Abstract
Previous studies have indicated that estrogen may delay disease progression and minimize the cognitive decline in patients with Alzheimer's disease (AD). However, the evidence for an estrogen deficiency in women with dementia and cognitive dysfunction is inconsistent. In the present review, a fixed effect meta-analysis revealed that the hormone replacement therapy (HRT) group exhibited significant improvements in Alzheimer Disease Assessment Scale-Cognitive subscale scores relative to those observed in the placebo group, suggesting that HRT is feasible for treating cognitive decline in patients with AD. However, no significant differences in Mini-Mental State Examination and Clinical Dementia Rating scale scores were observed between the 2 groups. The results of our systematic review indicate that HRT can improve cognitive function in female patients with AD. Due to limitations in sample size and the available literature, further multicenter trials with larger sample sizes are required to support these findings.
Introduction
First described by German physiologist Alois Alzheimer in 1906, AD is a chronic neurodegenerative disease characterized by irreversible memory loss, cognitive impairment, language dysfunction, and personality changes. The pathological hallmarks of AD include neurofibrillary tangles, 1 senile plaques, reductions in the number of cerebral cortical cells, and vascular amyloidosis of the internal cerebral artery. 2 Recent studies have indicated that more than 24 million people are affected by AD worldwide, and this figure is expected to double every 20 years. 3 An estimated 5.8 million Americans of all ages are living with Alzheimer dementia in 2019 and approximately 200,000 individuals under the age 65 who have younger-onset Alzheimer, though there is greater uncertainty about the younger-onset estimate. 4 This number will escalate rapidly in coming years, as the population of Americans aged 65 years and older is projected to grow from 55 million in 2019 to 88 million by 2050. 5
Epidemiological studies have demonstrated that decreases in serum levels of sex hormones occur following both female and male menopause; such decreases are consistent with the progression of AD. In vivo and in vitro studies have indicated that biochemical and neuropathological changes resulting from estrogen deficiencies are associated with changes in brain function in older adults and patients with AD. 6 -8 Recent studies have revealed that the protective effects of estrogen on the central nervous system typically occur via 3 mechanisms. The genomic mechanism is mediated by the estrogen nuclear receptor that promotes the growth of synapses, increasing the expression of nerve growth factors and receptors. In addition, a transmembrane receptor-mediated signal transduction pathway is known to alleviate the pathological changes associated with AD by reducing β-amyloid deposition. 9 Also, anti-inflammatory and antioxidant mechanisms can attenuate the toxicity of amyloid β (Aβ). 10,11
Testosterone can be converted into estradiol in vivo, indirectly enhancing the antioxidative effect of estrogen, improving cerebral circulation, and enhancing hippocampal function. Previous studies have demonstrated that testosterone can reduce amyloid aggregation in the brain. 12 Accumulating evidence suggests that decreases in serum levels of estradiol or testosterone increase the risk of developing AD, supporting the notion that hormone replacement therapy (HRT) can be helpful to this patient population.
Indeed, neuropathological, human, 13 and animal studies have highlighted the benefits of HRT among patients with AD. 14 Since estrogen inhibits the formation of Aβ oligomers, 15 it may delay the progression of AD. Additional research has indicated that estrogen may help to minimize cognitive decline and olfactory. 16 However, some studies including the first published large-scale study, the Women’s Health Initiative Memory Study, reported disappointing results for HRT therapy. The study showed that HRT increased the risk of dementia and was correlated with poorer cognitive outcomes in older postmenopausal women; these results were an unexpected contrast to previous findings. 17 -20 Nevertheless, a recent review indicated that HRT may still be an effective treatment for AD but further investigation is required. 21 In the present study, we aimed to review the safety of HRT and its efficacy for improving cognitive function in patients with AD.
Materials and Methods
Inclusion and Exclusion Criteria
For the present review, we included national and international studies published since the year 2000. All included studies utilized a randomized controlled double-blind design in which patients were diagnosed with AD in accordance with the National Institute of Neurological and Communicative Disorders and Stroke/AD and Related Disorders Association (NINCDS/ADRDA) criteria. 22 Studies were excluded due to poor quality, missing/repeated data, and a lack of clear AD diagnoses.
Search Strategy
We searched the following databases for relevant studies published between 2000 and 2017: China Academic Journals Full-text Database, Chinese Biological and Medical Database, VIP database, PubMed, Web of Science, Embase, and Medline. Search terms were as follows: AD OR dementia OR AD, cognitive function, and hormone therapy OR androgen OR estrogen.
Quality Assessment
Article quality was assessed in accordance with the Jadad scale (Table 1). 23 Each article was evaluated by 2 independent researchers and disagreements were resolved via joint discussions with a third party.
Jadad Scale for Reporting Randomized Controlled Trials.

Data Extraction
We extracted the following data from included articles: first author, date of publication, country of origin, experimental methods, total number of participants, results for the test/control groups, basic characteristics (age, gender, level of education, disease course, and so on), interventions (drug, dose, and treatment strategy), follow-up period, and adverse drug reactions.
Statistical Analysis
RevMan5.3 software was used to evaluate the heterogeneity of the included studies. A fixed effects model was used in the absence of heterogeneity (P ≥ .05, I2 < 50%), while a random effects model was used in the presence of heterogeneity (P < .05, I 2 > 50%). Mean differences and 95% CIs were also calculated. The results are shown as forest plots. Funnel plots were used to evaluate publication bias. The level of statistical significance was set at P < .05.
Study Retrieval
Of the 107 studies retrieved, 4 randomized controlled double-blind trials including 277 participants were finally incorporate into our analysis (Figure 1). A total of 160 and 117 participants were included in the HRT and placebo groups, respectively. The quality scores of all included studies were high (Table 2), given the maximum scores described in Table 1. The information extracted from each study is presented in Table 3.
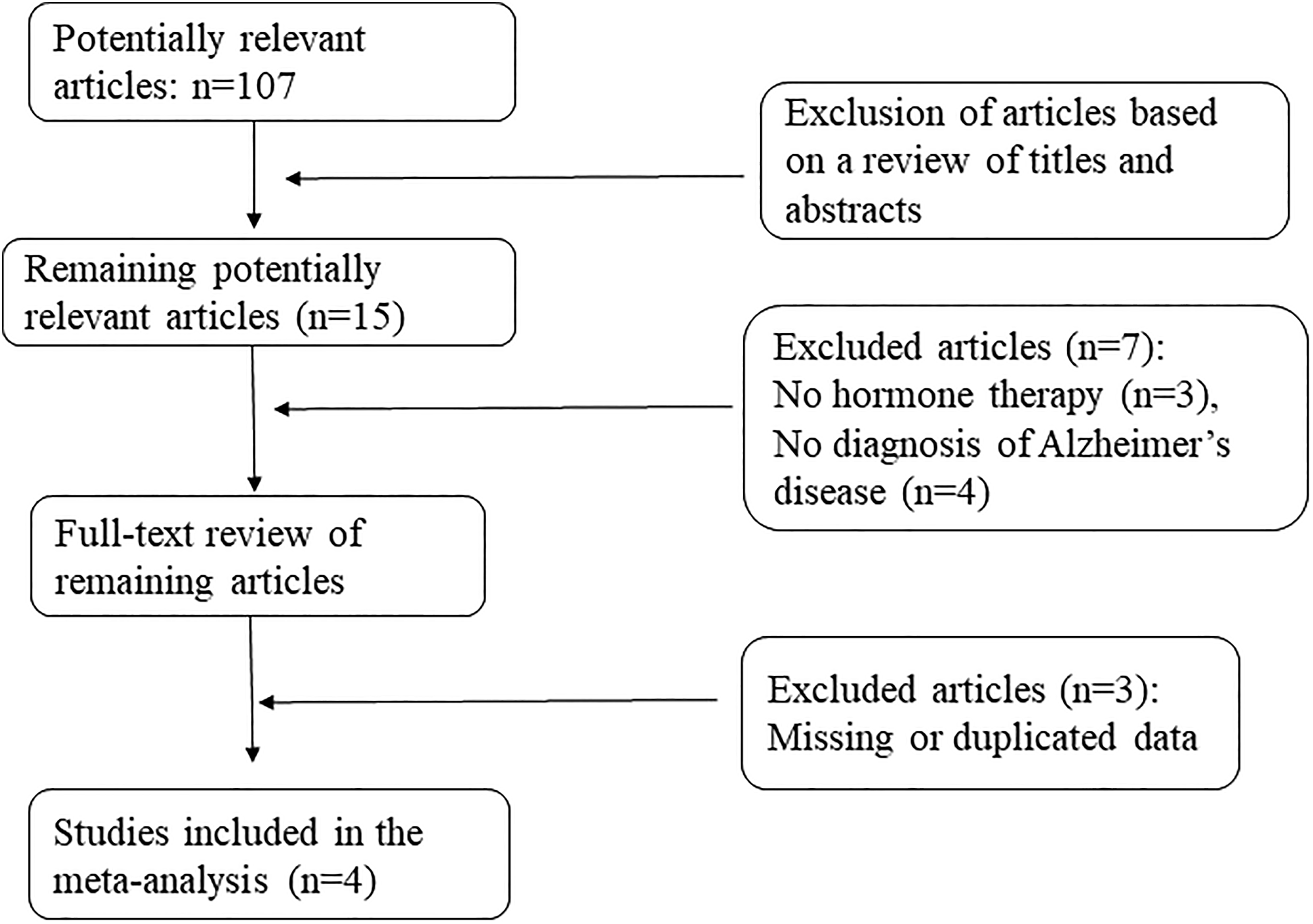
Flow chart outlining the search strategy and results of each step.
Study Quality Based on Evaluation Using the Jadad Scale.

Characteristics of the 4 Included Randomized Controlled Double-Blind Trials.

Abbreviations: ADAS-cog, Alzheimer Disease Assessment Scale-cognitive subscale; ADL, activities of daily living; CDR, Clinical Dementia Rating; CGIC, Clinical Global Impression of Change; HRT, hormone replacement therapy; IADL, instrumental activities of daily living; MMSE, Mini-Mental State Examination.
Results
Outcome Indicators
Mini-Mental State Examination score
We analyzed Mini-Mental State Examination (MMSE) data for 193 participants across 3 of the 4 included studies, as 1 study did not have MMSE data. Our results indicated that there was no significant difference in MMSE scores between the HRT and placebo groups among the studies (P = .79; Figure 2).

Forest plot of MMSE scores for experimental hormone replacement therapy and control groups. MMSE indicates Mini-Mental State Examination.
Alzheimer Disease Assessment Scale-Cognitive subscale (ADAS-Cog) scores
We analyzed ADAS-Cog data from 133 participants for the 2 studies that had ADAS-Cog data. Our analysis revealed significant differences in ADAS-Cog scores between the HRT and placebo groups (P = .004; Figure 3).

Forest plot of ADAS-Cog scores for experimental hormone replacement therapy and control groups. ADAS-Cog indicates Alzheimer Disease Assessment Scale-Cognitive subscale.
Clinical Dementia Rating (CDR) scores
We analyzed CDR scores for 144 patients in the 2 studies that contained CDR data. No statistically significant differences in CDR scores were observed between the HRT and placebo groups (P = .30) (Figure 4).

Forest plot of CDR scores for experimental hormone replacement therapy and control groups. CDR indicates Clinical Dementia Rating.
Risk of Bias
The risk of bias for each included study is summarized in Figure 5. For the fourth category, detection bias, and the sixth category, reporting bias, we were unable to obtain comprehensive results for the analyzed articles.

Risk of bias.
Publication Bias
Funnel plots, which analyze publication bias, are presented with the study precision on the y-axis and the study result on the x-axis. All funnel plots exhibited bilateral symmetry, indicating that there was no significant publication bias regarding MMSE (Figure 6), ADAS-Cog (Figure 7), or CDR scores (Figure 8).
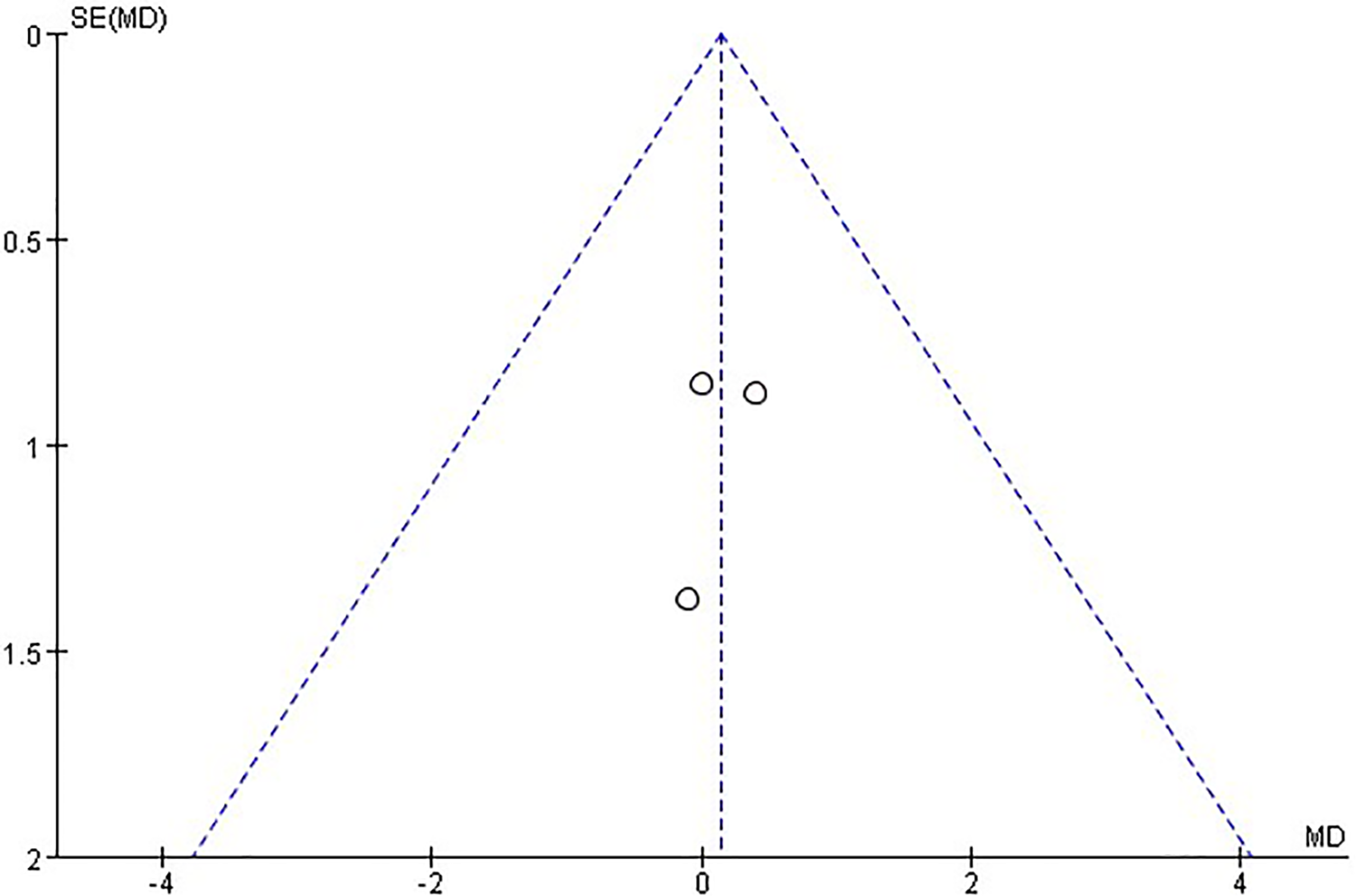
Funnel plot for MMSE score. MMSE indicates Mini-Mental State Examination.

Funnel plot for ADAS-Cog score. ADAS-Cog indicates Alzheimer Disease Assessment Scale-Cognitive subscale.

Funnel plot for CDR score. CDR indicates Clinical Dementia Rating.
Safety Analysis
Three of the 4 studies reported adverse events during the trials. The number of adverse events was similar in estrogen-treated groups and placebo groups. Also, there was no significant difference in treatment-emergent events between the groups. All 3 studies reported vaginal bleeding in the estrogen-treated group (44.0% in Wang et al, 27 5% in Mulnard et al, 26 and 3% in Valen Sendstad et al 24 ) but not in the placebo group. The reports did not state whether vaginal bleeding stopped after the trial. None of 3 studies performed a comprehensive gynecologic examination for the participants who experienced vaginal bleeding during the trial; therefore, it is difficult to ascertain the severity.
Reports have shown that women taking low dosages of estrogen (0.3-mg conjugated estrogen or its equivalent) are less likely to have undesirable side effects, such as vaginal bleeding or breast tenderness. 28 A recent review showed that conjugated estrogens/bazedoxifene (CE/BZA) treatment reduces hot flash frequency and severity, prevents bone loss, reduces bone turnover, improves vaginal maturation index ratings, promotes lubrication, enhances some measures of sleep, and enriches menopause-specific aspects of quality of life. 29 A random placebo trial revealed that adverse events, such as breast pain and vaginal bleeding, occurred significantly earlier with conjugated estrogens/medroxyprogesterone acetate (CE/MPA) versus CE/BZA or placeboes. 30 Therefore, we hypothesize that vaginal bleeding is related to the dosage and duration of estrogen treatment or its equivalent.
Discussion
Understanding the relationship between estrogen and AD may lead to novel therapeutic developments involving strategies utilizing hormone therapy. Estrogens and their receptors play key roles in modulating fatty acid content, utilization, and oxidation through their direct impact on intracellular signaling cascades within the central nervous system. 31 In the present meta-analysis, we evaluated the effects of HRT on cognitive function in patients with AD.
We compared MMSE and CDR scores between HRT and placebo groups, but no significant differences were observed. Developed in the United States in 1975, 32 the MMSE is a simple and user-friendly method for determining cognitive function in patients with a variety of neurological and psychiatric conditions. Although MMSE sensitivity and specificity values are 92.5% and 79.1%, respectively, 33 the rating has some disadvantages. Notably, higher levels of education have been associated with false negatives, whereas lower levels of education have been associated with false positives. For example, scores in patients with low literacy, primary school education, and high school education or greater have been reported as ≤17 points, ≤20 points, and ≤24 points, respectively. Furthermore, the MMSE is not sensitive to mild cognitive impairment, and the simple naming tests associated with the exam are often inadequate for evaluating memory function. Mini-Mental State Examination scores are also influenced by language dialects, often leading to false positives. The lack of change observed in our analysis may be due to the fact that MMSE scores do not account for the effects of dialect or education. High heterogenicity exists in CDR scores between the studies involved. In the study by Mulnardet al, 26 CDR scores between the 2 groups were significantly different but not in the study by Wang et al. 27 The dissimilarity may be related to the duration of treatment, which was 12 months in the study by Mulnard et al and 4 months in the study by Wang et al.
Our fixed-effect analysis revealed significant differences in ADAS-Cog scores between the HRT and placebo groups, indicating that HRT may be feasible for AD treatment. It has been shown that ADAS-Cog are more precise in measuring the severity of cognitive dysfunction than the MMSE, 34 which would hardly be affected by the educational level. 35 Some research studies indicate that ADAS-cog and its subscales could provide maximum information at moderate levels of cognitive dysfunction. 36-37 Thus, the result we got from the ADAS-Cog scores analysis above truly reveals differences in cognitive dysfunction between HRT and placebo group. More specifically, recent study suggests that estrogen-related receptor α plays a functional role in AD pathology. 38 Therefore, HRT may be a successful strategy to impede AD development. However, the present study has some limitations, including the small sample size of 4 randomized controlled trials. In addition, most studies did not report specific adverse events, limiting our ability to evaluate the safety of drug treatment. Furthermore, we observed some clinical heterogeneity in CDR scores among studies, which may have been due to human error when administering the assessment.
In summary, the results of our systematic review indicate that HRT can improve cognitive function in female patients with AD. Due to limitations in sample size and the available literature, further multicenter trials with larger sample sizes are required to substantiate these findings.
Footnotes
Authors’ Note
C.Z., Q.G.W., Y.L., S.L. conceived and designed the study. C.Z. collected the experimental data. C.Z and S.L. performed the statistical analysis. C.Z., Q.W., Z.W., and S.L wrote the manuscript. All authors read and approved the final manuscript. Qing Guang Wu contributed equally to this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China [grant numbers 81673619 and 81973497]. The authors are grateful to the Characteristic Key Discipline Construction Fund of Chinese Internal Medicine of Guangzhou University of Chinese Medicine for additional study funding.
