Abstract
Keywords
Background
The Alzheimer’s Disease Neuroimaging Initiative (ADNI) is a large consortium cohort study designed to examine the natural history of mild cognitive impairment (MCI) and establish biomarkers that predict progression to Alzheimer’s disease (AD). Vascular disease and other medical factors are the major contributors to cognitive decline and may have contributed both to cognitive status at baseline and cognitive decline and cardiovascular events during ADNI. 1
Gender, older age, hypertension, diabetes, smoking, atrial fibrillation, history of vascular disease, 2 and body mass index (BMI) have been well established as risk factors for vascular events. 3 Hypercholesterolemia over a wide range of cholesterol values has also been found to be a risk factor for vascular events, 4 while depression has recently been associated with the risk of vascular events. 5 Education has been found to be inversely associated with vascular events 6 and cognitive decline, but apolipoprotein E (APOE) genotype has been found to be associated with cognitive decline and stroke in a large meta-analysis. 7
In this longitudinal study, we aim to describe and compare baseline vascular risk factors and cardiovascular event rates among participants with and without cognitive impairment enrolled in ADNI.
Methods
The ADNI is a large consortium cohort study designed to test whether neuroimaging, biological, genetic, clinical, and neuropsychological markers can predict the progression of MCI and early AD. The ADNI was launched in 2004 by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, the Food and Drug Administration (FDA), private pharmaceutical companies, and nonprofit organizations. More than 800 participants, aged 55 to 90 years, have been recruited from 59 sites across the United States and Canada and followed for 2 to 3 years with neuroimaging and clinical assessments. Inclusion criteria included good general health, age 55 to 90 years, low Hachinski 8 and geriatric depression scores 9 and at least 6 grades of education. For additional information about ADNI, see www.adni-info.org.
Data
Data on screening diagnosis and baseline demographics, cardiovascular risk factors, medication, genetics, and longitudinal cardiovascular outcomes were collected from the ADNI clinical database (download version February 02, 2011). Hypercholesterolemia was defined as random blood cholesterol ≥11 mmol/L, medical history of hypercholesterolemia, or treatment with lipid-lowering medication. Diabetes mellitus (DM) was defined as random blood glucose ≥11 mmol/dL, medical history of DM, or treatment with glucose-lowering medication. Depression was defined as geriatric depression scale of ≥10 or treatment with an antidepressant. History of vascular disease was defined as a history of coronary artery disease, congestive heart failure, cerebrovascular disease, carotid artery stenosis, or peripheral vascular disease. Atrial fibrillation was defined by clinical history. Education was assessed as a continuous variable of years of education. Smoking was defined as the presence of any history of smoking. Hypertension was defined as systolic blood pressure >140 mm Hg, diastolic blood pressure >90 mm Hg, treatment with antihypertensive medication, or medical history of hypertension. All arterial blood pressure measurements were taken consistently in the same arm; when possible, the participant’s dominant arm was used. The forearm was placed at the level of the heart. Overweight was defined as BMI ≥25 kg/m2. Height and weight were measured by ADNI staff. APOE + was defined as the presence of one or more APOE4 allele.
Only participants who completed the baseline magnetic resonance imaging (MRI) were considered in this analysis. Eight participants were also excluded for failed MRI scan processing (n = 6) or missing baseline MRI data (n = 2). The primary outcome, time to first cardiovascular event (cardiovascular disease [CVD]), was defined as any myocardial infarction, stroke, revascularization, or cardiovascular death during follow-up and was obtained from the clinical database adverse event records. Death due to unknown cause was classified as cardiovascular if a noncardiovascular etiology was not specified. For each event, both event type and date were recorded.
Statistical Analyses
Pearson’s chi-square test was utilized for comparison of categorical variables and
Results
The analyzed cohort included a total of 810 consecutively enrolled participants composed of 226 healthy individuals, 400 participants with MCI, and 184 participants with AD. A maximum of 12 measurements per participant could be made over the period of 54 months. Average follow-up time was 31 months (range 0-54 months).
Baseline characteristics in the ADNI cohort are presented in Table 1. More participants (64%) in the MCI group were male (
Baseline Characteristics by Diagnosis for 810 Participants in ADNI Cohort a
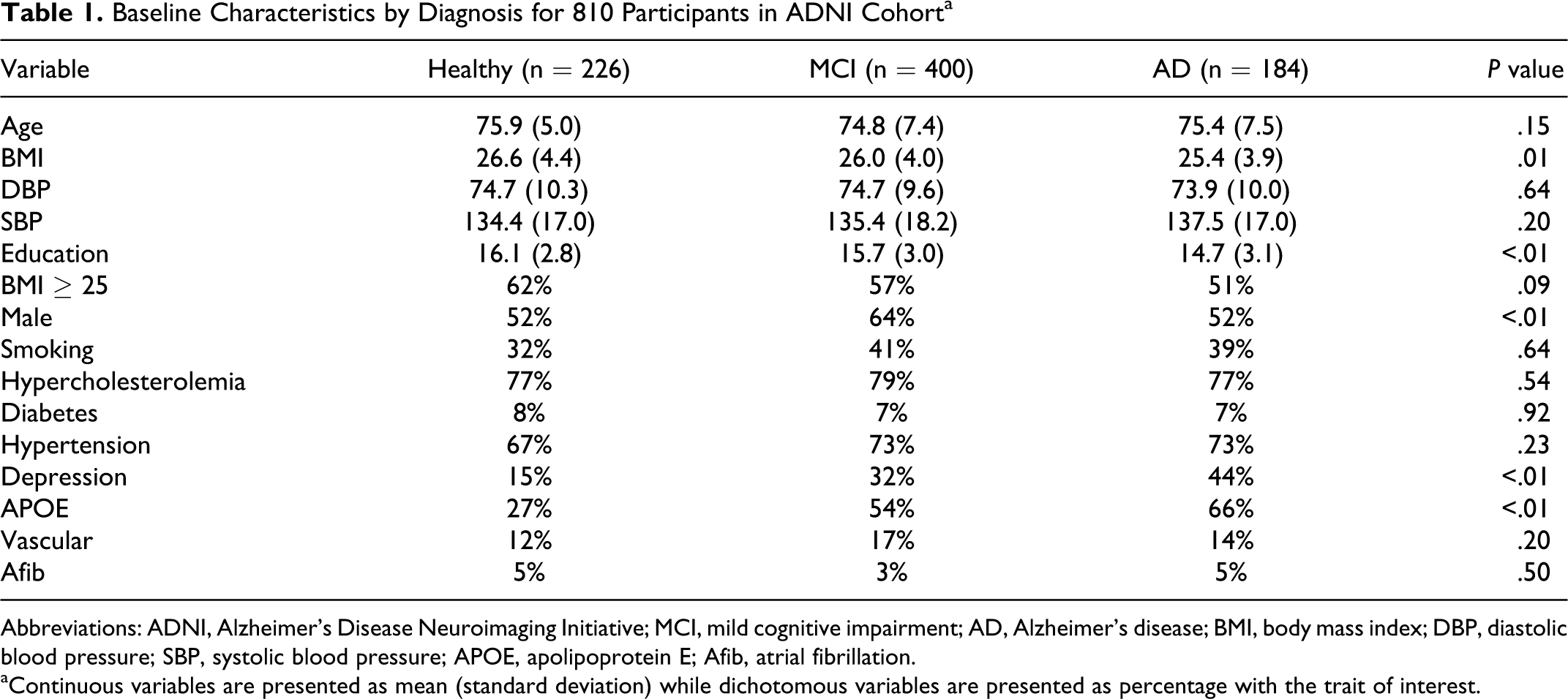
Abbreviations: ADNI, Alzheimer’s Disease Neuroimaging Initiative; MCI, mild cognitive impairment; AD, Alzheimer’s disease; BMI, body mass index; DBP, diastolic blood pressure; SBP, systolic blood pressure; APOE, apolipoprotein E; Afib, atrial fibrillation.
aContinuous variables are presented as mean (standard deviation) while dichotomous variables are presented as percentage with the trait of interest.
There were a total of 31 events including 11 strokes, 7 myocardial infarctions, 5 revascularizations, and 8 deaths. For descriptive purposes, Table 2 summarizes the CVD event rates among groups. The CVD event rates were 3.1% in the healthy individuals, 5% in the MCI group, and 2.2% in the AD group (Table 2).
Cardiovascular Event Type by Diagnosis
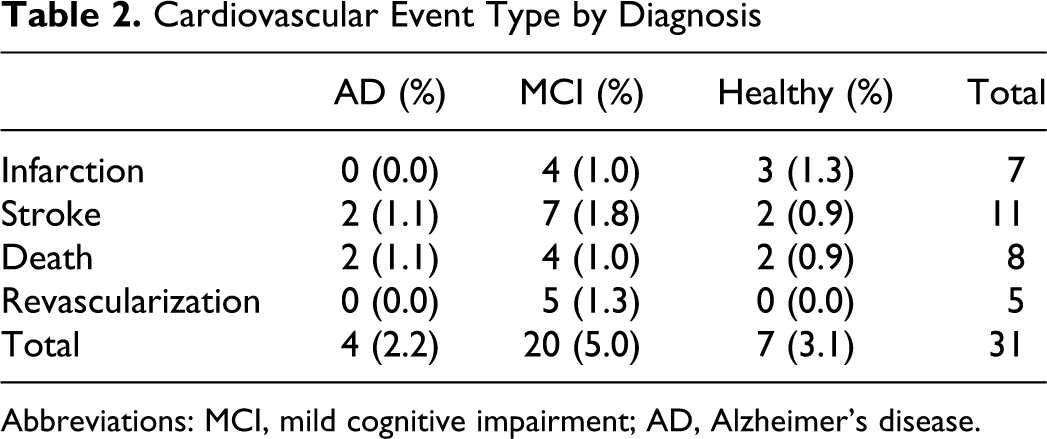
Abbreviations: MCI, mild cognitive impairment; AD, Alzheimer’s disease.
Table 3 presents the univariate and multivariate Cox proportional hazard models for the relationship between the hazard rate for the first CVD event and baseline characteristics. The univariate Cox model studies the effects of a covariate on the hazard of having a first CVD, without controlling the effects of other covariates. In contrast, the multivariate Cox model studies the effect of a covariate, after controlling for all the other observed confounders of this covariate. Male gender (
Univariate and Multivariate Cox Proportional Regression Hazards Model a
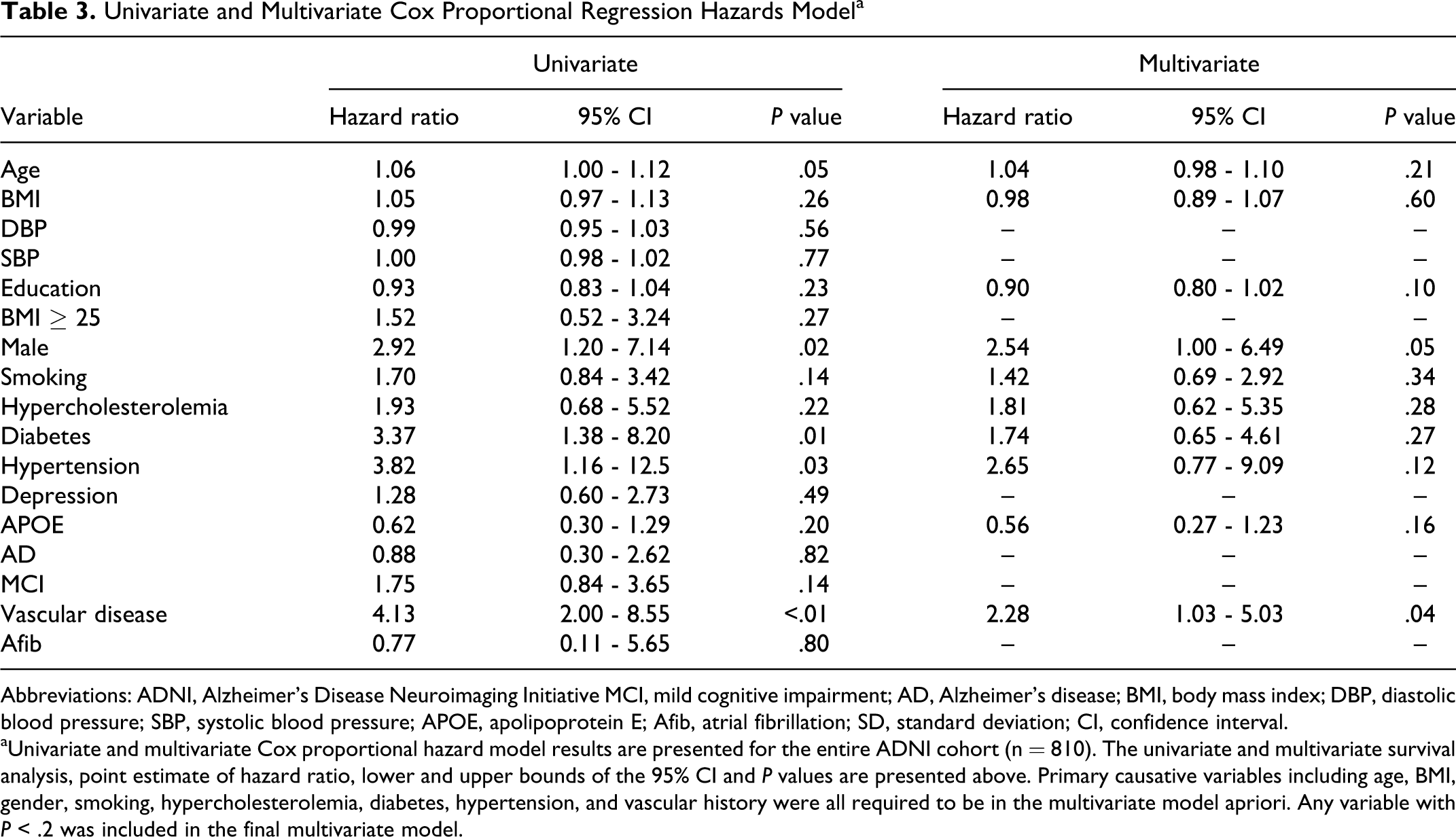
Abbreviations: ADNI, Alzheimer’s Disease Neuroimaging Initiative MCI, mild cognitive impairment; AD, Alzheimer’s disease; BMI, body mass index; DBP, diastolic blood pressure; SBP, systolic blood pressure; APOE, apolipoprotein E; Afib, atrial fibrillation; SD, standard deviation; CI, confidence interval.
aUnivariate and multivariate Cox proportional hazard model results are presented for the entire ADNI cohort (n = 810). The univariate and multivariate survival analysis, point estimate of hazard ratio, lower and upper bounds of the 95% CI and
Discussion
In this longitudinal study, we described the baseline cardiovascular risk factors and outcomes in ADNI. Despite the well-established relationship between vascular risk factors and cognitive impairment, the MCI and AD groups had comparable rates of CVD outcomes and most vascular risk factors with the healthy participants.
We also developed a multivariate model to describe the association between the baseline demographics and longitudinal outcomes. All the variables (except depression) that were expected to be associated with CVD were either significant or trending toward significance. Curiously, APOE + status was associated with less risk of CVD. The explanation for this is unclear: it may have been due to chance, covariance, survivorship bias, or selection bias (due to exclusion of patients with a history of stroke). Given the results of the multivariate model, male gender (
Event rates were low, at <2% per year, despite the mean age of 75 years. This may have been due to selection bias due to inclusion and exclusion criteria, survivorship bias, or the research environment placing the participants under heightened medical attention and follow-up. While there were no significant differences in the event rates among diagnostic groups, there were a number of baseline demographic differences between diagnostic groups.
Previously, others have reported on an association between smaller hippocampal size in AD and higher BMI in ADNI, 10 while others have reported on lower BMI being associated with faster cognitive decline in ADNI MCI participants. 11 We had previously reported on an excess of males in the MCI group, 12 which may have been due to recruitment bias. Risacher et al 13 also reported on lower educational level and higher APOE carrier status in the cognitively impaired participants. To our knowledge, this is the first report of the presence of cardiovascular risk factors and depression at baseline among the different diagnostic groups. One possible explanation for the lack of an association between CVD and depression is that the depression has been associated with both cognitive decline and pseudodementia, creating diagnostic uncertainty. Since the inclusion criteria included low geriatric depression scale, there may also have been selection bias. Limitations of this study include the study’s small sample size, limited length of follow-up, and imprecise definition of smoking and depression. Strengths of this study include its longitudinal nature and that baseline characteristics were systematically collected before events, minimizing recall, and selection biases. Further research is needed on the association between vascular risk factors and cognitive outcomes in the ADNI because differences in vascular risk factors may have contributed to cognitive decline and affected the results.
Footnotes
Authors’ Notes
All authors contributed equally to this study. Dr Xie, who is an assistant professor of biostatistics, performed the statistical analysis. Data used in the preparation of this article were obtained from the ADNI database (www.loni.ucla.edu/ADNI). As such, the investigators within ADNI contributed to the design and implementation of ADNI and/or provided data but did not participate in analysis or writing of this report. For a complete list of investigators involved in ADNI see: http://www.loni.ucla.edu/ADNI/Data/ADNI_Authorship_List.pdf.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ruland served as a speaker for Boehringer Ingelheim, consulted for Aztra Zeneca and both consulted and spoke for Otsuka.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Xie received funding from the University of Illinois at Chicago Center for Clinical and Translational Science, Award Number UL1RR029879 from the National Center for Research Resources. Data collection and sharing was funded by the ADNI (Principal Investigator: Michael Weiner; NIH grant U01 AG024904). The ADNI is funded by the National Institute on Aging (NIA), the National Institute of Biomedical Imaging and Bioengineering (NIBIB), and through generous contributions from the following: Pfizer Inc, Wyeth Research, Bristol-Myers Squibb, Eli Lilly and Company, GlaxoSmithKline, Merck & Co Inc, AstraZeneca AB, Novartis Pharmaceuticals Corporation, the Alzheimer’s Association, Eisai Global Clinical Development, Elan Corporation plc, Forest Laboratories, and the Institute for the Study of Aging, with participation by the FDA. Industry partnerships are coordinated through the Foundation for the National Institutes of Health. The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Disease Cooperative Study at the University of California, San Diego. The ADNI data are disseminated by the Laboratory of Neuro Imaging at the University of California, Los Angeles.
