Abstract
Histological differentiation is a major pathological criterion indicating the risk of tumor invasion and metastasis in patients with hepatocellular carcinoma. The degree of tumor differentiation is controlled by a complex interacting network of associated proteins. The principal aim of the present study is to identify the possible differentiation-related proteins which may be used for early diagnosis and more effective therapies. We compared poorly differentiated and well-differentiated hepatocellular carcinoma tissues by using 2-dimensional gel electrophoresis and matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Among the 11 identified protein spots, 6 were found to be upregulated in poorly differentiated hepatocellular carcinoma tissues and 5 were correspondingly downregulated. Immunohistochemistry was performed on 106 hepatocellular carcinoma tissues to confirm the results of the proteomic analysis. By using bioinformatic tools GO and STRING, these proteins were found to be related to catalytic activity, binding, and antioxidant activity. In particular, our data suggest that overexpression of peroxiredoxin-2, annexin A2, and heat shock protein β-1 was correlated with tumor invasion, metastasis, and poor prognosis, and therefore, these proteins may serve as potential diagnostic and therapeutic biomarkers.
Introduction
Hepatocellular carcinoma (HCC) is one of the most common cancers worldwide and the third common cause of cancer mortality with nearly 746 000 deaths per year. 1 Hepatocellular carcinoma is highly prevalent in Asia, especially in China. The incidence and mortality rates of HCC have increased substantially over the last decades. 2 Despite considerable improvement in the treatment modalities, the overall prognosis for patients with HCC is still poor. Radical resection for the early-stage disease is considered the best treatment option for HCC, but the overall survival is dismal, with the mean and median overall postsurgery survival being 38 and 29 months, respectively. 3 The reported 5- and 10-year overall survival rates were 34.4% and 10.5%, respectively, and the disease-free survival rates were 23.3% and 7.8%, respectively. 3,4 Patients with poorly differentiated HCC are more likely to develop invasive and metastatic diseases with much worse prognosis than those with well-differentiated tumors. 5 The molecular mechanisms dictating tumor differentiation in HCC are poorly understood.
In this study, we aimed to study the differential expression of the proteome between well-differentiated and poorly differentiated HCC tissues. By using 2-dimensional gel electrophoresis (2-DE) technique combined with matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS), we identified several differentially expressed proteins that might prove useful for the diagnosis and prognosis of patients with HCC.
Patients and Methods
Patients, Tissue Collection, and Preparation
Liver tissues were obtained from 20 patients (average age 51.2 ± 12.3 years) who underwent surgical liver resection for HCC at the First Hospital of Lanzhou University. Informed written consents were obtained from all patients and the study protocol was approved by the ethics committee of the First Clinical Medical College of Lanzhou University. The basic demographic data for all patients are listed in Table 1. None of the patients in this study received any preoperative therapy. Tumors were classified into poorly differentiated and well-differentiated groups according to the Specification for Diagnosis and Treatment of Primary Liver Cancer. 6 One hundred milligram of each sample was homogenized in liquid nitrogen and lysed in 0.5 mL lysis buffer (7 mol/L urea, 2 mol/L thiourea, 4% 3-(3-cholamidopropyl)dimethylammoniumpropane sulfonate (CHAPS), 65 mmol/L dithiothreitol, 0.2% Bio-Lyte 3/10 ampholyte, and 40 mmol/L Tris–HCl, pH 7.2) supplemented with 4% complete protease inhibitor cocktail (EDTA-free). The samples were then sonicated on ice for 10 cycles, each consisting of 5-second sonication followed by a 10-second break. After centrifugation at 16 000 rpm for 60 minutes at 4°C, the precipitation was removed and the supernatant retained for subsequent polyacrylamide gel electrophoresis, two-dimensional (2D-PAGE) analysis. Protein concentration was measured by the Bradford assay 7 (Tiangen, Beijing, China).
Patients’ Clinical Data.
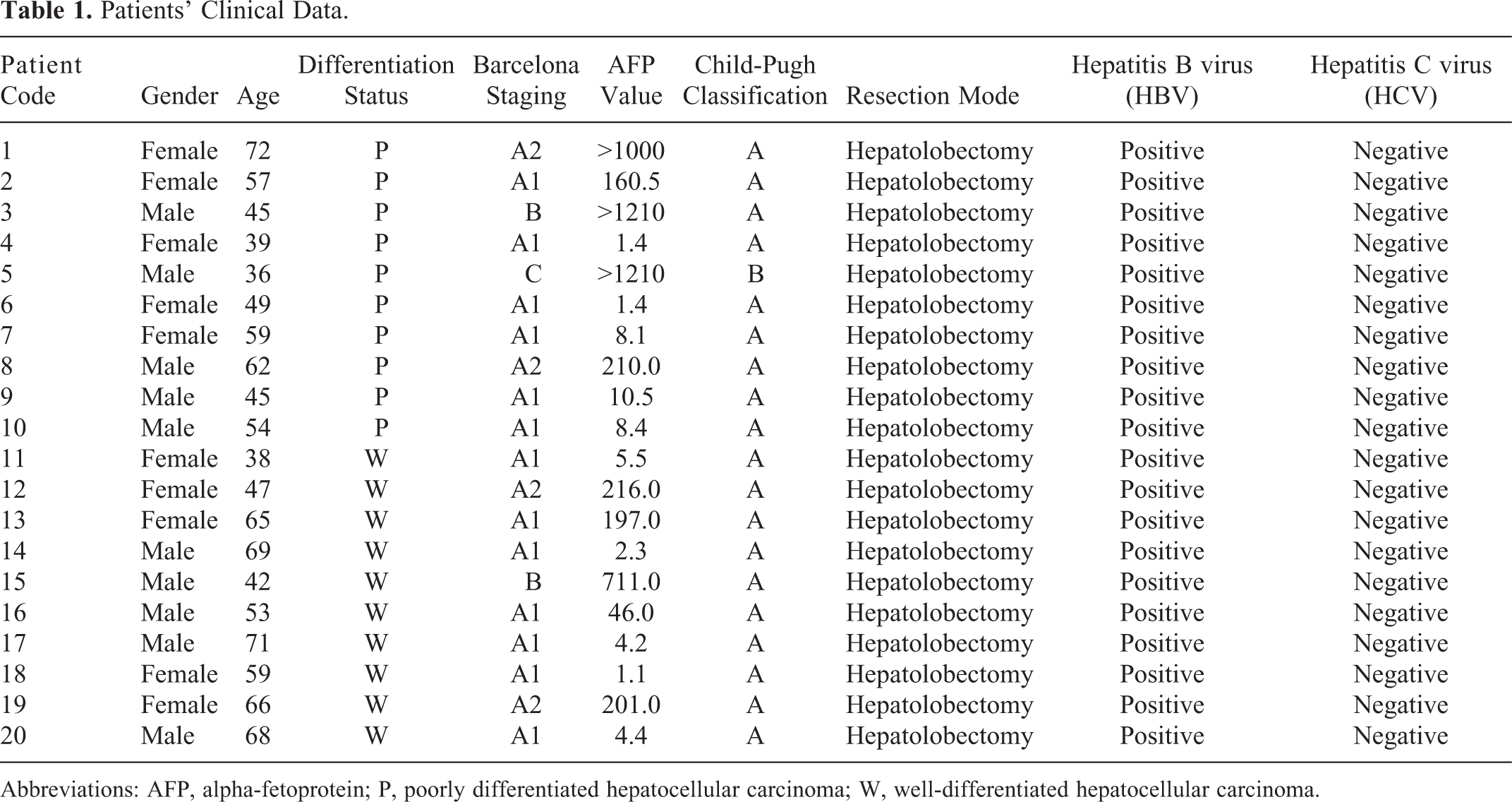
Abbreviations: AFP, alpha-fetoprotein; P, poorly differentiated hepatocellular carcinoma; W, well-differentiated hepatocellular carcinoma.
2-Dimensional Gel Electrophoresis and Image Analysis
The above-prepared protein samples were analyzed by 2-DE, and triplicate electrophoresis was performed for each pair of protein samples (well-differentiated and poorly differentiated). The 2-DE analysis of whole tissue lysates (100 mg) was conducted with isoelectric focusing (IEF) and electrophoresis units. Isoelectric focusing (the first dimension of 2-DE) was conducted with Bio-Rad PROTEAN IEF Cell (Hercules, California), according to the manufacturer’s instructions. Proteins were focused for 30 minutes at 250 V, 30 minutes at 500 V, 2 hours at 1000 V, then a gradient was applied from 1000 to 10 000 V over a period of 5 hours, and focusing was continued at 10 000 V for 6.5 hours to give a total of 95 kVh on an IPGphor. After IEF, pH gradient strips were equilibrated and the samples were subjected to the second-dimensional separation by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) in 1.0-mm-thick 10% polyacrylamide gels in a Bio-Rad PROTEAN II xi Cell system run at a voltage of 80 V per gel for the initial 30 minutes and 220 V for 8 hours thereafter. The second-dimensional SDS-PAGE was developed until the bromophenol blue dye marker had reached the bottom of the gel. The gels were stained using silver nitrate and then scanned with Bio-Rad VersaDoc 4000 imaging system. Automatic image analysis was conducted using the PDQuest 8.0 software (Bio-Rad), followed by manual matching. Protein spots with more than 2-fold change in density in a consistently increased or decreased direction were considered to be differentially expressed and were selected for further analysis.
The MALDI-TOF-MS Analysis and Database Searches
Protein spots of interest were cut out manually from the gel and put into siliconized microcentrifuge tubes. In-gel digestion, MALDI-TOF-MS, and database searching were performed by a commercial company (Shanghai Boyuan Biological Technology Co Ltd, Shanghai, China). The protein spots were analyzed with an ABI 4800 plus MALDI-TOF/TOF mass spectrometer (AB SCIEX, Les Ulis, France) after in-gel digestion. Peptide mass mapping was carried out using the Mascot V2.1 search engine (Matrix Science, London, United Kingdom) against the NCBI protein databases with a GPS Explorer software, V3.6 (Applied Biosystems, Waltham, Massachusetts). Proteins were successfully identified (Figure 1).
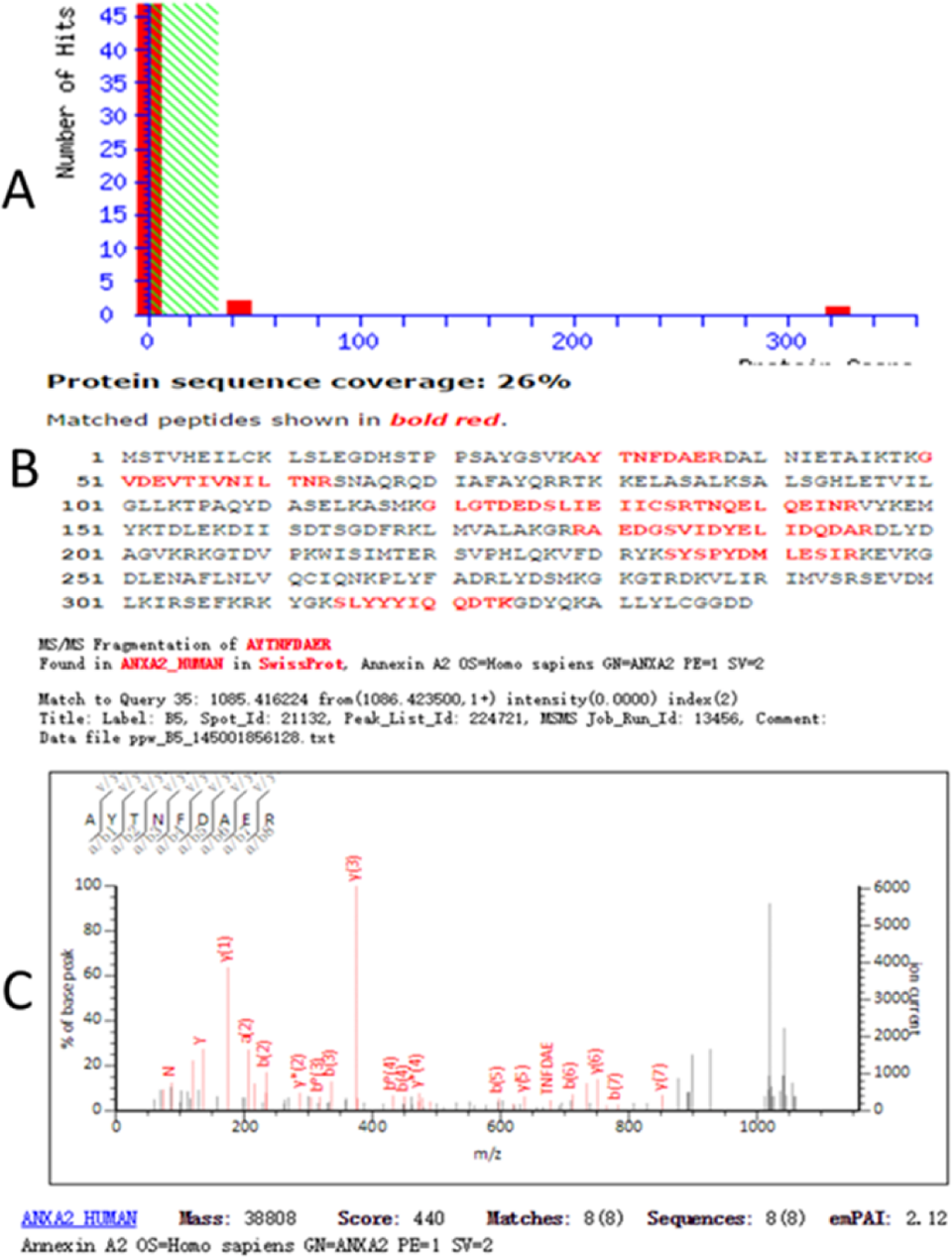
The result of the MALDI-TOF MS analysis of spots cut from the 2-DE gels confirms the protein was ANXA2. A, Mascot histogram of scores; B, protein amino acid sequence, the red parts indicate matching sequence; C, MALDI-TOF-MS of some peptides (MS/MS) maps. ANXA2 indicates annexin A2; 2-DE, two-dimensional gel electrophoresis; MALDI-TOF MS, matrix-assisted laser desorption ionization time-of-flight mass spectrometry.
Immunohistochemistry
To confirm the identified proteins, immunohistochemical staining was performed on 106 formalin-fixed, paraffin-embedded tissue samples from patients with HCC who underwent surgical liver resection at the First Hospital of Lanzhou University from June 2014 to May 2016. None of these patients received chemotherapy or radiation therapy before surgical liver resection. Paraffin-embedded tissues were cut into 4-μm sections using the CSA II system (DAKO, Glostrup, Denmark). For antigen retrieval, the sections were autoclaved in 10 mM citrate buffers (pH 6.0) at 120°C for 10 minutes. Rabbit anti-ANXA2 (Cat. No: YT0236), anti-HSPB1 (Cat. No: YM3356), anti-peroxiredoxin-2 (PRDX2; Cat. No: YT1706), and anti-calreticulin (CALR; Cat. No: YT0620)—all obtained from ImmunoWay Biotechnology Company (Plano, Texas)—were diluted at 1:300. Staining was assessed by 2 independent observers who were blinded to clinical data. Staining evaluation was based on the dominant differentiation area of the tumor tissues if varying degrees of differentiation are present in the entire tumor tissues. Five fields were randomly selected at high magnification (200×), and at least 1000 cells were counted. Positive staining is defined as yellow or yellowish brown granules being present within cell. The staining intensity was semi-quantitatively scored as follows: 0, no stain (colorless); 1, yellow stain; 2, brown stain; and 3, dark brown. Based on the percentage of positive staining, the extent of positivity was semi-quantitatively scored as follows: 0, less than 5% of cells are positively stained; 1, 6% to 25% of cells are positively stained; 2, 25% to 50% of cells are positively stained; 3, 51% to 75% of cells are positively stained; and 4, >75% cells are positively stained. The final positivity score was calculated as staining intensity × staining extent, with 0 being negative, 1 to 3 being weakly positive, 4 to 7 being positive, and 8 to 12 being strongly positive.
Bioinformatics Analysis
The bioinformatics analysis of the expression pattern was performed using the GENECARDS (http://www.genecards.org/), GO (http://geneontology.org/), and KEGG (http://www.kegg.jp/kegg/). Protein network for disease regulated proteins was also constructed using the STRING software (http://string-db.org/). The underlying database informs on known and predicted protein–protein interaction, and the constructed networks are based on active prediction methods of neighborhood, gene fusion, co-occurrence, co-expression, databases, and textmining.
Statistical Analysis
All experiments were repeated at least 3 times. Data from immunohistochemical staining were from a single experiment that was quantitatively similar to the replicate experiments. Statistical significance was determined using unpaired Student t-test. A P value of less than .05 is considered statistically significant.
Results
Profiling of Differentially Expressed Proteins in HCC by 2-DE
A 2-DE using nonlinear immobilized pH gradient (IPG) ranging from pH 3 to 10 was performed to separate the proteins extracted from well- and poorly differentiated HCC tissues. Approximately 650 to 700 protein spots were detected and 184 differentially expressed proteins were identified between the well- and poorly differentiated HCC tissues by 2-DE (Figure 2), in which 40 proteins showed >2-fold disparity in expression levels. The up- and downregulated protein spots exhibiting 4-fold difference in signal intensity (P < .05, t test, 95% confidence intervals) were excised from the gel.

Representative 2-D gel images of HCC tissues depicting identified protein spots that are differentially expressed between the poorly and well-differentiated tissues. 2D indicates 2-dimensional; HCC, hepatocellular carcinoma; P, poorly differentiated HCC group; W, well-differentiated HCC group.
Protein Identification by MALDI-TOF-MS
Eleven protein spots were identified, among which 6 were upregulated and 5 downregulated in poorly differentiated HCC tissues. Corresponding amino acid residue numbers are indicated on peaks that were matched to the identified protein based on a query of the database NCBInr. The upregulated proteins include glutathione-S-transferase ω-1 (GSTO1), PRDX2, annexin A2 (ANXA2), heat shock protein β-1 (HSPB1), haptoglobin (HPT), and enoyl-CoA-hydratase mitochondrial (ECHM). The downregulated proteins include fructose-bisphosphate aldolase A (ALDOA), CALR, fibrinogen β chain (FGB), serotransferrin (TF), and hemoglobin subunit β. The identification information data including isoelectric point (pI), molecular weight, accession number, and molecular weight search (MOWSE) score are summarized in Table 2.
Proteins Identified by MALDI-TOF-MS.a
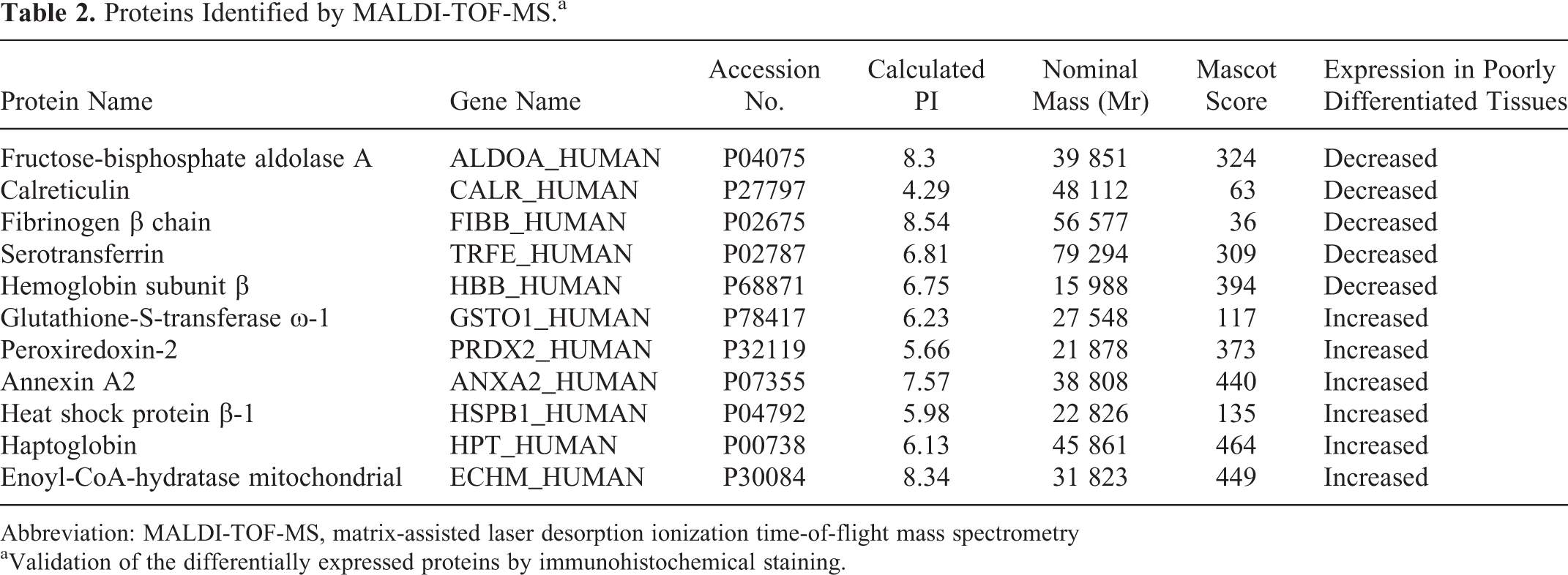
Abbreviation: MALDI-TOF-MS, matrix-assisted laser desorption ionization time-of-flight mass spectrometry
aValidation of the differentially expressed proteins by immunohistochemical staining.
The ANXA2, HSPB1, PRDX2, and CALR were validated by the immunohistochemical staining (Figure 3). The expression of ANXA2, HSPB1, and PRDX2 was closely correlated with the tumor differentiation status, which was decreased from poorly differentiated tumor to well-differentiated tumor (Figure 3C-H). The expression of CALR had an opposite pattern, which was increased from poorly differentiated tumor to well-differentiated tumor (Figure 3I-J). In addition, ANXA2 was mainly localized on the cytomembrane of HCC cells (Figure 3C and D). By the independent samples t test, the differential expression of all 4 proteins was statistically significant (P < .01; Figure 4).

H&E staining demonstrating the well-differentiated HCC (A) and poorly differentiated HCC (B). Immunohistochemistry for ANXA2 in poorly differentiated HCC (C), ANXA2 in well-differentiated HCC (D), PRDX2 in poorly differentiated HCC (E), PRDX2 in well-differentiated HCC (F). Heat shock protein β-1 in poorly differentiated HCC (G), HSPB1 in well-differentiated HCC (H), CALR in poorly differentiated HCC (I), and CALR in well-differentiated HCC (J). ANXA2, annexin A2; CALR, calreticulin; HCC, hepatocellular carcinoma; HSPB1, heat shock protein β-1; PRDX2, peroxiredoxin-2.

The semi-quantitative analysis of the expression levels of 4 proteins in poorly differentiated (P) and well-differentiated (W) HCC tissues as determined by immunohistochemical staining was performed by t test. HCC indicates hepatocellular carcinoma.
Bioinformatics Analysis
According to the GO (http://geneontology.org/) classification system, the biological roles of the differentially expressed proteins were categorized into 16 different categories (Figure 5A). By molecular functional analysis, ECHM, PRDX2, and GSTO1 were shown to be involved in catalytic activity (36%), FGB and CALR in binding (29%), TF in transporter activity (7%), ALDOA in enzyme regulator activity (7%), PRDX2 in antioxidant activity (7%), and GSTO1 in structural molecule activity (7%; Figure 5B). By biological process analysis, the largest proportion of differentially expressed proteins including CALR, GSTO1, ANXA2, HSPB1, and ECHM was in metabolic process (28.6%; Figure 5C). By pathway analysis, HSPB1 was shown to be involved in many signaling pathways such as vascular endothelial growth factor (VEGF) (P00056), p38 mitogen-activated protein kinase (MAPK; P05918), angiogenesis (P00005), and cholecystokinin A receptor (CCKR) (P06959). FGB was shown to be involved in plasminogen-activating cascade (P00050) and blood coagulation (P00011) and ALDOA in fructose galactose metabolism (P02744) and glycolysis (P00024; Figure 5D).

Pie chart depicting the identified proteins characterized by GO category. Proteins were classified in terms of their roles in protein class (A), molecular function (B), biological process (C) and pathway (D).
Analysis by active interaction sources from textmining, experiments, databases, co-expression, neighborhood, gene fusion, and co-occurrence by STRING indicated that ANXA2, PRDX2, and HSPB1 interacted with multiple proteins such as matrix metalloproteinase-2 (MMP2), MMP9, tumor protein p53 (P53), MAPK14, mitogen-activated protein kinase-activated protein kinase 2 (MAPKAPK2), and plasminogen activator. All of the proteins were shown to be involved in cell proliferation, invasion, metastasis, and the development of malignant tumors (Figure 6).
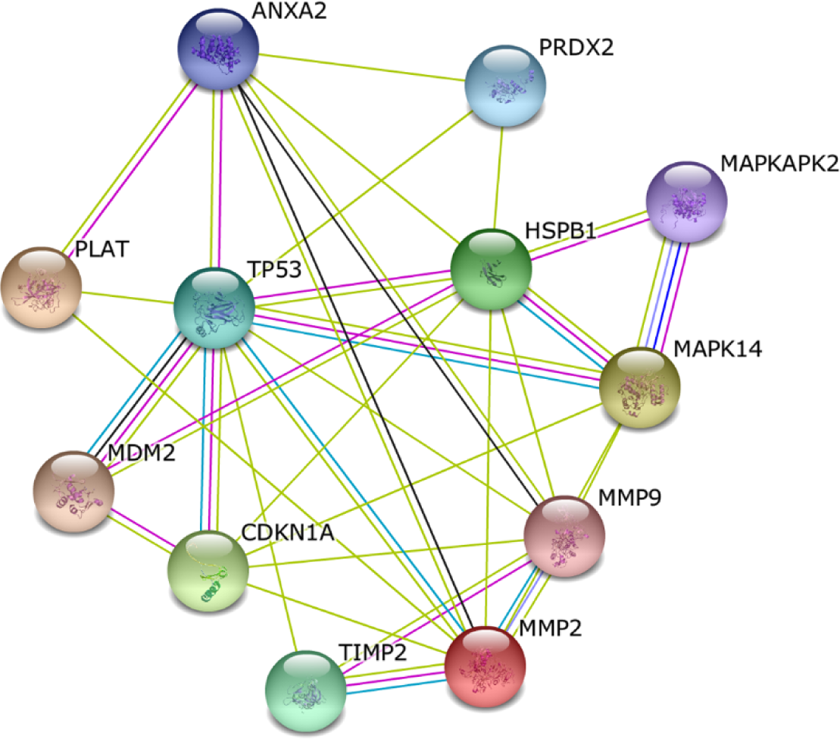
The simulated functional network of proteins that were differentially displayed in STRING database.
Discussion
Histological differentiation is an important factor affecting the prognosis of malignant tumors. Poorly differentiated HCC tissues tend to have stronger ability to invade the neighboring tissues and migrate to distant organs. 8 In this study, we identified 11 proteins that showed the most significantly differential expression between the well- and poorly differentiated HCC tissues. Six proteins were found to be upregulated in poorly differentiated HCC tissues. Glutathione-S-transferase ω-1, HPT, and ECHM were reported in many types of cancers such as cancers of lung, breast, head and neck, bladder, and colon, 9 –13 but their expression status in liver cancer has been rarely reported. In contrast, ANXA2, PRDX2, and HSPB1 have been well studied in HCC, and their biological functions have been shown to be directly or indirectly implicated in the malignant behavior of HCC cells.
ANXA2 belongs to ANXA protein family with membrane-bound and Ca2+-mediated capacities. 14 It is expressed in most eukaryotic cells. 15 ANXA2 may form single, heterodimer, and heterologous tetramers and plays a critical role in the invasion, metastasis, and angiogenesis of solid tumors. 16 ANXA2 has been reported overexpressed in differentiation status of many cancers, and as such dysregulation of ANXA2 has been reported to be involved in the development and metastasis of these cancers. 17 In Ras homologous member A (RhoA) and Ras-related C3 botulinum toxin substrates 1 (Rac1) signaling pathway, the phosphorylated ANXA2 together with Rho-associated protein kinase (Rho/ROCK)-mediated actin restructuring and cell adhesion promotes amoeboid movement and mesenchymal movement of HCC cell. 18 The ANXA2 binds to protein ligand P11 to form heterogeneous tetramers on the outside of the cell membrane and interacted with tissue plasminogen activator. The heterogeneous tetramers enhance the fibrinolytic enzyme on tumor angiogenesis and tissue reconstruction. 19 –21 Heterogeneous tetramers also activate MMPs, which facilitate the degradation of the extracellular matrix (ECM) and therefore promote tumor invasion and metastasis. 22 Previous research has shown that the overexpression of ANXA2 is correlated with poor prognosis in many types of cancer, such as gastric carcinoma, 23,24 colorectal cancer, 25,26 esophageal squamous cell carcinoma, 27,28 renal cell carcinoma, 29,30 lung cancer, 31 and multiple myeloma. 32 ANXA2 is an effective method for evaluating the extent of angiogenesis and for distinguishing HCC from nonneoplastic liver tissue, but the role of ANXA2 in neovascularization is not completely understood. 33 As demonstrated by immunohistochemical staining, ANXA2 protein is mostly located on the cell membrane. Overexpression of ANXA2 was present in poorly differentiated HCCs (P < .01). Although the source of ANXA2 in poorly differentiated HCC is not clear, the fact that ANXA2 is overexpressed in poorly differentiated HCC tissues indicates its role in mediating the invasion and metastasis of HCC.
HSPB1 is an important member of the HSP family. 34 Its main function is to protect cells from stress injuries, but it is also involved in cell differentiation and invasion, 35 signal transduction, 36 and apoptosis. 37 In VEGF signaling pathway, HSPB1 indirectly activates prion protein identification, causing intracellular accumulation of actin and alterations of cell morphology, thereby contributes to cell movement. 38 Heat shock protein β-1 has also been found to increase the activity of MMP2 and MMP9, leading to degradation of ECM and increased potential for invasion and metastasis of malignant cells. 39 Previous research has shown that silencing of HSPB1 significantly reduced the migration of invasive SK-Hep-1 cells, besides poorly differentiated HCCs tended to express more HSPB1 than well-differentiated HCCs. 40 HSPB1 together with its partners is suggested to contribute to tumor growth and progression in HCC by activating multiple signaling pathways responsible for the HCC proliferation and other malignant features. 41 In our study, immunohistochemical staining has revealed a significant overexpression of HSPB1 in poorly differentiated HCCs. Hence, HSPB1 may likely serve as a biomarker for early detection and therapeutic targets for HCC. More studies in animal model and large cohort of patients with HCC are warranted to confirm this aspect.
Peroxiredoxin-2 is involved in the regulation of redox generation, cell division, and differentiation. 42 It can enhance the cytotoxic activity of NK cells. 43 However, the role of PRDX2 on regulating tumor growth and development has not yet been clearly defined, and even in the same cancer, conflicting results have been published. It was reported that PRDX2 could scavenge reactive oxygen species such as hydrogen peroxide and promote tumor cell apoptosis. 44 In colorectal cancer, PRDX2 could promote cancer growth by stimulating vascular genesis and protecting cancer cells from oxidative stress–induced cell death. 45,46 However, overexpression of PRDX2 was also found to inhibit transforming growth factor (TGF) β1–induced epithelial–mesenchymal transition and migration of colonic epithelial cells. 47 In HCC, the role of PRDX2 is inconclusive. In our current study, overexpression of PRDX2 in HCC was mainly observed in poorly differentiated cancer tissues.
The downregulated protein CALR is a multifunctional protein mostly residing in endoplasmic reticulum (ER), where it functions as a molecular chaperone and a regulator for Ca2+ homeostasis. 48,49 Furthermore, non-ER CALR is also involved in the regulation of many biological functions such as gene expression, transcriptional regulation, RNA stability, immune response, cell proliferation, migration, adhesion, and apoptosis. 50 More evidences indicate that CALR has great impacts for the development of different cancers and the effect of CALR on tumor formation and progression may depend on cell types and clinical stages. 51 –53 In addition, CALR is required for TGF-β-stimulated ECM production. 54 In HCC cells, CALR can inhibit their growth, invasion, and cell cycle progression by downregulating CALR. 50 In our study, overexpression of CALR was mostly observed in the well-differentiated HCC tissue.
Network analysis and literature mining revealed that the identified proteins were functionally linked to certain signal pathways that are known to be frequently activated in HCC and many other malignant tumors. 55,56 By bioinformatics analysis, we propose that ANXA2, PRDX2, and HSPB1 may interact with MMP2 and MMP9 to enhance tumor cell invasion, and they may interact with P53, MAPK14, and MAPKAPK2 to accelerate tumor cell proliferation. They may interact with VEGF and p38 MAPK to promote the development of malignant tumors. Overexpression of ANXA2, PRDX2, and HSPB1 may indicate a poor differentiation of HCC tumors and a poor prognosis of patients with HCC.
Conclusion
Six proteins were found to be significantly upregulated in poorly differentiated HCC tissues, among them overexpression of ANXA2, PRDX2, and HSPB1 was significantly associated with invasion and metastasis and is closely correlated with the degree of histological differentiation. These proteins may play an important role in the development of HCC and may serve as potential diagnostic and therapeutic biomarkers for this cancer.
Footnotes
Abbreviations
Authors’ Note
Sheng Zhao and Gang Su contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Natural Science Foundation of China (31570509), Gansu Provincial Administration of traditional Chinese Medicine(GZK-2015-69), The provincial National Natural Science Foundation of Gansu (145RJA113), West Light Foundation of The Chinese Academy of Science [(2015) 90], The Health Industry Research Program Management Project of Gansu Province Fund (GWGL2010-23), and Science and Technology Project of China National Petroleum Corporation Lanzhou Petrochemical Company (LZSH201715-2).
