Abstract
The organic extract of Periplaneta americana L. (Dictyoptera; Blattidae) has been traditionally used in southwestern China as an alternative medicine against disorders such as hepatitis, trauma, gastric ulcers, burns, and heart disease. The present study describes bioassay-guided purification and chemotherapeutic evaluation of the 60% ethanolic fraction of P americana organic extracts (PAE60). The most effective cytotoxic fraction was determined by way of repeated in vitro screenings against 12 distinct cultured human carcinoma cell lines: Eca 109, BGC823, HO8910, LS174T, CNE, HeLa, K562, PC-3, A549, BEL 7404, HL-60, and KB, followed by in vivo antitumor assays of the lead fraction (PAE60). The complexity of enriched active fraction was qualitatively evaluated using thin layer chromatography. Reconstituted PAE60 was effective at inhibiting HL-60, KB, CNE, and BGC823 cell growth with IC50 values <20 µg mL−1. PAE60 reduced tumor growth in S180-bearing immunocompetent mice by 72.62% after 10 days following oral doses of 500 mg kg d−1 compared with 78.75% inhibition following 40 mg kg d−1 of cyclophosphamide (CTX). Thymus and spleen indices of S180-bearing mice treated with PAE60 were significantly greater (P < .05) than CTX treatment groups, suggesting potential immunomodulation of antitumor host defenses by PAE60. Antiviral activity was also investigated and PAE60 inhibited herpes simplex type-2 replication (IC50 = 4.11 ± 0.64 µg mL−1) with a selectivity index (CC50 to IC50 ratio) of 64.84 in Vero cells but was less effective on type-1 virus (IC50 of 25.6 ± 3.16 µg mL−1). These results support future clinical trials on P. americana as an alternative or complementary medicinal agent.
Keywords
Introduction
Cancer has become one of the leading causes of death in major geographic regions such as China, Europe, and the United States.1-3 Complementary and alternative medicine use has increased substantially in conventional and Western medical practices during the past decade4,5 but most notably for the treatment of cancer.6-8 Traditional Chinese medicine (TCM) therapies in particular have proven effective in tumor regression or in preventing carcinomas from further development. 9
Apart from more commonly known herbal and plant-derived medicines, natural secretions from insects and animals have demonstrated potential as alternative medicine therapies.10-14 Excretions from the larvae of the common green bottle fly, Phaenicia (Lucilia) sericata, for example, are known to possess potent antimicrobial and anti-methicillin-resistant Staphylococcus aureus effects. 15 Likewise, earthworm coelomic fluid has been extensively investigated for its cytotoxic activity. 16 There are currently a number of marine invertebrates (eg, sponges) that have become an abundant source of anticancer agents for testing in preclinical and clinical trials.17-20
As the knowledge base of invertebrate natural products is expanding, relatively less is known regarding arthropods. 21 One of the earliest documented practices in Western medicine was the use of the arachnid, Tarantula cubensis, to cure cancer of the tongue. 22 Evidence-based reports of “insect chemotherapy” appeared in 1972, when Pettit et al 23 discovered that a 95% ethanolic extract of the Pieridae family of butterflies leads to more than 70% inhibition of tumor growth in albino rats bearing Walker 256 carcinomas. Similar reports of arthropod extracts as anticancer agents emerged regarding the Asian rhinocerous beetle, Allomyrina dichotomus, 24 the North American yellow jacket wasp, Vespula pensylvanica, 25 and much later, the Texas lubber grasshopper, Brachystola magna. 26
We have been interested in potential chemotherapeutic properties of the American cockroach, Periplaneta americana, a species widely disseminated in China and throughout major cosmopolitan areas of the world. P americana has been extensively studied for its uncommon ability to resist environmental threats, which has contributed to its evolutionary presence for more than 300 million years.27,28 Additionally, P americana has been exploited in southwestern China and northeastern Brazil as an alternative naturopathic remedy for ulcers, burns, and heart disease.29,30 Medicinal use of P americana has remained consistent with classic descriptions in the ancient Chinese Pharmacopeia Shen Nong Ben Cao (circa 100
Based on recent ethnopharmacological reports31,32 we hypothesized that systematic fractionation of P americana extracts over chromatography columns would yield biologically active fractions for chemotherapeutic evaluation. Our long-range objective of identifying novel anticancer agents33-35 stimulated our interest in exploiting bioassay-guided purification 36 to expand our understanding of traditional Chinese/alternative medicines such as P americana extracts. The presence of an antimicrobial peptide in P americana 37 along with reported activity against hepatitis B 38 and Newcastle disease 39 suggest that antiviral activity against common herpes simplex could also be expected. Antiviral immune response peptides have been detected in other insects, such as Calliphora vicina, 40 and thus bioprospecting for similar activities in P americana appears promising. 41
The primary objective in this report was to employ bioassay-guided fractionation (BGF) to evaluate an enriched active component of P americana in 12 cultured human carcinoma cell lines and tumor-bearing mice. The secondary objective was to evaluate the bioactive P americana extract against model viruses (herpes simplex viruses type-1 and -2) in cultured African green monkey kidney epithelial (Vero) cells.
Materials and Methods
Chemicals
Polyamide gel (60-100 mesh); macroporous resins DA201, AB-8, and D101; silica gel (100-200 mesh), C-8, and C-18; strong anion-exchange basic styrene (type 201 × 7) and strong cation-exchange acidic styrene (type 001 × 7) were purchased from Huadong Chemicals Co Ltd (Hangzhou, Zhejiang, China) and Taizhou Si-qin Biochemical Materials Company (Taizhou, Zhejiang, China). Ethanol (EtOH), dimethyl sulfoxide (DMSO), carboxymethylcellulose sodium (CMC-Na), and other reagents were all commercial grade.
Cell Lines
Human promyelocytic leukemia HL-60 (SIBS #TCHu 23), human mouth epidermal carcinoma KB (SIBS #TCHu 73), human lung epithelial adenocarcinoma A549 (SIBS #TCHu 5), human hepatocellular carcinoma BEL 7404 (SIBS #TCHu 64), human nasopharyngeal carcinoma CNE (SIBS #TCHu 13), human cervical carcinoma HeLa (SIBS #TCHu 19), human chronic granulocytic leukemia K562 (SIBS #TCHu 29), human esophageal carcinoma Eca 109 (SIBS #TCHu69), human gastric adenocarcinoma BGC823 (SIBS #TCHu 11), human colon adenocarcinoma LS174T (SIBS #TCHu 32), human ovarian carcinoma HO8910 (SIBS #TCHu 24), human prostatic carcinoma PC-3 (SIBS #TCHu 41), and African green monkey kidney epithelial (Vero) cell lines were all obtained from the Cell Bank Type Culture Collection of the Chinese Academy of Sciences (SIBS, CAS, Shanghai, China). The mouse sarcoma-180 (S180) cell line was obtained from Nanjing Chinese Medical University (Nanjing, China). Cells were cultured in RPMI-1640 (Gibco, North Andover, MA) supplemented with 5% heat-inactivated fetal calf serum (Sijiqing Co, Hangzhou, Zhejiang, China), 100 units mL−1 penicillin and 100 µg mL−1 of streptomycin, at 37°C in 5% CO2 with 95% humidity.
Animals and Insects
Whole adult insects of P americana were collected from the Cockroach Cultivation Base of College of Pharmacy, Dali University, Yunnan Province, China. Insects were expertly examined by Professor Shunan Li at Dali University and voucher specimens housed in the College of Pharmacy, Dali University, Yunnan Province, China. Male ICR mice weighing 18 to 22 g (≈5 weeks old) were housed in our animal facility. Mouse S180 suspensions (2 × 107 cells mL−1) were implanted subcutaneously into one axilla of each mouse. The animals were maintained at 22°C ± 2°C with 40% to 70% relative humidity and free access to food and water. The experimental protocol was approved by the Animal Ethics Committee of Zhejiang University, in accordance with Principles of Laboratory Animal Care and Use in Research (Ministry of Health, Beijing, China).
Bioassay-Guided Purification of the Active Fraction of Periplaneta americana
Dried powder of crude P americana was subjected to 3 extractions with 60% EtOH–H2O, and the extracts were concentrated at 60°C using a rotary evaporator under reduced pressure. Crude P americana extract (PAE, 20 g) was loaded onto low-pressure glass 250-g resin columns (17 cm, inner diameter = 6 cm) with D101, AB-8, DA201, C-8, or C-18 packing materials, and each eluted with 0%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, and 95% EtOH in water solutions. For silica gel fractionation (100-200 mesh), an elution gradient of n-butanol–EtOH–H2O (4:1:1 to 3:1:1, v/v/v) was applied; for strong anion exchange, HCl–H2O (0:100 to 95:5, v/v) was applied; and for strong cation exchange, NH3–H2O (0:100 to 95:5, v/v) was applied. Eluates were evaporated and then freeze-dried to yield fractionated PAEs. Each sample was screened for cytotoxicity against 12 human carcinoma cell lines (see additional methods below). Fractionated PAEs with comparable activities (ie, <10% difference) were combined and resubjected to cytotoxicity assays. Combined fractions that maintained activity were purified on-column using a simplified solvent gradient. Eluates were dried and reconstituted for cytotoxicity screening. Extraction and fractionation of active PAEs was optimized for yield, assay performance, and reasonable cost of materials following triplicate experiments.
Human Carcinoma Cytotoxicity of PAE
Fractionated PAEs (50 µL) were dissolved in DMSO and diluted to different concentrations with culture media. Exponentially growing cells were seeded in 96-well plates at 5 × 103 cells per 100 µL of medium. After 24 hours, the PAE solutions were added to 96-well plates, which were incubated for 72 hours. DMSO was used as a negative control, and cisplatin (cis-diaminedichloroplatinum) (C-DDP) was selected as a positive control. MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] (Amresco, Solon, OH) in saline (5 mg mL−1) was added 72 hours after dosing and following incubation of the plates for another 3 hours, the optical densities (ODs) were read at 570 nm. Carcinoma cell toxicities were expressed as percent inhibition of cell growth and IC50 values obtained by nonlinear regression of log concentration–percentage inhibition data.
Vero cells were seeded in 96-well plates at 1.0 × 104 cells per 100 µL well and different concentrations of fractionated PAE in culture media were applied to wells after 48 hours. MTT in saline (5 mg mL−1) was added 48 hours after dosing and following incubation of the plates for an additional 3 hours, the ODs were read at 570 nm. The concentration causing 50% inhibition compared with untreated samples was calculated as the 50% cytotoxic concentration (CC50).
Qualitative Analysis of PAE60 Components
PAE60 was subjected to thin layer chromatography (TLC) and derivatization for qualitative analysis of fatty acids, steroids, amino acids, sugars, and aromatic compounds. For fatty acids, PAE60 in methanol was spotted on a silica gel plate (20 × 20 cm) and subjected to a 2D-TLC (dimension I, CHCl3–MeOH–28% NH3H2O–H2O, 100:65:7:3, v/v/v/v; dimension II, CHCl3–Me2CO–MeOH–AcOH–H2O, 10:4:3:2:1, v/v/v/v/v). The dried TLC plates were sprayed with 2′,7′-dichlorofluorescein or bromocresol green–bromophenol blue–KMnO4 and heated at 80°C for several minutes before viewing. Zlatkis reagent was used separately to assay steroids. 42 A Feigl reaction was used to check for quinonoid compounds. 43 For amino acid analysis, 15.0 mL of 6.0 M HCl solution was added to 200 mg of PAE60 in a sealed tube (100 mL), and the mixture was subjected to reduced pressure and heated at 110°C for 10 hours. After cooling, the hydrolysis products were filtered and dried before running on 2D silica gel TLC plates (20 × 20 cm; dimension I, n-BuOH–AcOH–H2O, 4:1:5, v/v/v; dimension II, CHCl3–MeOH–25% NH3H2O, 20:5:1, v/v/v). Dried TLC plates were sprayed with 0.1% ninhydrin–acetone reagent and maintained at 80°C for 20 minutes. Amino acid standards (Huadong Chemicals, Hangzhou, Zhejiang, China) were used for comparison of retention factors (Rf) to identify amino acids. For carbohydrate analysis, 10.0 mL of CHCl3 and 2.0 mL of n-BuOH were added to 500 mg of PAE60 in 50.0 mL water and the mixture was stirred vigorously for 20 minutes before centrifugation. The aqueous layer was spotted on 2 silica gel plates and developed in n-PrOH–NH3H2O (40:60:5, v/v/v) prepared at 4°C. One plate was dried and slowly immersed in 2% toluidine blue with 70% EtOH–AcOH (95:5, v/v) and the other was sprayed with anthrone agent.
Chemotherapeutic Evaluation of PAE60 in S180 Tumor-Bearing Mice
Tumor ascites fluid was removed from solid tumors in mice and diluted with saline (1:3 ratio) prior to subcutaneous inoculation of mice armpits with 0.2 mL (N = 40). After 24 hours, the animals were randomly divided into 4 groups (10 per group), and a homogenized formulation of PAE60 in 0.5% CMC-Na was administered to 2 groups via oral gavage at doses of 1500 and 500 mg kgbw−1 d−1, respectively (kgbw−1 d−1 means per kg of body weight per day) for 10 consecutive days. The third group was given 10 µL gbw−1 d−1·of 0.5% CMC-Na via oral gavage as a negative control. The fourth group received 40 mg kgbw−1 d−1 of cyclophosphamide (CTX) in saline via intraperitoneal injection as a positive control.
Tumor volumes (V) were measured daily on days 4 through 9 using Vernier calipers and calculated according to V = a × b2/2, where a and b represent the short and long tumor radius, respectively, on 2 different planes. Mice were sacrificed on the day 11 (ie, 24 hours after the last dose) with the spleen, thymus, and tumor of each animal harvested and weighed. The effect of treatment was recorded as tumor inhibition (%) = 1 − [tumor weighttreated/tumor weightcontrol] × 100, along with the spleen index (SI) = [splenic weight (g)/kgbw] and thymus index (TI) = [thymic weight (g)/kgbw].
Acute Toxicity Test of PAE60 in Healthy ICR Mice
Mice were given a single dose of PAE60 via oral gavage at 500, 1000, and 2000 mg kgbw−1 (N = 30, 10 per group) and placed in climate-controlled storage cages with adequate food and water. Animals were observed daily for changes and expired animals were examined postmortem to ascertain cause of death.
Antiherpetic Activity of PAE60 in Vero Cells
Herpes simplex viruses type-1 and -2 (HSV-1, HSV-2) were obtained from Nanjing Medical University (Jiangsu, China) and stored at −70°C until use. Virus stocks were titered on Vero cells seeded in 96-well plates with 2 mM
Statistical Analysis
In vitro screenings were performed in triplicate on separate days. Experiments in mice used 10 subjects per test. Data are presented as the mean ± standard error of the mean. Differences between means were assessed by one-way analysis of variance followed by Dunnett’s t test (P < .05) using SPSS v.11.0. The computer program IC50-calculator 45 was used for nonlinear regression of log concentration–response data.
Results
Bioassay-Guided Purification of the Active Fraction of Periplaneta americana
The identified procedure for isolating the most active fraction of crude P americana is shown in Figure 1. Dried powder of crude P americana (15.0 kg) produced an average PAE yield of 22% (w/w) following 3 extractions with 60% EtOH–H2O (50:1 each time). Crude PAE (200 g) was reconstituted in 1000 mL of distilled water and loaded on a polystyrene AB-8 column (2.0 kg, 20-60 mesh) for overnight absorption. The column was eluted with distilled H2O, followed by 60% EtOH–H2O (prior to elution with 95% EtOH–H2O for more lipophilic components). PAE60 possessed an approximately 30-fold improvement in potency over crude PAE; however, the yield (44.1 g) was only 1/75 of PAE (3310 g) and 1/340 of the raw insect materials (15.0 kg). Fractions had average yields of 73.15%, 22%, and 2.65% for PAE0, PAE60, and PAE95 respectively.

Preparative schedule for fractionation of alcoholic extracts of Periplaneta americana
Human Carcinoma Cell Toxicity of PAE
The toxicities of PAE at 70 µg mL−1 against 12 human carcinoma cell lines are shown in Figure 2. The aqueous fraction of PAE was either ineffective or exhibited less than 30% inhibition of carcinoma cell viability. The 95% ethanolic fraction generally performed better than the aqueous fraction, but PAE60 performed the best with approximately 40% to 85% inhibition on all tumor cell lines except HO8910. The IC50 values along with the relative potencies of PAE60 to C-DDP for the 12 carcinoma cell lines are listed in Table 1. The relative IC50 values of PAE0, PAE60, PAE95, and C-DDP were consistent with the cell viabilities in 70 µg mL−1. The results revealed that PAE60 was the most cytotoxic fraction of crude PAE for testing on tumor growth in mice.
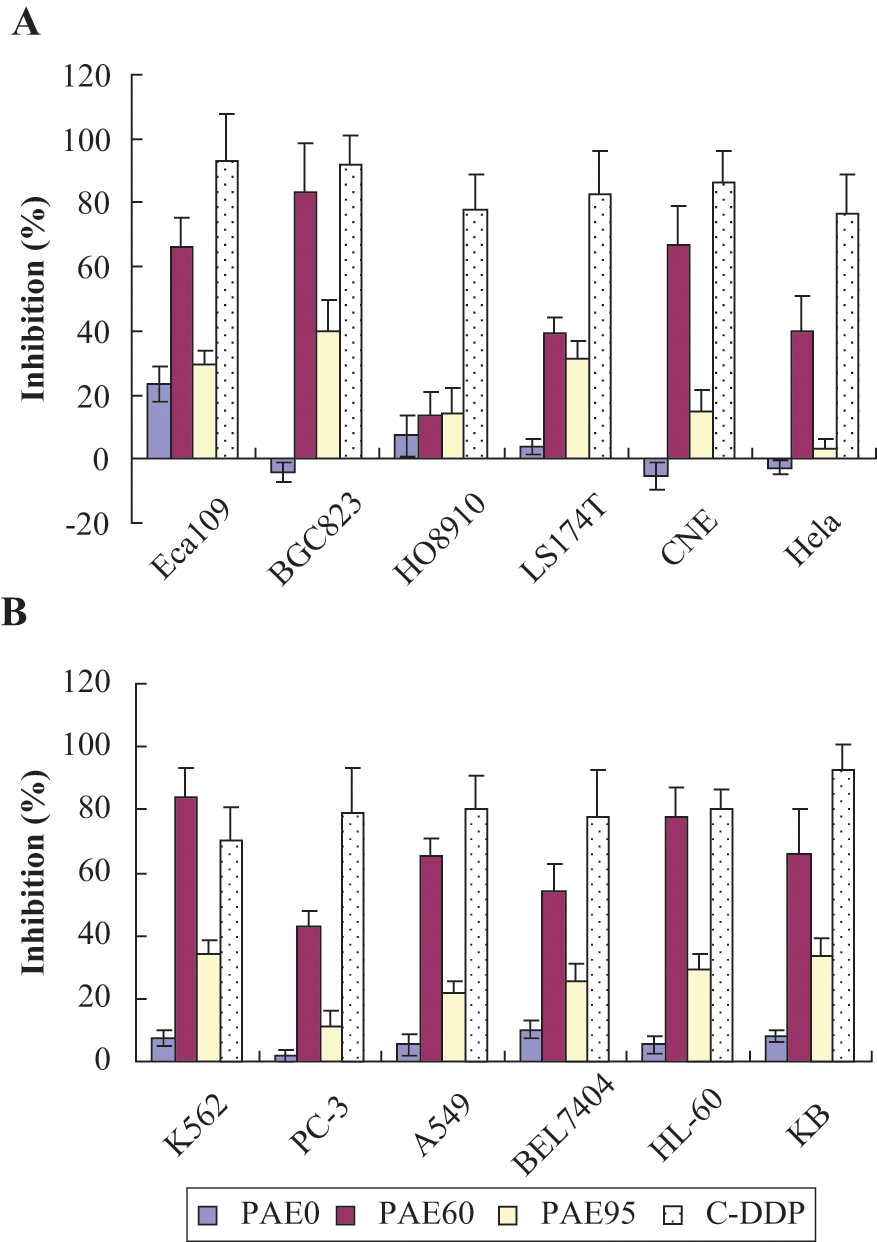
Cell toxicity of BGF extracts from Periplaneta americana against 12 human cultured carcinoma cell lines at 70 µg mL−1 (N = 3)
In Vitro Cytotoxicity (IC50) of BGF PAE and C-DDP Against 12 Human Carcinoma Cell Lines

Abbreviations: BGF, bioassay-guided fractionation; PAE, Periplaneta americana extract; C-DDP, cis-diaminedichloroplatinum.
Table 1 reveals that the K562 cell line was one of the most susceptible to PAE60. Owing to the stability of growth and passaging of K562 cells in our laboratory, we adopted this cell line to explore the dose- and time-dependent cytotoxic response to PAE60, shown in Figure 3.

Concentration–response profiles of PAE60 against K562 cells in vitro (N = 3)
Qualitative Analysis of PAE60 Components
The positive reaction to bromocresol green–bromophenol blue–KMnO4 and 2′,7′-dichlorofluorescein indicated the presence of saturated fatty acids and possibly some unsaturated fatty acids (as evidenced by a deep-blue nonfluorescence color). The reaction to Zlatkis reagent was slight, indicating an absence of cholesterol and sterol lipids in PAE60. The assay detected the presence of quinoid or naphthoquinoid compounds, as evidenced by the positive Feigl reaction. The amino acid composition was determined to be Asp, Ala, Arg, Glu, Gly, Ile, Leu, Lys, Phe, Pro, Ser, Thr, Tyr, and Val. Polysaccharides were indicated in PAE60 by positive reactions with toluidine blue and anthrone reagent.
Chemotherapeutic Evaluation of PAE60 in S180 Tumor-Bearing Mice
The antitumor activity of PAE60 in vivo is shown in Figure 4. Mice dosed at 1500 mg kgbw−1 d−1 showed 53.07% inhibition of tumor growth and weight relative to untreated animals. Mice dosed at 500 mg kgbw−1 d−1 showed 72.62% inhibition. The positive control, CTX, inhibited the tumors by 78.75% at 40 mg kgbw−1 d−1.

Tumor weights and tumor growth inhibition rates on day 11 of S180 tumor-bearing mice treated with CMC-Na (negative control), CTX (40 mg kgbw−1 d−1), PAE60 (L) (500 mg kgbw−1 d−1), and PAE60 (H) (1500 mg kgbw−1 d−1). Each value is the mean ± SEM (N = 10); *** indicates P < .005 when compared with the mean of the negative control group
The tumor growth kinetics from day 4 through 9 is shown in Figure 5. The untreated group and high-dose PAE60 exhibited similar growth rates at 71.1 and 71.4 mm 3 d−1, R 2 = .92 and .94, respectively, although the PAE60 (H) group was more effective at inhibiting early growth. CTX exhibited tumor growth of 44.7 mm3 d−1 (R2 = .84) and was not readily comparable to PAE60 (L) in the same manner because of a nonlinear growth profile (R2 = .62).
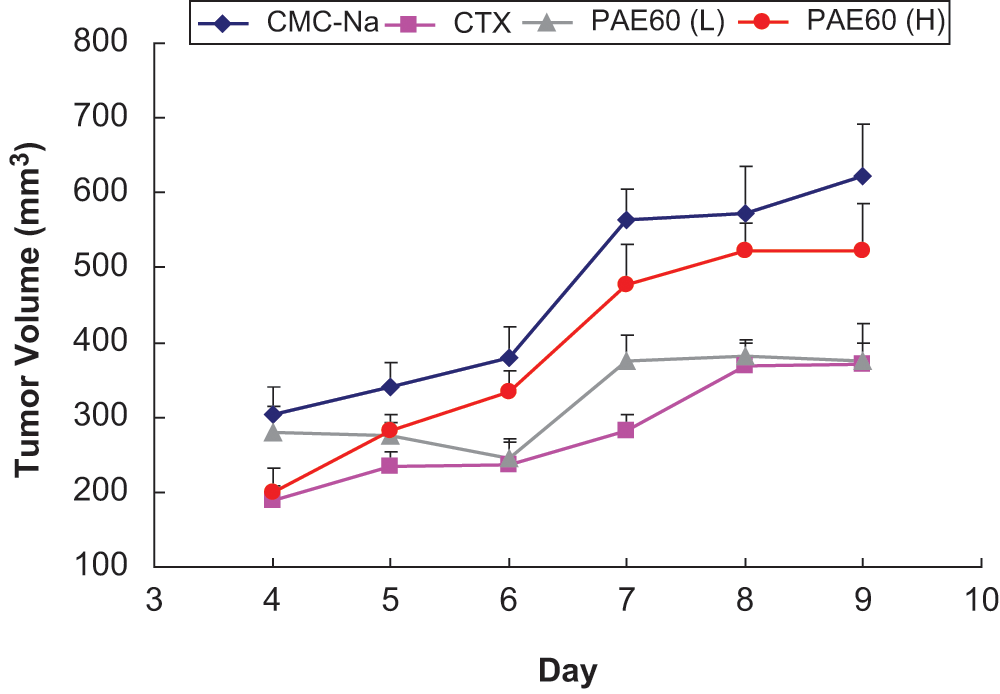
Growth kinetics of S180 tumor volumes for test groups: CMC-Na (vehicle control); CTX (cyclophosphamide positive control); PAE60 (L), 500 mg kgbw−1 d−1; PAE60 (H), 1500 mg kgbw−1 d−1
The spleen and thymus indices are listed in Table 2. The spleen and thymus indices of PAE60-treated mice were similar to the CMC-Na group and normal mice. Significant decreases in splenic and thymic weights were observed for the CTX-treated group compared with the CMC-Na group (P < .05). The results suggest an immunosuppressant effect of CTX compared with no immunotoxicity observed on lymphoid organs by PAE60.
Spleen Index and Thymus Index of S180 Mice Treated With PAE60 Versus Controls

Abbreviations: PAE60, 60% ethanolic fraction of Periplaneta americana organic extracts; CMC-Na, carboxymethylcellulose sodium; CTX, cyclophosphamide.
Dosages vs. body weight; each value is the mean ± standard error of the mean, n = 10.
”*” and “***” indicate P < .05 and P < .005, respectively, when compared with the mean of negative control group CMC-Na. “#” and “###” indicate P < .05 and P < .005, respectively, when compared with the mean of positive control group CTX.
The morphology of the tumors was assessed and is displayed in Figure 6. Figure 6A represents untreated excised tumors, which were blood-red in color and surrounded by rich blood vessels indicating the tumors were “healthy” and growing. Figure 6B shows CTX-treated mice tumors with a pale white color and with few blood vessels suggestive of tumors undergoing cell death (eg, necrosis or apoptosis). Figure 6C shows tumors exposed to 500 mg kgbw−1 d−1 of PAE60 where the color remained primarily blood red, although the tumor size was reduced and showed visual signs of dying tissue (associated with paleness of color). Tumors treated with 1500 mg kgbw−1 d−1 of PAE60 were still surrounded by blood vessels and maintained relatively normal proliferative growth (highly variable results) without much visual evidence of tissue or cell death (Figure 6D).

Relative size and color of S180 tumor groups following chemotherapy: (A) CMC-Na (untreated) group; (B) CTX-treated group; (C) PAE60 (L) (500 mg kgbw−1 d−1); and (D) PAE60 (H) (1500 mg kgbw−1 d−1)
Antiherpetic Activity of PAE60 in Vero Cells
The antiherpetic concentration–response relationship of PAE60 against HSV-2 is shown in Figure 7 (HSV-1 not shown). PAE60 exhibited concentration-dependent inhibition of cell damage due to HSV-2 infection. The efficacy (ie, the Emax as indicated by the height of the plateau on the dose–response curve) of PAE60 was comparable to ACV, but less potent with an IC50 of 4.1 µg mL−1 compared with 0.23 µg mL−1 for ACV. The maximum effect of PAE60 was reached with potentially minimal toxicity (ie, CC50 > 200 µg mL−1) as evidenced by its selectivity index (Table 3). PAE60 was less potent against HSV-1 (IC50 of 25.6 µg mL−1) compared with HSV-2.

Concentration (µg mL−1) versus percentage inhibition for PAE60 and ACV on HSV-2 replication in Vero cells. Data represent the mean ± SEM (N = 3)
Antiviral Potency (IC50) of PAE60 Against HSV-1 and HSV-2

Abbreviations: IC50, half maximal inhibitory concentration; PAE60, PAE60, 60% ethanolic fraction of Periplaneta americana organic extracts; HSV, herpes simplex virus; SI, selectivity index, CC50, cytotoxic concentration in 50% of cells.
Discussion
Bioassay-Guided Purification and Qualitative Analysis
The BGF procedure employed was designed to enrich the chemotherapeutically active extract components of P americana. Optimization of fractionation materials and elution schemes was based on 2 primary factors: preservation of cytotoxic potency and maximization of yield. Extracts optimized in this manner are thus required to be applied to a large number of samples, and our attempts to minimize the cost of the procedure enhance its potential as a viable commercialized medicine. The AB-8 column used is a partially polar macroporous adsorption resin and produced the most active fractions on most of the cancer cell lines tested with some loss of yield relative to other columns. The choice of extraction solvents in optimizing the fractionation was fairly broad; however, many solvents are expected to cause protein denaturation, which could reduce biological activity. Combining fractions with similar activity ensured the chemotherapeutic components would be fully present in the final product. Lower activity present in either the salt-rich aqueous fraction or more lipophilic extracts (ie, 95% ethanolic eluate) were therefore expected to be independently active from the mildly polar 60% ethanolic eluate.
Thin layer chromatography confirmed the presence of peptides and/or proteins in the active fraction, which support a hypothesis of immune function stimulant proteins in PAE60.46,47 The identification of quinoid compounds, however, did not rule out the activity of small molecules, as such compounds are well-known reactive toxicants. 48
Human Carcinoma Cell Toxicity of PAE
The human ovarian cancer line (HO8910) was relatively resistant to PAE, which supports a cell-specific effect or mechanism of action. 49 Ovarian cancer cell lines are reportedly less susceptible to cytotoxicity from immunoreactive proteins 50 and the results would be consistent with an immunomodulatory role of PAE60. PAE60 performed exceptionally well against human gastric adenocarcinoma BGC823 cells (83.4% inhibition), which are not particularly known for their resistance to anticancer peptides. 51 Put into context, BGC823 cells may be remarkably more sensitive to cytotoxic agents than most other carcinoma cells. 52 Patterns emerged from the in vitro results, such as the HL-60 and K562 leukemic cell lines as a group were generally highly susceptible to PAE60 followed by the epithelial/epidermal cancer lines KB, CNE, Eca 109, and A549 whereas cisplatin (a broad-spectrum DNA cross-linking agent) did not show any patterns of cell-type selectivity. The results suggested that the mechanism of action for PAE60 may be predisposed to certain cancer types, suggestive of receptor-based activity in lieu of direct cytotoxicity.
With use of C-DDP as a benchmark for sensitivity to treatment, the lowest relative potency IC50 ratio (indicative of the highest relative potency) was observed against nasopharyngeal carcinoma CNE cells (ie, PAE60/C-DDP = 6.0). The susceptibility to PAE60 according to relative potency exhibited the following sequence: CNE > A549 > K562 > KB cells. Three of the top 4 relative potency ratios were in epithelial-type cell lines (IC50 ratio <10), whereas the BGC823 gastric carcinoma cells with the lowest absolute IC50 produced a very weak IC50 ratio (>40) and was second from the bottom. The results highlight a hypersensitivity of BGC823 to broad-spectrum treatment not necessarily specific susceptibility of BGC823 cells to the mechanism of action of PAE60. The data overall suggest that the HL60 and K562 leukemic cell lines were generally most susceptible to PAE60.
In Vivo Chemotherapeutic Evaluation of PAE60
Abnormal manifestations of the test animals were not observed during the acute toxicity experiment, which was anticipated because of an established history of P americana as a dietary delicacy. 53 The medical utility of cockroaches has also reached clinical trials in the United States to evaluate the safety (and efficacy) of cockroach extract for sublingual immunotherapy in allergies and asthma. 54 Although acute toxicity was not expected, the potential or likelihood for adverse effects following chronic use and/or anaphylactic-like reactions remains, due in part to cockroach antigens being known to induce mammalian immune-cell hypersensitivities.55,56 The estimated LD50 was >2000 mg kgbw−1, and thus the in vivo therapeutic index of PAE 60 in normal ICR mice (2000 mg kgbw−1/500 mg kgbw−1) was comparable to an in vitro SI using Vero cell toxicity (CC50; Table 3) against most cancer cell lines in this study (ie, CC50/IC50 > 4). Although the in vivo results showed positive inhibition of S180 tumors, the inhibition was not dose dependent. PAE60 at 1500 mg kgbw−1 d−1 showed 53.07% inhibition relative to vehicle controls, but 500 mg kgbw−1 d−1 was more effective at 72.62% inhibition. Reduced chemotherapeutic-type effects seen at higher doses have been reported in the literature. For example, curcumin from the turmeric plant (Curcuma longa) reduces levels of glial fibrillary acidic protein, known to be immunoreactive with tumors, only at low doses. 57 In tumor-bearing mice, 7.5 mg kgbw−1 of phycobiliprotein from cyanobacteria inhibits H-22 tumors by 71.1% whereas 15.0 mg kgbw−1 causes 51.1% inhibition. 58
Several hypotheses may account for the observed inverted dose-response effects of PAE60. Both carcinogenesis and tumor inhibition are known to be multistage, multimechanism processes composed of different phases, and effective cancer chemotherapy is tightly associated with and favored by mechanistic optimization; that is, anti-angiogenesis, immunoregulation and direct cytotoxicity.59,60 Neither low- nor high-dose PAE60 appeared to stifle blood supply or reduce the proliferation of new blood vessels, thus PAE60 is not likely anti-angiogenic. Cockroach extracts fractionated for cytotoxicity could easily have multiple pharmacologically active components. It is plausible that the reduced response at the higher dose of PAE60 (as with other natural products) might be attributable to counteracting factors inherent in the diversity of compounds existing in the fraction. It is also feasible that the oral bioavailability of certain antitumor components might be reduced at higher doses relative to potential tumor-promoting constituents. Various factors could potentially counteract each other at higher doses of PAE60, resulting in reduced tumor suppression 1500 mg kgbw−1 compared with the low-dose group (though the difference is not statistically significant). The reduced effect seen with high-dose PAE60 could be indicative of a dose-sensitive tumor-stimulant property in a component of PAE60. For example, it was reported that BALB/c mice and human bronchial epithelial 16HBE140 cells treated with crude cockroach extracts have marked increases in tumor necrosis factor and interleukin-8 gene expressions respectively61,62 and are known tumor promoters.63-65 Likewise, various peptides are reported to possess cytotoxic capabilities against tumor cells;66,67 however, common hydrolysis into amino acids provides precursors for protein synthesis, which may promote tumor growth.68,69 Furthermore, fatty acids present in PAE may have the potential to inhibit tumor growth70,71 whereas higher levels of fatty acid intake are associated with reduced immune cell functions.72,73 The range of potential factors involved remind us that the tumoricidal process in vivo (eg, from anti-angiogenesis to immune response) is a complex and comprehensive interplay of physiologic and biochemical systems.
Inverted chemotherapeutic dose–response relationships in vivo (not corroborated by in vitro findings) suggest the mechanism of action may involve immunoregulation (with potential positive and/or negative feedback loops). For example, it was previously reported that 20 mg kgbw−1 of cyclophosphamide inhibits murine hepatoma MH129 tumors in immunocompetent but not immunodeficient mice, whereas high doses of 200 mg kgbw−1 are effective in both mice. It was determined that cyclophosphamide (a broad-spectrum apoptosis-inducing agent) is suppressive on the immune function of tumor-promoting T-cells (and splenocytes) at low doses, yet exerts a cytotoxic-mediated effect on tumors at high doses. 74 Alternatively, tumor progression in studied mouse models could be diverted by a paradoxical shift in immune response between primary adaptive and innate immune cell activation; this could trigger opposing antitumor or tumor-promoting reactions. 75
Thymus and spleen indices were obtained in this study to examine the immune function integrity of the animals in response to PAE60. The CTX-treated group presented significantly lower lymphoid organ weights than the CMC-Na-treated group, indicative of an immunosuppressive effect of cyclophosphamide in our experiments. The spleen and thymus indices in high- and low-dose PAE60 groups remained normal, thus PAE60 confers less toxicity on the thymus and spleen than cyclophosphamide. This lack of an immunosuppressant effect by PAE60 in light of limited visible tissue death or anti-angiogenic processes raises the likelihood that PAE60 operates through immunoregulation. 76 An overall mechanistic hypothesis is presented in Figure 8. It is unknown whether PAE60 follows any known tumor inhibitory pathways, and further mechanistic studies (eg, apoptosis and cell-signaling) on PAE60 are encouraged. 77

Hypothetical dose–response curves for individual components in PAE60 and the potential net effect: (curve a) tumor inhibition curve of PAE60 toward S180 sarcoma; (curve b) tumor promotion curve of PAE60 toward S180 sarcoma; and (curve c) real expressed compromised status of S180 tumor response
In Vitro Antiherpetic Activity
The antiherpetic potency of PAE60 in vitro was interestingly biased toward HSV-2 versus HSV-1. Although animals and plants are known to have potent and sophisticated antiviral and immune defense systems, antiviral compounds or extracts from other natural sources such as insects have not been extensively reported. 21 Antiviral extracts have been isolated from arthropods such as the moth, Heliothis virescens 78 and, more recently, the “killer caterpillar,” Lonomia obliqua. 79 Compounds from the firefly Photinus pyralis were reported to suppress HSV-1 plaques completely at 300 ng mL−1,80,81 and it has since become a source of mechanistically investigating modes of HSV infection. Since the total number of people aged 15 to 49 years living with HSV-2 infection worldwide in 2003 was estimated to be 536 million, 82 the opportunity exists for expanded research and development of PAE60 in antiviral chemotherapy.
Conclusions and Future Perspectives
PAE60 obtained through BGF of P americana caused >70% reductions in human carcinoma cell growth and S180 tumors and low toxicity in Vero cells. These results may raise the likelihood that the PAE60 comes into focus as an alternative or complementary medicine. Additionally, the wide-spectrum chemotherapeutic efficacy (ie, antitumor and antiviral properties) demonstrated by the 60% ethanolic fraction of P americana may succeed in drawing international attention and the interest of pharmacologists and biochemists, in addition to CAM researchers. Further investigations of PAE60 are necessary, such as the identification of active chemical structures and their relevant pharmacological assays, and are in progress.
Footnotes
Acknowledgements
We thank Dr Walee Chamulitrat (University of Heidelberg) and Professor Christopher H. K. Cheng (Chinese University of Hong Kong) for their critical reading of this article.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work is in part supported by the National Natural Science Foundation ((NNSF30560181 & 30860337), Yunnan Province Project (Lead high-level personnel training project No. 2009CI121) and by Key Programme of TCM Modernization of Yunnan Province (MOST 2008IF012).
