Abstract
Subjective memory complaints (SMCs) are commonly noted in memory disorder clinic patients. The present study sought to examine the presence of SMCs on the Cognitive Difficulties Scale (CDS) in older adults and to examine the relationship between CDS scores and current cognitive ability. Participants were 50 adults diagnosed with possible/probable Alzheimer’s disease (AD), 100 with amnestic mild cognitive impairment (MCI) and 84 cognitively healthy controls (HCs). Participants completed a neuropsychological evaluation and the self- and informant-reported CDS. Results indicated that greater self-reported SMCs were noted in the group with MCI ; however, self-reported CDS scores were associated with cognition in HCs only. Informant-reported CDS scores were predictive of cognitive ability in the diagnosis of MCI but not AD, indicating the importance of obtaining caregiver report in the evaluation of memory disorders. As AD is a neurodegenerative disorder, SMCs lose value in determining degree of cognitive impairment as disease stage increases.
Introduction
Early identification of mild cognitive impairment (MCI) and dementia has received much research and clinical attention because of the potential to develop early treatment interventions. Age-related changes occur in the brain, and Alzheimer’s disease (AD)-related pathology can be seen even prior to diagnosis of MCI. 1 -4 Early identification of individuals at risk of cognitive decline is important in order to implement appropriate treatments early in the disease process.
Older adults frequently report subjective memory complaints (SMCs). Subjective memory complaints were included in early criteria for MCI 5 and still remain important in the diagnostic process. 6 One study found that among individuals meeting criteria for a diagnosis of MCI, only 20.6% self-reported SMCs. 7 Decreased self-awareness of cognitive and functional limitations has been shown in AD, 8 but caregiver report can provide additional important information about possible dementia. A reliable office tool is needed to aid in prediction of those with memory complaints at risk of progression to dementia. The association between SMCs and subsequent MCI/dementia is unclear. In a meta-analysis of the SMCs’ literature, Mitchell 9 found that SMCs had a sensitivity of 42.8% and a specificity of 82.1% to predict subsequent dementia. In addition, 38.2% of those with MCI reported SMCs. The presence of SMCs has been linked to cognitive decline several years following initial assessment. 10 -12 Subjective memory complaints have also been linked with biomarkers of AD pathology, including amyloid-β deposition. 13,14 However, other research has shown SMCs are not entirely predictive of cognitive decline. 15 Still, several studies have shown evidence of pathology consistent with AD among individuals with SMCs, 16 -18 indicating a need to fully assess the relationship between SMCs and AD.
Performance on objective cognitive and neuropsychological tests has been compared to SMCs, in order to determine level of agreement. Results have been mixed. Some have found correlations between subjective and objective cognitive impairment, 8,19 -22 whereas others found no relationship. 23 -25 In some of these studies, SMCs were assessed via response to a single or multiple yes/no questions regarding memory difficulties. 26 It is possible that these studies might not have fully assessed the SMCs construct in order to compare it to objective test results.
The present study examined the presence and predictive value of self- and informant-reported cognitive complaints. Older adults, with and without a diagnosed memory disorder, completed the Cognitive Difficulties Scale (CDS), a measure of perceived cognitive dysfunction 27 and a neuropsychological evaluation. Informant ratings of memory complaints and daily living functional ability were also obtained. The study aims were as follows. First, we were interested in how diagnostic groups differed on the CDS and 2 of its subscales (attention and memory). The second aim was to examine whether the CDS, either the patient report or the informant report, could be used to provide information about SMCs and to predict current level of cognitive functioning.
Method
Participants
Participants were 234 community-dwelling older adults, aged 55 to 85. All participants were English speaking, had adequate hearing and visual abilities, and no history of significant psychiatric disorder or substance abuse/dependence in the past 2 years. Fifty participants with diagnosed probable or possible AD based on National Institute of Neurological and Communicative Disorders and Stroke/Alzheimer’s Disease and Related Disorders Associationdiagnostic criteria 28 were recruited from the Rhode Island Hospital Alzheimer’s Disease and Memory Disorders Center, an outpatient diagnostic and treatment clinic. Inclusion criteria were as follows: able to bring informant to study session, stable on antidepressant and/or dementia medications prior to testing, lack of structural changes on neuroimaging that could affect cognition, no current significant depressive symptoms (as assessed with the Cornell Scale for Depression in Dementia 29 ), no other history of neurological disorders impacting cognition, and Clinical Dementia Rating (CDR 30 ) scale score of 0.5 to 1.0. Additional information regarding selection criteria can be found elsewhere. 31 Of note, the primary purpose of the study was to examine the utility of a telephone-based screening for early cognitive impairment, and the specific design of the present study was determined by this original study.
One hundred participants were diagnosed with amnestic MCI, with inclusion criteria as follows: presence of memory complaints, memory loss on cognitive tests as evidenced by scores falling 1.5 standard deviations or more below age-adjusted means on any aspect of memory tasks (ie, learning, delayed recall, and recognition), a CDR score of 0.5, and relatively preserved ADL functioning.
Eighty-four healthy control (HC) participants were recruited from family members of patients, based on the original study design. Healthy control participants had a Mini-Mental Status Examination (MMSE 32 ) score within the normal range of >26, no history of neurological condition affecting cognition, and no significant current cognitive or depressive symptoms. Healthy control participants were not required to bring an informant to the testing session due to the design of the original telephone screening study. Patient diagnoses were agreed upon by consensus conference including neurologists, neuropsychologists, and neuropathologists.
Subjective Cognitive Complaints
The CDS
27
is a 38-item self-report measure of subjective complaints regarding immediate and delayed memory, attention, language, temporal orientation, and psychomotor abilities. Participants rate their agreement with each item on a 0 (
Procedure
All participants provided informed written consent. The study was approved by the institutional review board of Rhode Island Hospital. Participants attended an initial office visit, where they completed cognitive testing with a neuropsychologist. The evaluation included administration of the CDS (patient and informant report), MMSE, Dementia Rating Scale 2 (DRS-2), 34 and the Hopkins Verbal Learning Test-Revised (HVLT-R 35 ).
Statistical Analyses
All analyses were conducted using SPSS v.19 (IBM, Armonk, New York). Group differences in demographic variables were analyzed using 1-way analysis of variance (ANOVA) and Pearson’s chi-square tests as appropriate. Significant omnibus ANOVAs were followed up with Tukey’s HSD post hoc pairwise comparisons. To address the study aims, a series of hierarchical linear regression analyses were conducted for both the patient and the informant versions of the CDS. Age and education were entered in the first step, and self-reported or informant-reported CDS total, attention, or memory score in the second step. Criterion variables included MMSE; DRS-2 total scaled score, education-corrected total scaled score, and Initiation/Perseveration and Memory subtest scaled scores (these subtests were selected as they can help distinguish AD-type pathology from other dementias); and HVLT total learning and delayed recall
Results
The study sample was primarily caucasian (84% of HC, 98% of MCI, and 94% of AD) and female (68% of HC, 57% of MCI, and 61% of AD), with no significant differences between the groups on these variables (
Group Differences in CDS Scores
Data regarding performance on neuropsychological measures and the CDS are presented in Table 1. A significant difference in patient-reported CDS total scores was found,
Study Variables Presented as Mean (Standard Deviation).
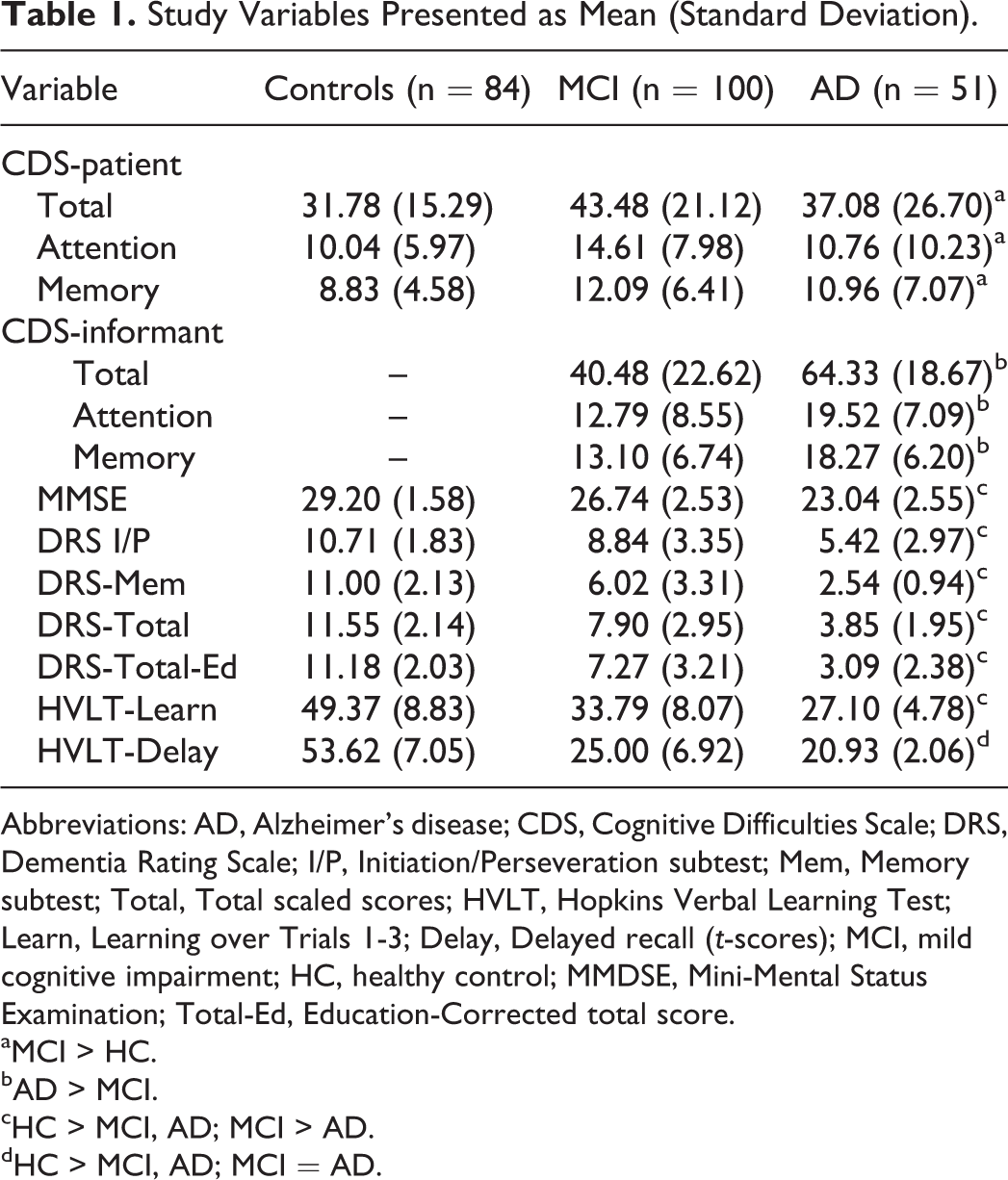
Abbreviations: AD, Alzheimer’s disease; CDS, Cognitive Difficulties Scale; DRS, Dementia Rating Scale; I/P, Initiation/Perseveration subtest; Mem, Memory subtest; Total, Total scaled scores; HVLT, Hopkins Verbal Learning Test; Learn, Learning over Trials 1-3; Delay, Delayed recall (
aMCI > HC.
bAD > MCI.
cHC > MCI, AD; MCI > AD.
dHC > MCI, AD; MCI = AD.
Analyses were also conducted on the informant-reported CDS. Informant-reported CDS total scores were higher in the AD than MCI group,
Differences in Demographics by SMCs
A series of independent-samples
Predicting Cognitive Performance From CDS Scores
Patient CDS scores
Only results with
Summary of Regression Analyses: Self-report CDS and Cognitive Task Performance.a
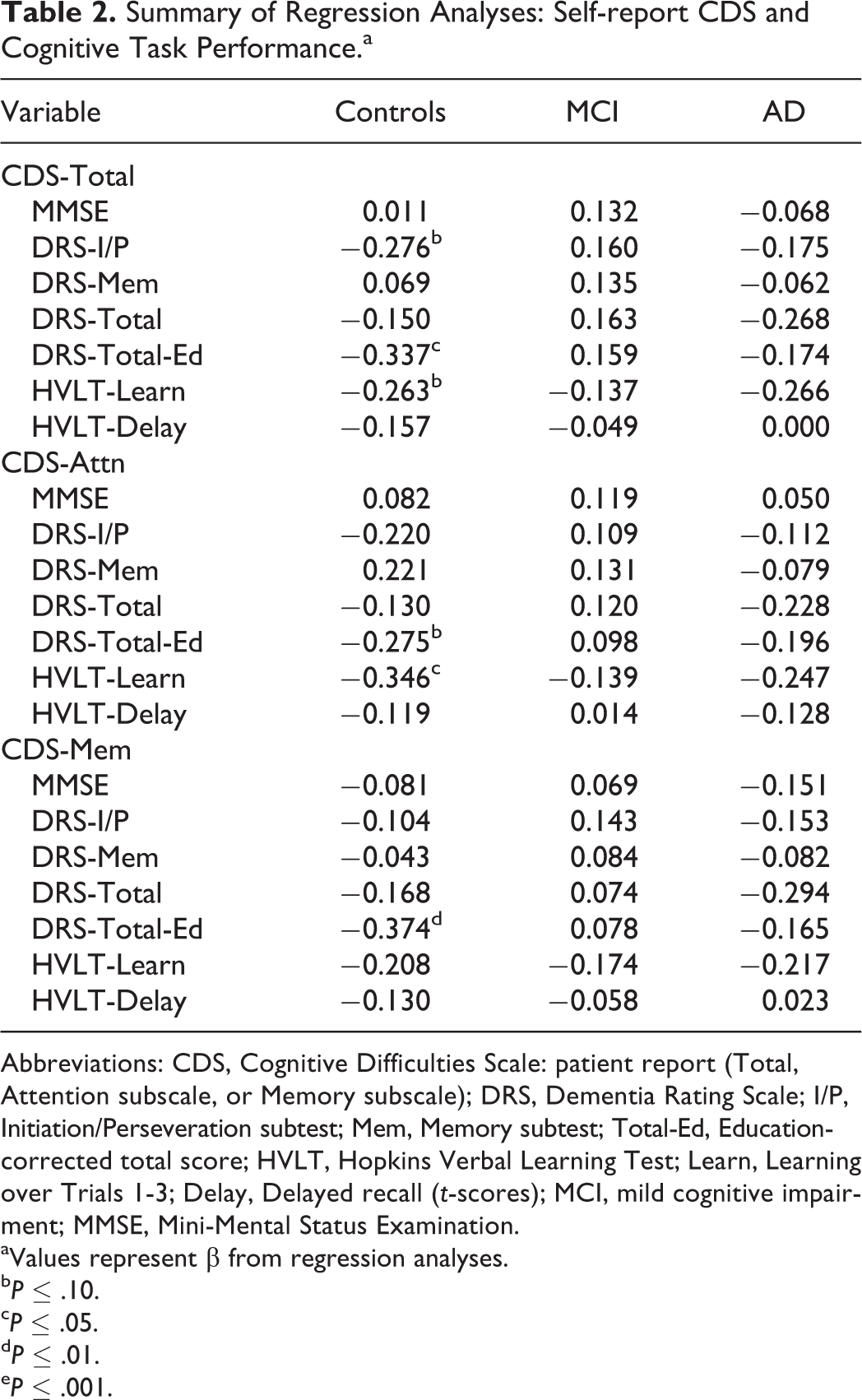
Abbreviations: CDS, Cognitive Difficulties Scale: patient report (Total, Attention subscale, or Memory subscale); DRS, Dementia Rating Scale; I/P, Initiation/Perseveration subtest; Mem, Memory subtest; Total-Ed, Education-corrected total score; HVLT, Hopkins Verbal Learning Test; Learn, Learning over Trials 1-3; Delay, Delayed recall (
aValues represent β from regression analyses.
b
c
d
e
Informant CDS scores
For the MCI group, informant-reported CDS total scores were predictive of MMSE scores,
Summary of Regression Analyses: Informant Report CDS and Cognitive Task Performance.a
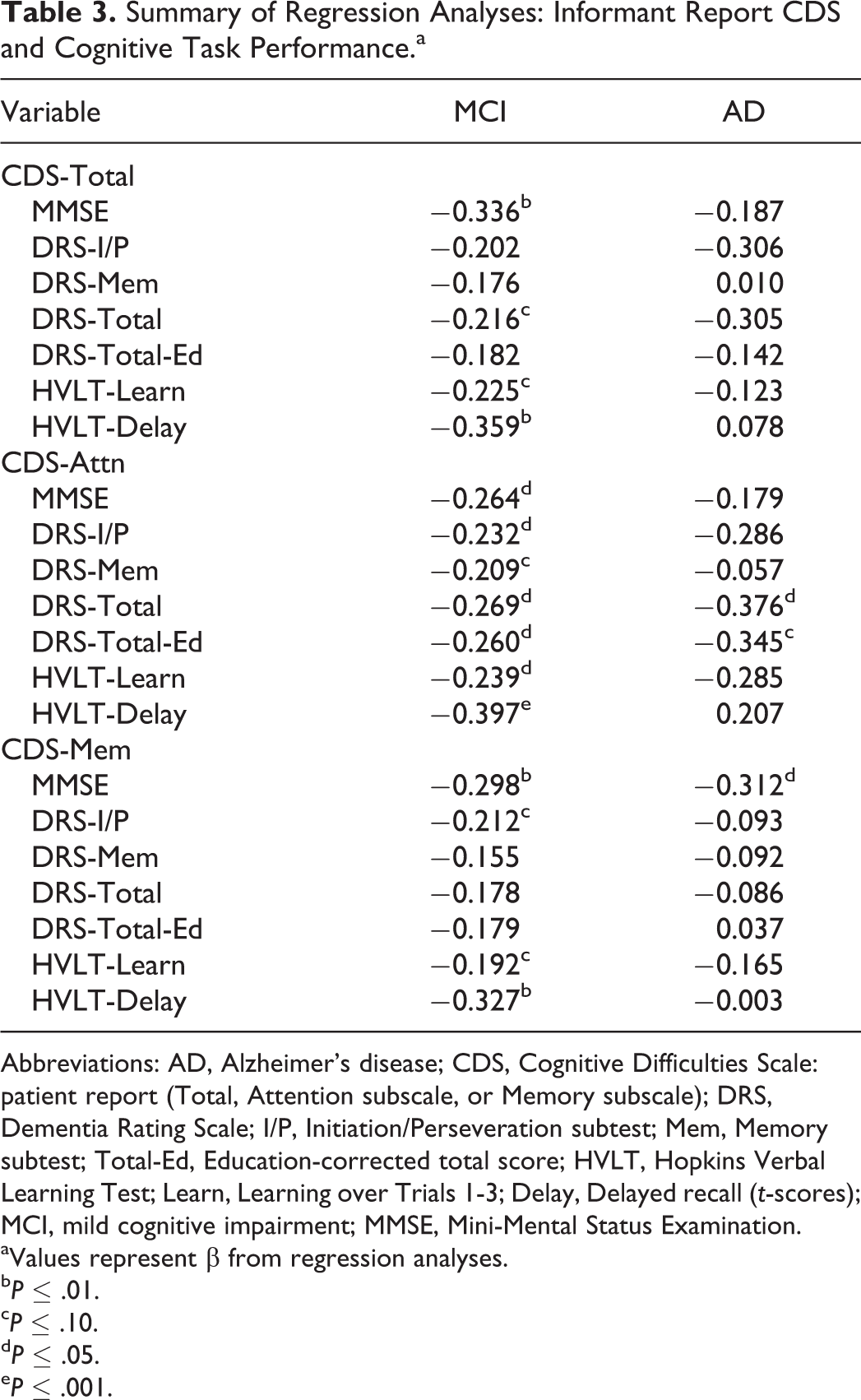
Abbreviations: AD, Alzheimer’s disease; CDS, Cognitive Difficulties Scale: patient report (Total, Attention subscale, or Memory subscale); DRS, Dementia Rating Scale; I/P, Initiation/Perseveration subtest; Mem, Memory subtest; Total-Ed, Education-corrected total score; HVLT, Hopkins Verbal Learning Test; Learn, Learning over Trials 1-3; Delay, Delayed recall (
aValues represent β from regression analyses.
b
c
d
e
For the AD group, no significant predictors emerged using the CDS total score (
Discussion
The present study sought to examine whether self- and informant-reported SMCs, assessed via CDS total and subscale scores, could provide additional information about concurrent cognitive ability. The study aims were to assess group differences on the CDS as well as the utility of the CDS to provide information about current level of cognitive function. First, we found that self-reported CDS scores were significantly higher among individuals with MCI than HCs; however, as level of impairment increased (ie, AD diagnosis), self-reported memory complaints on the CDS were no longer significant. Thus, as impairment increases, self-report ratings on the CDS are less helpful to distinguish between memory groups. However, administering the self-report CDS could help differentiate between memory complaints in HCs and those with MCI.
The second aim was to assess the utility of the CDS to provide information about cognitive function. We found that self-reported CDS scores were only predictive of concurrent neuropsychological test performance among HCs. Specifically, a high level of self-reported SMCs specific to memory was predictive of age- and education-corrected total score on the DRS-2, a commonly used screening instrument for MCI and dementia. When informant report was instead utilized, high levels of total, attention, and memory SMCs were predictive of worse MMSE scores and delayed recall on the HVLT in individuals with a diagnosis of MCI. Informant-reported SMCs were not predictive of concurrent neuropsychological performance in those with a diagnosis of AD.
These results suggest that the extent of current cognitive difficulties (HC vs MCI) and the source of information (self vs informant) may matter in the utility of the information to understand current cognitive status. Despite high levels of self-reported SMCs on the CDS in individuals with MCI, the self-report CDS did not predict concurrent cognitive ability in this group. Although some previous studies have found patient-reported level of SMCs predictive of dementia risk and diagnosis, 10 -14 others have shown no predictive utility of patient-reported SMCs. 15,23,36 Anosognosia has also been reported in MCI and AD. 8,23,37,38 The utilization of multiple sources of information in the diagnostic process is important, as the caregiver may be the individual noticing the most cognitive and functional change in the patient. Although we found significantly higher patient-reported CDS scores in those with MCI versus HCs, as well as significantly higher informant-reported CDS scores in those with AD versus those with MCI, we found few significant relationships between SMCs and objective test performance in these groups. Our lack of finding a link between patient-reported CDS scores and objective neuropsychological test performance in individuals with a diagnosis of MCI or AD may be related to this decreased level of cognitive awareness seen in individuals with memory impairment or to psychic denial, underlying the importance of obtaining informant report in the diagnostic process. As cognitive problems worsen, SMCs may lose value in the prediction of objective cognitive task performance. Measurement of SMCs (patient report) in cognitively intact older adults may then be a good method to identify those with subtle cognitive problems who are at risk of future cognitive decline.
Our results also provide some support for the use of the CDS to assess SMCs in individuals presenting for memory evaluation. How SMCs are assessed can affect results. 39 In some prior studies, SMCs were assessed via a single item or a brief, study-specific measure consisting of a few yes/no questions. 21 The CDS, which has been associated with cognitive difficulties in cardiovascular disease and other disorders, 40,41 may be beneficial as a brief, easy-to-understand inventory of SMCs that informants can complete for those presenting for neuropsychological evaluation of memory complaints. We, in addition to previous investigators, 8,19,20 found informant-reported SMCs predictive of lower concurrent neuropsychological test performance among individuals with a diagnosis of MCI, and thus the informant CDS may help elucidate the nature of current cognitive complaints and aid in early detection of cognitive decline in older adults. Both the self- and informant-reported CDS should be utilized in memory disorders clinics to fully assess cognitive complaints.
It is possible that depressive symptoms could have affected the results. Previous research has shown a 52.8% prevalence of SMCs in individuals with depression, and only a 28.7% prevalence of SMCs in those without depression. 42 Older adults with past and current depression are more likely to endorse SMCs, 43 -45 and depression may be more relevant to SMCs than current cognitive function. 46 -48 Higher self-reported SMCs may have been due in part to higher levels of depression in those with MCI, and self-reported SMC did not predict cognitive performance in this group. Thus, the higher self-reported SMC may reflect depression rather than self-awareness of cognitive impairments.
Several limitations may have affected the results of this study. We were unable to collect informant-rated CDS data on the HC group. Participants in this group were comprised of friends and relatives of patients in the MCI and AD groups, due to the design of the original screening study the data comes from. A follow-up study utilizing unrelated HC and patient samples is warranted. Our HC participants were also not required to bring an informant to study sessions. In future studies, obtaining informant-rated CDS data on HCs may help to determine whether informant-reported SMCs is specific to AD pathology. It is possible that in the present study, some of the HC endorsing high levels of SMCs had early AD pathology such as apolipoprotein E4 or other biomarkers. Data in the present study were collected at 1 time point, and more data are needed regarding how patient- and informant-reported SMCs may predict changes in cognition over a follow-up interval. Continued data collection is in progress to track changes in cognition in individuals in the HC and MCI groups, which would allow for such a comparison. The present study focused on the attention and memory subscales of the CDS. Future research should investigate whether other subscales provide additional predictive information about cognitive ability in HC, MCI, and AD. Several additional relationships between informant-reported SMCs and neuropsychological test performance were found in the present study; however, these did not hold up to our correction for multiple comparisons. Future research should investigate whether these relationships between SMCs and DRS scores hold in larger samples of MCI and AD.
In conclusion, the results of the present study indicate that individuals with MCI do report a high level of SMCs on the CDS and also provide evidence that self-reported SMCs (for HCs) and informant-reported SMCs (for those with MCI) on the CDS can provide information about concurrent cognitive status. Informant report of SMCs may also help clinicians to assess level of risk of further cognitive decline in patients with MCI. Finally, patient-reported SMCs are not correlated with objective neuropsychological test performance, underscoring the importance of obtaining objective cognitive tests in order to reliably identify MCI and AD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from Univita Health to Brian R. Ott.
