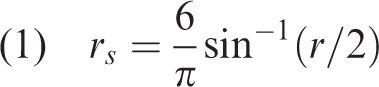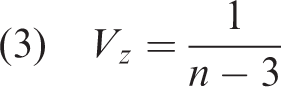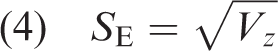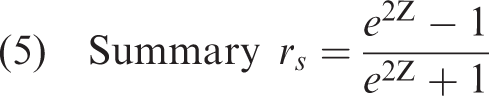Abstract
Objective
To evaluate the correlation between serum vitamin D levels and neurological function prognosis, including the overall cognitive level, in patients with ischemic stroke in a meta-analysis using Pearson or Spearman correlation coefficients.
Methods
PubMed, Cochrane Library, Web of Science, and Embase databases were searched for relevant studies published from inception to June 2025. Stata version 16.0. was used to conduct the meta-analysis.
Results
A total of fifteen studies involving 2030 participants aged 18–94 years were included. The meta-analysis demonstrated a significant inverse association between serum vitamin D levels and stroke severity and outcomes, as reflected by the admission Modified Rankin Scale score, admission National Institutes of Health Stroke Scale score, and Modified Rankin Scale score at 3 months (combined Fisher’s Z/r: −0.37/−0.35, 95% confidence interval: −0.68 to −0.06; −0.46/−0.43, 95% confidence interval: −0.55 to −0.37; and −0.21/−0.21, 95% confidence interval: −0.38 to −0.05, respectively). Higher serum vitamin D levels may be positively correlated with post-stroke cognitive improvement (combined Fisher’s Z = 0.34, 95% confidence interval: 0.24 to 0.45).
Conclusion
Serum vitamin D levels in patients with ischemic stroke were significantly negatively correlated with neurological function prognosis and, possibly, better cognitive outcomes.
Keywords
Introduction
Ischemic stroke (IS) remains the leading cause of disability worldwide and constitutes 85% of all cerebrovascular disease cases. 1 Recent Global Burden of Disease estimates indicate that the absolute burden of stroke-related disability has increased globally, with notable increases observed in several regions, including East Asia in recent years. 2 Although acute-phase reperfusion therapies such as intravenous thrombolysis and mechanical thrombectomy have considerably reduced mortality rates, short- and long-term outcomes remain heterogeneous. Over 40% of patients develop moderate to severe neurological disability and approximately 35% suffer from cognitive impairment, which significantly affects their long-term quality of life. 3 According to emerging evidence, nutritional and metabolic statuses may influence stroke prognosis beyond traditional vascular risk factors.4,5
Serum vitamin D plays a key role in calcium–phosphorus metabolism and bone homeostasis; additionally, it exerts neuroprotective effects through the activation of brain vitamin D receptors (VDRs), promoting neurotrophic factor production and suppressing nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–mediated neuroinflammation. 6 Several studies have examined the association between serum vitamin D levels and IS outcomes. For instance, Zeng et al. 7 have reported that serum vitamin D levels <38.4 nmol/L are significantly associated with poor 5-year prognosis (odds ratio (OR) = 6.36). Samarakoon et al. 5 have demonstrated that vitamin D deficiency increases the risk of poor 30-day outcomes (Modified Rankin Scale (mRS) score >2, p = 0.001). Similarly, the risk of recurrence is increased among patients with low serum vitamin D levels at the index stroke event. 8 Conversely, Xu et al. 9 observed that the protective effects of vitamin D were limited to nonhyperglycemic patients (hazard ratio (HR) = 1.58), with no significant impact under hyperglycemic conditions. Randomized studies have also produced mixed results, with high-dose vitamin D intervention failing to yield significant reductions in National Institutes of Health Stroke Scale (NIHSS) scores (p > 0.05).4,10 Although vitamin D deficiency has consistently been linked to elevated stroke risk,11,12 evidence regarding its impact on post-stroke neurological outcomes, including the NIHSS score, mRS score, and cognitive function, remains conflicting, with few studies providing a systematic quantitative synthesis.
Hence, we sought to elucidate the association between serum vitamin D levels and post-stroke neurological outcomes in IS patients through a systematic review and meta-analysis, with subgroup analyses performed to examine the sources of heterogeneity and inform prognostic assessment and therapeutic strategies.
Materials and methods
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 13 The study protocol has been published by the International Prospective Register of Systematic Reviews (PROSPERO, ID: CRD 420251077645). As the study was based on published data, ethical approval and patient consent were not required.
Search strategy
Two investigators independently searched PubMed, Cochrane Library, Web of Science, and Embase with no language restrictions. The search was conducted using the terms “Ischemic stroke” and “Vitamin D” and related expressions such as “Ischemic Stroke*,” “Ischaemic Stroke*,” “Acute Ischemic Stroke*,” “Cryptogenic Ischemic Stroke*,” “Cryptogenic Embolism Stroke*,” “Cryptogenic Stroke*,” and “Wake-up Stroke*.” An asterisk (*) was used as a truncation/wildcard symbol, where permitted by each database, to retrieve variations of the same word root (e.g. plural forms or alternative word endings). The full search strategy is detailed in Table S1. The search spanned from database inception to June 2025. Given the concerns that grey literature is typically not peer-reviewed and may lack adequate methodological descriptions and reliable data, our inclusion criteria included peer-reviewed journal articles. 14
Inclusion and exclusion criteria
The inclusion criteria were as follows: (a) patients with first-ever IS diagnosed using head computed tomography (CT) or magnetic resonance imaging (MRI), meeting the trial of Org 10172 in acute stroke treatment (TOAST) classification or World Health Organization criteria, aged ≥18 years; (b) serum 25-hydroxyvitamin D (25-(OH)D) levels measured within 24 h of admission; (c) functional outcomes assessed using at least one of the following scales: mRS, NIHSS, Mini-Mental State Examination (MMSE), and Montreal Cognitive Assessment (MoCA); and (d) study type: case–control studies, cohort studies, or cross-sectional studies.
The exclusion criteria were as follows: (a) studies involving non-IS patients or patients with diseases severely affecting vitamin D metabolism; (b) systematic reviews, meta-analyses, case reports, conference papers, animal studies, or letters; (c) studies with unavailable full-text articles; (d) duplicate publications or those without valid data; (e) nonEnglish language literature; and (f) low-quality studies with Newcastle–Ottawa Scale (NOS) scores <4 or Agency for Healthcare Research and Quality (AHRQ) scores <3.
Literature screening and data extraction
Two researchers independently screened the studies in accordance with the prespecified inclusion and exclusion criteria, and Endnote X9 was used for literature management. Any disagreements were resolved through discussion. Following the removal of duplicates, studies were screened by title and abstract, after which eligible articles underwent full-text review during the second stage of screening. Inclusion decisions were finalized jointly with basic data extracted and cross-checked for accuracy and standardized unit reporting. Discrepancies were resolved using input from a third reviewer. The extracted variables included study title, first author, year of publication, country, sample size, demographics, correlation coefficients, and outcome measures.
Literature quality assessment
Researchers assessed the literature against the inclusion/exclusion criteria. In cases of disagreement, the results were reviewed by a third investigator. The NOS was utilized for case–control and cohort studies to evaluate study population selection (0–4 points), group comparability (0–2 points), and exposure/outcome ascertainment (0–3 points). Scores of 7–9 were classified as high quality. Cross-sectional studies were evaluated using the AHRQ checklist, comprising 11 items rated as “Yes” (low risk, 1 point), “No” (high risk, 0 points), or “Unclear” (insufficient/ambiguous information, 0 points). Scores of 8–11 indicate high quality.
Outcome indicators
Stroke severity and functional outcomes were evaluated using the NIHSS and mRS. Cognitive function in patients with IS was evaluated using both MoCA and MMSE. Associations between serum vitamin D levels and stroke severity, functional recovery, and cognition were quantified using Pearson’s or Spearman’s correlation coefficients (r or rs). For the meta-analytic synthesis, r values were converted to rs as a unified outcome measure.
Statistical analyses
This study extracted baseline mRS and NIHSS scores within 24 h of admission and 3-month mRS scores. The baseline and 3-month mRS scores were analyzed separately to mitigate repeated measurement bias. Statistical analyses were conducted using StataSE (version 16.0; Stata StataCorp, College Station, TX, USA). The I2 statistic was used to evaluate heterogeneity, with I2 > 50% indicating substantial heterogeneity between the studies, and a random-effects model was used. Conversely, a fixed-effects model was employed if heterogeneity was low. The combined r values and 95% confidence intervals (CIs) for each study were calculated and presented as forest plots. Subgroup and sensitivity analyses were conducted to investigate the potential sources of heterogeneity. Publication bias was evaluated using funnel plots, Begg’s test, and Egger’s test, and statistical significance was set at α = 0.05 and p <0.05. Prior to meta-analysis, extracted correlation data were transformed according to standardized formulas:
Results
Literature search process
In total, 1218 articles were retrieved, including 191 from PubMed, 462 from Web of Science, 481 from Embase, and 84 from The Cochrane Library. After reviewing titles and abstracts, 1109 articles were excluded. Fifteen articles were included after full-text retrieval, reading, and quality evaluation. The literature search process and results are demonstrated in Figure S1.
Basic characteristics of the included studies
Fifteen studies were included, with four15–18 conducted in China, four19–22 in Egypt, three23–25 in India, and one each in Brazil, 26 South Korea, 27 Montenegro, 28 and Iran. 29 All were hospital-based studies with inpatient samples collected from 2014 to 2024 and included 2030 patients with IS in total, with sample sizes ranging from 25 to 364. Participants’ ages ranged from 18 to 94 years. Among these, five were case–control studies, six were cohort studies, and four were cross-sectional studies. The characteristics of the core study are presented in Table 1.
Basic characteristics and quality assessment of the included studies.

NOS: Newcastle–Ottawa Scale; AHRQ: Agency for Healthcare Research and Quality; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; K-MMSE: Korean Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; ELISA: enzyme-linked immunosorbent assay; CMIA: chemiluminescent microparticle immunoassay; CLIA: chemiluminescence immunoassay; ECLIA: electrochemiluminescence immunoassay.
Quality assessment
Of the 15 studies, five case–control and six cohort designs were evaluated using the NOS, with scores ranging from 6 to 9. Specifically, one study scored 6, three scored 7, five scored 8, and two scored 9. The four cross-sectional studies were assessed using the AHRQ checklist: two studies scored 7, one scored 8, and one scored 10. Detailed individual item scores for each study are available in Table S2. Overall, 12 studies were judged to be of high quality, demonstrating effective confounding controls, sound group comparability, and adequate follow-up. Lower-quality studies tended to have smaller sample sizes, limited confounder adjustment, incomplete follow-up reporting, and unclear case/control definitions. Nevertheless, the overall quality of the included studies was high.
Meta-analysis results of the included studies
Association between vitamin D levels and mRS scores in IS
Four studies explored the relationship between serum vitamin D levels and baseline mRS scores at admission for IS. As illustrated in the forest plot (Figure 1(a)), a significant negative correlation was noted (combined Fisher’s Z = −0.37, 95% CI: −0.68 to −0.06; combined r = −0.35, 95% CI: −0.59 to −0.06), indicating that elevated vitamin D levels are linked to improved functional status. Heterogeneity was substantial (I2 = 88.1%, p = 0.000). Five studies that examined the relationship between vitamin D levels and mRS scores at 3 months post-IS were included. The combined effect size indicated a negative correlation between vitamin D levels and mRS scores at 3 months (combined Fisher’s Z = −0.21, 95% CI: −0.38 to −0.05; combined r = −0.21, 95% CI: −0.36 to −0.05) (Figure 1(b)). Similar to the baseline analysis, this result demonstrated considerable heterogeneity (I2 = 57.7%, p = 0.051). Subgroup analysis (Table 2) indicated that income level, sample size, and study design contributed to heterogeneity, although these factors were insufficient to explain the variability.

Forest plots of the correlation (a) between vitamin D levels and baseline mRS scores and (b) between vitamin D levels and 3-month mRS scores in ischemic stroke patients. mRS: modified Rankin Scale.
Subgroup analysis of the correlation between vitamin D levels and mRS scores in ischemic stroke patients.

mRS: modified Rankin Scale; CI: confidence interval.
Association between vitamin D levels and NIHSS scores in IS
In total, 13 studies were included in the analysis of the correlation between serum vitamin D levels and NIHSS scores after IS. The combined effect size indicated a significant negative correlation between vitamin D levels and NIHSS scores (combined Fisher’s Z = −0.46, 95% CI: −0.55 to −0.37; combined r = −0.43, 95% CI: −0.50 to −0.35) (Figure 2). This study revealed moderate heterogeneity (I2 = 69.5%, p = 0.000, p < 0.0001). The subgroup analysis (Table 3) suggested that part of the heterogeneity stemmed from income levels and sample sizes. Studies from high-income countries reported more consistent results, whereas studies with larger samples exhibited greater heterogeneity, although the overall results remained significant. Regarding the study type, all cohort, case–control, and cross-sectional studies exhibited high heterogeneity. Furthermore, studies that used the enzyme-linked immunosorbent assay (ELISA) immunosorbent assay for vitamin D detection showed relatively low heterogeneity.

Forest plot of the correlation between vitamin D levels and NIHSS scores in ischemic stroke patients. NIHSS: National Institutes of Health Stroke Scale.
Subgroup analysis of the correlation between vitamin D levels and NIHSS scores in ischemic stroke patients.

NIHSS: National Institutes of Health Stroke Scale; CI: confidence interval; ELISA: enzyme-linked immunosorbent assay; CMIA: chemiluminescent microparticle immunoassay; CLIA: chemiluminescence immunoassay; ECLIA: electrochemiluminescence immunoassay.
Association between vitamin D levels and overall cognitive function after IS
Based on a fixed-effects model, the meta-analysis of two studies indicated a positive correlation between increased serum vitamin D levels and improved overall cognitive function post IS, with a combined Fisher’s Z value of 0.34 (95% CI: 0.24 to 0.45) (Figure S2). The two studies were highly consistent (I2 = 0.0%, p = 0.323). However, the limited number of included studies constrains the stability and generalizability of this association, and further validation is required before firm clinical conclusions can be established.
Publication bias and sensitivity analyses
Begg’s test (p = 0.734, p = 0.221), Egger’s test (p = 0.634, p = 0.620), and funnel plot analysis (Figure S3, Figure S4) were conducted to examine the correlation of serum vitamin D levels with baseline and 3-month mRS scores and revealed no evidence of significant publication bias (p > 0.05). Sensitivity analysis for the relationship between vitamin D and baseline mRS scores (Figure S5) revealed that exclusion of the study by Kim et al. 27 marginally altered the result stability (95% CI: −0.83 to 0.05); however, the overall negative correlation persisted. For the association between vitamin D levels and 3-month mRS scores (Figure S6), studies by Flifel et al. 20 and Alfieri et al. 26 notably influenced the aggregate effect sizes. Analysis of the correlation between vitamin D levels and NIHSS scores further supported the robustness, with Begg’s test (p = 0.951), Egger’s test (p = 0.957), and funnel plot (Figure S7) showing no significant publication bias. Further sensitivity analysis (Figure S8) showed that excluding individual studies did not substantially affect the pooled effect size for the correlation between vitamin D and NIHSS scores, which remained at approximately −0.46, indicating the robustness of the findings.
Discussion
IS poses a major public health challenge owing to its substantial prevalence, disability, mortality, and recurrence risks, which severely impair the patient’s quality of life and impose a major societal burden. 1 Functional prognosis after IS is influenced by multiple factors, with emerging data highlighting the neuroprotective potential of vitamin D. 30 In this study, we conducted a meta-analysis involving 15 studies (total sample size = 2030) and found that higher vitamin D levels within 24 h of hospital admission in IS patients are associated with lower NIHSS, baseline mRS, and 3-month mRS scores and better overall cognitive function.
Previous meta-analyses have predominantly highlighted the association between vitamin D levels and IS risk. Zhou et al. 31 found that vitamin D deficiency increased the risk of stroke onset, whereas Vergatti et al. 8 have reported that higher vitamin D levels at the index event are associated with a reduced risk of recurrence. Fusaro et al. 32 have reported that vitamin D supplementation may reduce the stroke risk by 13%. In contrast, our study specifically evaluated the relationship between serum vitamin D levels and functional outcomes, such as mRS score, NIHSS score, and cognitive function, providing direct clinical relevance for patient recovery trajectories. Similarly, a randomized controlled trial by Borowicz et al. 33 has reported that vitamin D3 supplementation (2000 IU daily) significantly improved Barthel Index scores in IS patients (β = 0.07, p = 0.006), aligning with our observed negative correlation between serum vitamin D levels and mRS scores. Furthermore, this study conducted a detailed subgroup analysis, covering large sample sizes, cohort studies, acute-phase patients, and middle-to-low-income populations, revealing differences across different groups. However, notably, Jarrar et al. 34 have reported that there were no significant functional improvements at 3 months after vitamin D supplementation, and other randomized controlled trials (RCTs) and systematic reviews similarly found limited benefits in post-stroke recovery over 3 months.35,36 These conflicting findings indicate that the role of vitamin D may be dose-dependent and nonlinear or even U-shaped effect. Both, a 2018 Polish expert panel guideline and a multi-expert consensus recommend defining serum 25(OH)D levels of 30–50 ng/mL as the optimal range associated with maximal health benefits for various outcomes.37,38
Vitamin D affects the severity and outcomes of IS via diverse biological mechanisms. During cerebral ischemia, vitamin D markedly increases VDR expression in the microglia and macrophages, suppressing proinflammatory cytokine secretion (e.g. tumor necrosis factor alpha (TNF-α) and interferon gamma (IFN-γ)), thereby limiting neuronal injury. 39 Vitamin D3 deficiency elevates matrix metalloproteinase (MMP-9) activity and downregulates tight junction proteins (occludin and claudin-5), resulting in increased blood–brain barrier permeability and greater cerebral edema. 40 Moreover, vitamin D mitigates oxidative stress by decreasing reactive oxygen species (ROS) generation, inhibiting caspase-3 and related apoptotic proteins, supporting brain-derived neurotrophic factor (BDNF) expression, enhancing neuroplastic capability, and contributing to both neurological and cognitive recovery. 41 It also promotes regulatory T-cell function while inhibiting Th17 proinflammatory cells, thereby modulating immune responses and diminishing secondary injury. 42 Collectively, these neuroprotective, anti-inflammatory, blood–brain barrier-stabilizing, antioxidant, anti-apoptotic, and immunoregulatory effects provide a biological basis for the beneficial associations observed with stroke outcomes.
We found that higher serum 25(OH)D levels were associated with better cognitive performance after stroke. However, owing to the relatively small number of included studies, these results are exploratory rather than confirmatory. Epidemiological studies have shown that vitamin D deficiency is linked to accelerated cognitive decline and an increased incidence of dementia.43,44 Vitamin D deficiency increases the risk of dementia and Alzheimer’s disease by approximately 1.5-fold and the risk of cognitive impairment by approximately 34%. 45 Vitamin D deficiency is common in patients with Parkinson’s disease and is associated with poor cognitive performance. Furthermore, it has been identified as an independent risk factor for post-stroke cognitive impairment.46,47 A recent meta-analysis of RCTs only showed a small but significant improvement in overall cognition with vitamin D treatment (Hedges’ g ≈ 0.13). However, the overall clinical trial evidence remains equivocal.48,49 The potential neuroprotective mechanisms of vitamin D include the upregulation of neurotrophic factors that support synaptic plasticity and neuronal survival (e.g. BDNF). It may further mitigate chronic neuroinflammation through modulation of immune response. 50 Vitamin D reduced oxidative stress and glutamate excitotoxicity. It also stabilized neuronal calcium homeostasis. Vitamin D helps maintain the integrity of the blood–brain barrier through both genomic and nongenomic pathways. This may counter barrier dysfunction observed in cognitive decline.50,51 To further clarify these associations, future studies should use standardized cognitive function measures corrected for education and stroke characteristics. Researchers should also combine dynamic vitamin D monitoring with biomarkers such as BDNF in IS patient cohorts in well-powered prospective intervention trials.
In the study results, significant heterogeneity was observed. We have already noted that differences in sample sizes, study designs, and inconsistencies in vitamin D testing methods may explain some of the inter-study variation. Some subgroups included only a few studies, which led to limited statistical power and explanatory capacity. Moreover, because many of the following clinical factors were not consistently reported in the included studies, it was not feasible to perform formal subgroup analyses on them. In the included studies, the enrolled stroke populations were heterogeneous. For example, Safari et al. 29 restricted inclusion to patients with acute IS involving the middle cerebral artery territory, whereas other studies recruited a broader and more generalizable cohort of hospitalized patients with acute IS. Clinically, differences in stroke subtype composition across studies may reflect distinct pathophysiological mechanisms, inflammatory responses, and recovery trajectories, which could, in turn, influence baseline vitamin D status. In addition, Masoud et al. 22 enrolled only thrombolyzed patients, while most other studies did not explicitly report acute-phase treatment details; such discrepancies in reperfusion therapy and related management may substantially affect short- and mid-term functional outcomes. Inflammatory status and nutritional phenotypes (e.g. obesity and malnutrition) are also important confounders that may vary across study populations. Both obesity and chronic inflammation have been associated with lower serum vitamin D levels. 52 However, many of the included studies did not report or adjust for these factors, potentially confounding our findings. Seasonal variation, latitude, and sun-exposure patterns may have further influenced baseline vitamin D levels. Among the included studies, only Kim et al. 27 considered season as a covariate and adjusted accordingly, whereas other studies did not explicitly address seasonality. Moreover, the studies included in the present meta-analysis spanned geographic regions from higher to lower latitudes with potentially distinct sunlight exposure profiles; however, these factors were rarely considered. Finally, different assays were used to measure vitamin D levels (e.g. ELISA, chemiluminescent immunoassay (CLIA), chemiluminescent microparticle immunoassay (CMIA), and electrochemiluminescence immunoassay (ECLIA)), and results were generally not standardized across methods, which could have led to inter-assay measurement differences of up to approximately 20%–40%. 53 Although a sensitivity analysis by assay type was attempted, the relatively small number of included studies available for each assay method precluded a meaningful evaluation. Collectively, these clinical and methodological sources of heterogeneity may have introduced residual confounding, measurement error, and between-study variability in effect estimates, thereby limiting causal inference. The high degree of heterogeneity reflects the complexity of real-world research and substantially limits the generalizability of the pooled outcomes.
This study had certain limitations that should be considered when interpreting the results. First, although we conducted subgroup analyses according to income level, sample size, and economic level across countries, these were insufficient to adequately explain or reduce the observed high heterogeneity. This may limit the confidence of the pooled effect estimates and affect the generalizability of the findings. Second, most of the evidence in this study comes from observational studies; therefore, no causal inferences can be drawn. Additionally, serum 25(OH)D levels measured within 24 h of admission may be influenced by acute-phase stress, inflammation, or treatment, and thus may not represent long-term vitamin D levels. Third, the detection platforms, calibration traceability, and threshold definitions for serum 25(OH)D levels were inconsistent across studies. This inconsistency may lead to measurement errors or misclassifications, affecting the comparability and precision of the effect sizes. Fourth, only two studies addressed cognitive outcomes; thus, the evidence is weak, and the combined results are exploratory. The limited number of studies on certain outcomes also weakened the publication bias test, potentially failing to fully exclude small-sample effects. Fifth, although there were no language restrictions in the search, only full-text English articles were included, and grey literature was excluded. This process may have caused language and publication biases and led to the omission of relevant evidence, particularly from regions such as Asia and the Middle East, where both IS and vitamin D deficiency are prevalent. Therefore, caution should be exercised while applying these conclusions to other healthcare systems and populations. Future studies should standardize the testing methods, thresholds, follow-up, and confounder control and employ multicenter prospective cohorts or RCTs to validate correlations and assess population differences.
Conclusion
This meta-analysis indicates that higher serum 25(OH)D levels at stroke onset are associated with milder neurological deficits (lower NIHSS/mRS scores) and possibly, better cognitive outcomes, with benefits most evident within an optimal range of 30–50 ng/mL. However, substantial heterogeneity and limited cognitive data warrant cautious interpretation, and causality cannot be inferred.
Footnotes
Acknowledgments
We thank the Cuiying Biomedical Research Center, Lanzhou University Second Hospital, and the Academician WorkStation of The Second Hospital & Clinical Medical School, Lanzhou University.
CRediT authorship contribution statement
Consent for publication
All authors have approved the manuscript and agree with its submission.
Data availability statement
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of interest statement
The authors declare no conflict of interest.
Ethics approval and consent to participate
As the study was based on published data, ethical approval and patient consent were not required.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 82201441, 82460245); Natural Science Funds of Gansu (grant number 22JR5RA1002); Second Hospital of Lanzhou University, and the “Cuiying Science and Technology Innovation” Plan for the Applied Basic Research Program (grant number CY2022-MS-A01); Cuiying Scientific Training Program for Undergraduates of Lanzhou University Second Hospital (grant numbers CYXZ2024-09, CYXZPT2025-35).
Supplemental material
Supplemental material for this article is available online.