Abstract
The current review aims to examine melatonin therapy for both sleep disturbances and cognitive function in dementia. We searched all randomized controlled trials published in Medline, Embase, the Cochrane Library, China National Knowledge Infrastructure, the Cochrane Dementia and Cognitive Improvement Group’s Specialized Register, and Clinical Trials.gov. The grading of recommendations assessment, development and evaluation framework was used to assess the quality of evidence. Seven studies were included (n = 520). Treated participants showed prolonged total sleep time (TST) by 24.36 minutes (
Introduction
Dementia is a common occurrence in the elderly population, accounting for 4.7% of the global population aged ≥60 years. The estimated population of individuals with dementia was across the globe is expected to double every 20 years and will reach an alarming 65.7 million in 2030. 1 Meanwhile, sleep disturbances become more common in late life and afflict between 19% and 50% of community-dwelling elderly patients affected with dementia. 2 –4 A deficiency in sleep may contribute to cognitive impairment (CI). 5,6 The mechanism of sleep deprivation in promoting dementia has been recently suggested by a pronounced effect of sleep on amyloid precursor protein metabolite clearance from interstitial space in the brain. 7 Furthermore, previous studies revealed that the majority of residents in long-term care facilities with dementia are also affected by mood disorders after disruption of normal sleep. 8 –11
Melatonin, the hormone secreted by the pineal gland, is believed to be effective in the treatment of sleep disturbance in healthy persons. 12 Disturbance in the circadian system and reduction of endogenous melatonin suggest a potential benefit of melatonin replacement therapy for dementia. Previous reviews suggested an improvement in circadian rhythm disturbances and delirium in dementia following melatonin replacement. 13,14 However, the efficacy of melatonin in both ameliorating sleep disorders and improving cognitive function in dementia is controversial in clinical studies to date. Therefore, we have performed a quantitative meta-analysis to evaluate the therapeutic effects of melatonin in patients with dementia and evaluate the clinical values of melatonin therapy for both sleep disturbances and cognitive function in dementia.
Methods
Data Sources and Literature Search
A systematic literature search was conducted on December 21, 2013, using the following databases: Medline, Embase, the Cochrane Library, and China National Knowledge Infrastructure. Search terms were employed as follows, but not limited to: “melatonin,” “N-acetyl-5-methoxytryptamine,” “dementia,” “Alzheimer Dementia,” “Dementia of Alzheimer’s type,” “Alzheimer’s Disease,” and “Vascular dementia”. A supplemental search that included ALOIS (the Cochrane Dementia and Cognitive Improvement Group’s Specialized Register), Clinical Trials.gov, and a manual search of bibliographic references of identified reviews and retrieved studies were undertaken. Corresponding authors were contacted for research details and missing data. Searches were not limited by publication date, but there was a language restriction to English and Chinese.
Study Selection
We included randomized clinical trials (RCTs) involving diagnosed dementia with medication including melatonin (any dose) versus placebo. Primary outcomes were sleep quality and assessment using the Mini-Mental State Examination, MMSE. 15 The following outcome measurements were employed to assess sleep quality: sleep efficacy, SE, that is, nocturnal total sleep time (TST)/time in bed × 100; or total time spent asleep while in bed, TST.
The MMSE is a short and reliable screening tool to assess cognitive function, with lower scores indicating greater impairment. We included secondary outcomes as follows: cognitive function measured by Alzheimer’s Disease Assessment-cognitive subscale, ADAS-cog, 16 and safety of melatonin use. The ADAS-cog measures cognitive decline associated with AD with the relevant aspects of memory, language, word recall, word-finding difficulty, and following commands (higher scores indicate poorer cognitive function). We excluded animal studies, case reports, nonrandomized trials, and trials without eligible outcome measurements. Finally, 7 articles were recruited (Figure 1).
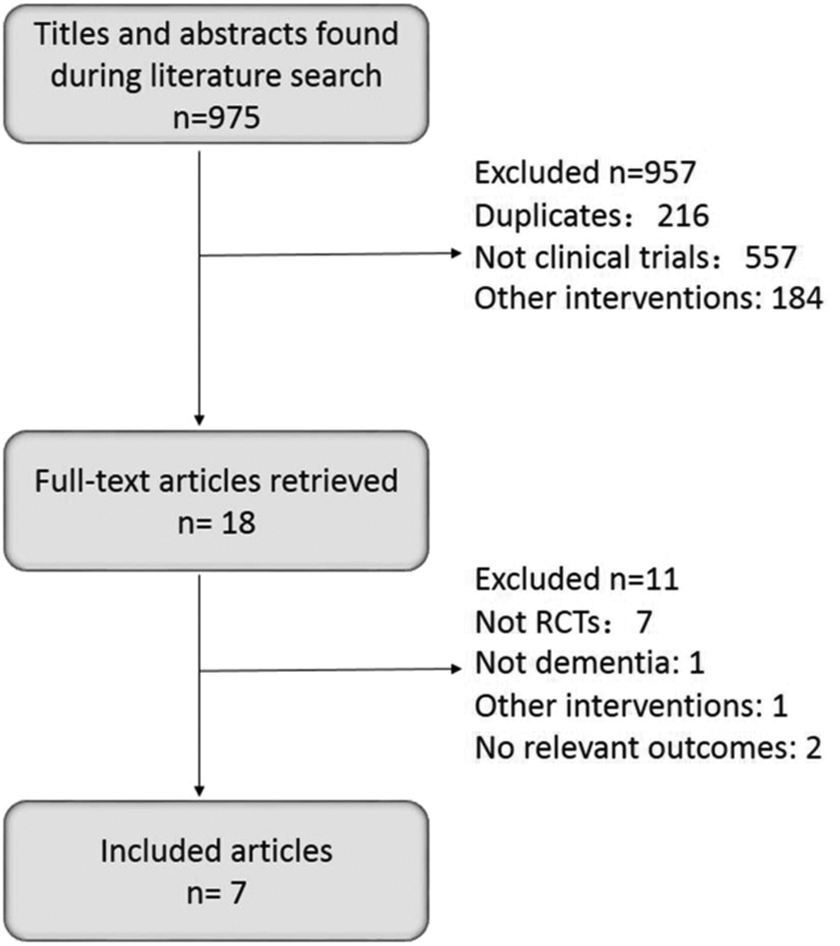
Identification of eligible studies.
Quality Assessment
Two team members (XJ and WLL) assessed the included trials independently with the Cochrane Collaboration’s tool for assessing risk of bias, 17 and the assessed domains included sequence generation; allocation concealment; blinding of participants and outcome assessors; incomplete outcome data; and selective outcome reporting.
We evaluated the quality of evidence according to grading of recommendations assessment, development and evaluation (GRADE) approach with GRADEpro software version 3.6. 18 This method rates the quality of evidence from high to very low. The quality of evidence would be downgraded according to the following aspects: limitations in design, inconsistency of results across studies, imprecision of overall estimates, indirect evidence, and publication bias.
Data Abstraction
Two team members (XJ and WLL) independently selected trials and extracted data from published reports according to a standardized spreadsheet. Disagreements were resolved through discussion. Important details including participant, intervention, comparator, and outcome were summarized (Table 1).
Characteristics of Included Studies.
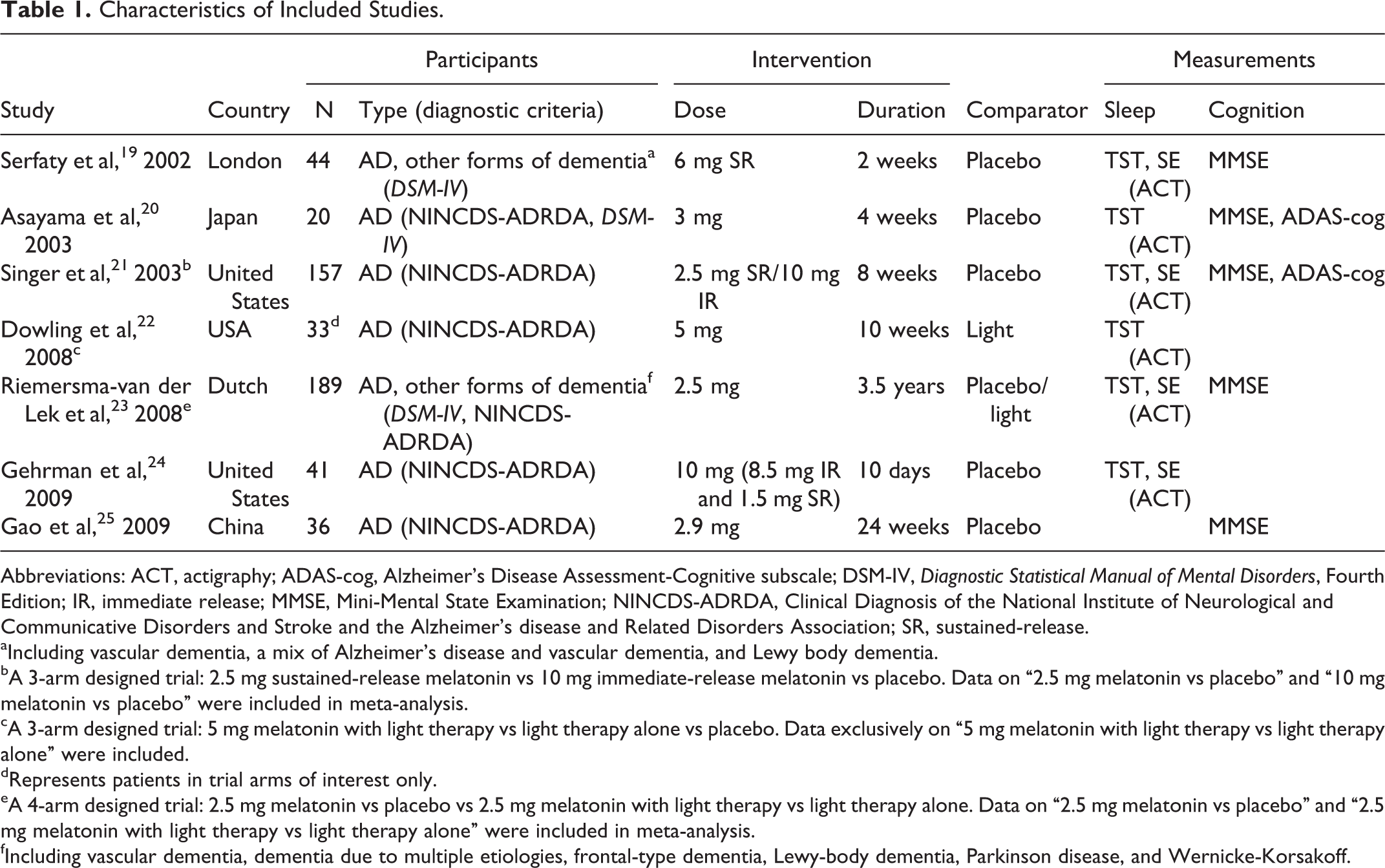
Abbreviations: ACT, actigraphy; ADAS-cog, Alzheimer’s Disease Assessment-Cognitive subscale; DSM-IV,
aIncluding vascular dementia, a mix of Alzheimer’s disease and vascular dementia, and Lewy body dementia.
bA 3-arm designed trial: 2.5 mg sustained-release melatonin vs 10 mg immediate-release melatonin vs placebo. Data on “2.5 mg melatonin vs placebo” and “10 mg melatonin vs placebo” were included in meta-analysis.
cA 3-arm designed trial: 5 mg melatonin with light therapy vs light therapy alone vs placebo. Data exclusively on “5 mg melatonin with light therapy vs light therapy alone” were included.
dRepresents patients in trial arms of interest only.
eA 4-arm designed trial: 2.5 mg melatonin vs placebo vs 2.5 mg melatonin with light therapy vs light therapy alone. Data on “2.5 mg melatonin vs placebo” and “2.5 mg melatonin with light therapy vs light therapy alone” were included in meta-analysis.
fIncluding vascular dementia, dementia due to multiple etiologies, frontal-type dementia, Lewy-body dementia, Parkinson disease, and Wernicke-Korsakoff.
Data Analysis
Meta-analysis was performed using Cochrane RevMan 5.2 Software (Review Manger Version 5.2). A random-effects method was utilized to pool studies, as it was considered a more conservative approach than alternatives. Heterogeneity was assessed using Cochran
Results
Study Characteristics
A total number of 520 patients were enrolled in the 7 studies selected. 19 –25 All studies were published in English, except 1 in Chinese. 25 Of the 7 studies, 6 were RCTs with 1 crossover trial design. 19 Due to a potential residual effect of melatonin on the progression of dementia, only the first treatment period of the crossover trial was included. Five studies compared melatonin versus placebo, 19,20,21,24,25 and 1 compared melatonin with light therapy to light therapy alone. 22 One study by Singer and coworkers 21 had 3 groups (placebo control vs 2.5 mg sustained-release melatonin vs10 mg immediate-release melatonin), and one 23 had 4 groups (placebo control vs melatonin alone vs light therapy with melatonin vs light therapy alone). Among the 7 trials, 5 employed patients with AD only, 20 –22,24,25 and 2 studies involved AD as well as vascular dementia or other types of dementia. 19,23
Risk of Bias and Quality of Evidence
Table 2 summarizes the risk of bias of included studies in this review. Most studies did not elucidate detailed methods of random sequence generation and allocation concealment. Under such circumstances, the procedures employed were judged as “low risk,” if the use of random sequences and allocation concealment was clearly described in relevant publications from the same investigators. 17 Most publications reported blinding of assessors and participants. Two studies were considered to have a high risk of bias for 1 item 19,25 and other reports had an unclear risk of bias in one aspect at least.
Risk of Bias for Included Studies.
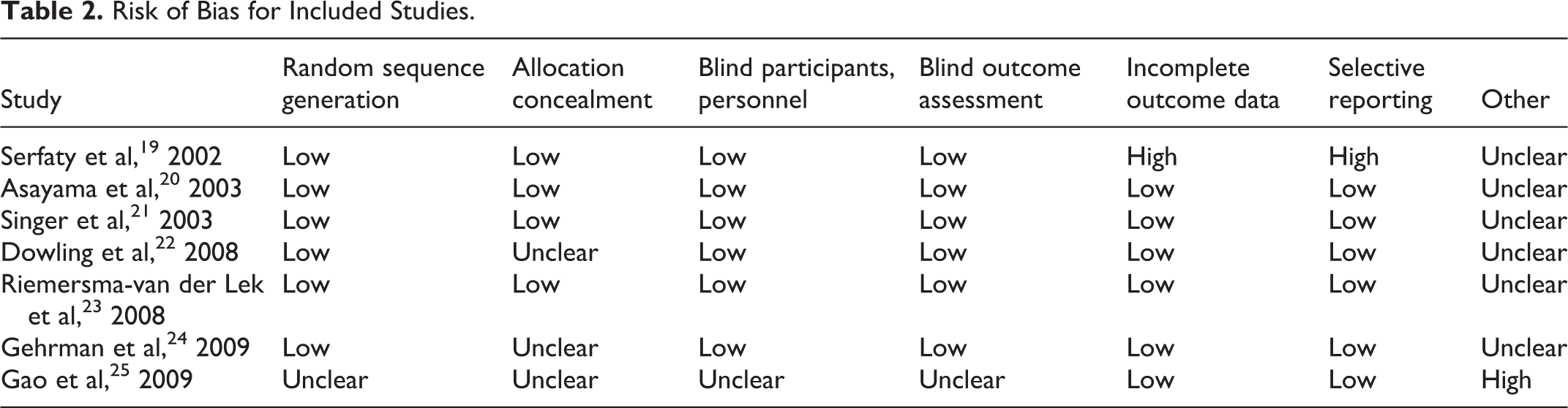
Based on the GRADE approach, downgraded factors were considered as follows: inconsistency resulting from heterogeneity, imprecision resulting from unavailable standard deviations, or a small number of participants (less than 400). We did not assess publication bias with funnel plots or regression analysis due to the dearth of eligible studies. 27 Finally, the quality of evidence was rated from moderate to low, as presented in Table 3.
Quality of Evidence, Adapted From GRADE Approach.a
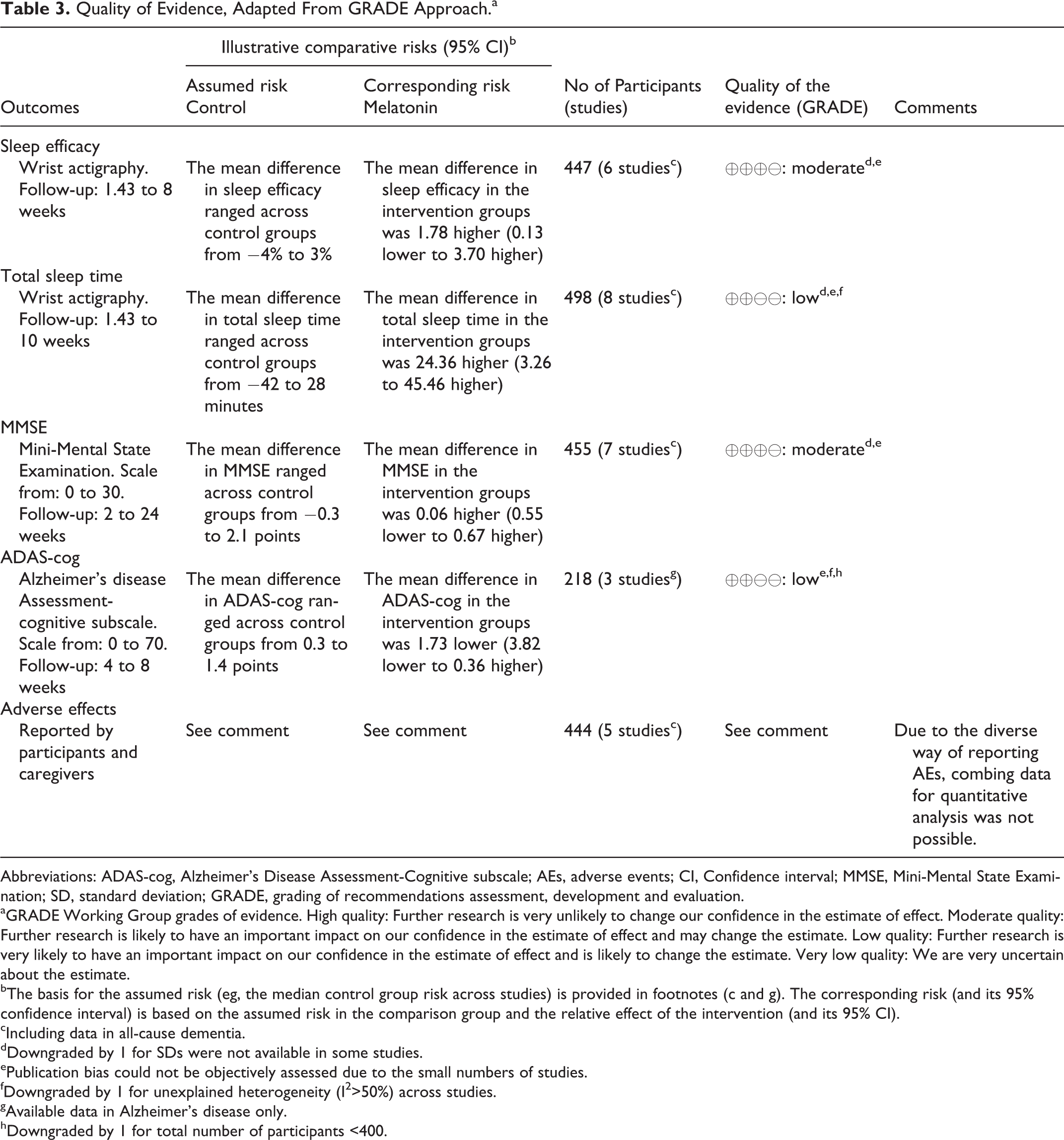
Abbreviations: ADAS-cog, Alzheimer’s Disease Assessment-Cognitive subscale; AEs, adverse events; CI, Confidence interval; MMSE, Mini-Mental State Examination; SD, standard deviation; GRADE, grading of recommendations assessment, development and evaluation.
aGRADE Working Group grades of evidence. High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate.
bThe basis for the assumed risk (eg, the median control group risk across studies) is provided in footnotes (c and g). The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
cIncluding data in all-cause dementia.
dDowngraded by 1 for SDs were not available in some studies.
ePublication bias could not be objectively assessed due to the small numbers of studies.
fDowngraded by 1 for unexplained heterogeneity (I2>50%) across studies.
gAvailable data in Alzheimer’s disease only.
hDowngraded by 1 for total number of participants <400.
Outcomes
Sleep
Sleep efficacy
Data on SE were available in 4 trials.
21
–24
Overall, there was a marginal enhancement in SE (MD = 1.78, 95% CI: −0.31 to 3.70, I2 = 25%,
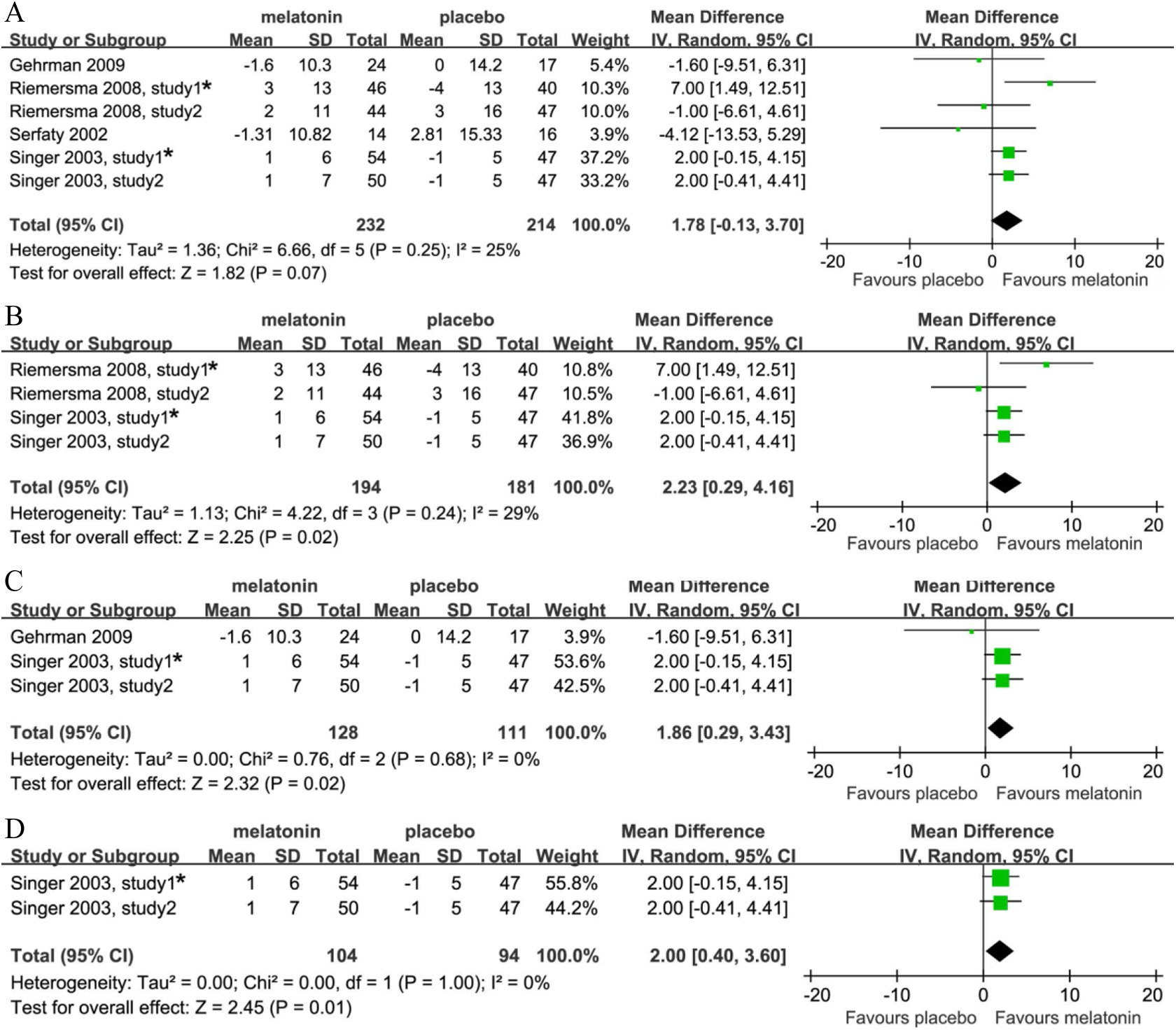
Effects of exogenous melatonin on sleep efficacy. (A) Melatonin treatment for 10 days to 8 weeks in patients with all-cause dementia; (B) melatonin treatment for at least 4 weeks in patients with all-cause dementia; (C) melatonin treatment for 10 days to 8 weeks in patients with Alzheimer’s disease (AD); and (D) melatonin for 8 weeks in patients with AD. *Riemersma 2008, study 1 represents “placebo vs melatonin” group. Reimersma 2008, study 2 represents “light therapy alone vs melatonin plus light therapy” group. Singer 2003, study 1 represents “placebo vs 2.5 mg sustained-release melatonin” group. Singer 2003, study 2 represents “placebo vs 10 mg immediate-release melatonin” group.
Total sleep time
Of all the 7 publications, 6 offered data on TST.
19
–24
Due to melatonin treatment, a statistically significant prolongation of 24.36 minutes (95% CI: 3.26-45.46, I2 = 59%,
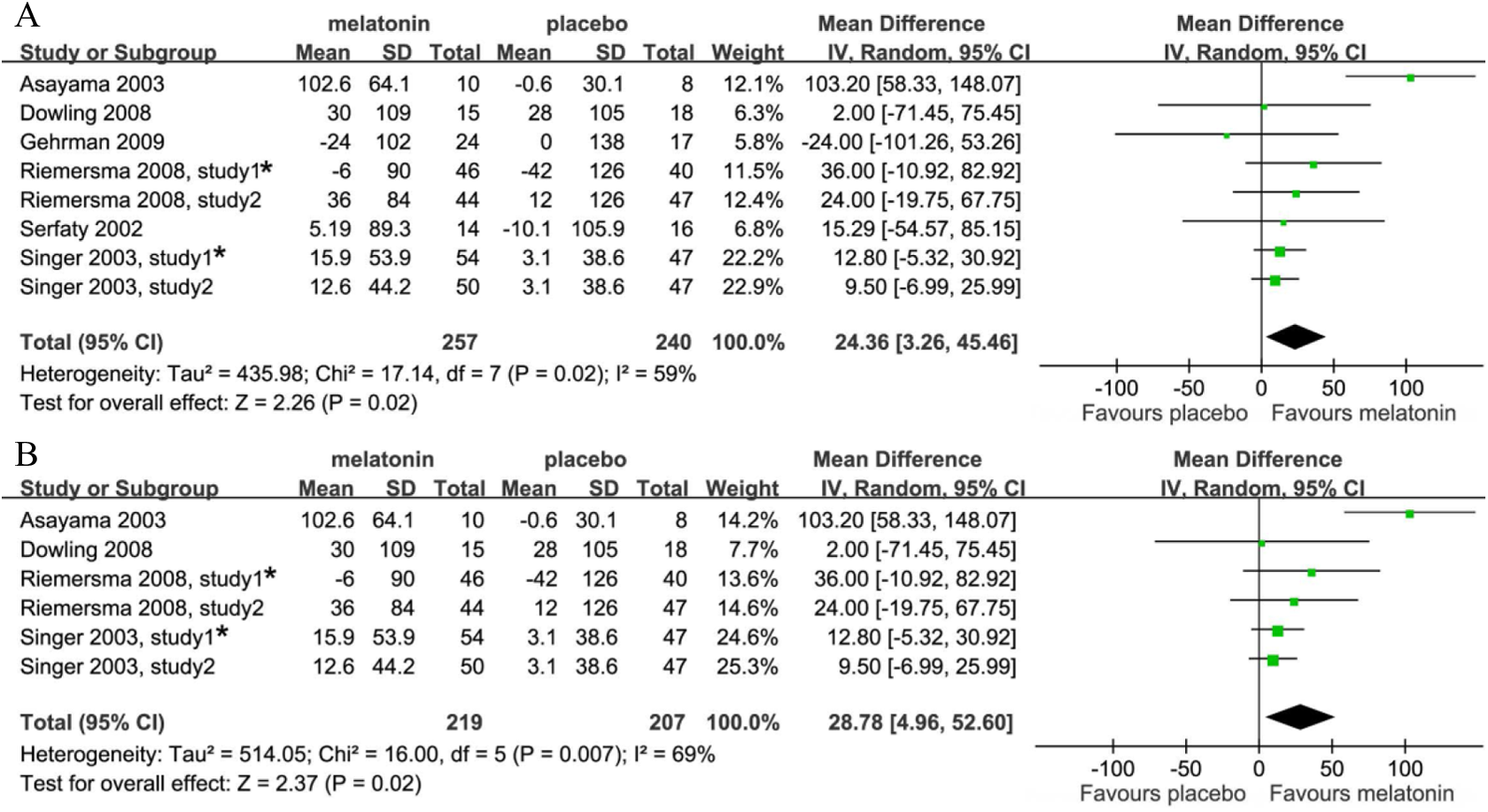
Effects of exogenous melatonin on total sleep time. (A) Melatonin treatment for 10 days to 10 weeks in patients with all-cause dementia and (B) melatonin treatment for at least 4 weeks in patients with all-cause dementia. *Riemersma 2008, study 1 represents “placebo vs melatonin” group. Reimersma 2008, study 2 represents “light therapy alone vs melatonin plus light therapy” group. Singer 2003, study 1 represents “placebo vs 2.5 mg sustained-release melatonin” group. Singer 2003, study 2 represents “placebo vs 10 mg immediate-release melatonin” group.
Cognition
Mini-Mental State Examination
Five studies reported MMSE.
19,20,21,23,25
Overall, melatonin improved MMSE by 0.06, which was insignificant in dementia of any etiology (95% CI: −0.55-0.67, I2 = 0%,
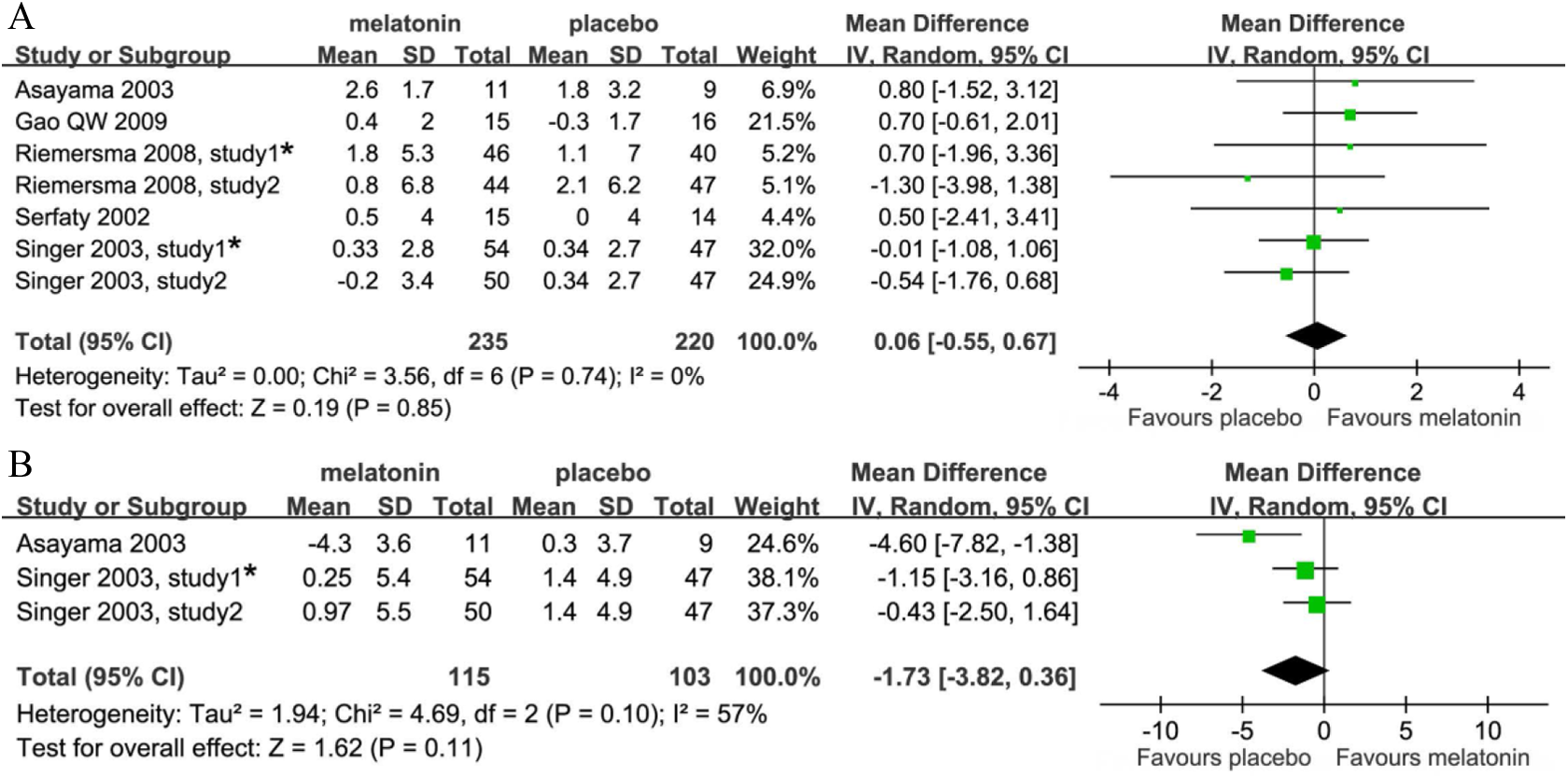
Effects of exogenous melatonin on cognitive function in patients with all-cause dementia. (A) Mini-Mental State Examination (MMSE); (B) Alzheimer’s disease Assessment-Cognitive subscale (ADAS-cog). *Riemersma 2008, study 1 represents “placebo vs melatonin” group. Reimersma 2008, study 2 represents “light therapy alone vs melatonin plus light therapy” group. Singer 2003, study 1 represents “placebo vs 2.5 mg sustained-release melatonin” group. Singer 2003, study 2 represents “placebo vs 10 mg immediate-release melatonin” group.
Alzheimer’s Disease Assessment-Cognitive subscale
Two trials assessed the impact of melatonin on cognitive function by ADAS-cog
20,21
and 1 trial used a 3-arm design.
21
All of them included patients with AD exclusively for 4 to 8 weeks of treatment. Similarly, quantitative synthesis did not present a significant improvement in ADAS-cog (MD = −1.73, 95% CI: −3.82-0.36, I2 = 57%,
Report of Adverse Events
Among the 7 studies, 3 of them collected data on adverse events (AEs) due to melatonin associated treatment.
21,23,19
In 1 study, a conclusion of no AEs was drawn, as none were reported by caregivers.
19
No severe AEs were found in 2 other studies.
21,23
One study reported AEs as abnormal behavior, aches/pain, falls, gastrointestinal distress, infection, respiratory/pulmonary symptoms, skin/subcutaneous tissue, and urinary symptoms, but no differences existed in mean number, severity, seriousness, or relatedness ratings of spontaneously reported AEs between placebo and the melatonin-treatment group.
21
One study revealed drowsiness and irritability as most commonly seen AEs, and symptoms of headache, hyperactivity, sweating, and trembling hands were reported more in melatonin-treated individuals than in the placebo group. However, all AEs failed to reach a significant
Discussion
According to published literature including case reports and open-label trials assessing the therapeutic value of melatonin in dementia, this study represents the first to quantitatively analyze these effects employing randomized controlled trials. The present meta-analysis suggested that patients with dementia eventually benefited from increased SE and TST after melatonin therapy, but without an improvement in cognition as measured by MMSE or ADAS-cog. In respect to Alzheimer’s type dementia, SE was enhanced by exogenous melatonin treatment; however, TST and cognitive function were not influenced at all.
We found that the most popular method for recording sleep used by previous reports was actigraphy (ACT) rather than polysomnography (PSG). Actigraphy is accepted as a reliable and uniform method for studying sleep and had an excellent correlation with PSG in dementia. 21,28 Although PSG is regarded as the gold standard, it requires in-depth understanding for adherence to guidelines of use, and poor adherence to these guidelines may have compromised its application in advanced stages of dementia. 29
Sleep quality is a broad concept comprising both objective sleep parameters and subjective sleep quality. In the present analysis, we evaluated both SE and TST from ACT as objective factors for their generally ascribed value in assessing the efficacy of hypnotic drugs in clinical trials. Both improvement in SE and extension of TST indicate a better subjective sleep quality and resulted in a subjective improvement in well-being of patients after awakening.
12
The present study suggests efficacy as measured by SE and TST in patients with dementia medicated with melatonin, with a more appreciable effect following a longer duration of therapy. Within the confines of the current studies analyzed in our meta-analysis, improvement in SE and prolonged TST were more likely significant following continued therapy for at least 4 weeks. No substantial differences were found when analyzing patients with AD exclusively and comparing to population samples of general dementia. In terms of other sleep parameters, the measurements and reports varied in different studies. The analysis of 3 studies indicated no difference in daytime napping as an effect of melatonin medication (MD = −11.08, 95% CI: −25.47-3.30, I2 = 43%,
The second finding of our meta-analysis is that melatonin treatment failed to improve cognitive function in patients with dementia. Among the included studies, only 1 reported beneficial effects of melatonin as measured by MMSE.
25
In this study, a significant (
Finally, we assessed the safety of melatonin therapy for people with dementia. Few studies have investigated AEs due to administration of melatonin. For those publications reporting AEs, the melatonin treatment group was similar to placebo, considering the incidence and type of AEs, indicating that melatonin was safe in patients with dementia. This finding supports previous studies of melatonin therapy in elderly people. 33 Melatonin provided a more tolerable choice for the elderly population compared to most benzodiazepines, which exert side effects that exacerbate impairment of memory and cognitive function.
There are certain limitations to be considered in our meta-analysis. Although SE and TST were analyzed, the dearth of literature and disunity in measurements do limit the application of quantitative analysis across other parameters including sleep onset latency, duration of awakenings at night, and daytime sleep time. Besides, the range of melatonin doses used is huge, from 1.5 mg to 10 mg. When combining data on TST for all-cause dementia, there is significant heterogeneity, particularly considering the study of Asayama and coworkers. The exclusion of this outlier decreased I2 with a similar result (MD = 12.33, 95% CI: 1.44-23.23, I2 = 0%,
The present meta-analysis concludes that melatonin may have a positive effect in ameliorating sleep disturbances in people with dementia, especially patients with circadian rhythm disturbances. Completion of randomized trials over extended periods, and across the wide range of doses utilized in the primary studies reviewed here, fulfilling sensitive measurements with regard to amelioration of cognitive decline are required to examine the possible effect of melatonin on functional ability and cognition in the future. Given the recent findings that sleep promotes amyloid clearance, 7 it would also be of interest for future studies of a broad scope and appeal to correlate sleep patterns of individuals over long periods of life to their postmortem amyloid plaque burden.
Footnotes
Authors’ Note
Jing Xu and Li-Ling Wang contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Basic Research Development Program of China (No. 2010CB945200, 2011CB504104), the National Natural Science Foundation of China (No. 81171027, 81200842, 91332107), National “Twelfth Five-Year” Plan for Science & Technology Support (No.2012BAI10B03), a Shanghai Key Project of Basic Science Research (No.09DZ1950400) and the Program for Outstanding Medical Academic Leaders (No. LJ 06003).
