Abstract
Objective
We estimated the conversion from cognitively normal to mild cognitive impairment (MCI) to probable dementia and death for underweight, normal, overweight, and obese older adults, where the timing of examinations is associated with the severity of dementia.
Methods
We analyzed six waves of the National Health and Aging Trends Study (NHATS). Body mass (BMI) was computed from height and weight. Multi-state survival models (MSMs) examined misclassification probability, time-to-event ratios, and cognitive decline.
Results
Participants (n = 6078) were 77 years old, 62% had overweight and/or obese BMI. After adjusting for the effects of cardiometabolic factors, age, sex, and race, obesity was protective against developing dementia (aHR=.44; 95%CI [.29-.67]) and dementia-related mortality (aHR=.63; 95%CI [.42-.95]).
Discussion
We found a negative relationship between obesity and dementia and dementia-related mortality, a finding that has been underreported in the literature. The continuing obesity epidemic might complicate the diagnosis and treatment of dementia.
Introduction
Alzheimer’s disease and related dementias (ADRD) are caused by a neuropathological cascade and are evident when cognitive decline impairs the ability to complete everyday tasks. 1 Since the disease has a long clinical course marked by progressive losses of brain matter, ADRD often requires substantial investment on the part of caregivers and increases the risk of mortality. Obesity is one possible modifiable correlate of dementia that is currently under-explored. Underweight, overweight, and obesity have been related to all-cause mortality risk and various poorer health outcomes, 2 but their impact on the risk of dementia remains a topic of some debate. For example, obesity has repeatedly been linked to the prevalence of dementia, with a higher midlife BMI proportionately 3 and consistently found to increase the risk of ADRD, 4 potentially indicating that metabolic alterations associated with obesity can harm the brain by causing apoptosis or necrosis. 5
A recently meta-analysis (n = 16,282) reported a 1.4-fold increased risk of dementia in obese older adults. 6 Yet, in a study of 14 different risk factors associated with cognitive impairment, in a lower risk of ADRD, 7 a result that was replicated in a more extensive study. 8 In light of these lingering questions, we seek to reconcile the discrepancies in previous findings and examine the cognitive trajectories of underweight, normal, overweight, and obese older adults. In the present study, we provide evidence of whether obese body mass is or is not a risk factor for dementia and dementia-related mortality. Therefore, obesity may or may not increase the risk of accelerated cognitive aging trajectories.
Literature Review
Obesity, often instrumentalized as body mass index (BMI) ≥ 30 kg/m2, is very common in the US. When severe, obesity is associated with an increased risk of insulin resistance, metabolic syndrome, and cardiovascular disease (CVD), which have also been shown to be risk factors for ADRD. 9 Previous statistical models of dementia did not consistently include these factors, like cardiovascular disease, type II diabetes, and vigorous physical activity that confound or mediate the relationship between BMI and dementia. Although some of these factors are implicated in the causal pathway between obesity and dementia, residual confounding may not be excluded. The lack of adjustment for level of education, or diabetes, which are strongly associated with BMI and dementia, likely contributed to the heterogeneity of previous findings. Additional co-morbid risks of ADRD and obesity include sex and race/ethnicity. Further, obesity and race/ethnicity are missing variables in the meta-analyses mentioned above. The exclusion of education and race/ethnicity variables is important to note because the prevalence of obesity is much higher in areas with lower educational attainment in the US. 10 Although some of these factors are implicated in the causal pathway between obesity and dementia; residual confounding may not be excluded. The lack of adjustment for level of education, or diabetes, which are strongly associated with BMI and dementia, likely contributed to the heterogeneity of previous findings.
Additional co-morbid risks of ADRD and obesity include sex and race/ethnicity. For example, obesity and race/ethnicity are missing variables in the meta-analyses mentioned above. However, BMI has long been recognized as a poor measure of adiposity and overall health, 11 and findings linking obesity and ADRD are not always clear. 12 In what has been termed the “obesity paradox,” those who are underweight also have greater all-cause mortality than those in the normal range, and below-normal BMI is a risk factor for ADRD. 13 These are not the only paradoxical finding in the literature: despite reports of overweight BMI in midlife being a risk factor for ADRD in late-life, 14 the cognitively protective benefits of being overweight in late-life have also been identified, and late-life weight loss can precede accelerated cognitive decline. These results counter the assumed linear relationship between BMI and late-life cognition, but further investigation is still needed to assess the impact of BMI on the dementing process from cognitively normal to mild cognitive impairment (MCI), to dementia, to dementia-related mortality. 15
Objective
In existing studies, these obesity paradoxes and limitations lack a consistent relationship between higher body mass and the onset of MCI and conversions to dementia. However, they also did not account for the potential that cognitive statuses vary by obesity status. Previous studies have limitations such as small community samples, relying on electronic health records, use of institutionalized samples of weight loss in nursing home settings, lack of racial or ethnic diversity, only focus on one transition from cognitively normal to MCI, or on those who convert from MCI to dementia. The obesity paradoxes and limitations demonstrate an ongoing need for additional study and a need to reconsider the relationship between higher body mass and the onset of MCI and dementia after accounting for the potential that misclassification rates vary by obesity status.
In response to these limitations, we aimed to estimate the hazards of conversion from normal cognition to mild cognitive impairment to probable dementia and death for underweight, normal, overweight, and obese older adults in an aging cohort where the timing of examinations may be associated with the severity of dementia. Persons with MCI may become impaired and later revert to normal or may stay mildly impaired for a long time without converting to dementia. Others may progress from cognitively normal to MCI and decline further into dementia (Figure 1). Using multi-state modeling, which uniquely allows us to account for misclassification errors, we help to rectify the limitations of other methodologies and estimate the probability that people might be misclassified as having MCI despite being normal. Four-State Cognitive Model with One Absorbing State.
76

Methods
Sample
The National Health and Aging Trends Study (NHATS) is an aging and health longitudinal survey of a nationally representative cohort of Americans 65 years and older. The NHATS has comprehensive sociodemographic, cognition, and self-report measures using validated criteria and is publicly available online https://www.nhats.org/) (NHATS). We utilized data from six waves (2011-2016) of the first cohort to present the incidence and conversion rates of older adults (N = 6078), with particular attention to the role of BMI on cognitive decline and eventual mortality. The NHATS study collects information on individuals living in communities and residential care settings, not from nursing homes. It allows proxy respondents to provide information for respondents with severe cognitive, speech, or hearing problems. The initial sample was recruited in 2011 and followed annually to 2016. Replenishment of the sample to maintain representation for the older Medicare population was conducted in 2015. In order to assess participants over a relatively longer observation, our analytic sample only targeted the first cohort enrolled in 2011, 16 excluding the refreshment sample beginning in 2015. After excluding participants who had merely one wave of cognitive status, we analyzed the impact of obesity on conversion from cognitively normal to MCI, then dementia, and eventually, death. IRB approval was granted before data analysis with a waiver of informed consent for secondary data analysis.
Measures
Cognitive Functioning
We classified our sample into probable dementia, MCI, or cognitively normal, with reasonably good sensitivity and specificity. 17 Participants were classified into mutually exclusive cognitive statuses: normal cognition, MCI, probable dementia, and death. Normal cognition included participants within one standard deviation from the mean on all cognitive functioning measures (tests of memory, orientation, and executive function). Memory was measured using a 10-word recall and delayed 10-word recall scores. Orientation was measured by identifying specific information such as the current date, President, or Vice President of the US Executive function was measured using the clock drawing test. The AD8 Dementia Screening Interview 18 assessed the memory, orientation, judgment, and function of the sample person reported by proxy respondents and provided a single measurement score. AD8 scores were available, including those who could be asked cognitive tests and judged by proxy, were also available. Following existing standards,19,20 MCI was defined by a score of ≤1.5 standard deviations below the mean on at least one of the three cognitive functioning enrollment domains: cognitive tests of memory, orientation, and executive function. Dementia was either self-indicated, proxy-reported physician’s diagnosis; AD8 scores ≥2; or scores ≤1.5 standard deviations below the mean on at least two of three cognitive functioning domains tested.
Covariates
Body Mass Index (BMI) was self-reported and is a ratio between weight and height in kg/m2 and was categorized into groups: underweight (below 18.5), normal (18.6-24.9), overweight (25-29.9), and obese (30 or greater) according to NIH classification and was measured at baseline. Diabetes status was diagnosed as diabetes status (yes/no.). Participants were asked to “please tell me if a doctor ever told you that you had diabetes?” For Vigorous Activities, participants were asked whether they spent time on vigorous activities that increased heart rate and made breathing harder in the last month, including working out, swimming, running, biking, or playing a sport. Cardiovascular disease status was whether the participant had a diagnosis of CVD (yes/no.). Participants were asked to “please tell me if a doctor ever told you that you had heart disease?” The age of the respondent was recorded at the initial interview. Sex was either male or female, with males as the reference group. Race/ethnicity included four categories: non-Hispanic whites, Black race, Other race, and Hispanic ethnicity. Education was categorized into three levels: less than high school, high school graduate, and college graduate, with having a high school diploma as the reference group.
Time
Time is the time interval in months between the starting point of the first survey and follow-ups. Informants reported death date during the follow-up, annual interview. Baseline age was included as a covariate to separate the effects of age and time. The interim date between the last and subsequent follow-up surveys was used for respondents who were lost to follow-up but did not have death data. We used the most frequent month of that year’s survey for missing values of the survey. We estimated the median month of the expiration year if there was a known death but were missing an accurate death date. We used a right-censoring event if the most recent cognitive status was missing and a death date.
Data Analyses
In examining time-to-event ratios and cognitive transitions, we used multi-state survival models (MSMs)21,22 and calculated the probability of diagnosis misclassification. An example of misclassification would be classifying a cognitively normal participant as having MCI. In prior cognitive research, estimating misclassification errors have been used to assess and rectify measurement errors. 23 In light of incomplete data, several strategies to loosen Markov chain assumptions have been proposed.24,25 Hidden and partially-hidden Markov models infer true states from observed data, the latter relying on auxiliary data that was collected in addition to the primary outcomes of interest (in this case, cognition).24,26
Following Sweeting et al.,
26
misclassification (matrix E) is an n x n matrix whose (r,s) function is:
Results
Sample Demographics
Sample characteristics (N = 6078), National Health and Aging Trends Study 2011-2016.

Effects of Body Mass Index on Transitions Between Cognitive States
Unadjusted Multi-State Models estimating the incidence of Mild Cognitive Impairment, Dementia, and Death, National Health, and Aging Trends Study 2011-2016.

Adjusted hazards ratios provided by Multi-State Models estimating the incidence of Mild Cognitive Impairment, conversion to dementia, and mortality in the National Health and Aging Trends Study 2011-2016.
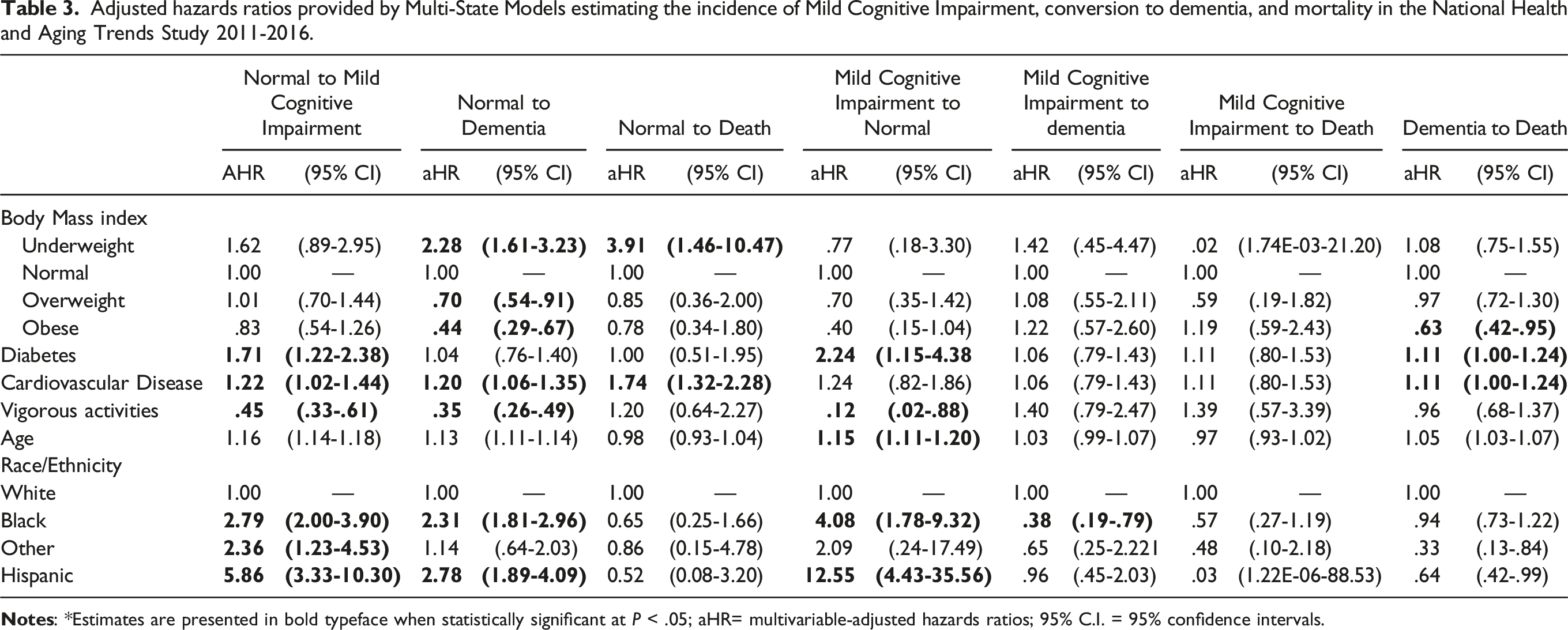
Both overweight and obese adults had a reduced risk of transitioning from cognitively normal to MCI and normal to dementia. Overweight adults had a reduced risk of transitioning to MCI by 33% (95%CI [14.2%-49.6%]) and dementia by 48% (95%CI [34.8%-58.1%]). Overweight BMI also showed a protective effect against mortality following an MCI state by 75%. Obese older adults had a reduced risk of transitioning from cognitively normal to MCI (HR=.55; 95%CI [.399-.753]) and normal to dementia (HR=.35; 95%CI [.26-.45]) also. Having an obese BMI was protective against transitioning from dementia to death (HR=.59; 95%CI [.39-.89]). Overall, obese, or overweight BMI was protective against transitions from cognitively normal to MCI and normal to dementia. At the same time, underweight adults were at increased risk of transitioning from cognitively normal to MCI and normal to dementia. After adjusting for the effects of diabetes, CVD, vigorous physical activity, age, and race/ethnicity, the protective effect of obesity in late-life against developing dementia (aHR=.44; 95%CI [.29-.67]) and dying from dementia (aHR=.63; 95%CI [.42-.95).
Having a diagnosis of CVD puts older adults at significantly increased hazards of converting to MCI (aHR=1.22; 95%CI [1.02-1.44]) and dementia (aHR=1.20; 95%CI [1.06-1.35]), as well as increased mortality hazard from all causes (aHR=1.74; 95%CI [1.32-2.28]). Transitions from cognitively normal to MCI were increased for Blacks (aHR=2.79; 95%CI [2.00-3.90]), Asian/Others (aHR=2.36; 95%CI [1.23-4.53]), and Hispanics (aHR=5.86; 95%CI [3.33-10.30])) yet faced reduced hazards of converting from MCI to dementia and significant for Blacks only (aHR=.38; 95%CI [.19-.79]). Older adults with diabetes were at a 71% increased risk of developing MCI (aHR=1.71; 95%CI [1.22-2.38]) and an 11% increased hazard of death from MCI (aHR=1.11 95%CI [1.001-1.24). Hispanics had 67% (16-87%) reduced mortality hazard from MCI to dementia, while Asian/Others had 36% (1-58%) increased hazard of converting to dementia.
Reversion from MCI to normal was increased up to four times for those with diabetes, older age (11-20%), up to nine times increased reversion for older Black adults, and 12.5 times increased for Hispanic participants. Vigorous activities reduced the hazard of reverting to cognitively normal by −88% (AHR=.12; 95%CI [.02-.88]).
Misclassification Probabilities of Adjusted Model
Model-Estimated Misclassification Probabilities for Cognitive Transitions.

Notes: All estimates were statistically significant at P < .05; 95% C.I.= 95% confidence intervals.
Discussion
We estimated hazard ratios of transition from cognitively normal to MCI, dementia, and dementia-related mortality in the US in underweight, normal, overweight, and obese older adults. Our results show the independent effect of underweight BMI on cognition apart from later-life co-morbidities, showing that underweight older adults are at increased risk of MCI and Dementia from a normal state. We also found that having an overweight or obese BMI in later life is protective against some complications of advanced cognitive aging, including MCI and dementia, and death. These findings are novel and report transitions across cognitive states and death in a longitudinal sampling of underweight to obese older adults.
Late-Life Obesity is Protective Against Cognitive Decline and Death
Elevated BMI was associated with a lower risk of cognitive decline and death when measured in late-life. The association between obese BMI and dementia persisted independently after accounting for aging effects, including aging-related diseases like CVD and diabetes. This is consistent with prior obesity-paradox literature that shows a complex association between BMI and cognition. 28 However, elevated BMI in midlife is typically associated with increased mortality and increased risk of cognitive decline. In the Whitehall II study, obesity (BMI≥30 kg/m2) at age 50 was associated with ∼2 times increased hazards of dementia later. The trajectories of participants' BMI differed between those with dementia and healthy controls. BMI decreases start eight years before an ADRD diagnosis using a case-control design. 29 However, elevated BMI in midlife is typically associated with increased mortality and increased risk of cognitive decline. decreases start ADRD. 29
Our results confirm that and provide robust estimates of the hazard of transitioning between cognitive states and death, with the possibility of reversion. However, this study was limited in under evaluation of ADRD cases as the data came from electronic health records and did not calculate misclassification, a strength of our study. Advances in cardiovascular health and public education have improved over the past 20 years, and it stands to reason that these improvements level the rising incidence of ADRD in the US. However, since 2013, increasing rates of obesity at earlier ages—thus increasing the number of potential dementia cases by 2050, growing to an estimated 13.8 million 30 to 152 million around the world. 31 Effectively, this creates a significant burden on women and minorities at increased risk of developing ADRD. 31
In prior research, the risk of dementia associated with obesity is either attenuated or reversed. 32 This attenuation is likely due to preclinical changes in weight in the years preceding the clinical onset of dementia, 33 estimated to result in a 10% loss of total body weight, 34 and observed in previous studies for more than a period of 2 to 4 years, five years, or up to 10 years 35 before a diagnosis of dementia. The exact mechanisms explaining the increased risk of dementia associated with obesity are poorly understood. On the one hand, obesity in midlife and at older ages is associated with brain atrophy. 36 On the other hand, obesity in late-life is a sign of possible resources available to the brain as people age and may be a proxy for better health after a mean age of 74 years old, as frailty becomes a threat. Potential avenues of further research include relating obese BMI to brain atrophy or preservation of mass, obese BMI to disruptions in cerebrovascular function, obese BMI and the development of amyloid pathology, obese BMI, and the breakdown in the blood-brain barrier (BBB), and systemic and neuroinflammation. At the same time, considerable improvements in CVD health and increased educational attainment per person over the second half of the last century are reasoned to be responsible for the leveling of the incidence rates of dementia.
Underweight Older Adults Face Increase Risk of Dementia and All-Cause Mortality
Our results also show increased conversion to dementia and all-cause mortality for underweight participants. Many people with dementia lose weight in the later stages. As we age, it is expected that people with ADRD lose 1.2 pounds of body weight per year compared to a loss of .6 pounds per year after age 65 in cognitively unimpaired adults. 37 This rapid weight loss is associated with faster disease progression and cognitive decline. 38 Late-life weight loss is attributed to reduced appetite, 39 increased physical movements and pacing, anorexia, 40 and increased metabolic demands in people with dementia. Several studies suggest that a lower late-life BMI is associated with an increased risk of incident dementia.41,42 However, other studies have suggested that late-life obesity is associated with smaller brain volumes (a marker of neurodegeneration and Alzheimer’s disease pathology) in individuals with MCI. 43 Increased BMI may be associated with an increased risk, and lower late-life BMI does not increase the risk of clinical dementia. 44
Our findings support prior works that show that unintentional weight loss at any age may indicate neurological disease and increased all-cause mortality. In the late stages of ADRD, researchers have observed patients losing two pounds of body weight per year, weighing less than their normal counterparts, and losing weight faster than healthy controls.45,46 Weight loss in late life is also related to small multiplicative effects of age, sex, metabolic health, and hypertension. Interestingly, other health risks related to BMI changes and dementia—depressive symptoms, marital status, anorectic medications, APOE ε4 expression, diabetes mellitus, and appetite change—were not related to weight loss and dementia. In previous studies, cardiovascular factors, such as myocardial infarction, transient ischemic attacks, stroke history, smoking, and hypertension, seem unrelated to weight loss and dementia. 46 Still, specific factors contributing to weight loss associated with cognitive decline are unclear.
Dual Processes Regarding Weight Loss Associated with Cognitive Decline
The prevailing hypothesis is that the apparent correlation between BMI and dementia is due to two processes: one, a direct connection between higher BMI and increased dementia risk, and two, a relationship confounded by weight loss during the prodromal phase. One hypothesis is that the apparent correlation between BMI and dementia is due to two processes: one, a direct connection between higher BMI and increased dementia risk, and two, a relationship confounded by weight loss during the prodromal dementia stage, making a harmful exposure appear protective via reverse causation. Supporting this hypothesis is the fact that clinical diagnosis of dementia is often preceded by a prolonged (20-30 years) prodromal phase, during which cardiometabolic abnormalities, including weight loss, are prevalent. 47 This results in a decreased BMI around the beginning of an observed dementia diagnosis. The studies supporting the two-process hypothesis are not generalizable to all US older adults and were limited by small sample sizes (N < 3000). These limitations make the estimates from prior studies susceptible to random error. Their lack of addressing the misclassification of participants in either models, or the data itself, biases their estimates toward underdiagnosis. Another limitation is that these studies did not attempt to define the etiological phase at the time of exposure measurement by stratifying the results by the length of time between the BMI assessment and the development of dementia. In response to these significant limitations, we utilized MSM models that a) used an extensive representative sample of older adults; b) we accounted for misclassification errors, thus addressing underestimation bias; c) we simultaneously estimated the conversions from MCI to dementia and dementia-related death, including lengths of time between cognitive states during the dementing process.
Our findings add to the current literature and model the dementing process and time spent in cognitive states before declining to dementia and, eventually, death. We found that having an underweight BMI in later life is associated with an increased risk of converting to MCI, dementia, and death, apart from the confounding effects of diabetes and CVD. Our results support that underweight older adults are at increased risk of cognitive impairment, and dramatic changes in BMI may be a useful clinical biomarker of prodromal ADRD. Interventions aimed at monitoring changes in BMI as we age into retirement are quick, non-invasive, and cost-effective measurements to monitor conversion to MCI before the onset of dementia. It would be helpful to characterize subtypes of dementias to determine if weight loss is ADRD specific or more general to other types of late-life brain disease.
Preventative Interventions Targeted at Cognitive Impairment
Cognitive impairment, dementia, and Alzheimer’s disease can be ameliorated with lifestyle changes. 48 As we stated earlier, observational studies have found several potentially modifiable risk factors for dementia, including hypertension, dyslipidemia, obesity in middle age, diabetes, smoking, physical inactivity, depression, and a lack of education. Randomized clinical studies are required to investigate whether therapies targeted at these risk factors may lower the incidence of cognitive decline and dementia in older adults. However, such trials are difficult to conduct due to cost, time, and methodological issues. However, to date, most preventative interventions have been studied in small community samples, focused on a single lifestyle component, and had null or modest outcomes. Given the complex nature of dementia and late-onset ADRD, multidomain therapies that target multiple risk factors and pathways may be required for optimum prevention to support effective lifestyle intervention techniques for various age groups and people with varying risk profiles. We agree with scholars that future research should concentrate on identifying successful and sustainable solutions. We also concur with scholars that future research should concentrate on identifying successful and sustainable in many geographic, economic, and cultural situations.
Nevertheless, to date, only three multidomain intervention trials have been completed. The primary results from two trials did not show a statistically significant benefit of preventive interventions. The other trial showed modest effects of the preventative interventions. Still, the most important factors influencing the onset of ADRD are often age-related, including 1) changes in genes associated with hypothalamic-pituitary-adrenal function, 2) perturbation of ribosomal and oxidative phosphorylation pathways, and 3) changes in insulin production. 49 The degree of lifelong stress or allostatic load mediates the magnitude of this aging impact, which starts early in life. 50 The brain is the ultimate mediator of stress-related death through. The brain is the ultimate mediator of stress-related death through hormonal changes that result in hypertension, glucose intolerance, CVD, and immunological issues. 51
Future Directions
Perhaps the best future direction regarding obesity and dementia incidence is developing targeted interventions toward reducing the incidence of T2 diabetes and vascular disease. Several studies have shown correlations between ADRD and CVD. 52 Normal brain function depends on the integrity of the vascular system. Age-related shifts in the vasculature result in autoregulation impairment, neovascular coupling, blood-brain barrier (BBB) leakage, and lower vascular tone. 53 These progressions may occur prior to cognitive impairment and, when combined with other variables, contribute to the neuropathology of ADRD. These progressions may occur prior to cognitive impairment and, when combined with other variables, contribute to the neuropathology of ADRD. While various vascular risk factors have been linked to the observed neuropsychology of ADRD, no single vascular risk factor has been shown to predict changes in any specific neuropsychological aspect. 54
Developing pharmaceutical H3 receptor antagonists is a plausible molecular approach to intervene in the pathophysiology of obesity and dementias, along with co-morbid sleep disorders and psychiatric symptoms. 55 Such antagonists are being researched for the treatment of several illnesses affecting 1) cognition disorders, such as Alzheimer’s disease, ADHD, and schizophrenia), and 2) sleep disorders, such as hypersomnia and narcolepsy), as well as energy homeostasis (e.g., obesity). Several pharmaceutical firms have concentrated their drug discovery efforts on the preclinical development of H3 receptor antagonists to treat attention deficit hyperactivity disorder, dementia, schizophrenia, obesity, and sleep disorders. 56 GSK-189254 (compound 12) is another newly disclosed chemical intended to treat dementia. 57 The GSK-189254 molecule is 1) highly selective and potent toward H3 receptors, 2) has good rodent and primate pharmacokinetic characteristics, and 3) is effective in dementia using several animal models. 58
Additionally, dementia is linked to several markers of microvascular endothelial dysfunction. First, dementia is more common in those with congestive heart failure, 59 and cerebral ischemia and stroke cause hypoxia, amyloid-beta deposition, and BBB dysfunction, leading to neurodegeneration. 60 Second, CVD may cause amyloid-beta deposition and influence the age at which ADRD develops, whereas amyloid-beta can cause cardiovascular deterioration. Third, increased amyloid-beta deposition has also been linked to small infarcts and microbleeds, which often go undiagnosed, thus providing methodological challenges. 61 Fourth, blood vessels in ADRD patients show degenerative alterations, amyloid-beta deposition, and loss of smooth muscle cells. 62 Last, blood vessels with collapsed and deteriorated endothelia are seen in more than 90% of ADRD patients and co-occur with amyloid-beta deposition.63-65
Strengths and Limitations
Our study is novel in using a longitudinal aging sample in the US with Markov modeling to explore the effects of underweight and obese BMI on the transition from cognitively normal to MCI, dementia, and death. Previous studies commonly used approaches are Cox proportional survival and regression models, but multiple barriers exist.66,67 Previous studies have limitations such as small community samples, rely on electronic health records, use institutionalized samples of weight loss in nursing home settings, lack ethnoracial diversity. Differently, our MSMs followed nonhomogeneous Markov processes to simulate the dementing processes, which solved the potential errors of observed time and missing values of cognitive state. We also fit our MSMs with a misclassification matrix to cope with potential misclassified errors of cognitive states, a technique many studies did not include in their estimations of conversion between cognitive states. Our approach using Markov modeling the dementing process is advantageous because MSM models ensure better control for period effects (the effects of calendar time on dementia diagnosis).
There are a few limitations in our research worth noting. Firstly, the study design of NHATS included oversamples of older aged individuals and Black participants on the Medicare enrollment file and a replenishment sample in 2015. Due to the constraints of the MSM package, our analyses did not weight for the complex survey design and attrition. Therefore, our estimates are not based on a nationally representative sample but rather on an oversampling of Blacks, Hispanics, and older adults over 65 years old. Our findings are not generalizable to the wider aging US population. However, they are more insightful to the cognitive transitions among Blacks and Hispanics with obese or underweight BMI, apart from co-morbid CVD and diabetes. Second, while there may be a concern that some proxy-reported dementia diagnoses brought a misclassification, which can be a potential bias, our MSM models accounted for misclassification of cognitive scoring to reduce potential errors. Thirdly, our four-state model for cognitive states contained a direct conversion from cognitively normal to probable dementia, ignoring the transitional MCI state. Participants could die from any cognitive state, thus accounting for mortality bias. Our model failed to converge when using continuous measures of BMI, or BMI in increments of 5, 10, and 15 kg/m2 increments. The resulting estimates had too many estimates and overflowed the model. Although we only used one measure of obesity (BMI), studies 68 have shown that BMI is the most suitable assessment of the risk of dementia and death. Moreover, while some studies use additional measures of adiposity, such as waist-to-hip ratios, body fat percentage, and skin-fold thickness, 69 such data is not gathered as part of the NHATS study.
MSM models did not include adjustments to cognition based on depression that may impact transition rates from cognitively normal to MCI to dementia and death. It is still unclear how much incidence rates are likely to be affected by depression and other related neuropsychological impairments. Depression is associated with inflammation in the brain and the body. 70 Likewise, higher BMI and having diabetes are also confounding factors in the inflammatory process. While depression was not included in the models due to the lack of consistent evidence presented in the current literature and our focus on metabolic dysregulation, we assert that the inclusion of both diabetes and BMI also serves as proxy indicators for increased inflammation in the body. 71 We identified BMI and diabetes as good indicators of poor health in Black and non-Black subsamples at increased risk of dementia in late-life following an extensive review of the literature.72,73
Nevertheless, since depressive symptoms are a common co-morbidity of cognitively impairing diseases, there remains some lack of clarity about the role of depression as a risk factor versus an indicator of the presence of neuropathology.74,75 Future work is needed to determine how the characteristics of dementia and metabolic dysfunction differ between Black, White, and Hispanic respondents to clarify whether depression plays a confounding or distinct role in inequalities ascribed to minority groups in this study. Future studies should examine cognitive transitions and confounders of neuropsychiatric disease (e.g., depression, anxiety, PTSD, psychosis) associated with dementia to elucidate the relationship between mental illness as a risk factor for transitions between cognitively normal, MCI, probable dementia, and death.
Lastly, our research is limited to one cohort from one study in the United States, with a unique historical relationship to obesity. Thus, we considered that our results could have been impacted by the NHATS sample characteristics as a cohort effect. More research from various cohorts will contribute to a better understanding of the relationship between underweight BMI and conversion to dementia.
Conclusion
There is clearly a protective effect between later-life obesity, dementia, and dementia-related mortality. While the widespread failure to stem the rise of obesity in an aging population is a concern, we showed the putative protective effects of obesity in later life against cognitive decline in older age, without complications of T2 diabetes and CVD. Therefore, obese body mass, itself, is not a risk factor for dementia and dementia-related mortality. Therefore, is likely not an increased risk of an accelerated trajectory of cognitive aging. This is a novel and innovative finding in aging and dementia research. We showed a negative relationship between obesity and dementia and dementia-related mortality, which has never been consistently found in the cognitive literature. Increasing age coupled with the negative cardiometabolic risks in mid-life and diverse demographic backgrounds likely compound trajectories of cognitive impairment and should be explored in future studies
Footnotes
Authors’ Contribution
YZ and SC analyzed the data. SC and GN interpreted the findings and results tables. GN conceptualized and wrote the manuscript, revisions, and approved the final manuscript. YZ and DH provided editorial advice to improve the manuscript and approved the final manuscript. SC provided senior experience and editorial advice to improve the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this analysis was provided by the National Institute on Aging (NIH/NIA RF1 AG058595; NIH/NIH R01 AG067621). The National Health and Aging Trends Study (NHATS) is sponsored by the National Institute on Aging (NIA U01AG032947) through a cooperative agreement with the Johns Hopkins Bloomberg School of Public Health.
Ethical Approval
This study was determined by the Ethics Board at Stony Brook University to be analyses of publicly available secondary data and therefore not human subjects research.
Informed Consent
Subjects have consented to the publication of the data.
Data Availability
The datasets generated and/or analyzed during the current study are publicly available.
