Abstract
We examined the interactive effects of apolipoprotein ∊4 (APOE-∊4), a risk factor for Alzheimer’s disease (AD), and diabetes risk on cortical thickness among 107 healthy elderly participants; in particular, participants included 27 APOE-∊4+ and 80 APOE-∊4− controls using T1-weighted structural magnetic resonance imaging. Regions of interests included select frontal, temporal, and parietal cortical regions. Among APOE-∊4, glucose abnormalities independently predicted reduced cortical thickness among temporoparietal regions but failed to predict changes for noncarriers. However, among noncarriers, age independently predicted reduced cortical thickness among temporoparietal and frontal regions. Diabetes risk is particularly important for the integrity of cortical gray matter in APOE-∊4 and demonstrates a pattern of thinning that is expected in preclinical AD. However, in the absence of this genetic factor, age confers risk for reduced cortical thickness among regions of expected compromise. This study supports aggressive management of cerebrovascular factors and earlier preclinical detection of AD among individuals presenting with genetic and metabolic risks.
Alzheimer’s disease (AD) afflicts nearly 10% of individuals older than 70 years; it accounts for the highest proportion of total dementia cases with up to 75% of individuals with dementia carrying an AD diagnosis. 1 Persons with AD demonstrate global and regional brain atrophy, with entorhinal and hippocampal atrophy among the earliest changes. 2 -4
Preclinical detection of AD among at-risk individuals has increasingly become an area of research importance, 5 given that targeted treatments to alter the trajectory of neurodegeneration may follow from such investigation. Two highly significant factors include genetic and cerebrovascular disease (CVD) risks. Researchers have investigated the utility of combining genetic or CVD risk factors with other biological markers, such as brain volume, 6 to facilitate earlier identification of disease. The most salient of the genetic risks strongly associated with the common form of AD is the apolipoprotein ∊4 (APOE-∊4) allele on chromosome 19. 7
The APOE-∊4 allele has a dose-related effect on AD, increasing risk and lowering onset age, 8 -11 whereas APOE-∊2 appears to confer protection. 12 Researchers have investigated the clinical utility of combining genetic risk with other biological markers, such as measures of brain volume, in the early detection of AD. 7,13,14 Using cross-sectional designs, magnetic resonance imaging (MRI) comparisons between nondemented APOE-∊4 carriers and noncarriers have yielded mixed results. 4,8,13 -19 Although some investigations have failed to detect volume differences among carriers versus noncarriers, 13,16,19,20 -22 several cross-sectional studies of nondemented persons with APOE-∊4 show that tissue loss appears to be dose dependent. 23,24 Structural MRI studies of nondemented APOE-∊4 participants have revealed brain gray matter (GM) and white matter (WM) changes that reflect AD neurodegeneration. 13,14
The research has demonstrated evidence for temporoparietal cortical thinning in AD throughout the disease course, 25 -33 with the greatest magnitude of difference in medial temporal, posterior cingulate, and temporoparietal 29 regions. The investigation has also revealed frontal brain changes in individuals at risk for AD. 33 -36
Conditions affecting the vascular system are also known to increase risk for AD 1,37,38 and result in similar morphological brain changes. 9 In particular, diabetes mellitus (DM) is associated with neuropathologic changes that are consistent with those documented in AD, 6,39 -43 and such changes can result from even subclinical levels of CVD risk. 11,44,45 Hyperglycemia, in particular, 15 may confer one of the strongest effects on the development of vascular complications. Previous research has indicated that the degree of glucose dysregulation is important in the resulting severity of cerebral dysfunction, although clinical severity of disease is not necessary for associated brain changes; for example, we have previously shown that cerebrovascular risk factors, even at subclinical levels, were associated with thickness abnormalities in a healthy community dwelling sample. 46
Of particular importance is the relationship between cerebrovascular abnormalities and APOE-∊4 on the development of AD. Prior work has also documented a synergistic impact of APOE-∊4 and diabetes on AD risk (with a 5-fold increase among those with both contributions). 16 In particular, research 47 -51 has documented that vascular burden among middle-aged individuals may interact with APOE-∊4 to increase the risk for degenerative decline (beyond what might be expected from this genetic risk alone). In particular, abnormal vascular or metabolic functioning and APOE-∊4 each increase the risk for dementia but when combined, risk is increased by approximately 4 to 5.5 times 52 and has been associated with cognitive impairments, 4,53 cerebral amyloid angiopathy, as well as increased neuritic plaques and neurofibrillary tangles. 16,22
Although no previous studies have examined the combined impacts of APOE-∊4 and glucose dysregulation on cortical thinning, widespread cortical thinning in healthy elderly participants has been documented in relation to CVD risk alone, 46 and interactive effects of amyloid-β and cerebrovascular risk (coronary risk profile and lipid abnormalities) have been shown in relation to parietal cortical thickness among older adults with and without mild dementia. 54 Research has also revealed that preferential thinning of parietal regions may only be demonstrated in the presence of the combined risk of amyloid-β and CVD risk versus a single isolated risk. 54 These findings are consistent with the pattern of regional cortical vulnerability documented in AD and suggest that the deleterious influence of amyloid-β is increased in the presence of vascular compromise. Therefore, research has supported an interactive impact of metabolic and genetic risk among posterior cortical association regions that are expected to be implicated throughout the AD-disease process.
The purpose of the present investigation was to examine the interactive effects of genetic (APOE-∊4) and CVD risk factors in the development of cortical thickness abnormalities that might be associated with preclinical AD in middle aged and older adults. We predicted to find reduced changes consistent with what has been observed in the earliest stages of AD, as well as what has been observed in association with CVD risk. Specifically, we expected to find decreased cortical thickness among temporoparietal and frontal association regions, particularly among APOE-∊4 individuals with abnormal glucose. Preclinical detection of AD is advanced through earlier identification of neuropathological changes, which may be associated with these subclinical conditions that increase AD risk, and therefore the purpose of the current investigation was to examine interactive effects of APOE-∊4 and diabetes risk among a sample of older adults with varying (mild-moderate) levels of vascular risk (glucose dysregulation) and without any signs of dementia versus healthy controls without this genetic risk.
Methods
Participants
The sample included 107 healthy elderly participants who underwent structural MRI. The current sample is similar to the samples used in prior studies conducted by our group. 44,55 Participants were recruited from 2 separate but overlapping studies examining the impact of CVD on brain structure/cognition. Twenty-nine participants were selected from a larger sample recruited by the Harvard Cooperative Program on Aging (HCPA) for the Claude Pepper Older American Independence Center (OAIC) of Harvard Medical School in response to an advertisement appearing in the HCPA newsletter asking for healthy community-dwelling older African Americans. Seventy-eight participants were recruited through the Understanding Cerebrovascular and Alzheimer’s Risk in the Elderly (UCARE) program; the participants were recruited through the Boston University Alzheimer’s Disease Center (BUADC) based on the initial criteria of being neurologically healthy based on a consensus conference diagnosis at the BUADC and of having a first-degree family relative with dementia. The participants were excluded due to history of traumatic brain injury (TBI) of “mild” severity or greater according to previous work, 56 history of 1+ TBI (due to cumulative neuropathological effects), diagnosis of dementia, severe psychiatric illness, or history of brain surgery/stroke. All participants were literate with at least a ninth grade education. Mini-Mental State Examination (MMSE) scores ranged from 23 to 30 (outside dementia range 57 ). One participant had an MMSE score of 23 and 3 had a score of 24, and results were not altered when eliminating these participants. Participants included 27 individuals positive for the ∊4 allele and 80 control participants (∊4−). A total of 25 individuals were heterozygous (∊3/4) and 2 were homozygous (∊4/4) for the APOE-∊4 allele. Two participants were excluded due to having the ∊2 allele, given its well understood protective role in AD. 12
Following informed consent, fasting blood was drawn and processed for analysis of glucose. In the current sample, the glucose levels ranged from 65 to 156 mg. According to the American Diabetes Association, 58 22.4% had “mildly” elevated glucose and 8.4% would be considered “diabetic”; thus 91.6% had normal to mildly elevated glucose. We also obtained a measure of glycosylated hemoglobin A1c (HbA1c). The HbA1c levels ranged from 4.9% to 8.4%, and 7.4% had HbA1c levels of 7+, indicating poorly controlled sugar levels in the months preceding evaluation. Of the total sample, 85 individuals were taking medications to control cerebrovascular risk (34.5% for blood pressure, 23.3% for cholesterol, and 7.5% for diabetes). Other laboratory measurements of cerebrovascular functioning were also explored to determine whether glucose demonstrated a unique relationship to outcome variables; these other variables included measures of blood pressure (mean arterial blood pressure, systolic, and diastolic blood pressure), cholesterol (total cholesterol, low-density lipoprotein, high-density lipoprotein, and triglycerides), and weight (body mass index). For all additional correlational analyses that included cerebrovascular measurements apart from glucose, P > .05.
Neuroimaging Protocol
Four participants were scanned using a Siemens 1.5 Tesla Sonata system (Siemens Erlangen, Germany), with the following parameters: magnetization-prepared rapid acquisition with gradient echo (MPRAGE); T1 = 1000 milliseconds, repetition time (TR) = 2.73 seconds, echo time (TE) = 3.39 milliseconds, flip angle = 7°, slice thickness = 1.33 mm, 128 slices, and field of view (FOV) = 256 × 256 mm. The remaining 103 participants were scanned on the upgraded Siemens 1.5 Avanto System, with slightly different parameters; MPRAGE: T1 = 1000 milliseconds, TR = 2.73 seconds, TE = 3.31 milliseconds, flip angle = 7°, slice thickness = 1.3 mm, 128 slices, and FOV = 256 × 256 mm.
Image Processing
Cortical thickness measurements were obtained by first conducting cortical reconstruction using the FreeSurfer image analysis suite, which is freely available for download online (http://surfer.nmr.mgh.harvard.edu/). Methods have been previously described in detail 59,60 ; see our previous work for a summary of this process. 46,61 Automated segmentation of the cortical surface, reconstruction of the pial surface, and calculation of cortical thickness were done using the FreeSurfer processing suite. 62 The neocortex was automatically subdivided based on gyral folding patterns 63 to yield regions of interest (ROIs). Twelve distinct ROIs were chosen a priori for investigation in order to examine the influence of AD-risk factors on temporoparietal abnormalities often implicated in AD; analyses also included a frontal control region; these ROIs included temporoparietal (entorhinal, supramarginal, inferior temporal, middle temporal, superior temporal, superior parietal, posterior cingulate, transverse temporal, precuneus, parahippocampal, and temporal pole) and frontal (superior frontal) cortical regions. Right and left hemisphere ROIs were averaged.
Statistical Analyses
Preliminary analyses included analysis of variance (ANOVA) and chi-square tests comparing genetic groups (APOE-∊4+ and APOE-∊4) on demographic and other related (eg, global cognition and glucose) variables. One way between-subjects ANOVA analyses were then run comparing genetic groups on cortical thickness ROIs. In order to evaluate the independent or modulatory impacts of genetic and cerebrovascular risks, a series of hierarchical multiple regression models were then created, and genetic groups were stratified for these comparisons. First, age was entered as a covariate (model 1), then diabetes risk (fasting glucose and HbA1c) was introduced as main effects (model 2). Dependent variables included cortical thickness values for several temporoparietal and frontal regions known to be implicated in the development of AD. In order to evaluate the specificity of these findings, we examined relationships for blood pressure, body mass index, and cholesterol. We did not control for multiple comparisons to allow for maximum exploration of study variables. The α value was set to .05.
Results
Average age of the total sample was M = 67.9 years (standard deviation [SD] = 9.1 years), and average education was M = 14.9 years (SD = 2.7). Males comprised 38.8% of APOE-∊4+ and 37.0% of APOE-∊4−. For the total sample, 33.6% were African American and 66.4% were Caucasian. Average fasting glucose was M = 95.3 (SD = 17.3) and average HbA1c was M = 5.8 (SD = 0.68). There were no significant differences between the APOE groups on any demographic variable, including age, education, gender, and ethnicity. There were also no differences between groups when considering the proportion of individuals with and without positive family history of AD. The APOE groups were also similar on global cognitive functioning as measured by the MMSE. Finally, glucose measurements (HbA1c, fasting glucose) did not differ between APOE groups. Due to the recruitment criteria for the BUADC study, there was a high prevalence (87.9%) of individuals presenting with a positive first-degree family history of dementia, placing them at higher risk of developing dementia, 64 see Table 1 for descriptive statistical data for demographic variables.
Descriptive Statistics by Group for Demographic and Diabetes Data.
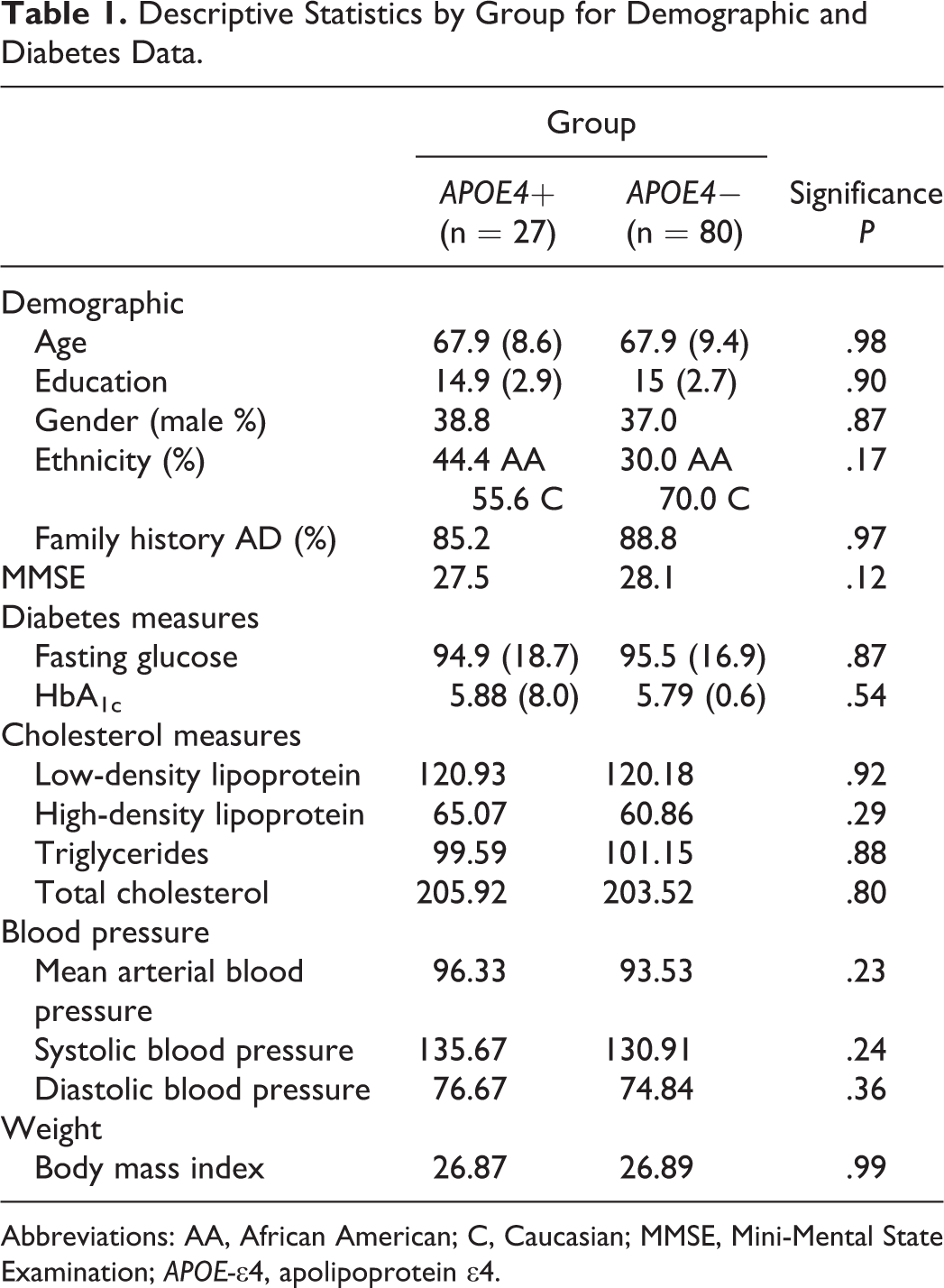
Abbreviations: AA, African American; C, Caucasian; MMSE, Mini-Mental State Examination; APOE-∊4, apolipoprotein ∊4.
One-way ANOVA analyses revealed no APOE group differences on any ROI for cortical thickness measurements (all P > 0.05; see Table 2). To further explore the relative impact of APOE-∊4 risk on cortical thickness, we conducted a series of hierarchical multiple regression analyses for the APOE group, entering both glucose variables and age into the model to determine the presence of potentially modulating variables in understanding cortical abnormalities in individuals with varying risks for AD. Multiple regression analyses revealed a distinct pattern of relationships which varied by group (APOE-∊4 vs controls). In particular, for the APOE-∊4 group, results revealed that glucose (when controlling for age) independently predicted decreased cortical thickness in temporal (parahippocampal [full model nonsignificant; fasting glucose, P = .037]; superior temporal [full model nonsignificant, HbA1c, P = .039]; and temporal pole [full model nonsignificant, fasting glucose, P = .032]) and parietal (supramarginal [full model, P = .029, HbA1c, P = .010]; posterior cingulate [full model nonsignificant, HbA1c, P = 0.047]) regions, see Figure 1 for scatterplots depicting these relationships. In contrast, for the control group, results revealed that age (when controlling for glucose) independently predicted decreased cortical thickness in several temporal (transverse temporal [full model, P = .029, age, P = .014]; superior temporal [full model, P = .002, age, P < .001]), parietal (precuneus [full model, P = .019, age, P = .005]; supramarginal [full model nonsignificant, age, P = .04]), and frontal (superior frontal [full model nonsignificant, age, P = .027]) regions, see Figure 2 for scatterplots depicting these relationships. Neither group demonstrated significant relationships of glucose (fasting or HbA1c) or age to entorhinal, inferior temporal, middle temporal, or superior parietal regions, see Table 3 for results of multiple regression analyses per group. There were no differences in results obtained on participants scanned using the upgraded Siemens 1.5 Avanto system versus the Siemens 1.5 Tesla Sonata system.
Descriptive Statistics by Group for Regional Thickness.
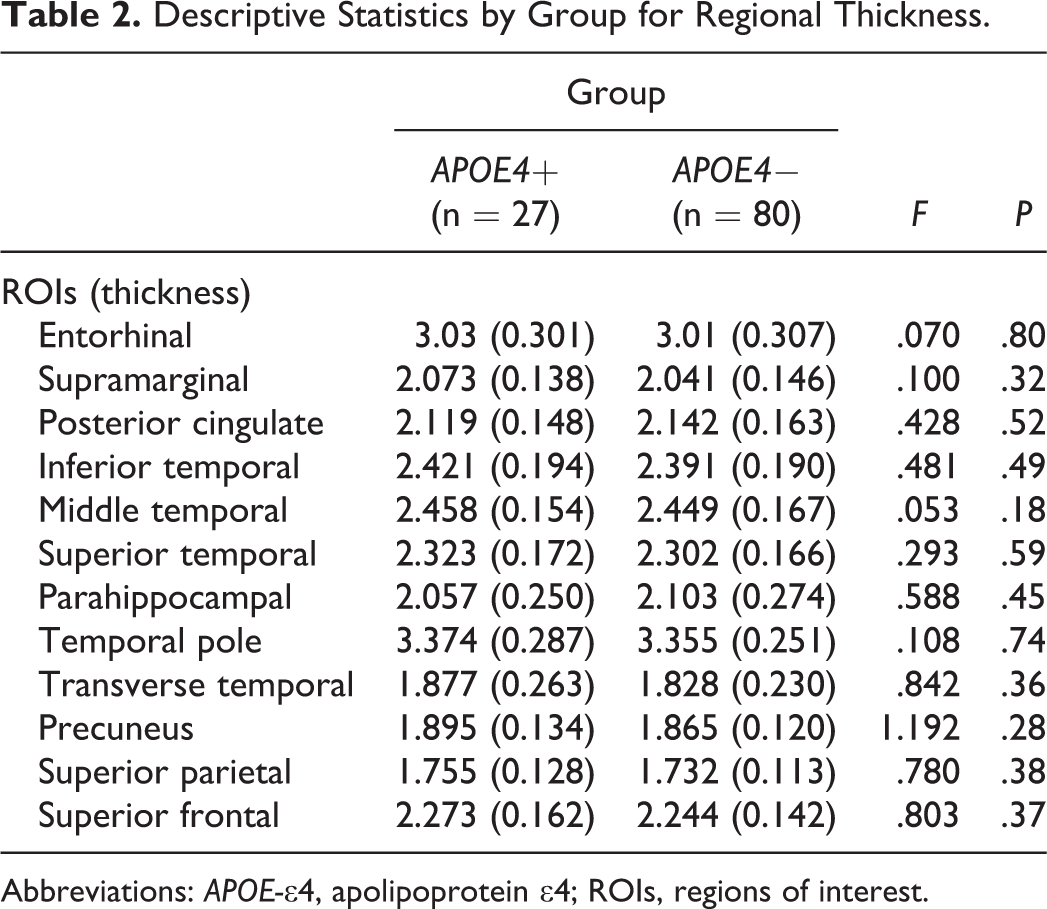
Abbreviations: APOE-∊4, apolipoprotein ∊4; ROIs, regions of interest.
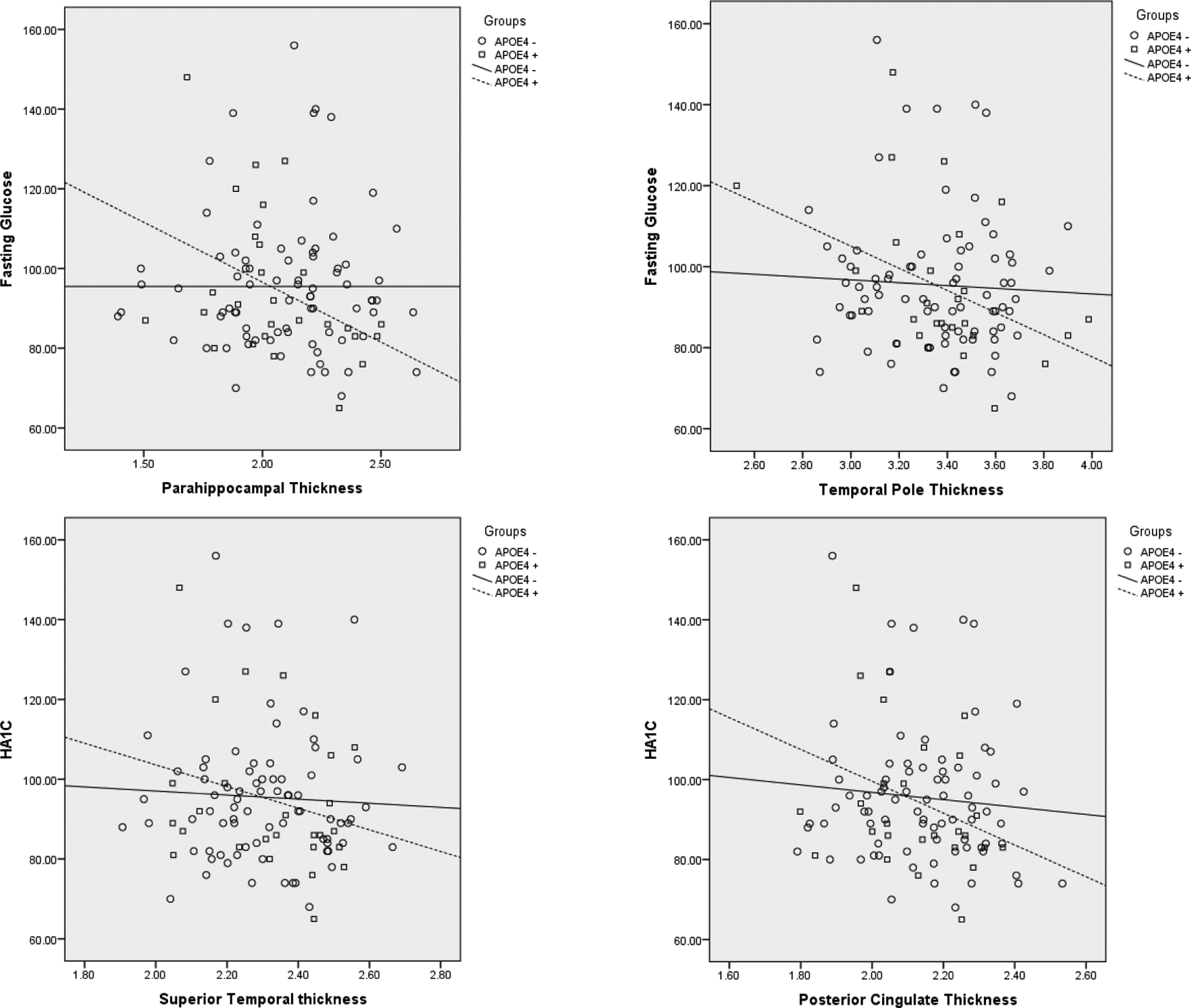
Significant relationships between cortical thickness and glucose among apolipoprotein ∊4 (APOE-∊4).
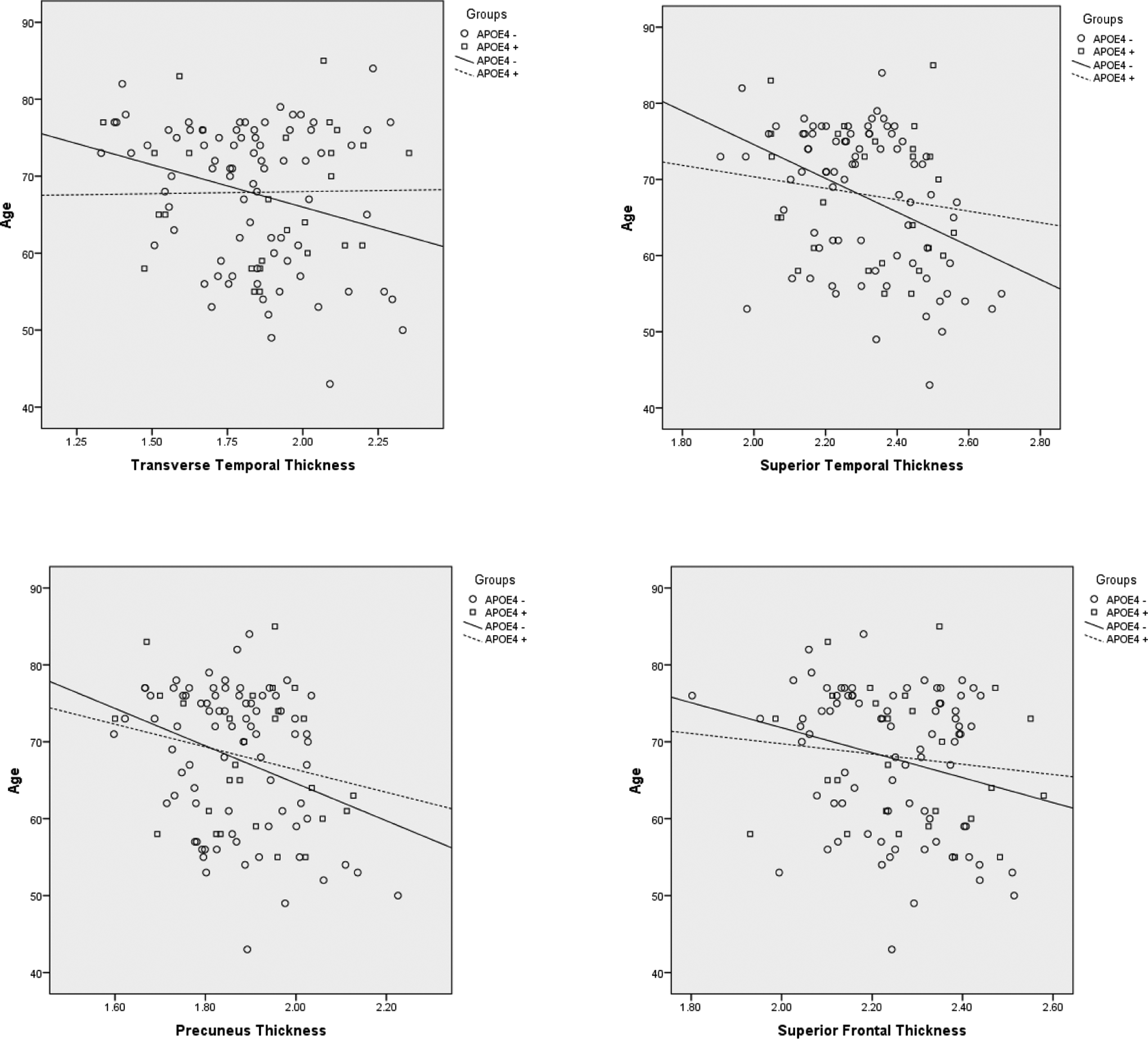
Significant relationships between cortical thickness and age among controls.
Direct and Interactive Relationships of APOE4 Status and Diabetes Risk on Cortical Thickness for Individual ROIs.a
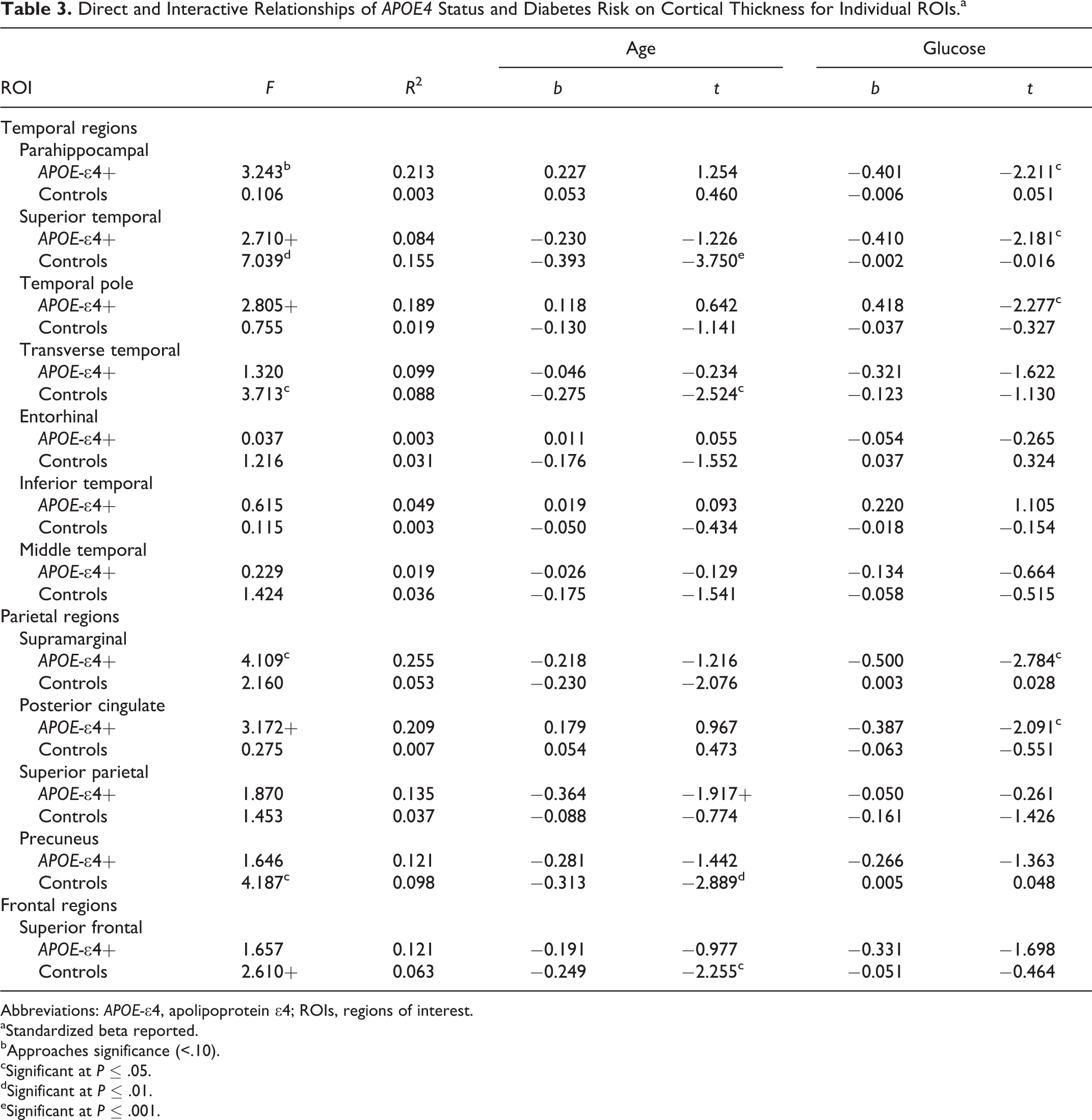
Abbreviations: APOE-∊4, apolipoprotein ∊4; ROIs, regions of interest.
aStandardized beta reported.
bApproaches significance (<.10).
cSignificant at P ≤ .05.
dSignificant at P ≤ .01.
eSignificant at P ≤ .001.
We also examined relationships to other cerebrovascular risk variables, including blood pressure (mean arterial blood pressure, systolic, and diastolic blood pressure), cholesterol (total cholesterol, low-density lipoprotein, high-density lipoprotein, and triglycerides), and body mass index; findings did not reveal significant relationships with cortical thickness for any of these variables (P > .05), suggesting a specific and preferential impact of glucose dysregulation on cortical tissue abnormalities in at-risk individuals.
Conclusions
The current results revealed that thinning of several temporoparietal brain regions was predicted by glucose dysregulation among APOE-∊4 participants, independent of age; in particular, significant negative relationships were found for supramarginal, posterior cingulate, parahippocampal gyrus, and temporal pole regions, with increased glucose predicting cortical thinning for the APOE-∊4 group.
These findings are consistent with previous work that has demonstrated widespread cortical thinning in relation to cerebrovascular and metabolic risk alone, even in the presence of subclinical risk, and interactively when combined with genetic risk. In particular, we have previously demonstrated that elevated cerebrovascular risk factors, including blood pressure and glucose, are associated with reduced thickness, even at subclinical levels, in an aging sample, 46 suggesting an influence of mild cerebrovascular risk alone. Other work has documented interactive (but not independent) effects of amyloid-β and vascular risk with associated reduced parietal cortical thickness 54 in older adults, indicating that the deleterious influence of amyloid-β on cortical integrity may be modified or exacerbated by the presence of vascular abnormalities.
The current results demonstrate that glucose dysregulation is particularly detrimental in individuals carrying the APOE-∊4 allele with regard to cortical thickness. Indeed, prior work has also documented an interactive impact of APOE-∊4 and diabetes on AD risk, whereas diabetes alone (in the absence of genetic risk) may be associated with less significant, yet moderate, risk for AD. 39 This interaction of metabolic and genetic risk has been documented previously by our group 67 and other work 49,51 and reflects neurodegeneration that is consistent with the expected pattern in early AD and supports a possible pathological mechanism for some at-risk individuals. This apparent association between APOE-∊4 and metabolic dysregulation may be related to the capacity of the ∊4 allele to modulate a variety of vascular factors; this risk may mediate the development of AD through Wallerian degeneration or by decreasing resting cerebral blood flow prior to dementia onset. 66
The combined impacts of both risks on brain structure have also been supported even when considering subclinically dysregulated glucose in the absence of clinical DM, with resulting impact on neuritic plaque formation, 52 and increases in amyloid precursor protein. 22 Our previous work 65 suggested interactive effects of metabolic and APOE-∊4 genetic risks on later myelinating WM integrity among healthy elderly participants at genetic risk for AD, but not among noncarriers; in particular, diffusion tensor imaging (DTI) investigation revealed that glucose measurements predict fractional anisotropy (FA) (a measure of white matter (WM) integrity) reductions among several later myelinating regions that may be affected in AD, including superior frontal, supramarginal, superior/middle/inferior temporal, precuneus, and superior parietal WM, but not on earlier myelinating control regions, including genu, splenium, and cerebral peduncles. Thus, the modulatory influence of CVD on APOE-∊4 risk may be exerted even in the absence of overt clinical diabetes, suggesting a milder severity of impact and an additional avenue for preclinical detection of disease. The findings documented here suggest that the impact of APOE-∊4 risk may extend beyond direct impact on WM microstructure and impact the integrity of cortical association regions.
Patients with pathologically confirmed AD (vs controls 25 ) and converters (MCI to AD 26 ) have demonstrated a similar pattern of cortical thinning among temporoparietal cortical regions that overlap with current findings. In particular, individuals at risk for or presenting with evidence of AD (given the presence of neuropathogical indicators or early clinical presentation) have demonstrated patterns of cortical thinning in temporal and temporoparietal areas that overlap with the present results; 27 -33 the greatest magnitude of differences has been reported in medial temporal, posterior cingulate, and temporoparietal (vs midfrontal 29 ) regions. These findings are also present in more advanced disease stages (patients with pathologically confirmed AD vs controls, 25 MCI to AD converters 26 ), with thinning in the posterior cingulate, superior temporal lobe, 25 and temporal pole. 26 Our current results support very early changes within these regions but only in the presence of the combined impacts of genetic and metabolic risks. In the absence of these dual impacts, and surprisingly, we did not find an effect of the APOE-∊4 group on temporoparietal cortical thickness, which may be reflective of related factors, including the very early preclinical disease stage of participants who participated in this sample and the discrepant APOE-∊4/control group sample sizes, and given the expected heterogeneity in resulting progression to AD. Taken together, these results support that regional involvement within several temporal and temporoparietal association areas may be expected to occur throughout the disease course and in individuals presenting with preclinical evidence for or significant risk of later developing AD.
Some regions which were expected to show reduced cortical thickness in the presence of APOE-∊4 status and cerebrovascular risk did not reveal significant findings, including several medial and lateral temporal regions (including the entorhinal cortex). Prior research has supported a significant role of the medial temporal lobe thinning in early AD, including the hippocampus and medial temporal lobes. 26,28,29,32,33 The reasons for this anomaly are unclear but may be reflected by the presence of very early disease without the emergence of clinical symptoms. It is also entirely possible that the presence of these combined risk factors precipitates the development of a unique set of abnormalities that presages classic AD-related neuropathology. Conversely, other work 30 has concluded that parietal and other posterior (vs medial temporal) regions may be compromised earlier in the disease course, which is consistent with our current results. It has been noted that cortical thickness may be a superior measurement of cortical atrophy versus measures of brain volume, and our findings may indicate an earlier neurodegenerative process, which is made visible by this more sensitive methodology. 35
Also surprising was the absence of cortical thickness abnormalities in frontal association areas among the APOE-∊4 group, given prior evidence which has implicated anterior regions among individuals at risk for AD. 30,33 -36 Investigation has also revealed frontal ventricular expansion or atrophy among patients meeting clinical criteria for MCI in both cross sectional 35,36 and longitudinal 36 studies. In addition, there is increased risk for frontal regional brain involvement among individuals with CVD or risk; in particular, studies conducted in our laboratory demonstrate vulnerability to frontal brain structures in healthy elderly participants and those with subtle cerebrovascular risks. 67
Particularly problematic for frontal brain integrity is the combined impact of genetic and cerebrovascular risks, and we have recently demonstrated an interaction between metabolic dysregulation and APOE-∊4 risk on WM dysfunction (including superior frontal 65 ). Investigation has documented relationships between WM abnormalities 68 and cortical thickness in frontal brain regions in MCI and AD, although this may vary by disease severity. 30 The development of WM abnormalities and its relationship with cortical atrophy may contribute to known patterns at brain dysfunction within both anterior and posterior association areas. Our sample in particular may have unique contributions for frontal brain compromise, given the presence of dual impacts for WM change (APOE-∊4, glucose dysregulation), which is known to be associated with abnormalities in this region. 69 However, it has been suggested that while these anterior brain changes may be present, they occur at a slower rate when compared to temporoparietal changes, 28,29,30,36 particularly among earlier AD disease stages. 28,29 Therefore, posterior versus anterior regions may demonstrate a more rapid rate of decline in the development of AD, although compromise within anterior regions may also be present. The earlier onset of more posterior changes, the preclinical nature of our current sample, and the absence of a direct investigation of brain WM (which may show changes prior to cortical brain dysfunction) may explain our failure to document frontal regional abnormalities in the APOE-∊4 group.
Results among the noncarrier group demonstrated that age contributes (independently of glucose measures) to cortical thinning of temporoparietal and frontal regions and particularly within superior frontal, precuneus, transverse temporal, temporal pole, and superior temporal areas; glucose variables were not predictive of cortical thickness within any region. These results demonstrate that glucose is particularly deleterious to cortical integrity in the presence of APOE-∊4 status, but without the presence of such genetic risk, glucose is not especially problematic beyond advancing age. Cortical thinning has been documented across healthy aging populations, and processes including cellular shrinkage and reduced dendritic arborization versus neuronal loss per se are believed to account for the cortical thinning that has been observed in advancing age. 70 Research has demonstrated that cortical thinning may follow a gradient, whereby abnormalities are accelerated within associated cortical regions and followed by dysfunction of primary sensory and motor cortices during later years, with prefrontal regions showing the most striking early abnormalities. 71 It has been suggested that developmentally later myelinating regions may be more associated with age-related volumetric atrophy than is the case for earlier myelinating regions, 55 which is in support of the current prefrontal cortical and temporal/temporoparietal findings for the healthy control group. Other research 61 has demonstrated that atrophy may occur in widespread fashion across the cerebral cortex within both early and later myelinating regions (eg, prefrontal cortex, precentral gyrus, and occipital cortex). The progression of cortical abnormalities attributable to healthy aging remains unclear given the presence of these conflicting data, although it is very likely that interactions between varying risk factors in advancing age contribute to and better predict cortical thinning patterns.
Despite the cross-sectional nature of this study, the current work suggests that examination of cortical thickness may reveal pathological brain changes, particularly in individuals with cooccurring cerebrovascular and genetic risks, before the more prominent cognitive dysfunction associated with AD becomes apparent, and also highlights the prominence of cerebrovascular brain changes that may be associated with a range of glucose dysregulation. Research during the preclinical phase is particularly important since cortical brain changes may precede behavioral manifestations of disease; therefore, it is during this critical period that investigative and clinical efforts may be most fruitful. Our results support earlier preclinical detection among this susceptible subgroup and suggest that such identification may serve as a critical AD biomarker.
Limitations
Several limitations deserve comment. First, bias may exist when considering the higher prevalence of first-degree family history of dementia (87.9%), who are known to be at higher risk for dementia 72 ; our data might thus reflect stronger associations between predictor variables and brain structure. This study also included a large proportion (33.6%) of African Americans, who are known to be at higher risk for vascular conditions. 69 However, this sample characteristic also allowed us to more directly tap factors relevant to vascular risk, which might not be feasible in a lower risk group. The current study is also limited by its cross-sectional design, as we are unable to determine how genetics and vascular dysregulation confer longitudinal brain changes, particularly at the time of disease conversion. Moreover, while the majority of participants presented with normal or mildly elevated glucose, a small percentage showed clinically elevated glucose and would be considered diabetic by clinical standards (ie, 8.4%, elevated glucose and 7.4%, elevated HbA1c); thus, this sample reflects a wider range of vascular compromise than might be suggested by a strictly subclinical sample. Additionally, data on the duration of vascular risk were not available, which has been shown to be related to WM integrity among DMII 73 ; thus, our ongoing work is now collecting this critical information. We also did not specifically collect data evaluating for the presence of mild psychiatric symptomatology, although individuals with severe psychiatric history were excluded; the failure to consider more minor impacts represents an additional limitation as these factors can often have impacts of brain structure and function. When considering insufficient within cell sample size (∊3/4 = 25 and ∊4/4 = 2), we were unable to examine dose-dependent relationships of the number of copies of the ∊4 allele on WM. Nevertheless, our results were not driven solely by the 2 homozygous participants. Similarly, our modest sample and discrepant group and individual cell sizes limited our ability to directly examine interactive impacts between genotype and cortical thickness or moderating impacts of glucose on these relationships. Instead, we present evidence of a distinct pattern of relationships that appears to differ based upon genetic status. It is strongly recommended that future work seeks to better understand these relationships with a sample size sufficient for this purpose. Finally, data from a small subsample of participants (n = 4) were collected using a different scanner, and while we believe that this reflects a weakness of the present study, our results were not driven by the impact of scanner differences or the few participants comprising this subsample.
Summary
Our results demonstrate that risk factors for cortical thinning, including APOE-∊4, glucose abnormality, and age, are differentially associated with patterns of cortical thinning across frontal, temporal, and parietal brain regions in middle-aged and older adults. In particular, among APOE-∊4 carriers, glucose functioning appears to be more important than other risks (eg, age) in maintaining cortical integrity across diverse temporoparietal brain regions, and the pattern of thinning associated with these risks is consistent with the changes demonstrated in the earliest stages of AD. However, for individuals presenting without this genetic risk (noncarriers), glucose abnormality is less salient in the study of cortical integrity, whereas advancing age demonstrates significance in predicting frontal, temporal, and parietal cortical integrity. These results demonstrate that the impact of cerebrovascular risk on cortical thickness integrity may vary by the presence or absence of the APOE-∊4 allele, a well-known risk factor for AD. This study supports aggressive management of cerebrovascular factors and earlier preclinical detection of AD among this susceptible subgroup, presenting with combined genetic and vascular risks.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Neurologic Disorders and Stroke (K23NS062148 and R01NS086882), the National Institute of Nursing Research (R01NR010827 and R01NS086882), the National Institute on Aging (P60AG08812 and P01AG004390), American Psychological Association Division 40, The Rosalind and Arthur Gilbert Foundation/AFAR New Investigator Award in Alzheimer’s Disease, and by Medical Research Service VA Merit Review Awards to William Milberg and Regina McGlinchey.
