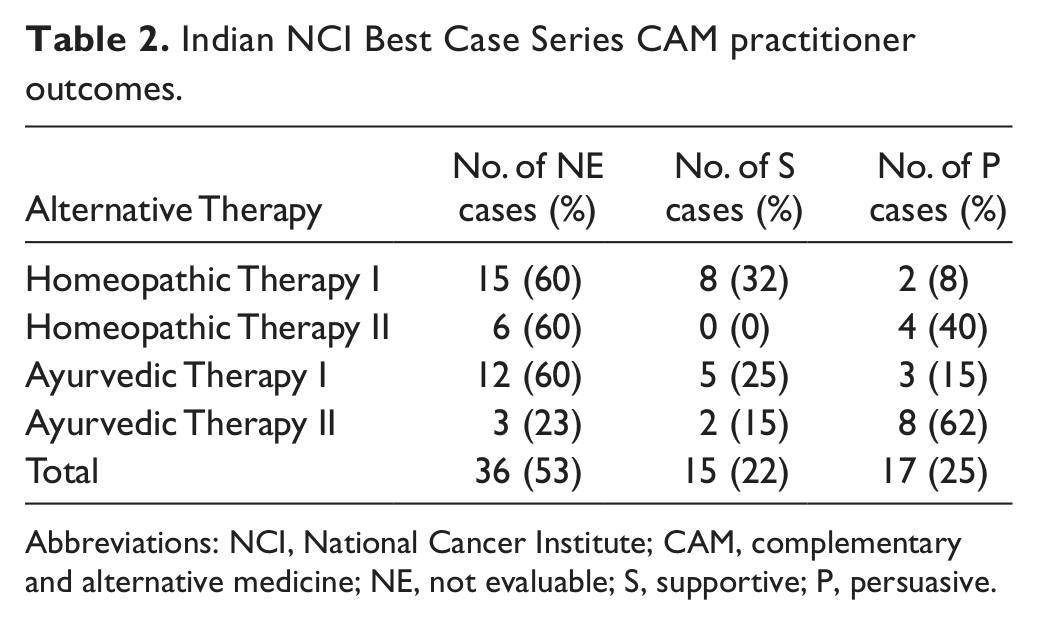
Abbreviations: NCI, National Cancer Institute; CAM, complementary and alternative medicine; NE, not evaluable; S, supportive; P, persuasive.
Table 3 (A-D) gives a comprehensive overview of the types of case reports submitted. The cases span a variety of tumor types, including lung, gallbladder, pancreatic, colon, esophageal, rectal, bone, soft tissue, lymphoid, brain, and breast cancers. Hematologic malignancies are also represented and include chronic lymphocytic leukemia (CLL), acute myeloid leukemia (AML), acute promyelocytic leukemia (APL), and acute lymphoblastic leukemia (ALL). Furthermore, the cases submitted represented an equal percentage of each of 2 types of alternative therapies.
Indian NCI Best Case Series.

Abbreviations: M, male; F, female; Y, yes; N, no; NE, not evaluable; S, supportive; P, persuasive.
For the first unconventional regimen, Homeopathic Therapy I, 25 cases were submitted and reviewed by the NCI BCS Program (Table 3A). The series spanned a variety of malignancies, including lung, pancreatic, gastric, and gallbladder cancers. Indeed, even a rare childhood nephroblastoma was represented (Wilms’s tumor). The age ranged from 2 to 81 years. There were 11 males and 14 females. Of the 25 cases, 2 (8%) were designated as P, 8 (32%) as S, and 15 (60%) as NE. The P cases had pathologic slides and radiographic imaging that were reviewed and confirmed by pathologists at the NIH. The S cases had radiographic images that were also reviewed at the NIH; however, although the original pathology reports confirmed cancer, the slides were not available for review and confirmation by the NIH.
The NCI Best Case Series Program received 14 case reports for a second unconventional regimen, Homeopathic Therapy II (Table 3B). The OCCAM chose and reviewed the 10 cases that met program criteria. Malignancies represented by this case series included small cell lung cancer, non–small cell lung cancer, cancer of the bile duct, and cancer of the esophagus. The ages ranged from 30 to 94 years. There were 6 males and 4 females. Of these 10 cases (Table 2), 4 (40%) cases were designated as P and 6 (60%) as NE.
A third unconventional regimen, Ayurvedic Therapy I (Table 3C), was represented by 20 cases that were submitted for review. This series also spanned multiple malignancies including cases of colorectal, pancreatic, esophageal, and head and neck cancers. Also included were peripheral nerve sheath and spindle cell sarcomas, as well as multiple myeloma. The ages ranged from 15 to 76 years. There were 15 male and 5 female patients. Of the 20 cases (Table 2), 3 (15%) were designated as P, 5 (25%) as S, and 12 (60%) as NE. As such, 60% of the case reports were either missing important information or had confounding variables. These designations were based on the review of cases and their fulfillment of program criteria as well as the confirmation of pathology and/or photographic images. Specifically, for the cases of head and neck cancers with visual lesions, photographs were used to document the evidence of tumor regression. In this case series, the persuasive cases had both imaging and pathologic slides reviewed and confirmed at the NIH, while the majority of supportive cases only had review of imaging and pathology reports. Unfortunately, original pathology slides were not available for review at the NIH.
Finally, a fourth Indian case series consisting of 13 reports, Ayurvedic Therapy II (Table 3D), was submitted to the program. The ages ranged from 3 to 74 years. There were 7 males and 6 females. Of the 13 cases (Table 2), 8 (62%) were designated as P, 2 (15%) as S, and 3 (23%) as NE. Once again, these designations were based on review of the case reports and primary source documentation, including pathology and radiology reports. The practitioner was not able to obtain a significant number of radiologic imaging and pathology slides from the patient’s conventional practitioners; however, all of the P cases had a diagnosis of leukemia, blood reports, and essential biomarkers available for review.
Discussion
In India, approximately 80% of cancer patients are diagnosed with late stage incurable disease. 7 Those who live in medically underserved areas are thus compelled to try unconventional and, oftentimes, more affordable therapies. 8 It has been found that approximately 70% of the Indian population obtains medical help from private physicians, while nearly half seek help from alternative or traditional medical practitioners. 9 Ayurvedic medicine, for example, is a system of traditional medicine native to India that has been practiced for nearly 3500 years. Reportedly, many cancer patients turn to it as a more affordable option or as a last resort after disease progression on conventional treatment. 10 Questions of efficacy, primarily due to the lack of data and ambiguities of therapeutic recipes, surround unconventional approaches, such as those presented herein.
The BCS methodology came into existence as a
The use of the BCS methodology by Jacobson et al 11 exemplifies and substantiates much of the challenges faced by our program. 11 For example, in compiling an acceptable case series for review, the NCI BCS Program requires pathologic confirmation of the cancer diagnosis, evidence that cancer was present at the time of the initiation of CAM treatment, no recent (<4 weeks) conventional treatment, and evidence of complete or partial response. 3 Though reasonable and necessary, these requirements are not easy to fulfill. Specifically, each criterion presents its own challenges of compliance. For example, our program experience indicates that the majority of patients have been initially diagnosed and treated by conventional oncologists. Subsequently, as treatment options decline, many patients pursue unconventional treatments. This presents a particular challenge for CAM practitioners who are compiling the case series, as required records are not readily accessible to them. Furthermore, since the case reports are retrospective, some cases may be relatively old, making it difficult to locate supporting information (eg, pathology slides and imaging). This challenge has been substantiated by Jacobson et al, 11 who also indicated that old charts and radiology films were often difficult to locate. Additionally, CAM practitioners, especially those who are not allopathic physicians, may require patients to interact routinely with their physicians. 12 Though necessary, this results in further dispersal of medical records that would need to be accessed for the completion of the case series submission. All of this may ultimately add up to a cumbersome and time-consuming retrieval process, seemingly unavoidable at times.
Another potential challenge faced by submitters is that data in a BCS are compiled from information accrued as part of routine clinical practice rather than in the context of a research setting; as such, information on potential confounding variables (such as earlier medical history or severity of disease) may be absent. 13 Furthermore, the most compelling candidates for a BCS are retrospective case reports of patients who have survived for many years after their initial diagnosis. Unfortunately, compilation of the information for these case reports also presents its own challenges. Specifically, the documentation trail is often cold; specimens, slides, and films may have deteriorated or been misplaced. Finally, treating physicians may have retired or relocated. 11 Unfortunately, because of these challenges, many “best cases” are ultimately not able to be reviewed under the auspices of the BCS Program, which necessarily requires primary source documentation for the purpose of validation.
It is also important to note that the BCS Program is subject to selection bias as a result of the process of self selection of “best cases” by CAM practitioners.2,3 Specifically, cases that are chosen are those that represent the best outcomes from a series of cases, and have complete documentation; therefore, as one might assume, a best case series is not designed for calculating a response rate. For instance, a report of 2 best-case series prepared for the Agency for Healthcare Research and Quality did not present response rates but recommended consideration of further studies. In both reviews, the cases involved cancers of several different organ/sites and a lengthy effort to obtain medical records. 14 To calculate a response rate, one would have to review the charts of all cancer patients seen at each clinic over a specific period of time and identify all complete and partial responses among them. In addition, one would have to eliminate all patients with recent or concurrent potentially curative conventional treatment and document the status of the cancer at the time of the initial visit to the unconventional practitioner. 11 Since this is not a part of the BCS review, response rates are appropriately not calculated.
Most important, the BCS approach is clearly
Finally, there are some unique challenges associated with obtaining and reviewing international cases, such as those described herein. The NCI BCS Program requires the submitting practitioners to obtain consent from the patients prior to sending case summaries to OCCAM. Subsequently, for those case summaries that meet the BCS criteria, researchers from the Case Review and Intramural Science Program of OCCAM must contact the subjects to explain the program and purpose of the review; however, the difficulty lies in the fact that approximately 35% of the population in India is uneducated. The time and technicalities involved in the consent process may be intimidating to subjects whose awareness about medical and legal rights is low. Moreover, language, cultural, and community barriers further increase the challenges of consent process. 15 The process may also be compromised by patient-centered barriers, such as age, education, and illness, and/or process-centered barriers, such as content and readability of a form, timing of discussion, and amount of time allotted for the process. 16
Several forms of traditional medical systems are available to the population in India. Notable among them are homeopathy and ayurvedic medicine. The practitioners of these medical systems treat a significant part of the population diagnosed with cancer; however, there is minimal collaboration between practitioners and researchers based in India. In 2007, NCI OCCAM examined the dynamics of this type of collaboration by organizing a conference titled “Cancer Researcher and CAM Practitioners: Fostering Collaborations; Advancing the Science.” The focus of this conference was to foster interaction and dialogue between the 2 groups with regard to priorities in CAM clinical research. Examples of successful partnerships were highlighted at the meeting, providing incentive for other future endeavors. This conference was attended by some homeopathic and ayurvedic practitioners from India. More conferences such as this are necessary in an endeavor to build bridges between CAM practitioners and researchers in India and the United States. Such partnerships will lay the foundation in furthering development of unconventional therapies as future mainstream cancer treatments.
Since the early 1980s, reports have emerged in the United States of cancer patients declining conventional cancer treatment and using complementary and unconventional approaches instead; however, the number of patients who make this decision is not well documented. 17 In 1984, Cassileth et al 17 examined those cancer patients who attended a university cancer center (n = 304) and those, recruited through various US media sources, who were receiving treatments from CAM practitioners or CAM clinics (n = 356). 17 Of the total (n = 660), 378 patients used CAM. Of these, 53 (14%) declined conventional treatment of any kind. In this review, 60% of the patients did not have any prior conventional treatment. We can speculate that one of the driving forces for the initial utilizations of cancer CAM therapies in India is a larger percentage of indigent population.9,18 Verhoef and White 19 conducted a qualitative study of 31 cancer patients who had declined all conventional cancer treatment for CAM between 1999 and 2000. They reported numerous motivations for the patient’s decision, including a negative experience with mainstream medicine, loss of family members or friends to cancer while on conventional treatment, CAM use before diagnosis, and a strong belief system in favor of whole-person healing. A variety of other factors also affect the decision to accept or decline standard treatment after a diagnosis of cancer. These include poor doctor–patient communication, the emotional effect of the diagnosis, perceived severity of conventional treatment side effects, a high need for decision-making control, and strong beliefs in holistic healing and the mind–body–spirit connection. van Kleffens and van Leeuwen 20 concluded that both medical and personal reasons play a role in a patient’s decision to refuse treatment but that personal values and experiences predominate. Their study also revealed that patients find quality of life to be very important and seem to believe that quality of life is incompatible with oncology treatment.
As previously mentioned in the Materials and Methods section, the second level of review of cases is currently done by an expert panel of oncologists. At the time these cases were reviewed by NCI BCS program, CAPCAM or DDG represented the second level of review. One of the homeopathic case series was deemed to be sufficient to warrant NCI initiated observational prospective research. However, a planned prospective observational study was not conducted because of our inability to obtain the necessary ethical and scientific regulatory approvals in India. The second homeopathic case series was deemed not to have enough convincing evidence to warrant NCI-initiated research. The 2 Ayurvedic case series were not recommended for a second level of review. This was because a significant number of cases did not have pathologic slides and/or radiologic imaging available for review at the NCI.
Conclusion
Claims of benefit for complex and practitioner-specific CAM are numerous and difficult to evaluate. OCCAM’s NCI BCS Program challenges CAM practitioners to submit their “best cases” to the same rigorous scrutiny applied to conventional cancer treatment. However, especially in settings where pathology, imaging, and charting are not used, are unavailable, or do not meet current conventional standards, most practitioners cannot document best cases. This article has highlighted the many hurdles that the cases faced and that any retrospective review of case reports (not just those involving CAM) must overcome.
For a variety of reasons, a significant portion of cancer patients in India, as well as elsewhere, seek care from CAM practitioners. Only if those practitioners can document their patients’ diagnoses, treatments, and outcomes can claims of success be evaluated. Researchers therefore may want to consider how they might help patients and practitioners to obtain such documentation prospectively, or in low-resource settings, how inexpensive and widely available technology (eg, cell phones) might be used to provide it.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential confliction of interest with respect to the authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this article was provided by the National Cancer Institute (NCI). The NCI did not participate in the design, analysis, interpretation of data, and writing of article.
