Abstract
Epstein–Barr virus (EBV) is a human virus with oncogenic potentials that is implicated in various human diseases and malignancies. In this study, the modulator activity of the potent herbal extract drug thymoquinone on EBV was assessed in vitro. Thymoquinone was tested for cytotoxicity on human cells of lymphoblastoid cells, Raji Burkitt’s lymphoma, DG-75 Burkitt’s lymphoma, peripheral blood mononuclear cells, and periodontal ligament fibroblast. Apoptosis induction was analyzed via TUNEL assay and activity studies of caspase-3. The effect of thymoquinone on EBV gene expression was determined using real-time polymerase chain reaction. We report here, for the first time, a promising selective inhibitory affect of thymoquinone on EBV-infected B cell lines in vitro, compared with lower activity on EBV negative B cell line and very low toxicity on human peripheral blood mononuclear cells and periodontal ligament fibroblasts. Moreover, the drug was found to efficiently suppress the RNA expression of EBNA2, LMP1, and EBNA1 genes. Specifically, EBNA2 expression levels were the most affected indicating that this gene might have a major contribution to thymoquinone potency against EBV infected cells. Overall, our results suggest that thymoquinone has the potential to suppress the growth of EBV-infected B cells efficiently.
Introduction
Epstein–Barr virus (EBV) is a double-stranded DNA virus, belonging to the γ-herpes virus family. EBV is a widespread human virus that infects more than 90% of the world adult population. 1 Following infection in immunocompetent individuals, the virus mostly establishes asymptomatic lifelong latency in memory B cells. 2 Also, it has been reported that the virus can infect both T cells and epithelial cells. 3 EBV has a potent transforming ability, being able to provoke uncontrolled proliferation of infected human B cells and has been associated with several diseases. It is the main causative agent of infectious mononucleosis 4 and has been linked with many types of lymphoid- and epithelial-cell malignancies, including Hodgkin lymphoma, Burkitt lymphoma, posttransplant lymphoproliferative disease,5-7 nasopharyngeal carcinoma, 8 and gastric cancer. 9
Human resting B cells from peripheral blood can be easily transformed by EBV in vitro into actively proliferating lymphoblastoid cell lines (LCLs), in which the continuous growth is driven by viral proteins. A restricted set of viral genes (latency Ш program genes) coding for 6 proteins (EBNA1, EBNA2, EBNA3a, EBNA3b, EBNA3c, and EBNA-LP) and 3 membrane proteins (LMP1, LMP2A, and LMP2B) have been reported to be expressed in LCLs in vitro. 10 However, 3 major genes were shown to have key roles in EBV-infected B cells growth and transformation, which are EBNA1, EBNA2, and LMP1. EBNA2 is the first EBV protein expressed after infection of B cells in vitro and plays a major role in cellular transformation. It is a trans-activator that regulates viral gene expression, being able to activate the promoters of EBNA genes, LMP1 and LMP2A. 11 EBNA1 is another crucial gene for EBV replication and persistence in B cells. It has a key role in replication and partitioning of EBV episomes in proliferating cells. 12 In addition, EBNA1 was detected in almost all types of latently infected cells and EBV-associated tumors. 13 LMP1 is a well-known viral oncoprotein that has been reported to induce lymphoma in transgenic mice expressing the gene in B cells. 14 Also, LMP1 inhibits apoptosis through upregulation of anti-apoptotic genes.15,16 In addition, LMP1 has been described to mimic the action of CD40 activation pathway and stimulate the nuclear factor-κB pathway. 17 Moreover, other studies using EBV mutants have described the key role of LMP1 for immortalization of B cells. 18
Despite the growing understanding of the role of EBV in the pathogenesis of disease, the optimal management of EBV-associated tumors remains unsatisfactory. The available strategies of treatment, for example, antiviral agents, monoclonal antibodies, and immune-based therapies are proceeding with encouraging outcomes, but they have lots of limitations in the clinic. 19
Thymoquinone (TQ; 2-isopropyl-5-methyl-1,4-benzoquinone) is the main bioactive component of the volatile oil of the medical plant Nigella sativa (Black seed), and it has been shown to excrete anticancerous, antioxidant, and anti-inflammatory activities both in vitro and in vivo.20-23 In this study, we sought to study the activity of TQ on survival and growth of EBV-infected B cells from different healthy donors and to evaluate its effect on the expression of the critical EBV genes.
Materials and Methods
Chemicals, Reagents, and Cell lines
Thymoquinone (Sigma, St Louis, MO) was prepared as a 10 mM stock solution in dimethyl sulfoxide. Aliquots of the stock were stored at −80°C, and the appropriate working concentrations were prepared with the cell culture medium immediately prior to the experiments. Culture medium (RPM1-1640 and DMEM), fetal bovine serum (FBS),
Epstein–Barr Virus Stock Preparation
B95-8 Marmoset cell line was maintained between 3 × 105 and 9 × 105 cells/mL by diluting the cells 1:3 every 3 to 4 days in RPM1-1640 (supplemented with 10% FBS and 2mM
Generation of EBV-Transformed B Cell Lines (Lymphoblastoid Cell Line)
Peripheral blood mononuclear cells (PBMCs) from 4 different healthy adults were separated by gradient centrifugation (3200 rpm, 12 minutes) over Ficoll-Histopaque (Sigma, St Louis, MO). Then, 2 × 106 mononuclear cells from each donor were suspended in 2 mL of transforming medium containing 1.6 mL RPMI-1640 (supplemented with 20% FBS, 2mM
Human Mononuclear Cells, Periodontal Ligament Fibroblasts Cells Isolation, and Culturing
Peripheral blood mononuclear cells from 5 healthy volunteers (aged between 27 and 54 years) were separated by gradient centrifugation (3200 rpm, 12 minutes) over Ficoll-Histopaque. The cells were washed twice in RPM1-1640 culture medium, and then the mononuclear cells were suspended in culture medium and used directly in cytotoxicity experiments. The periodontal ligament fibroblasts (PLF) cells were isolated and cultured according to the protocol. 24 The 10th to 15th passages of PLF cells were used in the experiment.
Cell Cytotoxicity Assay
The EBV-infected B cell lines; human lymphoblastoid cells (LCma-81) and human Burkitt’s lymphoma cell line (Raji), EBV-negative human Burkitt’s lymphoma cell line (DG-75 and BL41), PBMCs and PLF cells were all subjected to cytotoxicity assay. For suspended cells, briefly, the cells were seeded at a density of 4 × 104 per well in 96-well plates in appropriate medium, then treated with increasing concentrations of thymoquinone (0.4-200 µmol/L). For adherent cells (PLFs), 1 × 104 cells were seeded in each well. Colorimetric Cell Titer 96 non-Radioactive Cell Proliferation Assay (Promega, Madison, WI) was used to detect cells proliferation in each well according to manufacturer’s instructions. Briefly, 15 µL of dye solution were added on each well. The cells were incubated at 37°C and 5% CO2 for 4 hours. Then, 100 µL of the solubilization solution were added to solublize formazan precipitate. The absorbance was recorded at 570 nm using a colorimetric plate reader (Sunrise-Basic, Tecan, Grödig, Austria). Cellular proliferation was expressed as a percentage of cell viability of TQ-treated cells relative to untreated controls. The IC-50 concentrations were obtained from the dose–response curves using GraphPad Prism software 5 (San Diego, CA).
Detection of Apoptosis by the TUNEL and Caspas-3 Assays
The terminal deoxyribonucleotidyl transferase–mediated dUTP nick end labeling (TUNEL) assay was performed using DeadEnd Colorimetric TUNEL System (Promega, Madison, WI). Briefly, 8 × 105 lymphoblastoid cells were treated with TQ for 24 hours. The cells were collected, washed, and suspended in phosphate-buffered saline (PBS) and were added on poly-
The activity of caspase-3 was determined by a caspase colorimetric assay kit (R&D Systems, Minneapolis, MN) according to the manufacturer’s protocol. Briefly, TQ-treated cells were lysed in cold lysis buffer. Then, the cell lysates were tested for protease activity using a caspase-specific peptide conjugated to the color reporter molecule p-nitroaniline. The color reaction was quantified with a spectrophotometer at a wavelength of 405 nm. Caspase enzymatic activities in cell lysates are directly proportional to the color reaction.
RNA Extraction, DNase Treatment, and RT-PCR
RNA was extracted from TQ treated 2 × 105 lymphoblastoid cells using conventional Trizol-chloroform method (Invitrogen, Carlsbad, CA). Around 1 µg of RNA was treated with 1 unit of RQ1 RNase-Free DNase (Promega, Madison, WI) according to manufacturer’s instructions. All the treated RNA was added to the reverse transcription-PCR (RT-PCR) to produce the cDNA. Briefly, 10 µL of treated RNA was mixed with 2 µL dNTPs (10 mM) and 1 µL random primers (500 µg/mL), heated at 65°C for 5 minutes, and then put on ice for 2 to 3 minutes, followed by adding 4 µL of M-MLV-5X buffer, 40 units of RNasin ribonuclease inhibitor R and 200 units of M-MLV reverse transcriptase (Promega, Madison, WI). The components in the tube were mixed gently and incubated for 5 minutes at 25°C, for 40 minutes at 42°C, and 15 minutes at 70°C.
Quantitative Reverse Transcription PCR
All quantitative RT-PCR analyses were performed using the iCycler iQ-BioRad instrument (BioRad, Hercules, CA). Each reaction included a 25 µL reaction mixture containing 10 pmol of each primer (Table 1), 12.5 µL of 2 × SYBR Green PCR Master Mix (GoTaq qPCR Master Mix, Promega, Madison, WI), and 2 µL of the template cDNA. The typical amplification program included hot start activation at 95°C for 2 minutes, followed by 40 cycles of denaturation at 95°C for 15 seconds, annealing, and extension at 60°C for 1 minute. To exclude the presence of nonspecific bands and primer dimers; the RT-PCR samples were run on 2% agarose gel electrophoresis. The housekeeping gene β-actin was used as a control for normalization. To generate a standard calibration curve, a template cDNA from LCLs was used. RNA expression ratio of a certain gene was calculated from the standard calibration curve and threshold cycles (Ct) of both drug treated and untreated LCLs’ mRNA, using the following equation:
Primer Sequences Used for Amplification of Epstein–Barr Virus Genes EBNA1, EBNA2, and LMP1 a

The cellular β-actin primers were obtained from Quanti Tect primer assay (Qiagen, Germantown, MD)
Statistical Analysis
Results are presented as means ± standard deviation. The differences between groups were compared by one-way analysis of variance followed by Tukey’s post hoc test and considered significant at P < .05. The statistical analysis was carried out by using SSPS version 16.0.
Results
Thymoquinone Efficiently Inhibits Survival and Growth of EBV+/− B Cell Lines
The effects of TQ on the survival of B cell lines (LCma-81, Raji, and DG-75) were measured by the MTT assay. The survival of cells was notably inhibited after treatment with increasing concentrations of TQ (Figure 1). The calculated IC-50s of TQ on LCma-81 that were established from 4 different donors were 2.5 ± 1.1, 1.42 ± 0.2, 0.99 ± 0.02, and 1.73 ± 0.24 µmol/L. IC-50 of TQ on Raji after 24 hours was 11 ± 2.1 µmol/L, and IC-50s of the 2 EBV negative cell lines DG-75 and BL41 cells after 24 hours were 20 ± 2.1 and 16.24 ± 0.31 µmol/L, respectively.

Cytotoxic effect of thymoquinone (TQ) on Epstein–Barr virus (EBV)–infected B cell lines (LCma-81 and Raji) and EBV-negative B cell line (DG-75). Exponentially growing cells were seeded in 96-well plates. Cells were treated with increasing concentrations of TQ, and incubated for 24 hours. MTT satin was added on the cells for additional 4 hours. Cellular proliferation was assessed by the ability of cells to convert the soluble salt of MTT into an insoluble formazan precipitate. Data represent the mean ± SD of 3 independent experiments each performed in triplicate
Thymoquinone Has Low Toxicity on Normal Human PBMC and PLF Cells
To assess the cytotoxicity of TQ on normal cells, we tested the effect of TQ on the viability of human PBMCs and PLF cells. TQ showed very low toxicity on those cells; with calculated IC-50s of 18 ± 4.1 and 85 ± 6.5 µmol/L on PBMCs and PLF cells, respectively (Figure 2).

Comparison of TQ cytotoxicity on lymphoblastoid cells (LCma-81) and normal cells (PBMC and PLF) assessed by MTT assay. The data represent the mean ± SD of 3 independent experiments, each performed in triplicate
Thymoquinone Induces Apoptosis in EBV-Infected B Cells
The ability of TQ to induce apoptosis was measured by TUNEL and caspase-3 assays. Using the TUNEL protocol, more apoptotic nuclei were stained with dark brown to black color in TQ-treated cells as compared with the control cells (Figure 3). In addition, the measurement of caspase activation was carried out to determine the induction of caspase-3, which plays a central role in the execution phase of cell apoptosis. Indeed, caspase-3 was found to be significantly upregulated by three- to fourfold (P < .05) in response to an increase in TQ doses after 6 hours of treatment (Figure 4). At 2 hours treatment, the upregulations were only significant at the 6 µmol/L level (P < .05).
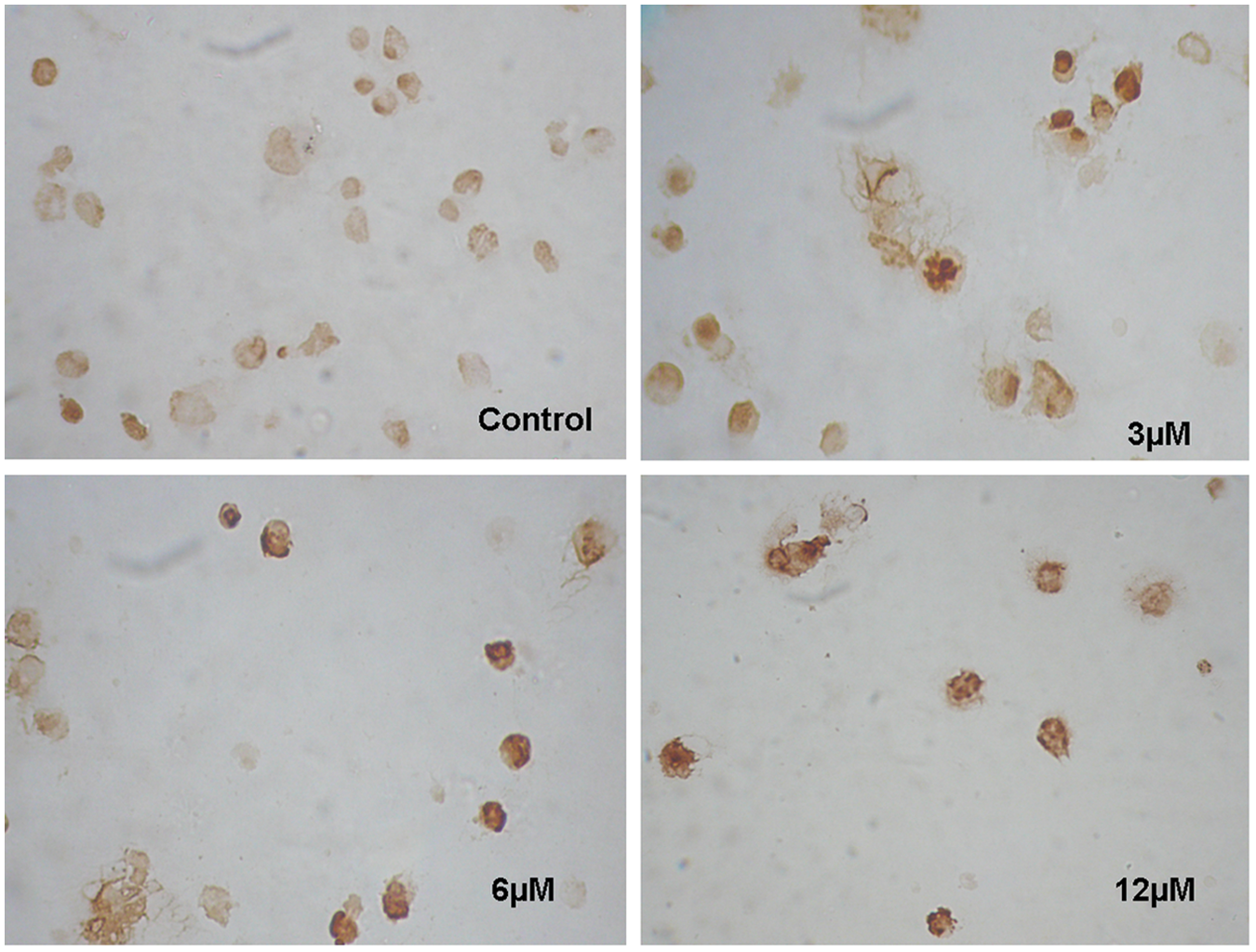
TUNEL staining after incubation of thymoquinone (TQ) with human lymphoblastoid cells. The LCma-81 cells were treated with increasing concentrations of TQ for 24 hours after which the cells were subjected to TUNEL assay. Untreated cells were used as control in the experiment. Apoptotic nuclei were stained with dark brown color

Effect of thymoquinone (TQ) on caspase-3 activity. The level of caspase-3 was upregulated in lymphoblastoid cells (LCma-81) after 2 and 6 hours treatment with increasing concentrations of TQ. Data represent the mean ± SD of 2 independent experiments. *Statistically significant (P < .05)
Thymoquinone Inhibits EBV Gene Expression
The effects of TQ treatment on EBV gene expression have been determined. Total RNA was extracted from LCma-81 cells that had been treated with increasing concentrations of TQ. The cDNA for 3 viral genes, namely EBNA1, EBNA2, and LMP1, was quantified using Q-PCR. The housekeeping gene β-actin was used as a control for normalization. The EBNA2 gene was the first among the 3 viral genes to be significantly affected at 8 hours following TQ treatment (P < .05). EBNA2 was also the most inhibited at the 3 time periods: 8, 12, and 24 hours after treatment with 12 and 25 µM drug doses (Figure 5). Furthermore, LMP1 RNA was observed to be decreased significantly at 12 hours after TQ treatment (P < .05), whereas EBNA1 was the last gene to be significantly affected at 24 hours (P < .05).
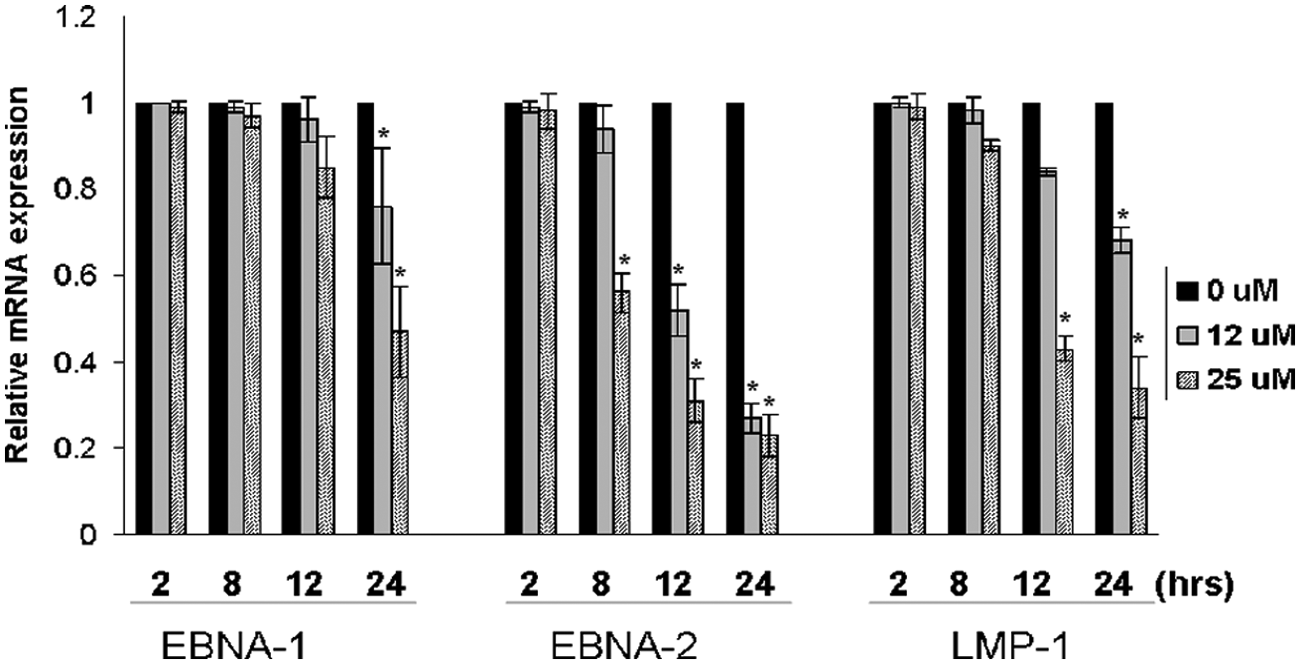
Effect of thymoquinone (TQ) on Epstein–Barr virus (EBV) gene expression. The graph shows a comparison of relative mRNA expression of EBV genes after treatment with increasing concentrations of TQ at different periods of time. The results represent the mean ± SD of 2 independent experiments each performed in duplicate. *Statistically significant (P < .05)
Discussion
The present study reveals a new effect of TQ. It shows that TQ efficiently suppresses the survival and growth of both EBV+/− B cell lines. Interestingly, TQ exerts the highest activity against human lymphoblastoid cells (LCma-81). Also, TQ shows significant higher activity against EBV-infected human Burkett’s lymphoma (Raji) in comparison with noninfected cells (DG-75 and BL41). Moreover, TQ has a low toxicity against normal cells in vitro. In fact, very low toxicity of TQ was observed on cultured PBMCs and normal fibroblasts.
To assess whether TQ is able to kill cells by apoptosis, we have performed 2 different assays that detect apoptosis at 2 levels: morphological and enzymatic. Using TUNEL assay, we obtained typical morphology of apoptotic cells after treatment with TQ. Furthermore, detection of caspase-3 activity was performed to confirm apoptosis, and interestingly caspase-3 activity was significantly (P < .05) increased in dose- and time-dependent manner.
Comparing the obtained TQ IC50s against LCma-81 and Raji with DG-75 and other reported tumor cell lines in the literature reveals a significant variation. For example, on average, TQ is approximately 10 times more potent against LCma-81 than the DG-75 and myeloblastic leukemia HL60 cell lines. 25 This selective killing ability raises a high suspicion that TQ may target the invasive EBV virus genes. Accordingly, we used real-time PCR to test the effect of different TQ concentrations and times of exposure on the expression level of the most important EBV genes. Clearly, the expression level of EBNA2 was the most significantly affected, showing both time and concentration dependence. For example, EBNA2 was the only gene to show a significant amount of inhibition at 8-hour treatment (P < .05) and it was also the most downregulated gene after treating the cells with 25 µM for 12 and 24 hours (Figure 4). The second most significantly affected gene was LMP1. Because of the fact that the expression of LMP1 gene, as many other genes in the lymphoblastoid cells, is known to be under the control of the EBNA2 gene,26,27 LMP1 inhibitory effect may not have resulted from direct TQ interaction but rather from losing the cotranscriptional role of EBNA2. This transcriptional regulatory activity of EBNA2 over LMP1 is exerted through intermediate specific DNA binding proteins that interact with the Cp promoter region of LMP1. EBNA1 is the least affected gene, only scoring a significant expression downregulation after treating the lymphoblastoid cells with 25 µM of TQ for a maximum time of 24 hours (Figure 5). Keeping in mind that EBNA2 does not have a direct trans-activation activity seen with LMP1, the very late inhibitory effect of TQ on the EBNA1 expression may be linked more to the consequences that follow inhibition of the expression of the lymphoblastoid cell most important genes, EBNA2 and LMP1. Taken at face value, this finding implies that TQ inhibits EBNA2 specifically and most probably this inhibition has a major contribution to its high potency against lymphoblastoid cells. This explanation does agree with the study of Farrell et al, 28 who have shown that switching EBNA2 expression by a nuclear antigen peptide inhibits the EBV-induced growth proliferation. Also, this finding can be strengthened by many other studies that illustrated the importance of EBNA2 for lymphoblastoid cell survival and EBV immortalization of B-cells. 10
Conclusions
From a therapeutic point of view, most EBV-associated tumors respond poorly to intensive chemotherapy regimens, and the presence of the EBV genome within these tumors increases the chances of developing strategies directed at viral targets. 29 Accordingly, TQ provides a promising agent that has the ability to significantly inhibit the transcription of EBNA2 and LMP1 and to a lesser extent EBNA1. Importantly, TQ has not been reported to cause suppression of bone marrow function, 30 and therefore it might not add to the hematopoietic toxicity that is often associated with other chemotherapeutic agents. To our knowledge, this is the first report that describes the TQ activity and potential to induce apoptosis in EBV-infected cells. Further investigation should be conducted to reveal the exact mechanism of action.
Footnotes
Acknowledgements
We are grateful for Prof Paul Farrell for the EBV+/− cell lines. We would like to thank Dr Sana Najeh for her assistance in isolation and culturing of periodontal ligament fibroblasts.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

