Abstract
The purpose of this study is to assess the effect of vitamin D replacement on cognitive function in older adults. A total of 560 patients who underwent comprehensive geriatric assessment including Global cognitive assessment, Basic Activities of Daily Living (BADL), and Instrumental Activities of Daily Living (IADL) twice in 6-month period were retrospectively reviewed. Oral cholecalciferol was replaced to patients with vitamin D deficiency routinely. In baseline cognitive scores, BADL-IADL scores were lower in the severe deficiency group than in the deficiency and adequate groups (P < .05). With regard to the relation between changes in cognitive functions, BADL-IADL scores on the 6-month versus baseline, no difference was determined in patients with and without dementia (P > .05). Vitamin D replacement may not improve cognitive performance in older adults, even if vitamin D is raised to adequate level, suggesting that longer term replacement therapy may be needed to improve cognitive function.
Introduction
Cognitive dysfunction and dementia cause great socioeconomic health burden in societies. Although dementia has recently become one of the most popular topics in the scientific community, there is no effective medical treatment yet. 1 For this reason, it is important to identify and prevent reversible risk factors. Many etiologic causes, such as cardiovascular risk factors, vitamin deficiencies, depression, thyroid dysfunction, cancer, may lead to the development of dementia. 2
One of the suspicious factors is vitamin D, which plays a primary role in systemic calcium homeostasis and bone health. Mainly affecting the intestine, kidney, and bone, 3 biologically active 1,25-hydroxy vitamin D (1,25 (OH)2 D) has receptors on many different nucleated cells. Accordingly, vitamin D is also shown to have effect on the immune system, insulin resistance, the cardiovascular system, and the central nervous system. 4 –7 Vitamin D deficiency has been found to be associated with orthostatic hypotension, depression, depression, sarcopenia, and frailty, which are important causes of mortality and morbidity in older adults, 8,9 as vitamin D has many effects on more than one system. Therefore, the relationship between vitamin D and cognitive functions is one of the most studied topics. Vitamin D has been reported to play a neuroprotective role by regulating the release of neurotrophic factors such as neurotrophin 3, nerve growth factor, and glial cell-derived neurotrophic factor, 10,11 increasing antioxidant pathways 12 and reducing inflammatory biomarkers. 13 New evidence suggests that vitamin D may affect mood and cognitive functions in older adults. 14 It has been reported that low 25 (OH) D levels are common in Alzheimer’s disease (AD) and depressed patients. 15,16 Nevertheless, the current evidence in the literature about the relationship between cognitive performance and vitamin D is controversial. 16 Furthermore, it is not clear whether vitamin D supplementation in older adults is also effective on cognitive functions. 17
In this study, we aimed to show the relationship between cognitive performance and vitamin D level and the effect of vitamin D supplementation on cognitive functions in older adults.
Materials and Methods
The files of 1684 patients who applied to geriatric outpatient clinics and underwent comprehensive geriatric assessment (CGA) from January 2015 to March 2017 were retrospectively reviewed. Demographic characteristics, blood pressure levels, comorbidities, drugs, polypharmacy, cognitive status, activity of daily living indices, and laboratory data were obtained from the hospital records. The investigation was approved by the local ethics committee and conformed to the Declaration of Helsinki.
Inclusion Criteria
The study included patients who underwent CGA twice at 6-month intervals.
Exclusion Criteria
Exclusion criteria were determined as severe illnesses that can cause unstable major medical illness (eg, acute cerebrovascular event, sepsis, gastrointestinal bleeding, acute renal failure, acute respiratory failure), major primary psychiatric disorders (schizophrenia, bipolar disorder, or recurrent major depressive disorder), age <60 years, and toxic levels of vitamin D (even at baseline and/or follow-up level ≥150 ng/mL). Patients with missing data in hospital records were also not included in the study.
Patients’ Characteristics
Patients’ demographic characteristics including age, sex, duration of education, accompanying systemic diseases, and the number of drugs used were recorded. Hypertension, coronary artery disease, congestive heart failure, diabetes mellitus, hyperlipidemia, peripheral arterial disease, and cerebrovascular disease were obtained from patients’ reports. Dementia and depression were diagnosed according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition criteria. 18
Comprehensive Geriatric Assessment
In an attempt to assess global cognitive function, the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA) scales were selected for those who had less than 11 years of education and more than 11 years of education, respectively. Patients were also assessed using the Geriatric Depression Scale (GDS), 19 the Basic Activities of Daily Living (BADL) and Instrumental Activities of Daily Living (IADL), the clock-drawing test (CD), and the Clinical Dementia Rating Scale. 20 The change in points was obtained by subtracting the initial value from the follow-up value, and the delta (Δ) value was specified for each test score—(Δ) value: (6-month evaluation–baseline evaluation).
Vitamin D Levels
25 hydroxy-vitamin D [25 (OH) D] levels were used to assess the vitamin D status of the patients. 21 Serum 25-hydroxy D vitamin radioimmunoassay was measured by the autoanalyzer diagnostic modular system (Roche E170 and P-800, Roche Diagnostics International AG, Rotkreuz, Switzerland). Vitamin D-level status was accepted as <10 ng/mL severe deficiency, 10 to 20 ng/mL deficiency, and ≥20 ng/mL sufficient in the baseline. 21,22 Oral cholecalciferol was routinely replaced to patients with vitamin D deficiency. Cholecalciferol was administered 50 000 IU once weekly for 6 weeks, followed by 5000 IU cholecalciferol per week for severe D vitamin deficiency. 3 Vitamin D deficiency was treated only with maintenance treatment. After 6 months, these 3 groups were divided into 2 groups according to the levels of vitamin D of ≥20 ng/mL and <20 ng/mL.
Statistical Analysis
Analysis of the data was done in the SPSS 22 program. Descriptive statistics are shown as mean ± standard deviation for normal distribution, median for unaltered distributions (maximum from minimum), and number of cases and percentage for nominal variables. When the group number was 2, the significance of the differences between the groups was examined by t test, and mean values were investigated by Mann Whitney U test. When the number of groups was more than 1, the significance of differences between the groups in terms of averages was investigated by analysis of variance (ANOVA), and the significance of the medians was determined by the Kruskal-Wallis test. Nominal variables were assessed by Pearson χ2 or Fisher exact test. Variances in the multiple groups were assessed by post hoc Tukey test. Binary logistic regression was used to assess the relationship between vitamin D-level status and baseline cognitive function. For P < .05, the results were considered statistically significant. We calculated that minimum 310 participants were required to show cognitive effects of vitamin D on cognitive functions at the level of 5% acceptable error and 95% confidence. 23
Results
In this retrospective follow-up study, 560 patients were included. The characteristics of the patients according to the baseline vitamin D-level status are shown in Table 1. The average age of the study group was 74.9 (8.17) years. Female gender and lower education level were higher in the group with severe vitamin D deficiency (P ≤ .01). The proportion of dementia in the group with severe vitamin D deficiency was also higher (P = .01). The difference between the groups was not significant in terms of comorbid disease except dementia (P > .05). At the same time, the results of the baseline comprehensive geriatric evaluation are summarized in Table 1. Mini-Mental State Examination, MoCA, BADL, IADL, and clock drawing scores are lower in the group with vitamin D deficiency.
Patients’ Characteristics According to Basal Vitamin D Value Groups.
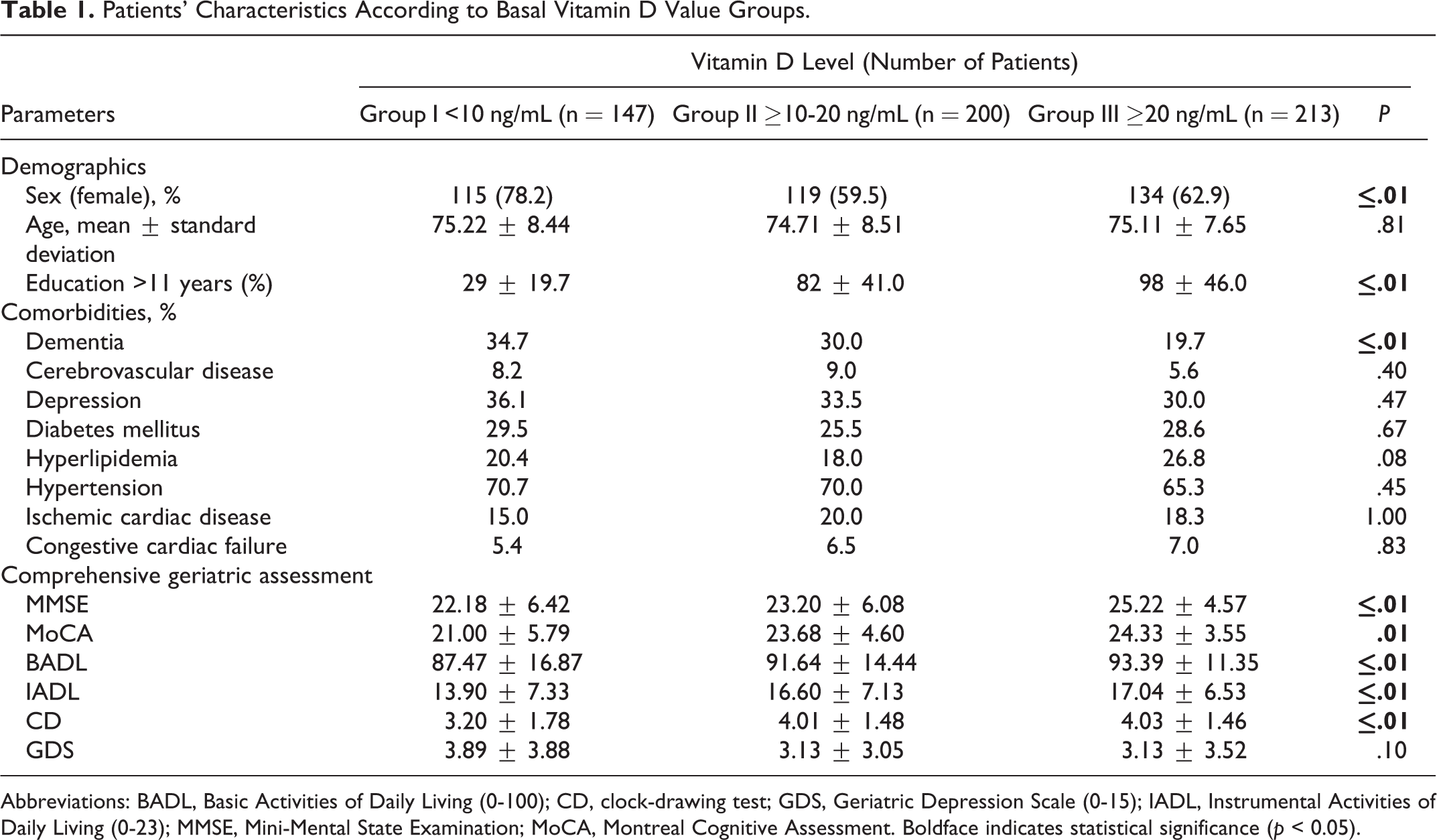
Abbreviations: BADL, Basic Activities of Daily Living (0-100); CD, clock-drawing test; GDS, Geriatric Depression Scale (0-15); IADL, Instrumental Activities of Daily Living (0-23); MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment. Boldface indicates statistical significance (p < 0.05).
Since these scores are highly skewed, traditional ANOVA and linear regression cannot be used without compromising the interpretability of the data using some complex transformations. For this reason, the response variables were divided into 2 for all logistic regression models (MMSE score: [<24, ≥24], 24 MoCA score: [<21, ≥21], 25 BADL: [<90, ≥90], 26 IADL: [<20, ≥20] 27 ) and used for backward shifting. For all models, the relationship between cognitive performance and vitamin D was adjusted for age, gender, education (years), body mass index, serum calcium, phosphorus, hemoglobin, thyroid stimulating hormone, folate, vitamin B12, and the environment that the patients live in.
The likelihood of being in the high cognitive performance group is 2.19 times greater than the vitamin D deficiency group controlling other covariates for the normal vitamin D group. There was a significant positive effect on baseline BADL score of vitamin D concentration as a continuous variable. Specifically, a unit increase in Vitamin D provides a 4% increase in the likelihood of having a high BADL score. Patients with normal vitamin D levels were more likely to have higher IADL scores than the deficient group. The cross-sectional relationships between vitamin D status and cognitive scores and functionality are shown in Table 2.
Associations Between Vitamin D Status and Global Cognitive Scores and Functionality.
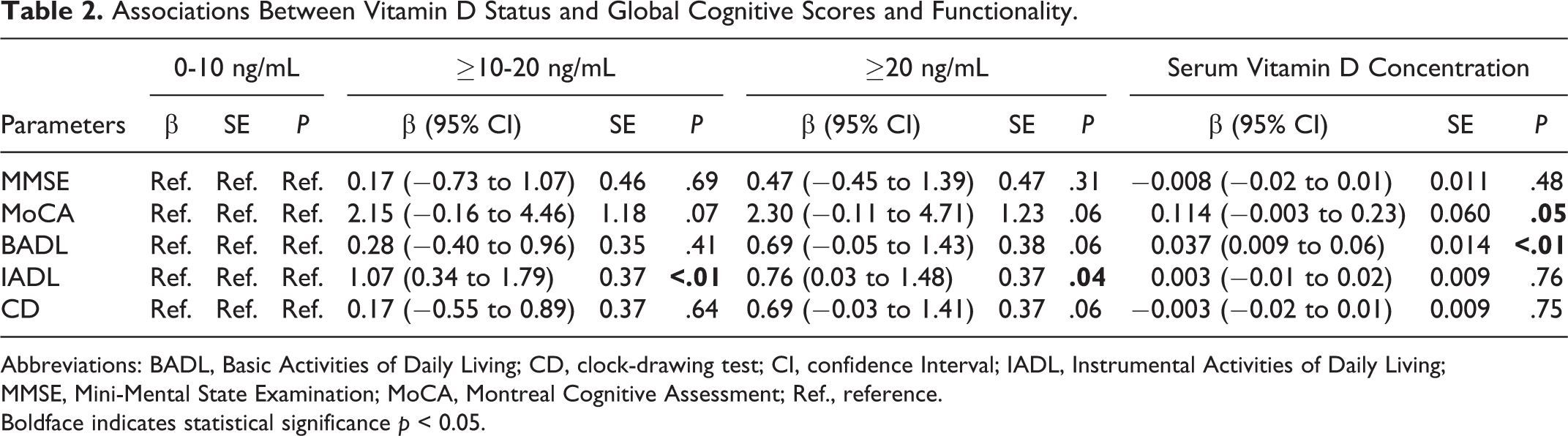
Abbreviations: BADL, Basic Activities of Daily Living; CD, clock-drawing test; CI, confidence Interval; IADL, Instrumental Activities of Daily Living; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; Ref., reference. Boldface indicates statistical significance p < 0.05.
No statistically significant difference was observed in nondemented patients with ≥20 ng/mL or <20 ng/mL of vitamin D levels when compared with follow-up neurocognitive test results at the sixth month. Comprehensive geriatric assessment details at the sixth month were summarized by baseline vitamin D-level groups for nondemented patients in Table 3.The changes in MMSE, MoCA, GDS, BADL, IADL, and CD scores were not significant for the nondemented 6 groups according to baseline and following vitamin D status. In addition, at the end of the sixth month of follow-up, all of the performance of the nondemented patients with vitamin D deficiency were observed to be lower than at the end of the sixth month even if they reached adequate vitamin D levels (P < .05).
Follow-Up Evaluations and Delta (Δ) Changes in Patients Without Dementia.
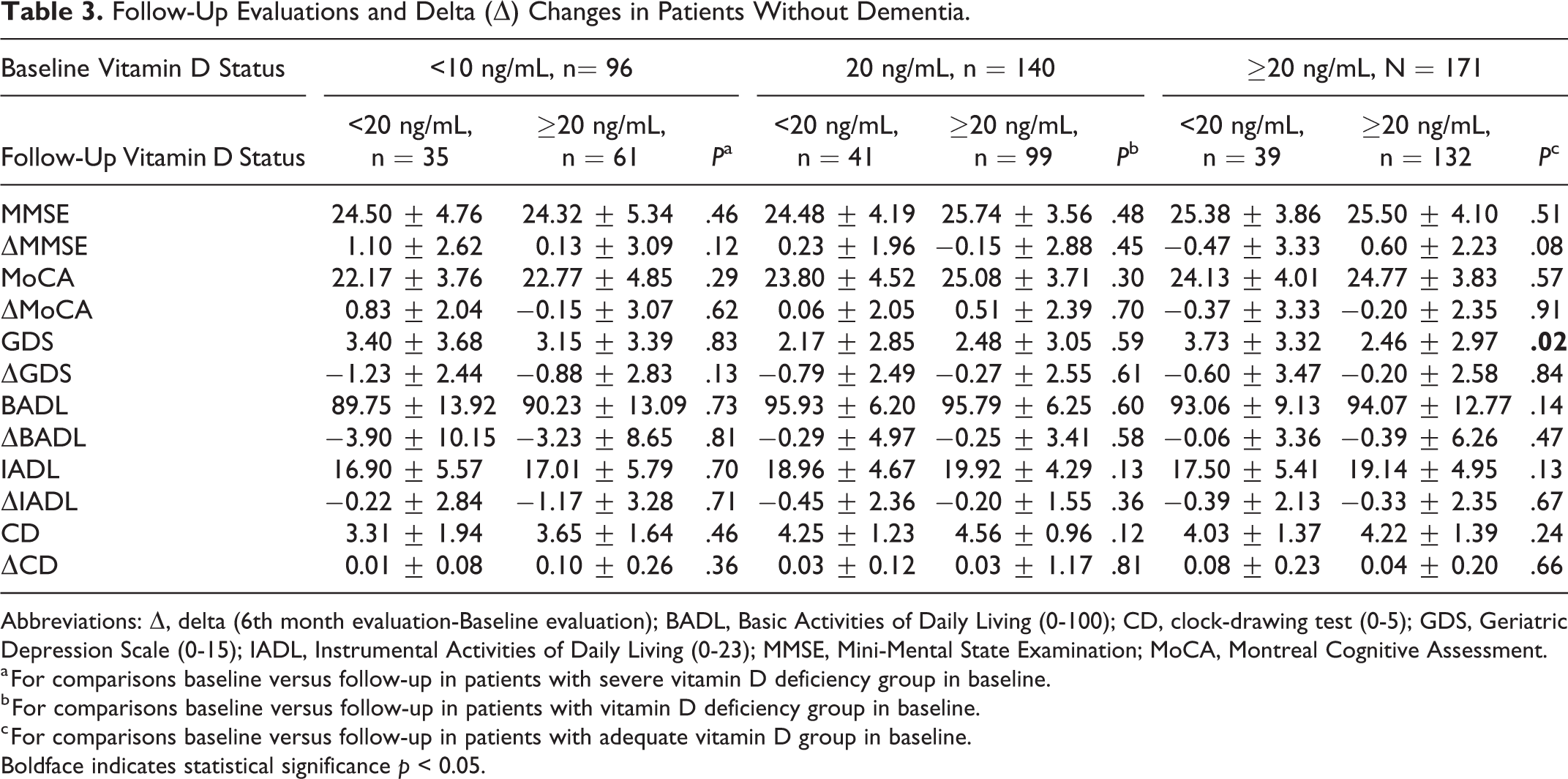
Abbreviations: Δ, delta (6th month evaluation-Baseline evaluation); BADL, Basic Activities of Daily Living (0-100); CD, clock-drawing test (0-5); GDS, Geriatric Depression Scale (0-15); IADL, Instrumental Activities of Daily Living (0-23); MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment.
a For comparisons baseline versus follow-up in patients with severe vitamin D deficiency group in baseline.
b For comparisons baseline versus follow-up in patients with vitamin D deficiency group in baseline.
c For comparisons baseline versus follow-up in patients with adequate vitamin D group in baseline. Boldface indicates statistical significance p < 0.05.
Owing to the difference in terms of sex and dementia in the severe deficient group at the baseline, we evaluated both gender and patients with dementia separately. There was no difference in MMSE, MoCA scores, and comprehensive assessment parameters between male and female groups (P > .05). There was no statistically significant difference in dementia group after vitamin D replacement. Similar results were also obtained when vitamin D deficiency was accepted as <30 ng/mL.
Discussion
In this study, it was demonstrated that older adults with higher vitamin D level were more likely to have better cognitive performance than those lower vitamin D level. However, vitamin D replacement had no effect on cognitive functions or ADLs of patients with or without dementia, although vitamin D levels were increased from baseline severe deficient to adequate levels in the sixth month.
Vitamin D is an active hormone with many endocrine and paracrine effects on different tissues. Active vitamin D enhances intestinal calcium absorption in the small intestine and interacts with the vitamin D receptor–retinoic acid x-receptor complex (VDR-RXR) to increase the expression of the epithelial calcium channel. The VDR-RXR, with muscle mass and strength enhancing effects, also stimulates the proliferation and differentiation of muscle cells, allowing skeletal muscle cells to be protected from insulin resistance. 28 Furthermore, individuals with insulin resistance have increased risk of the development of dementia. 29 Patients may have limited sun exposure due to physical and cognitive dysfunction. In other words, physical and cognitive dysfunction may accelerate the deficiency of vitamin D.
Dementia and cognitive impairment cause dependence and loss of functioning, which increase the cost of health care and the need for care in older adults. 2 Recent data suggest that there may be a relationship between cognitive functions and vitamin D deficiency. 16 Modulation of cognitive processes in aging and neuropathophysiology of dementia is complex. It has been reported that vitamin D has a neuroprotective effect on neuronal integrity by detoxification routes, anti-inflammatory effects, and neurotrophin synthesis. 30 Vitamin D is also associated with reduced β-amyloid 1-42 concentrations in cerebrospinal fluid. 31 Genetic linkage to late-onset AD has been assigned to the 12q13 chromosome containing the gene for VDR28. 32 In addition, vitamin D may also regulate systolic blood pressure, cardiac autonomic activity, immunological functions, cellular differentiation and proliferation, insulin resistance, diabetes mellitus, and hypertension. 33,34 All these factors support that vitamin D may be indirectly related to cognitive decline and dementia.
On the other hand, it was reported that the total MMSE score was not related, but the executive function was associated with vitamin D concentration. 14 In addition, intervention studies of vitamin D supplementation did not show any benefit in cognitive functions. 17,35,36 However, it is reported that patients with AD had lower concentrations of vitamin D than those without AD and that MMSE scores were lower in individuals with lower 25 (OH) D concentrations in an extensive meta-analyses. 16
Consistent with previous analyses, low vitamin D levels were associated with poorer cognitive function and dementia in this study 37 ; however, in the following period, it was shown that vitamin D supplementation did not have clear benefit, nor was the prevention of cognitive impairment in older adults, which may be suggested by various explanations. First, since both age-related changes in pharmacokinetics and pharmacodynamics can lead to a reduction in the effect of treatments, the replacement of vitamin D in older adults may not be as effective as in nonolder patients. 38 Second, as vitamin D has nuclear receptors and mainly acts by gene expression, the effect of vitamin D on cognition may be seen in the long term. 39 For this reason, 24 weeks may be too short to clearly reveal the effect of vitamin D replacement, and long-term study may be an appropriate approach. Thus, vitamin D replacement can be particularly important at Middle Ages before symptoms develop in later life 37 and should be part of successful aging. Third, it was previously reported that psychomotor and executive functions are most susceptible to fluctuations in vitamin D, 40 but only global cognitive score was evaluated in this study. Vitamin D may be associated with particularly some domains of cognition. More detailed neurocognitive tests might have been sensitive to show change in cognitive subdomains.
This study has many powerful features such as sample size, being a longitudinal study, and assessment of patients’ global cognitive functions. Moreover, all comorbid diseases that could affect cognitive functions as well as vitamin B12, folic acid, and thyroid hormone levels were considered in patients with and without dementia, which is another powerful aspect of this study. On the other hand, it has some limitations like seasonal fluctuation effect of Vitamin D, as patients are included in the study in different seasons. Moreover, when interpreting serial assessments, the roles of practice effects and test–retest reliability on follow-up performance may cause some considerations on the results. We wanted to minimize this effect by assessing patients with objective scales. Another limitation is that we examined only global cognitive score; however, it could have been better if the detailed cognitive evaluation with subdomains could have been performed, and 6 months follow-up may be insufficient to show the effect of vitamin D on cognitive functions. Additionally, due to the small sample size of patients with dementia, our results related to dementia may not be generalized.
Conclusions
Vitamin D deficiency seems to be associated with dementia and cognitive impairment in older adults. However, even if vitamin D levels of patients with severe vitamin D deficiency were increased to an adequate level, the cognitive performance of these patients did not reach to the cognitive performance of patients whose vitamin D levels were adequate from the beginning. For this reason, patients should be screened and treated for vitamin D deficiency as a component of “successful aging” from Middle Ages.
Footnotes
Authors’ Note
Esra ATES BULUT performed manuscript writing and data collection; Pinar SOYSAL supported statistical analysis, manuscript writing, and conceptualism; Idil YAVUZ made detailed statistical analysis; Suleyman Emre KOCYIGIT contributed data collection; and Ahmet Turan ISIK designed the study and performed manuscript writing and conceptualism.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
