Abstract
This is a case presentation of a primary central nervous system B-cell lymphoma in a 69-year-old woman with chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids and chronic immunosuppressive treatment. The patient had been diagnosed as having probable chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids, according to international criteria. Afterward, regular clinical and imaging examinations and blood tests were performed. The patient presented with primary central nervous system B-cell lymphoma 3 years after the initial diagnosis of chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids. Brain tissue histology was indicative of diffuse giant B cells, Epstein–Barr virus positive, and non-Hodgkin lymphoma. The nature of chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids is obscure, driving the formulation of many hypotheses about its causes. In our opinion, the presented case supports the putative neoplastic nature of the disease, at least in the long term, and/or along with Epstein–Barr virus involvement, which is known that have been related to other immune-mediated diseases such as multiple sclerosis and malignancies, especially with specific human leukocyte antigen haplotypes. Further research is needed and close monitoring of such patients is strongly recommended.
Keywords
Introduction
Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS) is a recently circumscribed immune-mediated disease characterized by distinct radiological features and a great response to glucocorticosteroid administration. It is a rare syndrome with male predominance (3:1) and middle age of onset.1–3
CLIPPERS may simulate other immune-mediated disorders such as multiple sclerosis (MS), angiitis, and rheumatological diseases with central nervous system (CNS) involvement, as well as paraneoplastic, and infectious or parainfectious diseases such as hepatitis B or Epstein–Barr virus (EBV) infection.4–6 A relationship between CLIPPERS and neoplasms such as primary lymphoma of the CNS has also been described.1,7 The possible role of EBV infection in the perpetuation or initiation of lymphoma in the clinical context of CLIPPERS has not been addressed extensively before. 8
Description of the clinical case
A 69-year-old woman diagnosed with probable CLIPPERS 9 and without other remarkable medical histories (except anxiety, asymptomatic thyroid nodule, and tympanoplasty), presented with progressively worsened dizziness, headache, decreased visual acuity, diplopia, nausea, vomiting, and walking instability, for 7 days.
Three years ago, at the age of 66, the patient’s brain and spinal cord MRI revealed multiple T2-hyperintense, enhancing lesions in the posterior cranial fossa and spinal cord (Figure 1(a)–(f)). Blood and CSF analysis were normal except for mild elevation of CSF protein. The immunologic blood tests (including paraneoplastic antibodies, anti-MOG, anti-AQP4, and anti-GFAP antibodies) and CSF and CSF-PCR studies, including EBV, were negative. The cytologic examination of CSF did not reveal malignant cells and CSF flow cytometry disclosed a predominance of T lymphocytes: 76.3% CD3(+)CD4(+) and 20.3% CD3(+)CD8(+). The patient received intravenous and per os corticosteroids with clinical and radiological remission and ultimately the diagnosis of probable CLIPPERS was set 10 (Figure 1(g)).

Serial MRI. Initial MRI (a–f), 3 years before current admission reveals multiple T2-hyperintense, punctuate lesions in the pons and the cerebellum on axial T2-FLAIR image (arrows a), enhancing on axial T1 post-contrast administration (arrows b). Sagittal T1 post-contrast images (c–f) show multiple foci of contrast enhancement in the pons, the cerebellum, and the medulla oblongata (arrows c), as well as in the cervical (arrows d), thoracic spinal cord (arrows e) and in the conus medullaris (arrow f). Resolution of the abnormalities following corticosteroid treatment is evident on sagittal T1 post-contrast image 2 months later (g). Six months after the initial MRI a new T2-FLAIR hyperintense area is noticed in the left middle cerebellar peduncle (arrow h), while smaller hyperintense foci appear in the cerebellum and the spinal cord. Axial brail T1 (i) and sagittal cervical spine T1 (j), both post-contrast administration reveal lesion enhancement in the left cerebellar peduncle, the cerebellum (arrows i), and the spinal cord (arrow j). Response to corticosteroids was found on follow-up MRIs (not shown). Finally, MRI 3 years after the initial scan shows a T2-FLAIR hyperintense (arrow k), enhancing (arrow l) mass lesion with increased relative perfusion on cerebral blood volume map (arrow m) in the left cerebellar hemisphere and the ipsilateral cerebellar peduncle. High Cho concentration, decreased NAA, lipid (short arrow n), and lactate (long arrow n) peaks were found on MR spectroscopy (n).
Six months later, a few new punctuate T2-hyperintense enhancing lesions in the cerebellum and cervical spinal cord and a larger lesion in the left cerebellum peduncle appeared (Figure 1(h)–(j)). Six doses of Cyclophosphamide were infused with total clinical and radiological remission and subsequently per os Azathioprine (50 mg three times per day) was prescribed, as maintenance therapy.
Three months before the recent admission (32 months after the initial MRI scan), regarding the patient’s clinical and imaging stability, the dose of Azathioprine was reduced to 25 mg three times per day.
At the time of admission, the clinical examination revealed saccadic smooth pursuit, decreased visual acuity of the left eye (OD 20/70), mild muscle weakness in the left extremities, head tremor, cerebellar ataxia of the left arm, brisk right tendon reflexes and a Babinski sign 11 on the right plantar.
Brain MRI (Figure 1(k)–(n)) was performed and showed a mass lesion in the left cerebellar hemisphere with peripheral enhancement, perilesional edema, and mass effect on the fourth ventricle. Increased relative cerebral blood volume was observed on MR perfusion and a large choline peak, reversed choline/creatinine ratio, and markedly decreased N-Acetylaspartate, lipid, and lactate peaks were found on MR spectroscopy. The MRI findings were suggestive of a primary CNS tumor, most probably lymphoma. Additionally, thoracic and abdominal scanning with computerized tomography was performed with no systematic findings. The patient was treated with dexamethasone, per os, with dramatic clinical improvement.
Biopsy—partial resection—histology
Consequently, the patient was transferred to another’s Hospital Neurosurgery Clinic. A biopsy of the lesion was done and concurrently, the debulking of the tumor was attempted to lessen the rapidly evolving symptoms that appeared due to the mass effect.
Histologic examination revealed large lymphocytes with one to three nucleoli in between inflammatory cells (Figure 2(a)), strong CD20 (Figure 2(b)), and ki-67 (Figure 2(c)) expression in the large B lymphocytes and positive chromogenic in situ hybridization for EBV-encoded small RNAs (Figure 2(d)). The above-mentioned findings were indicative of diffuse giant B cells, EBV positive, and non-Hodgkin lymphoma. The patient after biopsy and mass resection was dismissed and we lost further follow-up.
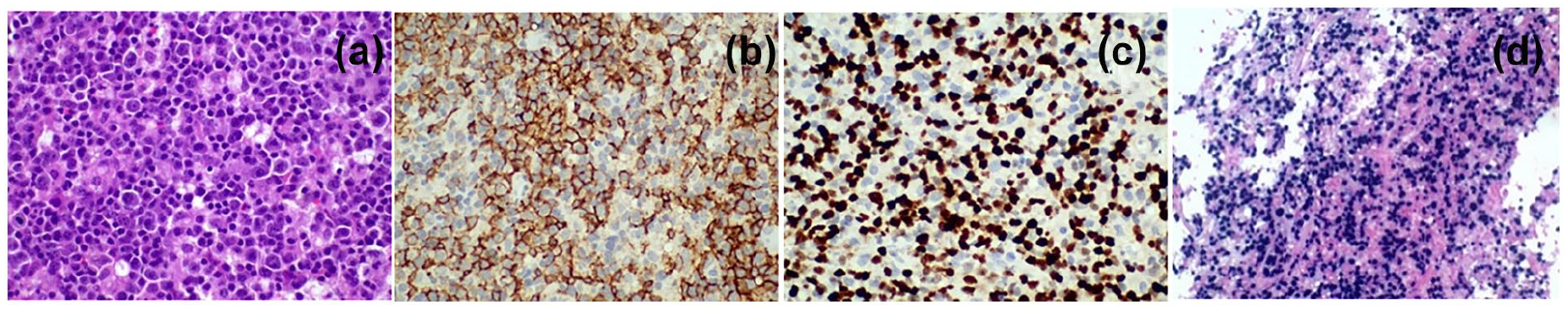
Biopsy. Large lymphocytes with one to three nucleoli in between inflammatory cells, HE ×400 (a), strong CD20 expression in the large B lymphocytes, IHC ×400 (b), strong ki-67 expression in the large B lymphocytes, IHC ×400 (c), in situ hybridization for the EBERs: diffuse positivity in large B cells (d).
Unfortunately, the patient’s husband informed us of her death, 2 months later.
Discussion
CLIPPERS has been associated with many pathologies of CNS, such as neoplasms, autoimmune inflammatory, and infectious diseases. Recently, a possible correlation of CLIPPERS with a human leukocyte antigen (HLA)-B27 has been implied 12 , and the development of B-cell lymphoma after CLIPPERS and EBV infection has been reported. 7
We present a case of PCNSL in a CLIPPERS-diagnosed patient, provoking concern about the possibility of the neoplastic nature of the syndrome. PCNSL accounts for 4%–7% of all brain tumors. The presence of EBV-positive diffuse B-cell lymphomas is related to older age, male sex, and immunosuppression. It is rare in immunocompetent individuals and is associated with chronic EBV infection, though its causality, in the pathogenesis, has not been proved. 13 Studies support the hypothesis that EBV could possibly lead to lymphomagenesis. In particular, the EBV-infected B cells go unchecked by T cells due to immunosenescence, noted during aging, and ultimately leads to uncontrolled proliferation.14–16 Moreover, EBV infection could lead to the development of solid tumors, such as nasopharyngeal carcinoma, especially in susceptible people with particular HLA polymorphisms. 17
It is remarkable that our patient had an episode of radiological deterioration 6 months after the diagnosis of CLIPPERS, similar to the present clinical manifestation, although a far milder, and T2-hypertensive, T1-hypotensive, gadolinium enhancement lesion, in the left cerebellar peduncle; in the same position of B-cell lymphoma’s.
It is reasonable to consider that CLIPPERS were a harbinger of CNS lymphoma, and the sixth-month relapse was the same B-cell lymphoma that responded to the above-mentioned immunosuppressants (Cyclophosphamide and Corticosteroids).
As far as its correlation with neoplasms, it is not clearly defined if CLIPPERS is an early stage of a neoplasm, a paraneoplastic disease, or if the chronic immunosuppressive treatment of the syndrome or/and the EBV involvement are responsible for the sequential malignancy.
Unfortunately, we cannot support a hypothesis of possible EBV seroconversion, after the diagnosis of CLIPPERS, due to a lack of serum EBV antibodies at the time of CLIPPERS diagnosis and CSF-PCR and serum EBV antibodies at the time of EBV-positive diffuse giant B-cell lymphoma diagnosis.
It would be under serious consideration the fact that in our patient, who had not had any medical history of immunosuppression, the first relapse occurred soon after the initiation of corticosteroids (six months), with a relatively bigger lesion in the left cerebellar peduncle, at the same position with the presented lymphoma. Thus, it is reasonable to assume that the CLIPPERS itself was the later presented lymphoma.
The detailed and closer follow-up, of such patients, is warranted due to the putative malignant nature of the CLIPPERS syndrome, a fact that has to be clearly confirmed in more cases in the future. It could be also useful for testing of EBV genome and antibodies from the initiation of CLIPPERS diagnosis and further on, to clarify its possible pathogenetic role.
Conclusion
The advances in radiologic methods and equipment, improving the detection capabilities, at very early stages of the diseases, may be responsible for the emerging CLIPPERS cases, which probably constitute an early common manifestation of different underlying conditions. In the future, the detailed description and close monitoring of these cases, with EBV included, from the beginning of the diagnosis may reveal subtle distinctive features that could potentially differentiate underlying causes, with putative fatal outcomes.
Footnotes
Author’s contribution
M.A. conceived the idea and planned the accomplishment of the article, as the head of the Department. M.-E.E. contributed as the physician of the patient. Professor L.S. contributed as director of the Neurologic Department. G.S. is the neurosurgeon who excises the tumor. G.V. and P.T. are the neuroradiologists who assessed the magnetic resonance imaging and spectroscopy. E.-A.S. assayed the histopathology of biopsy specimen. A.V. reviewed the article. E.K. was the patient’s physician and the article’s writer.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
